Abstract
Migraine groups have impaired ability to identify global motion direction in noisy random dot stimuli, an observation that has been used as evidence for cortical hyperexcitability. Several studies have also suggested abnormalities in cognitive processing, particularly in the domains of attention, visuo-spatial processing and memory. This study aimed to determine whether poor performance by migraineurs in motion coherence tasks could be explained by non-visual cognitive factors such as attention. Twenty-nine migraineurs and 27 non-headache controls participated. Global motion coherence thresholds were measured along with measures of neuropsychological function, using the Repeatable Battery for the Assessment of Neuropsychological Status (RBANS). The migraine group had significantly higher motion coherence thresholds than controls. No significant difference in attention or any other RBANS index score was found between groups. Index scores did not correlate with motion perception thresholds. This study does not support inattention or other cognitive abnormality as an explanation for motion perception anomalies in migraine.
Introduction
A number of studies have identified differences in cortical visual processing in people with migraine when compared with non-headache controls (1–6). Visual processing has been targeted because the visual symptoms that are commonly associated with migraine, either in the form of aura or other more subtle symptoms such as blur, indicate that the visual pathways are involved in migrainous pathophysiology. One visual task that has repeatedly shown impairments is the ability to identify global motion direction from the integration of local motion cues embedded in noise (1–4, 7). The stimulus for this task is comprised of random dots, where a proportion of the dots move in a common (signal) direction while the remainder move in random directions. In normal observers, the ability to integrate global motion yields a signal:noise ratio that is constant at threshold in low density patterns (8). It has been proposed that cortical neurons are hyperexcitable in migraine, either due to hyperexcitability itself or secondary to reduced cortical inhibition (5, 7, 9–12). Neuronal hyperexcitability is predicted to increase the level of neural noise and to result in impairments in integrating local cues into a global motion direction when embedded in noise.
Cognitive function has been studied in migraineurs with variable results (for review see (13)). The most consistent findings point to deficits of attention, visuo-spatial processing and memory in migraineurs (14, 15). Other studies have found no differences in cognitive function between migraineurs and controls (16–19). It has been suggested that the results vary due to differences in subject selection criteria, the potential influence of migraine medications on performance and differences in test procedures (13, 14).
If cognitive abnormalities exist in migraineurs, it is possible that the visual deficits reported may arise from non-visual factors such as deficits in attention. The purpose of the current study was to compare global motion perception to neuropsychological measures in migraineurs when measured between migraines. We chose a cohort of young, otherwise healthy migraineurs with relatively mild migraine symptomatology, to match the characteristics of groups where we have identified motion perception deficits previously. Neuropsychological measures may be more likely to be impaired in individuals with severe migraine symptomatology such as those selected from specialist clinics (13, 14). We wished to avoid possible confounds of medications (13, 20, 21) or other systemic or psychiatric comorbidities that are likely in a more severe migraine group (22).
Materials and methods
Participants
Twenty-nine people with migraine (11 male) and 27 non-headache controls (nine male) participated. Subjects were recruited via written advertisement sent to staff and students of the University of Western Australia. The median age of both groups was 26 years (range 19–47 for controls, 19–43 for migraineurs). All migraine participants were required to meet the International Headache Society (23) classification criteria for either migraine with aura (MA, n = 16) or migraine without aura (MoA, n = 13). A measure of headache severity was obtained via the Migraine Disability Assessment (MIDAS) questionnaire (24). Patients report the number of days that headaches have prevented daily activities and rate pain severity on a scale of 0–10, where 0 is no pain and 10 ‘pain as bad as it can be’. The MIDAS scores and classifications of the migraineurs are shown in Table 1. The average pain rating was 6 and ranged from 3 to 9. Subjects also reported via an alternate questionnaire an estimate of the number of migraines that they had experienced in the past year. The median was eight, with a range of 1–40 migraine events. Age at first migraine event was also retrospectively reported. The mean duration of migraine experience was 13 years and ranged from 1 year to 32 years. Non-headache controls were not permitted to experience any form of headache more often than once every 3 months and had never experienced migraine.
Migraine Disability Assessment (MIDAS) questionnaire scores for the participants with migraine

Subjects were also required to meet the following visual and ocular inclusion criteria: best corrected visual acuity of 6/7.5 or better, refractive errors less than ±5.00 diopters sphere and ±2.00 diopters astigmatism, normal anterior eye and ophthalmoscopic examination, no history of systemic disease known to affect ocular or cognitive function with the exception of migraine and no current medications known to affect visual field sensitivity or contrast sensitivity.
