Abstract
Introduction: Deep brain stimulation (DBS) of the posterior hypothalamus has been found to be effective in the treatment of refractory chronic cluster headache (CCH).
Methods: We report the long-term outcomes of five patients with refractory CCH on whom stimulation of a modified target of approximately 3 mm in radius, which included the posterolateral hypothalamus, the fasciculus mammillotegmentalis, the fasciculus mammillothalamicus and the fasciculus medialis telencephali, was performed. The stereotaxic coordinates were 4 mm from the third ventricle wall, 2 mm from behind the mid-intercommissural point and 5 mm from under the intercommissural line.
Results: All patients became pain-free for 1–2 weeks after the procedure, but then needed an average of 54 days to optimize stimulation parameters. After a mean follow-up of 33 months, two remain pain-free, two have an excellent response (>90% decrease in attack frequency) and in one the attacks have been reduced by half. There were no serious adverse events. Permanent myosis and euphoria/well-being feeling were seen in three patients. Other adverse events, such as diplopia, dizziness, global headache of cervical dystonia, were seen transiently related to an increase in stimulation parameters. Attacks reappeared transiently in two patients as a result of cable rupture and when the stimulator was disconnected.
Conclusions: Our results supports the efficacy of DBS in very refractory CCH with a slightly modified hypothalamic target conceived to avoid the lateral ventricle wall so as to extend the stimulated brain area and to decrease the morbidity of potential haemorrhagic complications.
Keywords
Introduction
Cluster headache (CH) is an infrequent neurovascular primary headache, which is included in the group of trigeminal autonomic cephalgias. Its clinical picture is characterized by severe unilateral headache episodes lasting 30–120 minutes and accompanied by autonomic signs or symptoms secondary to a parasympathetic hyperactivity and sympathetic hypoactivity (1). About 10–15% of CH patients develop the chronic form (CCH) (1), of which 10–20% do not respond to any type of medication (2).
The exact aetiology of CH is unknown. However, a number of clinical, laboratory and, recently, functional neuroimaging data strongly suggest that CH attacks are generated in the hypothalamus (3,4). As a consequence of this evidence, neurostimulation of the posteroinferior hypothalamus has been carried out in selected, refractory CCH patients in the past decade. Results with this new surgical approach have been reviewed recently for a total of 52 patients (5). Although in general this technique has been shown to be effective in almost two-thirds of cases, the results of these groups are not homogenous.
The purpose of this article is to show the clinical outcomes on five refractory patients with CCH who were treated with a modified posterolateral anatomical target of deep hypothalamic stimulation. This area was chosen mainly to try to stimulate several fasciculi that exchange autonomic information between the hypothalamus and the brainstem and to avoid the damage to the third ventricle wall.
Patients and methods
Patients
Clinical characteristics of the patients and treatments prior to surgery

CH: cluster headache, CCH: chronic cluster headache, F: Female, M: Male. Data are shown as mean ± SD or as a range.
They all had been treated, without success, with maximum therapeutic doses and adequate trials of verapamil, topiramate, valproate and lithium (as well as other miscellaneous medications) alone and in combination. Four had also failed thermocoagulation or gamma-knife procedures and one had not responded to bilateral suboccipital stimulation for 1 year. They all used domiciliary oxygen but needed several daily injections of subcutaneous sumatriptan (except patient 4, who suffers from ischemic cardiomyopathy) and variable daily dosages of steroids. Three patients had developed serious complications due to the chronic consumption of steroids (Table 1). Neurological examination, including neuropsychological battery, brain magnetic resonance imaging (MRI) and the rest of the pre-operative tests were normal in all patients.
Patients signed the informed consent for this treatment, which had been previously approved by our ethics committee for patients refractory to all other treatments, showing no psychiatric disorder and being stable in the CCH situation for at least 2 years. They all were asked to fulfil a headache diary for at least 6 months before and all the time after the operation. Patients were seen by the neurosurgeon (FS) and the neurologist with interest in headache (JP) every 2 weeks the first 2 months and then at least once every 3 months.
Surgical technique
All patients were referred to our centre (which is national reference for the functional neurosurgical treatment of both refractory neuropathic pain and movement disorders) by neurologists specialized in headache. All patients were operated in the same centre by the same team and with the same general surgical technique used for surgery for movement disorders (7).
