Abstract
Objective: To evaluate the efficacy of Coenzyme Q10 (CoQ10) supplementation in the prevention of migraine in children using a placebo-controlled, double-blinded, crossover, add-on trial. Background: CoQ10 has been demonstrated to have efficacy in migraine prevention in adults but lacks pediatric research with more rigorous methodology. CoQ10 has been observed to be deficient in a significant number of children and adolescents presenting to tertiary headache centers. CoQ10 has the potential to modify both the inflammatory changes that occur during recurrent migraine and the alteration of mitochondrial function. A deficit of CoQ10 could thus affect the response to treatment and clinical characteristics of migraine in children and adults. Methods: One-hundred-and-twenty children and adolescents with migraine headache were randomized in a crossover, double-blind, placebo-controlled, randomized, add-on study to receive a placebo or CoQ10 (100 mg) supplement for 224 days. Data for 76 patients were available at the crossover point and 50 were analysed at the endpoint. Response to treatment, overall headache improvement, and headache disability were assessed. Results: Both the placebo and CoQ10 groups showed reduced migraine frequency [F(1, 60) = 15.68, p < 0.001], severity [F(1, 54) = 8.09, p = 0.006], and duration [F(1, 45) = 6.27, p = 0.016] over time. CoQ10 treated patients had a significantly greater improvement in frequency from subject reported baseline starting within 4 weeks of initiation. No group differences comparing the first 4 weeks of treatment with the last 4 weeks of treatment were found in migraine frequency [F(1, 60) = 2.34, p > 0.05], severity [F(1, 54) = 0.06, p > 0.05], or duration [F(1, 45) = 0.14, p > 0.05]. Conclusions: Overall, results of the study demonstrate that children and adolescents with migraine improved over time with multidisciplinary, standardized treatment regardless of supplementation with CoQ10 or placebo. There was no difference in headache outcomes between the CoQ10 and placebo groups at day 224. Due to the improvements seen in weeks 1–4, CoQ10 may lead to earlier improvement in headache severity, but given the sample size this conclusion warrants further investigation with a larger sample.
Migraine headache is a debilitating illness that affects up to 10.6% of children 5–15 years old (1), with 75% of children reporting recurrent headaches by the age of 15 (2). The prevalence of migraine increases through the teen years, with approximately 28% of girls and 15% of boys reporting migraine headache (3). Migraines, however, are under-reported, under-diagnosed and under-treated at great cost to society (estimated as high as 17.2 billion US dollars per year due to lost productivity) (3). Disability due to migraine and impact of quality of life can be significant in children (4).
There are several FDA-approved preventative headache therapies for adults. The effectiveness of these therapies in children has been initiated, but has not been fully evaluated in children and adolescents, with no medications currently approved for the prevention of migraine in children. Nutraceuticals have been suggested as a potentially effective preventative treatment for migraine headache in adults. Nutraceuticals such as coenzyme Q10 (CoQ10), magnesium, and riboflavin have been suggested to either have a direct impact on headache occurrence or indirectly improve headaches by correcting a deficiency that allows other preventative agents to be more effective (5–12).
Two small studies of adults with migraine have demonstrated the effectiveness of CoQ10 (5–6). One study demonstrated the effectiveness of 150 mg in an open-label study in 32 adults with episodic headaches (5). The other study examined the effectiveness of 100 mg three times a day dosing in 42 adults with episodic headache compared with placebo (6). Both studies demonstrated that CoQ10 was well tolerated.
Additional research is needed to further evaluate the efficacy of CoQ10 as a treatment in pediatric migraine prevention with a more rigorous methodology. The current study was a placebo-controlled, double-blinded, crossover trial evaluating the efficacy of Q10 supplementation in the prevention of migraine in children.
