Abstract
Aim: This study aimed to revisit previous findings of superior processing of motion direction in migraineurs with a more stringent direction discrimination task and to investigate whether increased internal noise can account for motion processing deficits in migraineurs.
Methods: Groups of 13 migraineurs (4 with aura, 9 without aura) and 15 headache-free controls completed three psychophysical tasks: one detecting coherence in a motion stimulus, one discriminating the spiral angle in a glass pattern and another discriminating the spiral angle in a global-motion task. Internal noise estimates were obtained for all tasks using an N-pass method.
Results: Consistent with previous research, migraineurs had higher motion coherence thresholds than controls. However, there were no significant performance differences on the spiral global-motion and global-form tasks. There was no significant group difference in internal noise estimates associated with any of the tasks.
Conclusions: The results from this study suggest that variation in internal noise levels is not the mechanism driving motion coherence threshold differences in migraine. Rather, we argue that motion processing deficits may result from cortical changes leading to less efficient extraction of global-motion signals from noise.
Introduction
Migraine is a common neurovascular condition, characterized by intermittent headaches that affects between 10% and 15% of the population (1,2). Visual aura symptoms including scotomas or blind spots, zigzags of light and stars are reported by about a third of migraineurs, typically before the onset of the headache (3,4). Neuroimaging studies have demonstrated that cortical areas V3a and MT, thought to be involved in motion processing (5–8), are two of the first areas to display cortical spreading depression (9–11). In addition to this, functional MRI studies have demonstrated that migraineurs have increased cortical thickness in areas V3a and MT (also called V5 and involved in motion processing) (7,8,12). Research examining visual processing in migraine has revealed higher motion coherence thresholds (13–19) and also higher coherence thresholds for form detection (17). However, Antal et al. (16) found superior motion direction (up/down) performance in migraineurs compared with controls for a briefly presented (48 ms), 100% coherent display. An up/down judgment is a coarse test of direction coding but this result is at odds with the notion of poorer processing of all aspects of global motion. As it is unclear whether this result arises from the use of a coarse direction discrimination task or from the 100% coherence level, or both, the current study revisits this finding with a more stringent direction discrimination task using both 100% and a lower level of coherence. It remains unclear what is driving the observed performance deficits. One possibility is that the threshold variations could arise from neuronal hyperexcitability in cortical motion areas (V5 and V3a) (19–22), which may result in an impaired ability to integrate local cues into a coherent global motion percept (23). Evidence for hyperexcitability in the visual cortex of migraineurs has come from Transcranial Magnetic Stimulation (TMS) where individuals with migraine required a lower intensity of TMS pulses delivered over visual cortical area V5 to induce phosphenes compared to controls (21,22,24–30). Further evidence for hyperexcitability was found in a study by Antal et al. (31) using functional MRI, which showed significantly stronger activation in the MT complex in response to moving visual stimuli.
There are at least two ways in which the visual cortex could become hyperexcitable: through increased neural excitation or through weaker inhibitory mechanisms. A more excitable system may also result in an increase in internal noise, where internal noise can be defined as the portion of neuronal firing that is unrelated to the presence of signal, creating a baseline level of noise in cortical responses (32). It is unclear whether increased internal noise could account for motion and form coherence performance differences observed in migraineurs.
A direct method for estimating internal noise was used by Levi et al. (33) while investigating amblyopia. The N-pass method requires the observer to complete several runs of identical stimuli which contain both signal and noise. The observer's response to the identical stimuli is recorded (33–36). Internal noise is assumed to be the source of variation that produces inconsistent responses across different passes of identical stimuli, therefore this variation can be used to estimate the proportion of internal noise (35). Expressing the estimates as a proportion also allows for comparison across visual tasks, providing researchers with a metric to illustrate how the proportion of internal noise changes across tasks, cortical visual areas and participant groups.
The current study aimed to (i) investigate a potential mechanism underlying the observed motion processing deficit when the signal is not 100% coherent and (ii) revisit previous findings of superior motion direction processing in migraineurs with a more stringent discrimination task using both 100% and a lower level of coherence.