The stereotaxic coordinates used were: 4 mm lateral to the third ventricle wall, 2 mm behind the mid-intercommissural point and 5 mm under the intercommissural line. Implantation of the tetrapolar electrode (model no. 3389, Medtronic, Minneapolis, MN, USA), always ipsilateral to the pain side, was performed under local anaesthesia plus sedation and multi-unit recording. A minimum of three recordings were conducted on all patients. The first recording was to locate the subthalamic nucleus (X: 12 mm lateral to the intercommissural line, Y: 2 mm behind the mid-intercommissural point and 5 mm under the intercommissural line). The second was to situate the anterior edge of the subthalamic nucleus, and the last one was to look for activity in the target point. This last recording had no specific pattern and always had low bioelectrical activity. An electrophysiological pattern compatible with red nucleus activity was not obtained in any of the recordings. As explained in the informed consent form, for 4 days after the electrode implant and by means of its external connection, neurophysiological studies were carried out for the purpose of investigating the maximum threshold and the side effects of neurostimulation, sleep patterns and other types of neurophysiological changes (8) On day 5 of the implant, all patients underwent a control MRI in accordance with the standards of limitation for the MRI head specific absorption rate (9). The images were obtained using a SIGNA (General Electric Medical Systems, Milwaukee, WI) MRI equipment of 1.5 tesla with CVI gradients (40 mT/m, 150 mT/ms).
The Cartesian coordinates of the active contact were measured in the MRI from the central point of the radiological artifact produced by the active contact at the mid-intercommissural point. The anatomical structures that theoretically remained within a 3 mm radius of action from the centre of the active contact were projected onto various atlases (10–13) using the same reference points (intercommissural line and related planes). Twenty-four hours later and under general anaesthesia, an Itrel 3-type neurostimulator (Medtronic, Minneapolis, MN, USA) was implanted in the infraclavicular region ipsilateral to the electrode.
Results
Measurement of electrode locations
The mean stereotaxic coordinates of the effective active contact, as measured by the MRI, were 4.83 ± 0.65 mm lateral to the third ventricle wall, 1.40 ± 0.56 mm behind the mid-intercommissural point and 4.16 ± 0.40 mm below the intercommissural line.
Clinical results
Postsurgical results are shown in Table 2. All patients dramatically improved after the initial operation, even with the stimulator not in operation. However, this improvement was transient, lasting up to 2 weeks in all cases. After this initial period, they required repetitive adjustments of the amplitude and pulse with of the stimulation to maintain a prolonged stimulation effect.
Postoperative axial MRI at the lowest electrode contact of our patients (patients 1 to 5 from left to right). T2-weighted axial MR image shows the location of the active contact (black arrow). In all of the images, contact can be seen in front of and slightly lateral to the red nucleus (asterisk) and lateral to the fasciculus mammillothalamicus (white arrow). Cartesian coordinates of the active contact, stimulation parameters and clinical outcomes X: distance in mm from the lateral wall of the third ventricle, Y: distance in mm from behind the mid-intercommissural point, Z: distance in mm from under the intercommissural line. Follow-up time: pain-free period starting from the optimization of the stimulation parameters. Values are shown as mean ± SD

The average time to optimize the neurostimulation parameters was 54 ± 19 days. The mean follow-up time period has been 33 months (range 12 to 48 months). Two patients remain totally pain-free, in two both the frequency of the attacks and the need of subcutaneous sumatriptan have decreased more than 90%, while in patient number 5 the number of attacks is halved. Without exception, all patients could give up steroids. Four patients have taken verapamil after the procedure. Two patients remain on verapamil (patients 1 and 5) as they are not totally pain-free, while in patient 4, who is pain-free, verapamil was kept due to hypertension and ischemic cardiomyopathy. Patient 2 has taken verapamil only temporally due reappearance to cluster headache attacks, after the neurostimulator had to be switched off when he was diagnosed and underwent surgery for adenocarcinoma of the floor of the mouth. No other preventatives have been needed.
Adverse events
There was no significant comorbidity related to the surgical procedure. After an excellent response for 18 months, patient 1 required a consultation for reappearance of several daily cluster headache attacks. A rupture of the intracerebral electrode was diagnosed by skull X-rays and a new intracerebral electrode was implanted.
Regarding treatment-related adverse events, persistent myosis and euphoria/well-being feeling were seen in three patients, while occasional dizziness (three patients), blurring vision/diplopia (two patients), concentration difficulties (one patient), cervical dystonia (one patient), generalized headache (one patient) and increase in appetite (one patient) were seen transiently when the stimulation threshold was increased. Patient 1 had two typical cluster headache attacks on her right side for the first time just after the procedure. During the follow-up none of the patients had alterations in heart rate, blood pressure, respiratory function or changes in sleep-wake cycles. Neuropsychological examination showed objective changes after the procedure in any patient.