Methods
Subjects
One-hundred-and-twenty subjects were recruited for a placebo-controlled, double-blinded, crossover, add-on study of 100 mg of CoQ10 versus placebo. One-hundred-and-twenty subjects were recruited based on past research and an expected 20% dropout rate. A dose of 100 mg was based on a limited number of reports at study initiation in pediatric cardiac disease that demonstrated the safety of 1–3 mg/kg/day. Subjects were 6 to 17 years old, inclusive, who met the International Classification of Headache Disorder-II (ICHD-II) for migraine with or without aura either episodic or chronic, who were recommended to start prophylactic therapy (>3 headaches per month or PedMIDAS ≥ 20). Exclusion criteria included subjects who previously failed CoQ10 therapy for migraine prophylaxis, those who previously discontinued CoQ10 due to adverse events, those who are taking CoQ10 or had taken CoQ10 within 14 days prior to enrolment, and subjects with continuous headaches. Children in the study continued to receive the full multidisciplinary therapy that included acute therapy, prophylactic therapy, and biobehavioral therapy.
In the first stage of the study, subjects were equally randomized to be treated with either CoQ10 or placebo. After 16 weeks subjects were crossed over to the alternate therapy. The study design is shown in Figure 1.
Study recruitment.
The study was reviewed and approved by the Cincinnati Children’s Institutional Review Board.
Experimental design
Randomization was used to avoid bias in the assignment of subjects to treatment, to increase the likelihood that known and unknown subject attributes (e.g. demographics and baseline characteristics) were evenly balanced across treatment groups, and to enhance the validity of statistical comparisons across treatment groups. Subjects were randomized according to a 1 : 1 ratio (CoQ10:placebo). Randomization was provided by the hospital’s investigational pharmacy, who distributed the CoQ10 or placebo. The investigator entered the qualified subject’s identification in numerical order, thereby assigning the subject to one of the two treatment groups. The number assigned to a subject was retained by the subject for the duration of the study. The randomization code was not revealed to study subjects, investigators, or clinical staff until all subjects had completed therapy.
All subjects completing the study received both CoQ10 and placebo. Changes in frequency from reported baseline were analysed in 4 week blocks, while other outcome measures were obtained at routine visits approximately every 8 weeks.
The pre-randomization/screening
Subjects needed an average of more than 3 migraine-days per month during the 3 months prior to screening to be included in the study. Subjects could have daily headaches, but not continuous headaches. Initial headache frequency was based on the baseline report of the average number of headaches per month for the previous 3 months. The study was explained and informed consent was obtained from the subject’s parent or legal guardian. Subjects capable of giving assent also signed the informed consent. Preventative and acute headache medications were permitted for headache prevention and symptomatic relief of migraine and non-migraine headache episodes.
Double-blind phase
Subjects who met the enrolment criteria were randomized in a double-blind fashion, according to a 1 : 1 ratio, to one of two treatment groups (CoQ10 or placebo). In this crossover design, the double-blind phase had two periods, with the patients assigned to one of the two arms at initiation; subjects received either CoQ10 first or placebo first. All subjects receiving CoQ10 were given the 100 mg chewable tablets, and all subjects receiving placebo were given a matching placebo. At day 112 (visit number 3) subjects were crossed over to the alternate treatment arm. No washout period was used in this study. A minimum body weight of 35 kg was required for study participation.
Headache characteristics
Baseline headache characteristics for subjects including headache frequency per month, average severity, average duration, and associated symptoms were obtained prior to randomization. These features were also collected at each study visit. Baseline monthly frequency for the month prior to initiation and crossover were used for response changes. Exact headache frequency during the treatment phase was determined using headache calendars that were reviewed at each study visit. The prospective headache calendar included items assessing 1) headache frequency – indicating whether or not they had a headache each day; 2) headache intensity on a 0–10 numeric rating scale; and 3) headache duration in hours. Numeric pain rating scales and headache diaries are commonly used in treatment studies (16).
CoEnzyme Q10 levels
Serum levels of CoQ10 were obtained at study initiation, crossover, and study completion. This included total CoQ10, reduced CoQ10, percent reduced CoQ10, percent oxidized CoQ10, and CoQ10 corrected for cholesterol levels. CoQ10 was measured by high performance liquid chromatography (HPLC) analysis. Five separate values are reported: total CoQ10 in µg/ml (ref. range = 0.5 to 1.5 µg/ml), CoQ10 in the reduced state (ref. range = 0.48 to 1.45 µg/ml, CoQ10 in the oxidized state (ref. range = 0.02 to 0.05 µg/ml), the percentage in the reduced state (ref. range = 94% to 98%), and a CoQ10/cholesterol ratio (ref. range = 0.30 to 0.80). At the end of the study (day 224), CoQ10 levels were examined. If the final CoQ10 levels revealed a low total CoQ10, or low Q10 corrected for cholesterol, a recommendation for the child to start CoQ10 was made to the parent.