Motion processing was assessed using the global motion coherence threshold task previously used by McKendrick and Badcock (13), but combined with the N-pass method to estimate the internal noise level to determine whether the threshold difference between groups was due to differences in internal noise levels. Two additional tasks assessed the observer’s ability to determine the spiral angle, the pitch of a logarithmic spiral, in a global stimulus with either 100% coherence or with added external noise. The spiral motion task, requiring angular judgments, also allowed an examination of whether higher motion coherence thresholds for migraineurs might be attributable to poorer identification of motion flow fields.
As difference in coherence thresholds for form processing have also been reported, we incorporated a global form task that used a glass pattern arranged in a spiral shape requiring participants to make judgments regarding the spiral angle (37). This task was chosen as it is comparable to the spiral motion task (both requiring judgments of angle), allowing direct comparisons of performance and internal noise estimates across motion and form tasks. This allowed us to determine whether migraineurs have both global form and motion processing deficits when coherence thresholds are not being assessed, that is, at suprathreshold coherence levels more characteristic of natural viewing.
Methods
Participants
A total of 28 participants aged between 19 and 40 years completed the experiment. Of the 28 participants, 13 experienced migraine (4 had migraine with aura, 9 had migraine without aura) and 15 were headache-free controls. A power analysis was performed (SigmaStat, V3.0, SPSS Inc) using data from studies that had previously used a motion coherence task with migraineurs (13–15,17). The power analysis indicated that 11 participants were sufficient in each group to have 80% power for detecting a medium sized effect (0.5) (38) when using the traditional 0.05 criterion of statistical significance.
The criteria for headache-free controls specified that they must have never experienced a migraine and have less than four spontaneous headaches (i.e. not related to sickness or tension) per year. Migraine participants were asked if they had a formal diagnosis of migraine from their general practitioner or a neurologist. The International Headache Society criteria (39) for migraine were used to ensure that participants met criteria for migraine either with (MA) or without (MO) aura. The frequency of migraine events was between 1 and 50 per year (mean 13.44, SD 13.85) and the duration of migraine history was between 2 and 23 years (mean of 14.83, SD 5.69). In order to assess migraine severity, the Migraine Disability Assessment (MIDAS) was used (40). Scores on the MIDAS are usually interpreted as follows: Grade 1, minimal or infrequent disability (score 0–5); Grade 2, mild disability (scores 6–10); Grade 3, moderate disability (scores 11–20); and Grade 4, severe disability (score 21+). The MIDAS scores in the current study ranged from 0 to 39 with a mean of 19.58 (SD 9.81). Participants were not allowed to be taking any preventative migraine medication and were at least 4 days after migraine cessation at time of testing to ensure no residual effects of medication or the migraine episode such as fatigue. Participants were asked to call and report any migraines they experienced in the 4 days after testing. No participants called. Normal findings in a comprehensive eye examination, which included slit lamp biomicroscopy, ophthalmoscopy and tonometry, were required. All participants had normal or best corrected-to-normal vision of 6/7.5 or better for the experimental working distance, refractive errors less than ± 5.00 D sphere and ± 2.00 D astigmatism. Participants were also required to be free from any medications or diseases that are known to effect visual or cortical function. Human research ethics approval for the study was provided by the Human Research Ethics Committee (The University of Melbourne). Participants provided written informed consent before participation and all aspects of the protocol were in agreement with the tenets of the Declaration of Helsinki.
Apparatus and general procedure
The stimuli were drawn using MATLAB 7 (Mathworks, Natick, 2005) on a host Pentium computer and the images were loaded on to a ViSaGe graphics system (Cambridge Research Systems). The stimuli were displayed on a Sony Trinitron G520 monitor set at a resolution of 1024 × 768 pixels (34° 08’ × 25° 36’), with a frame rate of 100 Hz and a mean luminance of 45 cd/m2. Luminance calibration was performed using an Optical OP 200-E photometer (head model number 265) and associated software to linearize the luminance response (Cambridge Research Systems, Kent, UK). The stimuli were viewed binocularly and all participants were refractively corrected for the viewing distance. The observer viewing distance was maintained using a chin rest.
