Abstract
The purpose of this study was to evaluate the efficacy of topiramate in the treatment of chronic migraine. This was a double-blind, randomized, placebo controlled, parallel-group study. Patients suffering from chronic migraine with analgesic overuse were randomly assigned in a 1 : 1 ratio to receive topiramate or placebo. Following a baseline phase of eight weeks, the study drug was titrated in 25-mg increments over one week to 50 mg daily. Titration phase was followed by a 8-week maintenance phase. Number of days with headache during a 28-day period was the efficacy variable. At baseline, there was no difference in the number of days with headache between patients treated with topiramate and those treated with placebo (mean ± SD: 20.9 ± 3.2 and 20.8 ± 3.2, respectively). During the last 4 week-maintenance phase, topiramate-treated patients experienced a significantly lower 28-day headache frequency in comparison to those treated with placebo (mean number of days with headache ± SD: 8.1 ± 8.1 vs. 20.6 ± 3.4, P < 0.0007). Topiramate at low doses proved to be an effective therapeutic approach to reduce headache frequency in patients with chronic migraine and analgesic overuse.
Introduction
The term chronic daily headache (CHD) refers to a group of disorders characterized by high frequency headaches, often associated with considerable disability (1). One of the most frequent conditions included in the CDH group is the drug-induced rebound form that can occur in migraine patients with analgesic overuse (chronic migraine with analgesic overuse). There is still no satisfactory therapeutic approach for these patients. Pathophysiological considerations suggest that the possible basis for the shift from episodic to chronic headache can be progressive damage to the central nociceptive system (2). Topiramate is a relatively new antiepileptic drug whose effects include action on the GABA and glutamate systems (3). This fact could explain its possible efficacy in the treatment of pain. In this study we sought to verify the possible advantage of topiramate treatment in a group of patients with a severe form of chronic migraine with analgesic overuse.
Subjects and methods
We included 28 consecutive patients referred to our Headache Centre who were diagnosed as affected by CDH (4). Specifically, we selected subjects suffering from chronic migraine with acute medication overuse (5). All included patients had a history of migraine without aura attacks (6) before the occurrence of chronic migraine for at least 10 years. All patients previously had failed to respond to at least four preventive adequate doses of medication for an adequate duration. Exclusion criteria were: use of carbonic anhydrase inhibitors, history of renal calculi, pregnancy or lactation. Patients with neurological diseases or taking any prophylactic treatment for headache at the moment of our observation were also excluded. Patients maintained a daily headache diary, recording frequency, duration, severity of their headaches and abortive drug use for the entire study period. At the moment of inclusion, an assessment of disability was performed by means of the Migraine Disability Assessment (MIDAS) questionnaire (7).
The study comprised the following phases:
An 8-week baseline phase;
A 1-week titration phase in which patients were randomized on a 1 : 1 basis to receive topiramate or placebo;
An 8-week maintenance period, divided in two 4-week phases.
The length of the baseline period was established as 8 weeks to obtain a mean of a 4-week period headache frequency in order to optimize the real quantification of the chronic migraine severity. In fact, at the first visit, some patients reported a certain variability in the number of days with headache from one month to another.
The following therapeutic scheme was used: 1 tablet containing placebo or 25 mg topiramate for seven days and then a further tablet was added (placebo or 25 mg topiramate). Each patient received the entire quantity of tablets containing topiramate 25 mg or placebo necessary to complete the study at the moment of the randomization. The dose of topiramate (50 mg) or matching placebo remained constant during the following 8 weeks of the study. During the study period patients were asked to reduce analgesic administration and in any case to not take it more than once a day.
The efficacy end point was the reduction in the 28-day headache frequency from the baseline phase to the first and second 4-week period of the maintenance phase.
An interim history, review of the headache diary and a report of any adverse event was performed every two weeks of the maintenance phase. Moreover, patients were allowed to have telephone contact at any time during the study.
All patients gave their informed consent to participate. The study was approved by the Local Etichs Committee.
Statistical analysis was performed by means of a Multivariate analysis of variance (MANOVA) with repeated measures. The Ducan's multiple range test was used for the posthoc comparisons. The rates of patients who, respectively, reported 50%, 75% and 90% reduction in headache frequency in each group, after the first and second 4-week period of treatment were compared using a χ2 test. The odds-ratio (with 95% confidence limits) of days with headache reduction was also computed.
Results
Table 1 shows the characteristics of patients. There was no difference regarding demographic and baseline characteristics. Comorbidity for fibromyalgia and psychiatric disorders was not found in any patient. Preventive medication for migraine had been interrupted at least three months before our observation. All patients had severe disability defined as grade IV at MIDAS: more than 21 days with significant reduction in social and work activities in the last three months. Table 2 reports the individual and mean number of days with headache in the two groups of patients during the baseline and the treatment periods. For the baseline phase, the mean between the first and second 4-week period was considered. The number of days with headache was stable in all patients but three (one in the topiramate group and two in the placebo group). In these cases, there was a difference of two days with headache between the first and second 4-week period of the baseline phase. All patients but one receiving topiramate completed the study. The reason for the drop-out was related to gastric intolerance that occurred during the titration phase. Other patients reported some transient and mild in severity side-effects that did not lead to further drop-out (Table 3). No patient was able to completely withdraw analgesic use. However, they all respected our suggestion to limit their use to no more than once a day.