Discussion
This small series supports the reported efficacy of hypothalamic DBS for the treatment of refractory chronic cluster headache patients using a slightly modified hypothalamic target 4 mm lateral to the ventricular wall with the aim of increasing the stimulated area and avoiding potential bleeding secondary to the ventricular damage (2,5,14–22). After a mean follow-up of 33 months, the longest in the literature, two of our stimulated patients remain pain-free, two have a dramatic reduction in attack frequency and the fifth, with only 12 months of follow-up, has an average reduction in frequency of 50%. All our patients suffered from multiple daily attacks (three to eight attacks daily) for more than 2 years (2 to 15 years), during which all relevant drugs, alone and in combination and at high doses, were tried and produced no significant benefit. They were also refractory to thermocoagulation (two patients, two procedures in one patient and four in the remaining one), gamma-knife (one patient, two procedures) and bilateral occipital nerve stimulation (1 patient). Among other drugs, before hypothalamic DBS three patients had been taking daily steroids for more than 1 year. All three had developed serious complications (see Table 1). Although four patients remained on verapamil (in one patient because of ischemic cardiomyopathy and not because of cluster headache attacks, and in another only transiently), they all were able to stop the remaining preventatives, including steroids, with a very clear improvement in medication-related adverse events. Without exception, they could return to work. Perhaps as a result of a direct impact of the tetrapolar electrode causing a microlesion or a neuronal shock (23) and concurring with previous series, all patients became transiently (up to two weeks) pain-free after the implantation of the DBS. It took, however, almost 2 months of repeated adjustments of stimulation parameters to obtain permanent relief. Our efficacy results, with an average success of 86%, are even better than those found in the previously reported patients with hypothalamic DBS implants (5,14). There were two clinical data that, in our opinion, reinforce the efficacy of hypothalamic DBS: the reappearance of the attacks after 18 months in patient 1, which was solved after the ruptured electrode was replaced, and the worsening of the attacks when the stimulator was transiently switched off in patient 2.
Hypothalamic stimulation was well tolerated by our patients. There were no serious complications. Coinciding with previous reports, the most common adverse events were myosis, dizziness, euphoria/well-being and double vision. Except for myosis and the euphoria/well-being feeling, all the remaining adverse events were transient and related to an increase in the stimulation parameters. Also concurring with previous series, one patient had, for the first time, typical cluster headache attacks for a few days on the contralateral side.
We have stimulated new target of a brain volume of approximately 3 mm in radius, which included a lateral hypothalamic area (LHA) and the fasciculi mammillotegmentalis (FMTG), mammillothalamicus (FMTH) and medialis telencephali (FMTL) or medial forebrain bundle. The surgical target selected initially by the group from Milan (14,17,19,24,25) is based on the theory of posterior hypothalamic nucleus (PHN) dysfunction as the origin of CH (4). It has not been firmly established, however, that the PHN is the primary anatomical generator of CH attacks (17–26). The PHN has vague limits, continuing on the lateral side of the nucleus hypothalamicus lateralis (NHL) (10) or the LHA (27). There is no consensus as to whether the volume of stimulated brain tissue forms a part of the PHN or of the periventricular/periaqueductal grey matter (5,24,28,29). Some authors even believe that there is no contact whatsoever located in the PHN but in posterior hypothalamic region (25). In addition, a shift of just 2 mm lateral to the intercommissural line, chosen as the lateral coordinate, increases the risk of injuring the ventricular ependyma and may lose the area of stimulation (17). This target is the same one used in the treatment of aggressivity. Electrical stimulation of this point can provoke sympathetic responses, rotating movements of the eyeball and encephalographic changes, among other responses (30).