Participants completed a total of three visual tasks across two testing sessions each lasting 90 minutes: a motion coherence task, a spiral motion task and a spiral global form task (see below for stimulus and task details). The stimuli and the task for each experimental condition was shown to the participant before the commencement of testing and a practice run was completed to ensure that they were not responding randomly. The order of the tasks for each participant was randomized across testing sessions to control for the possibility of order effects. All participants were carefully observed by the experimenter during the testing sessions for compliance and fatigue. No obvious effects of practice on the tasks were observed by the experimenter. In all experiments, the method of constant stimuli (MOCS) was used with seven signal levels and a total of 20 trials per level. Experiments were conducted in a darkened room (< 1 cd/m2 reflected from surfaces) to minimize reflections and interference from other light sources.
So that the internal noise associated with each task could be calculated, the stimuli used on the first run of each condition were saved and then re-ordered and re-presented to the observer. Therefore, in run two and run three of the task, the observer saw identical combinations of stimuli and external noise to those presented in run one but in different trial sequences. A procedure using three runs was chosen, as Levi et al. (41) reported that three runs provided approximately equal reliability to using four runs, but with fewer trials.
In some conditions external noise (described in detail in the experimental sections below) was added to the stimuli in the spiral form and motion tasks to determine whether the level of external noise affects individuals with migraine differently from controls.
Motion coherence task
The stimulus consisted of 100 white dots (75 cd/m2) displayed within a 10° aperture on a black background (0.5 cd/m2). The dots were 8.6 arc min in diameter and had a rectangular profile that was centered on 0 cycles per degree (c/°). The 1st lobe of the sinc function had an area of –7 to 7 c/°. This task is identical to the one used previously (14,16,17). The dots were randomly placed within the aperture initially and then were given the appearance of movement using an eight frame motion sequence in which each frame was shown for 50 ms. The dots moved at a velocity of 2.86°/s. A percentage of the dots moved in a common direction (to the right of the screen). The rest of the dots were moving in random directions, but the chosen direction was not permitted to be within 10° of the signal direction so no noise dots could move in exactly the same direction as the signal dots. The stimulus in the reference interval contained random motion. If the displacement moved the dots outside of the aperture they were randomly re-plotted within the aperture for the next frame. No dots were permitted to be signal dots if the necessary displacement moved them outside of the aperture. Dot overlap was precluded. Any signal dots that overlapped were instead treated as noise dots and another dot was chosen as signal. The dots had a long lifetime but their role as signal or noise dots was randomly allocated on each frame transition. The viewing distance was 40 cm.
The task was a temporal two interval forced choice (2IFC) procedure in which participants were required to decide which of the two presentations contained the coherent flow (to the right). The MOCS procedure was used to control the number of dots moving coherently to the right of the screen. The MOCS levels ranged from 3 to 21% coherence, and this was consistent across all observers. The participants indicated their choice using a button box. Participants completed three runs, each consisting of 140 trials.