Disability assessment
Disability was assessed at each study visit using the Pediatric Migraine Disability Assessment (PedMIDAS) (4). The PedMIDAS is a measure that evaluates the impact of headaches on school, home, play, and social activities in the past 90 days. Six developmentally appropriate questions constitute the PedMIDAS and they are designed to be answered by the adolescents in consultation with their parents. The PedMIDAS uses a total score grading scale of none (0–10), mild (11–30), moderate (31–50), and severe (>50) to determine clinical significance.
Data analysis and efficacy
The intent-to-treat population for efficacy analyses included all randomized subjects who received at least one dose of CoQ10 and had provided at least one efficacy evaluation.
Efficacy analysis included both a comparison of the reduction in the number of headache days per month at baseline for the 3 months prior to study initiation to the last 4 weeks of treatment and a comparison of the reduction in the number of headache days per month (28 days) during the first 4 weeks for each treatment group compared with the last 4 weeks in each crossover analysis.
Additional secondary efficacy analyses included reduction in the monthly migraine frequency during the treatment period relative to first 4 weeks, the reduction in the number of non-migraine headache episodes, reduction in number of migraine-days and total headache-days (migraine plus non-migraine) during the entire double-blind phase, and the reduction in headache frequency and headache frequency from reported monthly baseline. Migraine frequency was obtained from the patient’s report on the calendar of the headache meeting all of the criteria for migraine. Additional analyses were conducted on the percentage of responders (subjects with a 50% or more reduction in mean monthly number of migraine-days, total headache-days, migraine episodes, and non-migraine headache episodes); reduction in the severity of migraines (using the 0–10 numerical scale); reduction in the duration of migraines; reduction in associated migraine symptoms (e.g. photophobia, phonophobia, nausea, vomiting, and abdominal pain); the reduction in disability using PedMIDAS; and the change in the quality of life using PedsQL 4.0.
The difference in the monthly rate of migraine-days was analysed using an analysis of covariance. The percentage of responders were analysed using a logistic regression. In both cases, baseline rate was treated as a covariate and treatment and center as qualitative independent factors.
Other secondary outcomes (i.e. reduction in the associated symptoms and quality of life assessments) were analysed using an analysis of covariance.
In addition, comparisons of headache frequency were made using one-sided t-tests between patient reported baseline prior to randomization and each 4 week time period (weeks 1–4, weeks 4–8, weeks 9–12, and weeks 13–16) and for reported baseline at crossover and each subsequent 4 week time period (weeks 17–20, weeks, 21–24, weeks 25–28, and weeks 29–32).
Fifty subjects were initially randomized to each group with an expectation of a 20% dropout rate and power calculated as 80%. Recruitment was increased to 60 in each group to account for a higher than expected dropout rate and to have enough statistical power to detect meaningful differences. However, higher than expected participant drop-out limited statistical analyses and generalization of findings.