Migraineurs were tested at least 4 days (range 5 days to 6 months) after the cessation of a migraine to avoid possible transient effects on performance due to medication or fatigue. Migraineurs were not permitted to be taking ongoing preventative migraine pharmacotherapy.
All subjects reported English as their first language (spoken at home since infancy) and all had completed at least 13 years of education. Subjects provided written informed consent in accordance with a protocol approved by the University of Western Australia Human Research Ethics Committee and in agreement with the tenets of the Declaration of Helsinki.
Global motion perception
The global motion stimulus was the same as we have described in detail elsewhere (1, 3). The task was performed using custom software written in Matlab 6.0 (Mathworks, Natick, MA, USA) and displayed using a Cambridge Research Systems VSG 2/5 graphics card (Cambridge Research Systems, Rochester, UK) on a gamma corrected Philips (Model GD-402) 21-in grey-scale monitor (100 Hz refresh rate). Subjects viewed the monitor monocularly with their preferred eye from 75 cm, with appropriate refractive correction, using a chinrest.
The global motion stimulus consisted of 100 moving random dots displayed within a 10° circular aperture. The dots were white (75 cd/m2), 8.6 min arc in diameter and moved at a velocity of 2.86°/s over a black background (0.5 cd/m2). A percentage of dots moved downwards (signal) while the remaining dots (noise) moved in random directions, excluding ±10° of the signal direction (downwards). An eight-frame movie sequence was displayed to create the motion percept. Each frame was shown for 50 ms with no interframe interval. Different dots were chosen to be signal dots on each frame. This aspect of the stimulus design minimizes the utility of tracking individual dots (a local motion cue) to establish the percept of coherent global motion (8). Instead, the motion information must be globally integrated to perceive the coherent motion signal. Global integration of local motion cues is a property of cortical area V5 (25).
Global dot motion stimuli were presented in a two-interval forced choice design. After an auditory tone, a 400-ms stimulus was presented. A 500-ms interstimulus interval followed, then a second tone, then another 400-ms stimulus. One of the stimuli contained a coherent motion signal and the other only random motion. Subjects chose which of the presentations contained coherent motion. The signal level commenced at 50 dots (of 100 dots). The number of signal dots was varied according to a staircase strategy. Three consecutive correct responses resulted in a decrease in signal, whereas every incorrect response resulted in an increase in signal dots. The three-down, one-up design results in the staircase converging on the approximate 79% correct performance level (26). Initially the staircase stepsize was eight dots, which was halved on the first three reversals, resulting in a final stepsize of one dot. The staircase terminated after seven reversals and the mean of the last four reversals was calculated. Two staircases were interleaved and the mean of the two staircase results was taken as the threshold for each subject.
Cognitive assessment: Repeatable Battery for the Assessment of Neuropsychological Status
The Repeatable Battery for the Assessment of Neuropsychological Status (RBANS) is a relatively brief (approximately 30 min), standarized cognitive assessment battery which was developed to characterize cognitive decline in older adults and as a neuropsychological screening battery for younger adults (27, 28). In addition to providing a global assessment of cognitive function (Total score), the RBANS yields separate index scores across five relatively independent cognitive domains (Language, Attention, Immediate memory, Delayed memory and Visuo-spatial construction), thus allowing us to test whether cognitive deficits in migraineurs are specific to visual motion performance or also involve impairments in attention, memory and visuo-spatial functions as suggested by previous studies (14, 15). Previous work reports average reliability coefficents (split-half) of RBANS indices in the 0.80 range, whilst the average coefficient (across all age groups tested) for the RBANS Total score was 0.94, indicating adequate test reliability (28). Similarly, examination of the pattern of intercorrelations of RBANS Index scores showed a modest correlation (as expected) between immediate and delayed memory Index scores whilst remaining correlations were low to moderate (0.2–0.4), supporting the proposal that these scores are measuring distinct cognitive constructs (28).
Within our study, The RBANS was administered and scored by one of the authors (M.G.) for all subjects. RBANS assessment was conducted prior to the measure of motion perception.