Our coordinates were selected on the basis of four factors. First, the description of three areas in the posterior section of the lateral wall of the third ventricle described by Sano et al. (31) was used: an internal area of 0 to 1 mm that demonstrated parasympathetic responses to electrical stimulation, a medial area of 1 to 5 mm that has sympathetic responses and a third lateral area of more than 5 mm that presents parasympathetic responses. Second, stimulation of a point in the posterior hypothalamus located at 3 mm under the mid-intercommissural point and 5 mm from the lateral wall of the third ventricle provoked parasympathetic responses. Third, a point located at 4 mm from the third ventricular wall, 2 mm behind the mid-intercommissural point and 5 mm under the intercommissural line, would be located in the pre-rubric region of the tegmentum and would cross both the FMTH and the FMTG (12,13). The FMTG originates from the mammillary bodies and is orientated in an upward direction and its anterior-superior section winds behind the red nucleus to subsequently descend towards the tegmentum and ends in the nucleus tegmentalis dorsalis (Gudden) and in the nucleus reticularis tegmenti pontis (Bechterew) (13). Finally, the nucleus tegmentalis dorsalis sends efferents towards the nucleus salivatorius superior, nucleus salivatorius inferior and nucleus solitarius (13). In summary, the FMTG exchanges autonomic information between the hypothalamus and the vegetative nuclei of the cranial pairs.
As shown by the French group, the coordinates of the effective active contacts of other authors were 2.8 ± 1.0 mm lateral to the intercommissural line, 1.97 ± 1.8 mm posterior to the mid-intercommissural point and 2.85 ± 1.5 mm under the intercommissural line (19) or 2.98 (1.16) mm lateral to the intercommissural line, 3.53 (1.97) mm behind the intercommissural point and 3.31 (1.97) mm under the intercommissural line (25). Concurring with these measurements, the volume of neuromodulated brain by active contact would include the FMTL (19), the mesencephalic grey matter, the red nucleus, the fasciculus retroflexus (Meynert) and the fasciculus longitudinalis dorsalis (Schütz), among others (31), indicating that this target would be closer to the mid-intercommissural point than that used by the other authors (14,17,20). This fact confirms that the target in the group from Milan was getting closer to this point, from 6 to 2 mm posterior to the intercommissural point and from 8 to 5 mm under the intercommissural line (21,29).
In our patients, the mean stereotaxic coordinates of effective contact were 4.83 ± 0.65 mm from the third ventricular wall, 1.40 ± 0.56 mm from behind the mid-intercommissural point and 4.16 ± 0.40 mm under the intercommissural line. The mean difference between the surgical and final coordinates were 0.83 ± 0.65 mm for X, 0.6 ± 0.57 mm for Y and 0.83 ± 0.41 mm for the Z coordinate. This difference is due to the fact that the error in a deep brain electrode implant can be 1.5 ± 1 mm (32,33).
Therefore, the active contact would be located in the pre-rubric area of the tegmentum, taking approximately 1 mm in front of the FMTL and 1.5 mm at the NHP and NHL, at 1 mm in front of and 1 mm over the FMTG, and 1 mm behind and 2 mm inside the FMTH (11,12). If we take the measurements of the tetrapolar electrode into consideration (model no. 3389, Medtronic, Minneapolis, MN, USA) and the electrical parameters used, the cerebral volume under the field of action of the active contact would be a sphere of approximately 3 mm in measured radius (data provided by Medtronic, Minneapolis, MN, USA), which would have the above-mentioned anatomical structures in its field of action (Figures 2 and 3). In the future, the previous and our target areas might be stimulated along the same stereotactic trajectory with a proper multicontact electrode inserted between the two targets to compare the two sites and to boost the therapeutic possibilities for CCH.