Results
Demographics
Patient demographics and headache characteristics
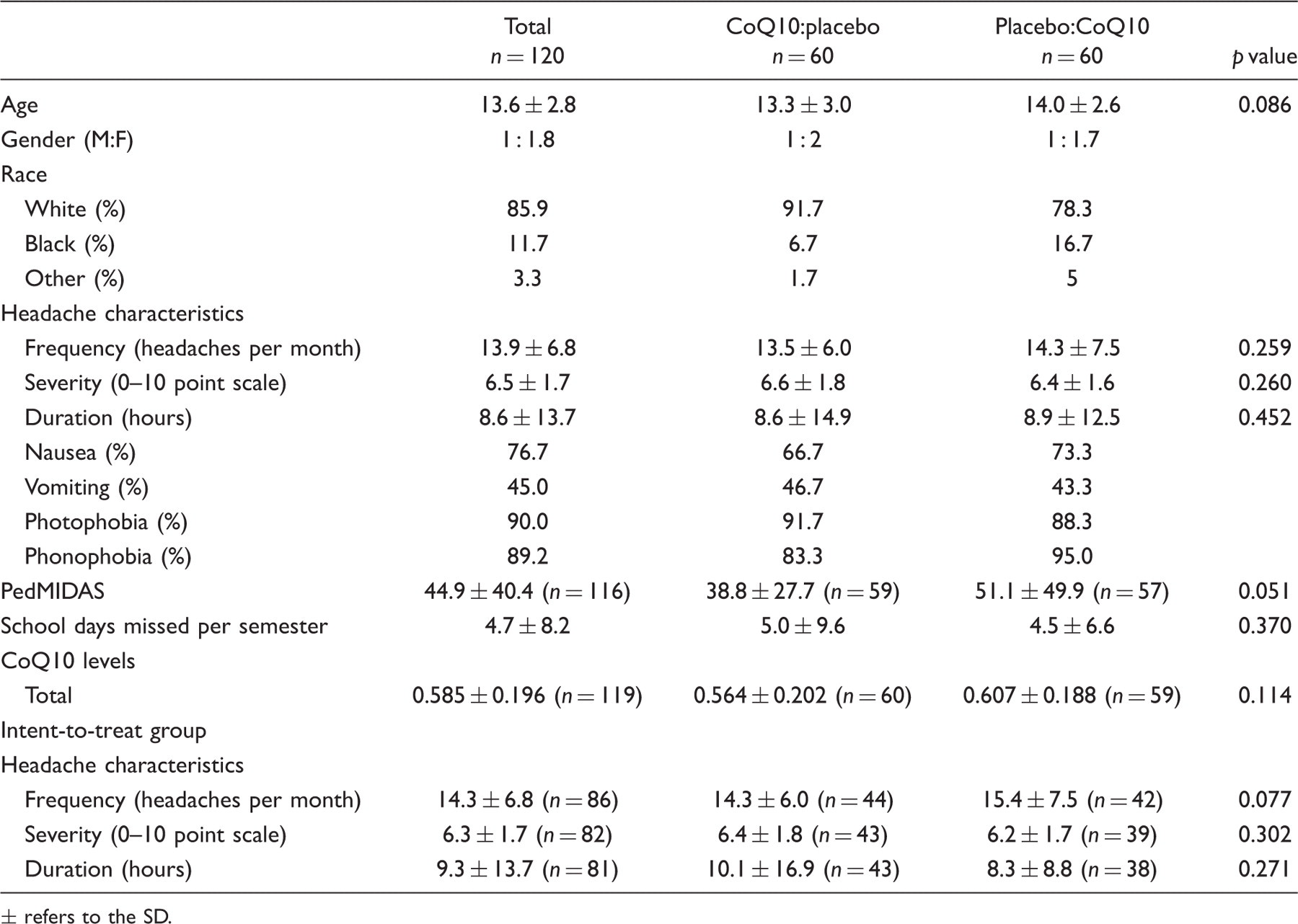
± refers to the SD.
During enrolment, 197 of the 317 potential patients were not eligible for participation. The most common reasons for ineligibility were: weight below 35 kg, reporting continuous headaches, taking preventative medications, or chose not to participate in the study. Forty-eight study participants dropped out at crossover. The most common reasons for drop out at crossover were because participants were lost to follow up or did not return to clinic in a timely manner. An additional 22 participants dropped out at the end point of the study because those participants did not return to the clinic at the established time. Final sample sizes used in analyses based on completion of week 16 were: for analyses with full data – completion of crossover: headache frequency and migraine frequency, n = 62 (34 CoQ10; 28 placebo), severity, n = 56 (30 CoQ10, 26 placebo), duration n = 47 (27 CoQ10, 20 placebo). For intent-to-treat analyses: headache frequency, n = 85 (44 CoQ10; 41 placebo), migraine frequency n = 84 (44 CoQ10, 41 placebo), severity n = 83 (43 CoQ10, 40 placebo), duration n = 76 (39 CoQ10, 37 placebo). The flow of participants through the study is shown in Figure 1.
Initial headache characteristics
Initial reported baseline headache frequency for the 120 subjects was 13.9 ± 6.8 The initial severity for the entire study population was 6.5 ± 1.7 and the initial duration was 8.6 ± 13. Initial PedMIDAS was 44.9 ± 40.4. See Table 1 for additional headache characteristics.