Spiral motion task
This stimulus consisted of 100 dots with a 2D Gaussian luminance profile on a gray background (45 cd/m2). The standard deviation of the 2D Gaussian defining the contrast of each dot was 4’ and had a maximum luminance of 90 cd/m2. The stimulus had a dot density of 1.27 dots per square degree of visual angle. To create the spiral motion pattern, the dots were initially positioned at random on the points of intersection of an invisible polar grid comprising 100 radii equally spaced in polar angle and 100 concentric circles. The radii of the circles were given by An illustration of how the spiral motion pattern was constructed. α represents the spiral angle; β(n) represents the added external noise angle specific to each dot pair drawn from a uniform distribution. All dot pairs in the pattern were oriented at α to the local radius when there was no external noise. When external noise was added all dot pairs were oriented at an angle of α + β(n) relative to the local radius.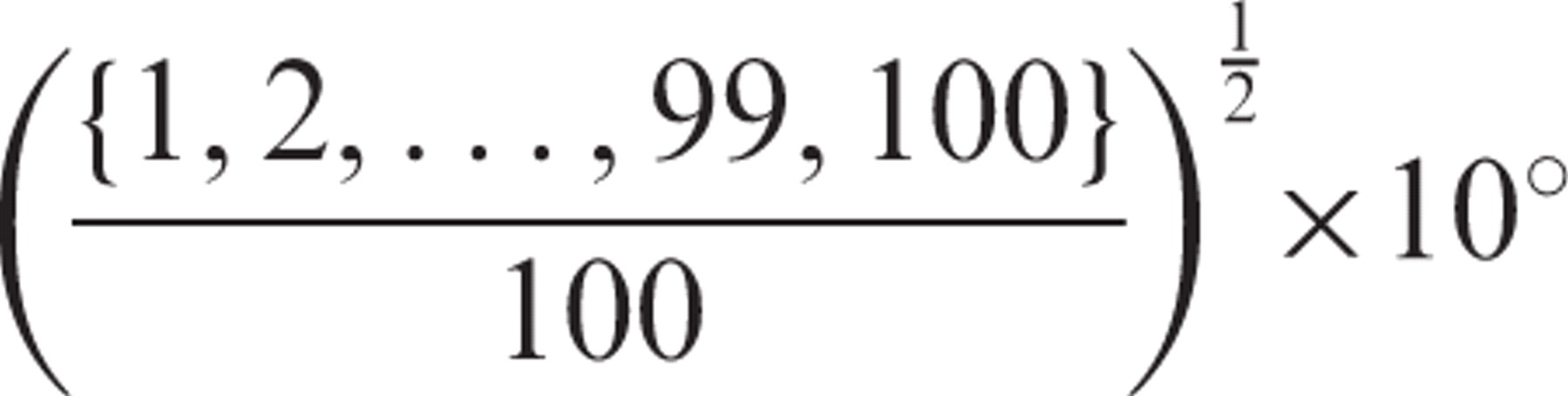

There were two experimental conditions: 100% coherence (i.e. no external noise) and added external noise. In the latter condition, orientation noise, randomly drawn from a uniform distribution with a spread of ± 64°, was added to the motion direction of the dots. The external noise was redrawn from the distribution for each dot individually and done for each stimulus presentation within an initial run. The stimuli were presented for a duration of 500 ms.
For both conditions, the task was a two interval forced choice (2IFC) procedure in which participants were required to decide in which of the two presentations the dots moved in the tightest or most closed spiral. The reference pattern always had a spiral angle of 45° (37). A MOCS procedure was used to adjust the orientation of the dot pairs relative to the local radius. The MOCS steps were centered around 45°. The participants indicated their choice using a button box. No feedback was provided. Participants completed three runs with 100% coherence and 3 runs with the added external noise.
Spiral form task
This stimulus consisted of 200 bright Gaussian dots on a gray background. The standard deviation of the 2D Gaussian defining the contrast of each dot was 4’ and the dots had a maximum luminance of 90 cd/m2. The stimuli were presented for 500 ms and were created using dot pairs, which had a center to center separation of 24’ of arc, placed within a 10° circular aperture. The dot density of the pattern was 1.27 dots per square degree of visual angle. The midpoints between the dots of a pair were arranged to be at a point of intersection of one of 100 radii equally spaced in polar angle and 100 concentric circles increasing in area linearly. The radii of the circles were given by Equation 1. The viewing distance was 65 cm.