Demographic and baseline characteristics of patients

28-day headache frequency at baseline (B) and in the first (T1) and second (T2) four-week phase of the maintenance period in patients receiving topiramate or placebo

†Drop-out from the study.
Side-effects reported by patients during the treatment period
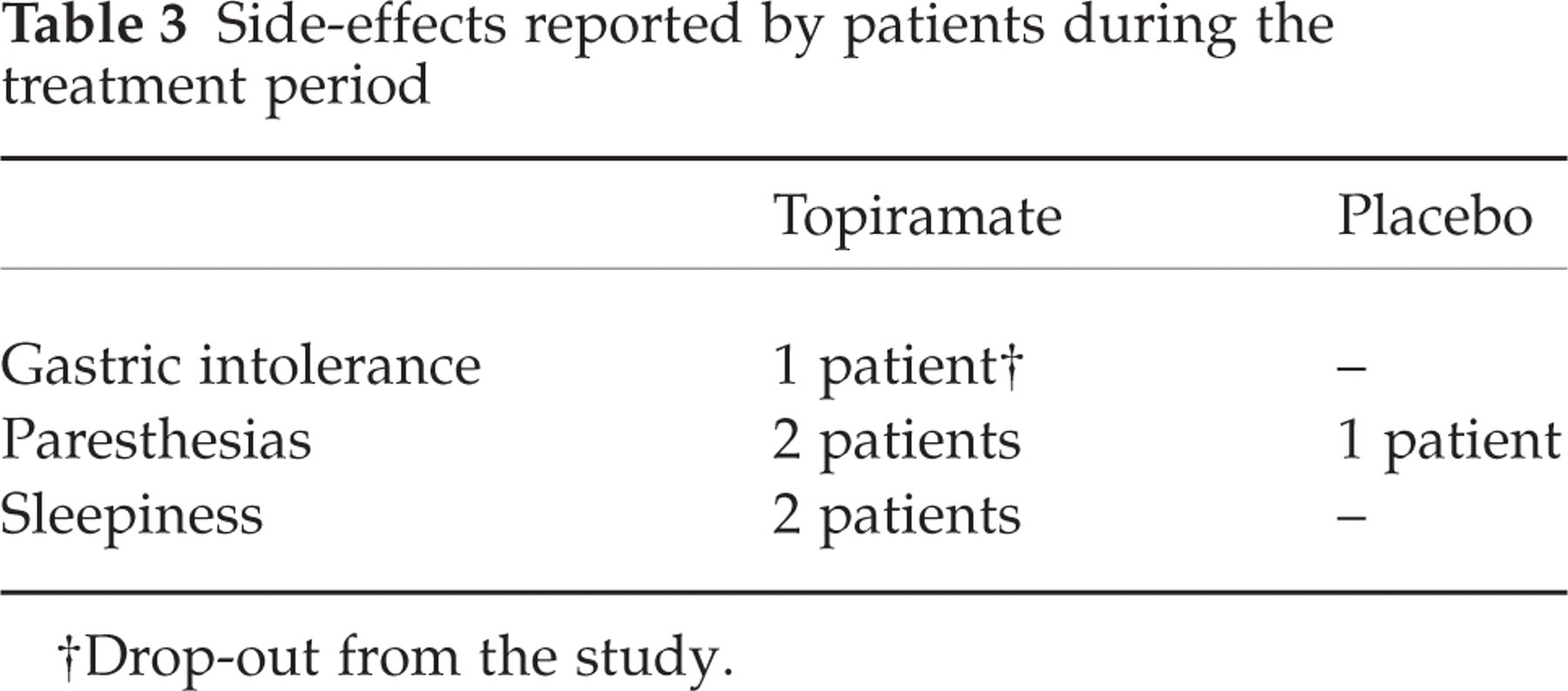
†Drop-out from the study.
The ‘time per treatment effect’ was statistically significant (F (2, 50) = 14,4; P < 0.00001) (Fig. 1). Figure 2 shows the mean number of days with headache during the two phases of the maintenance period. The intragroup comparison showed a significant progressive reduction in the number of days with headache only in the topiramate group (F (1,25) = 14,5; P < 0.008). The posthoc effects showed that there was no difference in the headache frequency at baseline between the two groups (topiramate group: mean ± SD 20.9 ± 3.2; placebo group: mean ± SD 20.8 ± 3.2). The mean number of days with headache in the topiramate group measured at the end of phase 1 of the treatment period (15.3 ± 7.5) was significantly lower with respect to the baseline period (P < 0.03) and to the placebo group at the same time(20.3 ± 2.8, P < 0.005). The mean number of days with headache in the topiramate group measured at the end of phase 2 of the treatment period (8.1 ± 1.8) was significantly lower with respect to the baseline period (P < 0.0007) and to the placebo group at the same time(20.6 ± 3.4, P < 0.0005). Finally, the mean number of days with headache in the topiramate group was significantly lower at the end of phase 2 with respect to the end of phase 1 of the treatment period (P < 0.03).