Results
Consistent with previous studies (1, 3) we found no significant difference in global motion thresholds between the MA and MoA groups, so data for migraineurs were pooled for further analysis (t(27) = −0.835, P = 0.411). Similarly, no significant difference between the MA and MoA groups was identified for any of the index scores from the RBANS assessment (F(1,5) = 0.145, P = 0.70).
Figure 1 shows group means (±SD) for the migraine and control groups for the global motion task (Fig. 1A) as well as all individual subject data. Index scores from the RBANS assessment (Fig. 1B–G) are plotted in identical format in Fig. 2. For both tasks, the distribution of the group data did not differ significantly from a normal distribution (Kolmogorov–Smirnov test, P > 0.05). Mean migraine motion coherence thresholds were significantly higher than those of controls (t(54) = 2.1, P = 0.04), consistent with previous reports (1–4, 7). No significant difference was found between migraineurs and controls for the cognitive assessment (
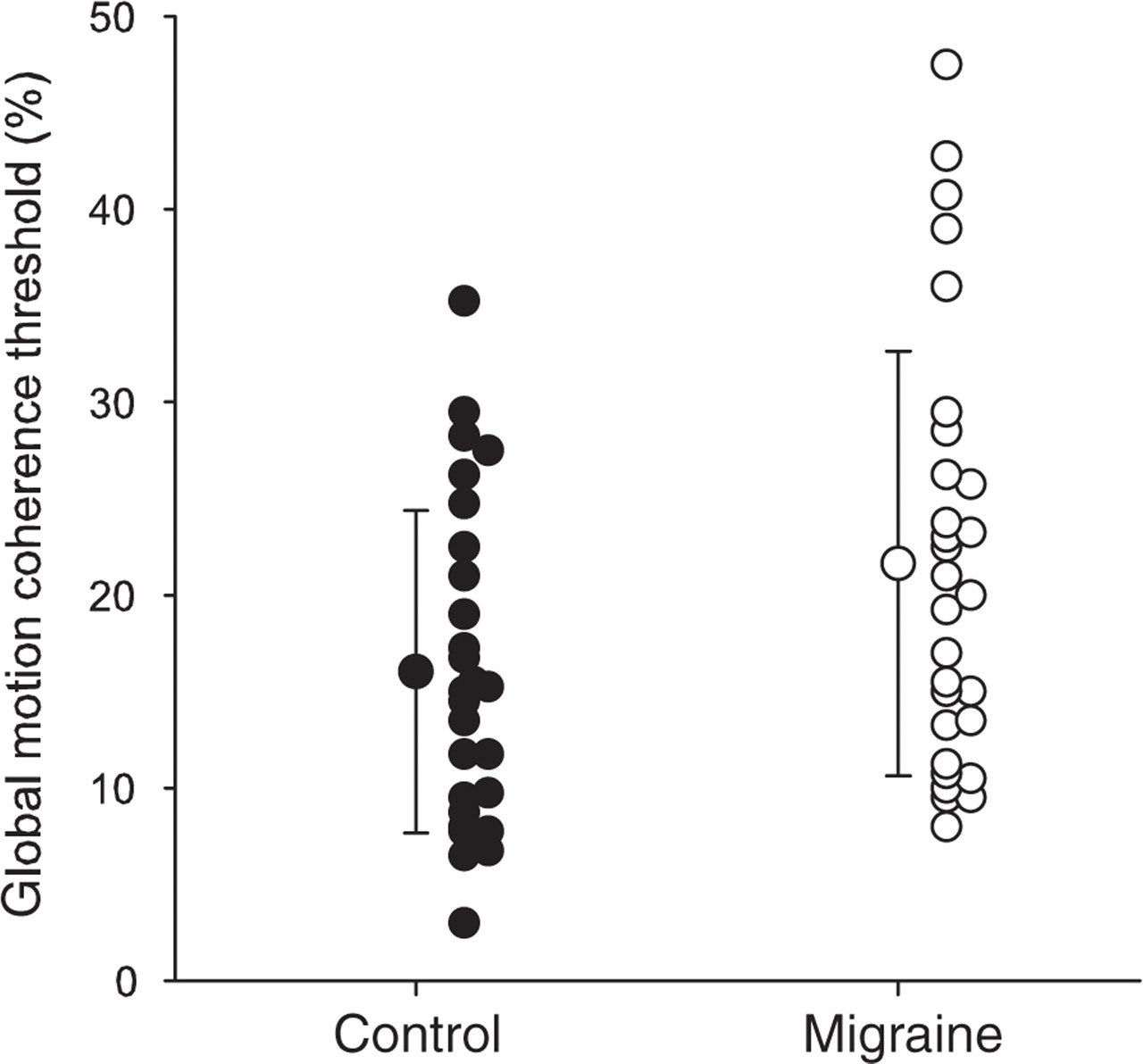
Global motion coherence thresholds for the control (•) and migraine (○) groups. Means and standard deviations are shown along with all individual data.