The dot pairs were assigned at random to a subset of the intersections but each radius and circle was used only once in each pattern. This system resulted in a constant density for the dot pairs on average, but minimized the probability of overlap between dots of adjacent pairs. As shown in Figure 1, orientation variation (β(n)) was added to the dot pairs to introduce external noise to all dots. The amount of external noise added to the dot pairs was drawn from a uniform distribution with a spread of ± 40°. The noise was redrawn for each dot pair individually, for each stimulus presentation in the first run, but the identical stimuli were presented in subsequent n-pass runs. Adding external noise to the orientation of the dot pairs had the effect of making the judgment of the spiral angle in the pattern more difficult (Figure 2). The amount of added external noise was smaller than that added to the spiral motion task as pilot studies showed a smaller amount of noise was required to produce at least a doubling of performance thresholds on this task. When the external noise is low, the main source of variability is the additive internal noise and thresholds remain relatively constant; however, as external noise increases above the level of the intrinsic internal noise, multiplicative internal noise becomes the main source of variability in the threshold estimate and thresholds increase according to a power function (42–45). Therefore, by using no external noise and external noise that is large enough to have an impact on performance, differences in either additive or multiplicative noise will be visible.
(A) An example of the spiral form stimuli with 100% coherence, where the pattern on the right (test stimulus) is more tightly closed than the pattern on the left (reference stimulus). In (A) the reference has a spiral angle of 45° and the test stimulus has a spiral angle of 63°. (B) An example of stimuli with a 45° spiral angle for the reference stimulus on the left and a 66° spiral angle for the test stimulus on the right. Both stimuli in (B) have added external orientation noise drawn from a uniform distribution with a spread of ± 40°.
The task was a two interval forced choice (2IFC) procedure in which participants were required to decide in which of the two presentations the stimulus formed the tightest or most-closed spiral. The reference pattern always had a spiral angle of 45°. A MOCS procedure was used to adjust the orientation of the dot pairs relative to the local radius. The MOCS steps were centered around 45°. The participants indicated their choice using a button box. No feedback was provided. Participants completed three runs with no external noise and three runs with up to ± 40° of added external noise.
Results