(A) Coronal schematic drawing at 1 mm behind the mid-commissural point. The location of the tetrapolar electrode is represented, showing its relationship with neighbouring structures; the posterior hypothalamic area (PH), subthalamic nucleus (STN), lateral hypothalamic area (LH), fasciculus mammillothalamicus (MT) and fasciculus mammillotegmentalis (mtg). Reprinted with permission from reference (12). (B) T2-weighted coronal MR image showing the location of the implanted electrode in patient 1. The deepest contact is at 4.4 mm from the wall of the third ventricle and 4 mm under the AC-CP line. (C) Coronal diffusion tensor tractogram, which has been co-registered and overlaid onto the T2-weighted images, demonstrating the relationship of the main white matter tracts around the electrode influence area. Sagittal neuroanatomy atlas section with radiological correlation. (A) Schematic drawing of a sagittal section through the mammillary bodies and red nucleus showing the major white matter tracts of the diencephalic area. Modified with permission from reference (13). 1: red nucleus, 2: mammillary bodies, 3: column of fornix, 4: tractus mammillothalamicus, 5: tractus mammillotegmentalis, 6: tegmentum mesencephali. (B) T1-weighted sagittal MR image (patient 1) showing the location of the active contact of the implanted electrode. (C) Sagittal diffusion tensor tractogram, which has been co-registered and overlaid onto the T2-weighted images, demonstrating the relationship of the main white matter tracts around the electrode influence volume.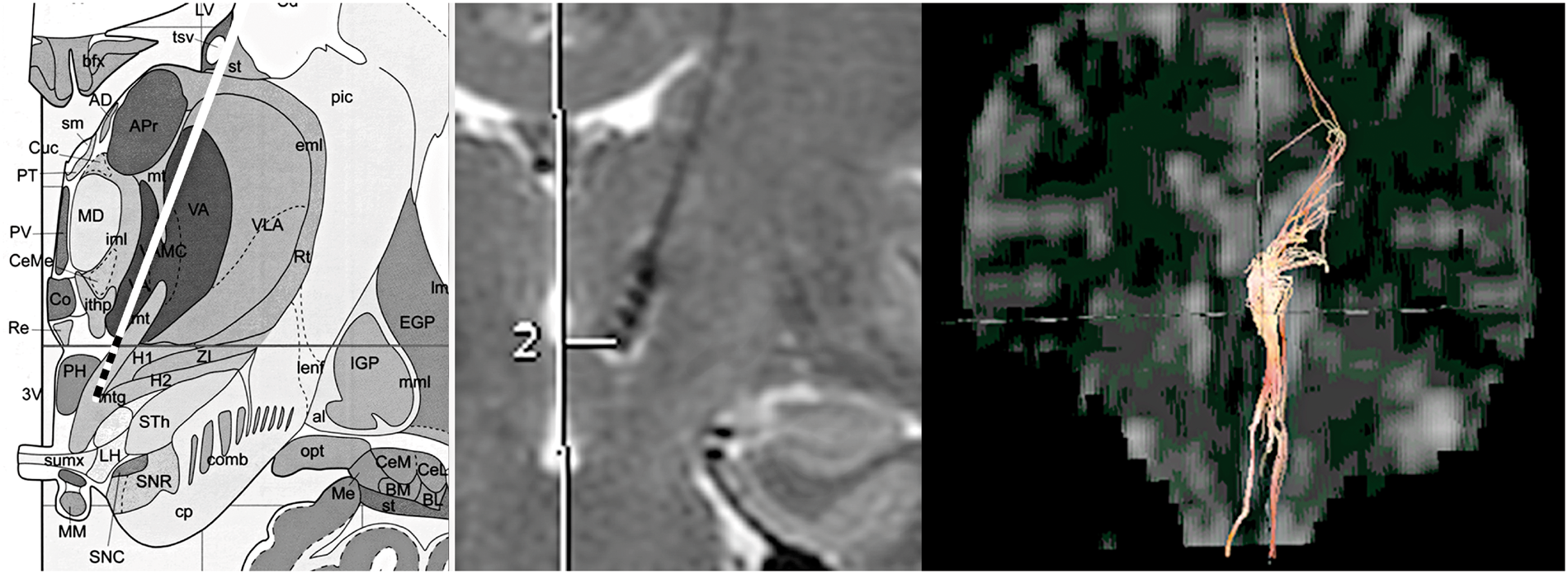

Although there is no consensus on how cerebral neurostimulation provokes its therapeutic effects on CCH (34–36) or on other trigemino-autonomic cephalalgias (37–40), we believe that by neuromodelling the FMTG, whether directly or through the NHL and NHP, brainstem parasympathetic hyperactivity would be attenuated. On the other hand, stimulation of the FMTL could modulate the brain areas involved in the emotional component of pain, such as the anterior cingulate cortex, the insula and the contralateral thalamus (5), and the painful experience of CH could be modulated through the memory circuit and anterior nucleo-mammillary tubercles of the thalamus-hippocampus. Likewise, stimulation of the FMTL could model the mesocorticolimbic dopaminergic reward system (41) being able to produce mood changes by affecting the frontobasal areas involved in conduction and provokes an additional effect on the affective pain element. This mechanism is probably the reason for the ‘calming effect’ that three of our patients reported and that was also observed by other authors (18).
In summary, stimulation of all these structures would exercise a rather complex neuromodelling effect on hypothalamo-brainstem neuronal circuits and cortical structures involved in pain development in CH (34). Although the number of cases in our series is limited, the good clinical response of our patients leads us to suggest that the target currently used so far needs to be slightly modified. Further studies are needed to confirm our outcomes.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflicts of interest
The authors declare that there is no conflict of interest.