Comparison between control (•) and migraine (○) groups for the Repeatable Battery for the Assessment of Neuropsychological Status (RBANS) index scores. Means and standard deviations are shown along with all individual data. Higher RBANS index scores represent better performance.
Pearson product moment correlation coefficients relating each of the Repeatable Battery for the Assessment of Neuropsychological Status (RBANS) index scores to the performance on the global motion coherence task

Discussion
This study confirms that group differences in motion perception exist between people with migraine and non-headache controls at times between migraine events (1–4, 7). It also confirms previous reports of no difference in cognitive processing in migraineurs (16–19). Our findings suggest that the global motion deficits are likely to be perceptual in origin rather than an epiphenomenon of reduced attention or other cognitive abnormality.
It has been suggested that neuropsychological measures are more likely to be impaired in individuals with severe migraine symptomatology and that cognitive changes might result as a longer term effect of cumulative damage caused secondary to the migraine process (13, 14). If global motion coherence deficits are due to neuronal hypersensitivity (1, 7), which is presumably an innate part of the migraine condition, whereas cognitive differences arise cumulatively, it is not surprising that we did not find differences in our relatively young and mild migraine cohort as these two phenomena may be expected to have different causes.
The motion coherence task used in this study requires global integration of local motion signals to extract the coherent motion direction. Current understanding of the human visual system indicates that such global integration is likely to occur in extrastriate cortical area V5 (29–31). There is also recent evidence for additional processing of global motion in area V3A (32). Perceptual global integration deficits in migraineurs are not selective for motion processing, as we have recently also demonstrated deficits in the ability to integrate global structure from local form cues presented as Glass patterns (1). While the processing of motion and form information does not occur entirely independently, current understanding indicates that the predominant processing of form information is separate from that of motion and that global form extraction is likely to require neural processing at extrastriate area V4 (33). Evidence of involvement of extrastriate areas in migraine is also available from recent studies using transcranial magnetic stimulation (TMS) (34, 35). Other perceptual deficits have been identified in migraineurs, including enhanced sensitivity to high-contrast masking patterns (36, 37) and differences in pattern adaptation (38). Taken together, these results argue for a generalized cortical neural processing deficit in migraineurs which involves both primary and extrastriate visual cortical areas.
An important, but to date unanswered question, is whether the identification of motion perception deficits in migraine has significance in terms of either indicating migraine susceptibility or predicting functional visual performance in a day-to-day setting. As motion coherence thresholds are presumably elevated due to anomalies in cortical neural processing, it is possible that these measures may provide information regarding susceptibility to migraine or alternatively provide a metric for assessing efficacy of treatment. To date, studies measuring global motion perception in migraineurs have used relatively small sample sizes (<40). Substantial overlap in performance exists between migraineurs and controls. No relationship has been identified between motion coherence performance and migraine characteristics such as headache frequency or years of migraine experience. These results should be viewed with caution, as much larger samples are probably necessary to answer these questions conclusively. It has been established that motion coherence deficits are not exclusive to those migraineurs who experience visual aura (1, 3, 7). Regarding the issue of functional visual significance, the ability to process motion information is important for day-to-day tasks such as driving. We have recently demonstrated that migraineurs can identify the direction of heading from simulated optic flow with similar precision to controls (39). Further research is required to determine whether the motion coherence deficits experienced by some migraineurs result in perceptual difficulties in a real-world setting, such as when driving.
This study confirms the results of previous works that have found that visual motion perception is impaired in people with migraine when stimuli are presented embedded in noise. We found no evidence for significant cognitive abnormalities in young, otherwise healthy people with migraine at times between their attacks. Nor do our data support inattention as an explanation for motion perception abnormalities in migraine.
Acknowledgements
NHMRC Australian Clinical Research Fellowship no. 139150 (A.M.M.).