A cumulative normal distribution was fitted using non-linear regression across MOCS levels for each condition in each experimental task (Prism 5.02, Graphpad Software). The sigma value obtained from the cumulative normal distribution was adopted as the threshold measure (in which an increase in sigma shows a decrease in performance) for both the spiral motion and form tasks. Threshold defined in this way represents the spiral angle for 68.2% correct performance. For the motion coherence task the mean of the cumulative normal distribution was adopted as a measure of threshold as observer performance on this task ran from 0.5 to 1. Therefore, the mean represents 75% correct. The migraine with and without aura groups were compared and no significant differences were found on any experimental task, although the migraine with aura group is small. Therefore, the two groups were collapsed into one migraine group for analysis.
As expected, for the spiral motion task and the spiral form task, thresholds increased when external noise was added to the stimulus (Figure 3) for both groups (compare horizontal pairs indicating mean and 95% confidence intervals, Figure 3a with 3b and 3c with 3d).
Threshold estimates (sigma) for individual participants in the control group (black circles) and the migraine group (open triangles) on a spiral form task, a spiral motion task with and without external noise (in degrees) and a motion coherence task (MCT). The group mean threshold is denoted by the solid black line and the error bars are the 95% confidence intervals.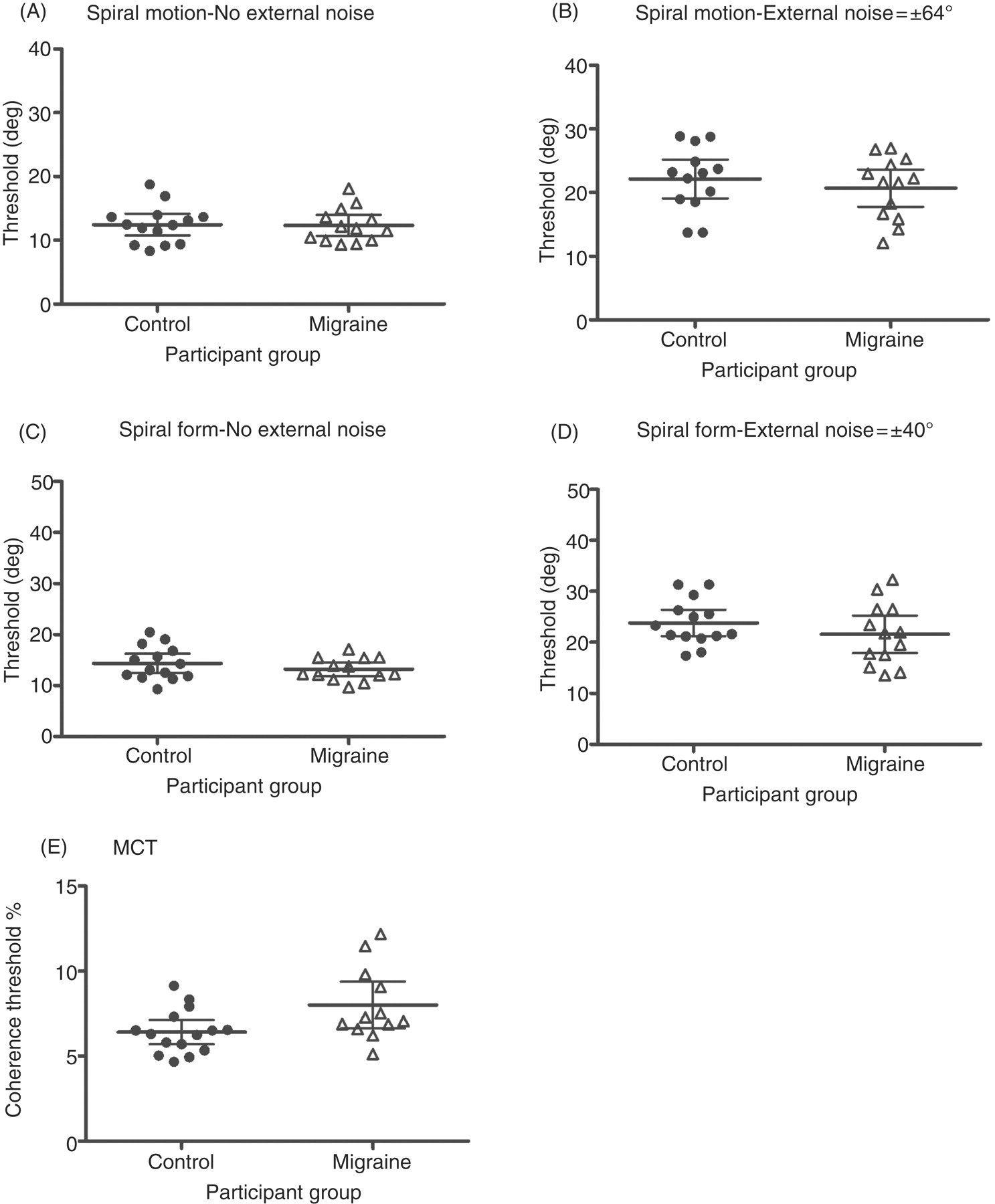
Thresholds for the MCT task were then compared across groups using a t-test to determine whether there were any performance differences. There was a significant difference in thresholds between migraineurs and headache-free controls on the motion coherence task (t(25) = 2.36, p < 0.05), where migraineurs required a greater number of coherently moving dots to correctly identify the pattern containing signal (Figure 3e).
A mixed design, two-way analysis of variance (group × noise level ANOVA) was performed separately on the threshold data from the spiral form task and the spiral motion task. The design of the ANOVA used thresholds as the dependent variable. There were two independent variables, each with two levels. The first was the within-groups factor, noise (no noise or added external noise) and the second was the between groups factor, participant group (migraine or control).
The thresholds and (95% confidence intervals) for controls and individuals with migraine for the spiral form task and the spiral motion task

Analysis of the threshold data from the spiral motion task showed a significant within-groups effect of added external noise (F(1, 25) = 90.12, p < 0.001). This indicates that performance on the task was poorer when there was additional noise added to the stimulus. There was no significant effect of group (F(1, 25) = 1.83, p > 0.05), indicating that individuals with migraine performed at the same level as controls (Table 1 and Figure 3). There was also no significant group by noise-level interaction (F(1, 25) = 2.60, p > 0.05).