Changes in the mean number of days with headache (calculated over a 28-day period) during the different phases of the study in patients treated with topiramate (•) or placebo (○).

Mean number of days with headache during the first (phase 1) and second (phase 2) 4-week maintenance phases in patients treated with topiramate or placebo. □ Mean; shaded boxes show standard error and error bars show SD .
At the end of phase 1 of the treatment period, the 50% responder rate was 28% (4 patients) in the topiramate group and 0% in the placebo group (odds ratio: 55.6, 95% c.l. 0.1–31121). At the end of phase 2 of the treatment period, the 50% responder rate was 71% (10 patients) in the topiramate group and 7% (1 patient) in the placebo group (odds ratio: 32.5, 95% c.l. 3.1–337). Patients reporting more than 75% reduction in headache frequency were 6 (42%) in the topiramate group and 0 in the placebo group (odds ratio 36, 95% c.l. 1.76–733), whereas those referring a greater than 90% benefit were 4 (28%) in the topiramate group and 0 in the placebo group (odds ratio 10.8, 95% c.l. 0.5–228).
Discussion
The possibility of using topiramate for pain treatment is based on some of the central effects of the drug that include the blocking of voltage-sensitive sodium channels and the kainate/d-amino-3-hydroxy-methyl-4-isoxozole propionic acid (AMPA) species of glutamate receptor, the action on the GABA neurotransmitter system and the increase in human cerebral GABA concentration (8, 9). Accordingly, there is some evidence suggesting that topiramate is effective in the treatment of various chronic pain conditions (10). There is also a limited experience in the use of this drug in primary headache prophylaxis with encouraging results in migraine treatment (11) and contradictory results in cluster headache patients (12, 13). In this study, we evaluated the possible effect on pain relief in a group of patients with CDH. Chronic daily headache is common enough to be considered a public health problem and its impact in reducing the quality of life is significant (14). Therapeutic studies have not supported very encouraging results (15, 16). One possible reason is that CDH refers to different conditions including both primary and secondary headaches whose underlying pathogenetic mechanisms are presumably not the same (1). This implies that each type of CDH would require a different therapeutic approach. For this reason, in this study we focused our attention on patients with a similar clinical condition. In fact, we only included subjects with a history of migraine without aura attacks preceding the transformation to chronic migraine associated with analgesic overuse. In a recent study with magnetic resonance imaging, abnormally elevated iron levels have been found in the periaqueductal grey matter (PAG) of episodic migraine sufferers as well as in patients with CDH with drug overuse (17). This finding has been interpreted as a consequence of impaired iron homeostasis, possibly associated with neuronal damage or dysfunction (18). In the group of CDH patients, the extent of the iron accumulation was strongly associated with the duration of the disease. The functions of the PAG are complex, but there is a lot of experimental and clinical evidence suggesting that its activation may alter the balance of descending influence of on/off neurons on pain transmission, enhancing the on pain-facilitating network (19, 20). Based on these considerations, it seems reasonable to hypothesize a possible beneficial effect on CDH of management strategies directed at interacting with the central mechanisms of pain control and more generically to counteract central nervous system hyperexcitability. This kind of strategy has already been considered. In fact, different studies have shown that the use of divalproex sodium can exert a beneficial effect in some patients with CDH (21, 22).
Our findings suggest that topiramate administration may add a new therapeutic strategy for chronic migraine patients. In the present study, the drug was effective in reducing the frequency of headache in a large percentage of patients. These results are in accordance with those of some preliminary investigations in which topiramate at various doses was used in patients with different kinds of severe persistent intractable headache (23–25). Two other aspects of our study need a comment. The first is related to the important reduction of days with headache occurring in one patient in the placebo group and the second is the lack of drop-outs for patients of both groups who did not show any change in headache frequency during the treatment period. For the first point it is not possible to rule out the effect of a spontaneous variation of the severity of the disease. Regarding full adherence to the study protocol, an important factor in avoiding drop-outs for a poor response to treatment was probably the fact that all patients were carefully followed and allowed to contact at any time the physicians involved in the clinical evaluation. This probably had a psychologically supportive and motivational effect.
In our study the CNS-related side-effects were negligible. Only one patient dropped-out for gastric intolerance. The very good tolerance profile can probably be explained by the fact that the doses used were lower than those usually needed in epilepsy management.
The encouraging results of this investigation suggest it would be opportune to carry out wider studies to confirm our data. In this study, we limited our evaluation to the effect of 50 mg of topiramate. It is possible that an increased dose regimen would lead to further benefits. In fact, at least three patients treated with topiramate did not report a significant decrease in headache frequency. Further, it would be interesting to evaluate whether the positive response to topiramate can be considered persistent in chronic migraine patients even after the discontinuation of treatment or whether chronic or periodic drug administration is necessary.
Footnotes
Acknowledgements
We thank Prof MG Ceravolo and Dr M Capecci for their invaluable help in the statistical analysis