Given that thresholds are a combination of efficiency and internal noise (43,44), a breakdown of the components of performance was required as thresholds could be the same between groups but with higher or lower efficiency paired with more or less internal noise to give the same outcome. Therefore, internal noise was estimated using the N-pass technique (35,36). Observer responses on each trial are correlated across two runs, pairing all runs with each other (run 1 with run 2; run 1 with run 3; and run 2 with run 3). This gave three correlation estimates that were then averaged, giving a single average estimate of the proportion of internal noise (random noise) to consistent noise. An examination of internal noise estimates showed that there was no significant difference in estimates between migraineurs and headache-free controls on the spiral motion task with or without external noise (t(25) = 0.88, p > 0.05), (t(25) = 0.88, p > 0.05), the spiral form task with or without external noise(t(25) = 0.66, p > 0.05), (t(25) = 0.68, p > 0.05) nor the motion coherence task (t(25) = 0.16, p > 0.05) (see Figure 4). Further analysis showed no significant correlation (using a criterion of p > 0.05) between thresholds and internal noise estimates for either group on any experimental condition.
Mean internal noise estimates for individual participants in the control group (black circles) and the migraine group (open triangles) on a Spiral Form Task, a Spiral Motion Task with and without external noise (in degrees) and a Motion Coherence Task (MCT). The mean internal noise estimate for groups is denoted by the solid black line and the error bars represent the 95% confidence intervals.
Recalculated internal noise estimates (and 95% confidence intervals) for controls and migraineurs on a spiral form task, a spiral motion task with and without external noise and a motion coherence task

Discussion
This study aimed (i) to determine whether increased internal noise could account for performance differences observed on a global motion coherence task used previously (13–15) and equivalent results by others (16) and (ii) to determine whether a difference in internal noise levels was a factor influencing performance when migraineurs perform visual tasks requiring global processing. To estimate internal noise the N-pass method was used (36). Two further tasks were used that required participants to discriminate, on the basis of the spiral angle, between stimuli containing spiral or angular motion or equivalently spiral global form. These were conducted with 100% signal coherence levels and also with added orientation noise to allow comparison with a previous result suggesting that migraineurs may be superior in judging direction of global motion flow when 100% coherent signals are presented (16). We compared the performance and also estimated internal noise levels across visual tasks.
Consistent with previous research (13,15,16), there was a significant difference in thresholds on the motion coherence task, with the group of migraineurs requiring more coherently moving dots to correctly identify the presence of coherent motion than controls.
However, we found no significant threshold differences between groups on the spiral motion task or the spiral form task. Thresholds increased in the presence of external local-orientation noise but this was consistent across groups. The finding that migraineurs perform comparably to controls on measures of spiral angle discrimination in the motion task contrasts with a previous study (16) that reported superior up/down motion direction processing in migraineurs when the display was 100% coherent. That study required participants to identify only whether the dots were moving up or down and used a very brief presentation time (48 ms). In contrast, we measured the ability to detect small changes in the angle of spiral global motion with 100% coherence, as a more demanding test of motion flow discrimination. Therefore, using a finer motion flow discrimination task and longer presentation time (500 ms) than that previously used with 100% coherence, our results suggest that migraineurs do not display superior global motion processing than controls.
A comparison of the thresholds from the spiral motion task and the motion coherence task suggest that performance differences between groups are not generalized across motion processing but are specific to certain aspects of motion processing. Migraineurs have intact angular motion processing as measured by the spiral motion task once the signal is specified, but a deficit in the motion coherence threshold task. The results also indicate that angular processing is intact in the form pathway of individuals with migraine as measured by the spiral form task. In parallel to these findings a previous report suggests that migraineurs also have higher spiral form coherence thresholds (17).
Therefore, the results suggest that migraineurs’ ability to integrate local motion signals into a coherent global motion signal is poorer than that of controls. However, when the signal level is high, migraineurs may adequately integrate the local motion signals to make comparable motion flow judgments to controls. This suggestion is supported by previous research (46), which examined optic flow. That study found higher motion coherence thresholds for the migraine participants but comparable suprathreshold heading performance when there was a strong motion coherence signal.
Examination of the internal noise estimates obtained from the N-pass method showed that there was no significant difference between groups on any of the tasks used. Previous analysis indicates that the study had sufficient power to find a medium sized effect (0.5) (38). Therefore, the finding that internal noise estimates do not differ between migraine and control participants indicates that increased internal noise is not the mechanism underlying performance differences. Instead, the motion coherence sensitivity loss may be the result of structural cortical differences. There is recent evidence from a study using diffusion tensor imaging that indicated anatomical differences in the visual cortex of migraineurs, including increased cortical thickness in visual area MT, also known as V5, and V3a, which are involved in motion processing (12). Evidence that structural variation in the size of a cortical area can influence perceptual judgments has been presented (47) for cortical area V1. In our experiments the cortical changes would need to lead to less efficient extraction of global motion signals from noise, which could lead to threshold increases without requiring increased internal noise. The impact of such structural differences cannot be resolved by the current study.
A recent study by Wagner et al. (48) found evidence for decreased efficiency for migraine without aura participants and evidence for increased multiplicative internal noise in people with migraine with aura underlying the observed performance deficits. They used a contrast detection task with luminance noise added to degrade the display. In order to estimate internal noise, the perceptual template model (45) was used. This model partitions internal noise into additive (independent of the stimulus) and multiplicative (scaled by the magnitude of the stimulus) noise and also provides information about how efficiently the observer is using the available signal. Participants in the study were required to detect a luminance target in the presence of luminance noise. The results indicated that the migraine group had higher thresholds than controls when the external noise was present, but that thresholds were no different when there was no external noise present. The migraine with aura participants had strong deficits in external noise exclusion, whereas the migraine without aura group had only a minor deficit in external noise exclusion, as evidenced by higher thresholds for the migraine with aura participants.
It was argued (48) that the results suggest that migraineurs, particularly the aura group, experience higher variability of neuronal activity than headache-free controls and that the perceptual template model indicated that people who experience migraines have an increase in the internal noise induced by the added external noise, that is, multiplicative noise. Based on this assertion, if there is a generalized increase in internal noise it would be expected that group differences in performance might be present in the current study with high external noise levels. It is important to note that this suggestion does not require the internal noise estimates to be comparable across cortical areas, nor are we trying to suggest they are, but it requires the estimates for the control and migraine groups to differ. We found no evidence of increased internal noise, additive or multiplicative, for either of the spiral discrimination tasks, despite significant differences in motion coherence thresholds for the migraine population. Although we had a small number of migraine with aura participants and it was that group which showed a difference in the Wagner et al., study (48).
An important point of difference between our study and the previous study by Wagner et al. (48) is that they added external luminance noise over the top of the display, degrading the visibility of the stimulus, whereas the external noise added in each experiment in the current study was applied to the critical stimulus dimension (orientation) but did not vary the visibility of the elements of the stimuli. Therefore, it may be the case that migraineurs do not have a generalized increase in internal noise but rather have a specific deficit only when the stimulus visibility is degraded. To determine whether there is a conflict between our results and the previous study (48), it would be desirable to conduct tasks using several noise types, including their task, within a common group of participants. This research is underway.
In conclusion, the results of this study suggest that migraineurs have an abnormality in the intermediate stage of motion processing, but that it is not a generalized motion processing abnormality, as shown by comparable performance to controls when making angular judgments on the spiral motion task. Furthermore, the results suggest that a generalized increase in internal noise is not the mechanism underlying performance differences on a motion coherence task. The motion coherence sensitivity loss may be the result of structural cortical differences, but if so they must result in decreased efficiency. Given previous findings (48) that found increased internal noise associated with processing of luminance, further investigation into the effect of different types of external noise on the visual performance of migraineurs is warranted. This would allow a full characterization of visual processing and its dependence on the level of internal noise in migraineurs, but it is clear that there is no uniform increase in internal noise.
Footnotes
Funding
This work was supported by NHMRC Project [grant #509208 to AMM and DRB].