For the intent-to-treat group (those patients who initiated the study with at least one return study visit with calendar data), the initial reported baseline headache frequency for the 86 subjects was 14.3 ± 6.8. The initial severity for the intent-to-treat group was 6.3 ± 1.72 (n = 82) and the initial duration was 9.3 ± 13.7 (n = 81). Initial PedMIDAS was 46.2 ± 42.9 (n = 83).
For the subjects for whom full analysis was available (calendar data for weeks 1–4 and weeks 13–16), the initial reported baseline headache frequency for the 63 subjects was 14.6 ± 6.8 (n = 63) (CoQ10 arm = 13.3 ± 6.4 (n = 33), placebo arm = 16.0 ± 7.0 (n = 30), p = 0.055). The initial severity for the subjects with full data was 6.2 ± 1.8 (n = 59) (CoQ10 = 6.5 ± 1.9 (n = 32), placebo = 5.9 ± 1.8 (n = 27), p = 0.107) and the initial duration was 7.2 ± 9.5 (n = 59) (CoQ10 = 7.2 ± 10.2 (n = 33), placebo = 7.2 ± 8.7 (n = 26), p = 0.5). Initial PedMIDAS was 41.7 ± 33.7 (n = 60) (CoQ10 = 35.6 ± 26.2 (n = 32), placebo = 48.7 ± 40.0 (n = 28), p = 0.072).
Outcome analysis
Headache frequency
Headache frequency was significantly reduced (p = 0.03) for all subjects (CoQ10 and placebo) from baseline to weeks 1–4 (chronic migraine: Figure 2; episodic migraine: Figure 3; Table 2). At week 1–4, subjects receiving CoQ10 had significantly fewer headaches compared with placebo in the total study (p = 0.03) as well as the chronic migraine group (p = 0.04). However, when the change from initial baseline was determined, no significant differences were seen at the first time points for those with chronic migraine because of a statistical difference between the CoQ10 and placebo groups at baseline. For the episodic migraine, there was no difference during the first stage, with both CoQ10 and placebo improving similarly, but after the crossover, those subjects crossing from placebo to CoQ10 continued to improve, while those subjects crossing from CoQ10 to placebo worsened. The difference between groups at weeks 17–20 was statistically significant (p = 0.01). For the chronic migraine, this reversal did not occur, but the difference between the CoQ10 and placebo seen in the first stage was eliminated.
Headache frequency for participants with chronic headache. Headache frequency for participants with episodic headache. Headache frequency response for episodic and chronic migraine Significant p values. Q/P, Q10/P = CoQ10 and placebo arms of the trial. ± refers to the SD.


The repeated measures ANOVA with headache frequency as the outcome variable produced a significant main effect for time, F(1, 60) = 31.39, p < 0.001. Both the CoQ10 and placebo groups reported a reduced headache frequency at 224 days. The time x condition interaction was not significant, F(1, 60) = 1.28, p > 0.05. Patients in the CoQ10 group did not report fewer headaches over time than those in the placebo group. Similar results were found for the intent-to-treat analysis, with a significant main effect for time, F(1, 83) = 32.11, p < 0.001, and no significant interaction, F(1, 83) = 1.97, p > 0.05.
Migraine frequency
For migraine frequency, there was a significant main effect for time, F(1, 60) = 15.68, p < 0.001. Both the CoQ10 and placebo groups reported a reduced migraine frequency at 224 days. The time x condition interaction was not significant, F(1, 60) = 2.34, p > 0.05. Similar results were found for the intent-to-treat analysis with a significant main effect for time, F(1, 82) = 18.80, p < 0.001, and no significant interaction, F(1, 82) = 2.23, p > 0.05.
Headache severity
The results for headache severity showed a significant main effect for time, F(1, 54) = 8.09, p = 0.006, but no significant time x condition interaction, F(1, 54) = 0.06, p > 0.05. Both treatment groups reported a reduced headache severity over time. Patients in the CoQ10 group did not report less severe headaches than the placebo group. The intent-to-treat analyses showed similar results with a significant main effect for time, F(1, 81) = 9.41, p = 0.003, and no significant interaction, F(1, 81) = 0.15, p > 0.05.
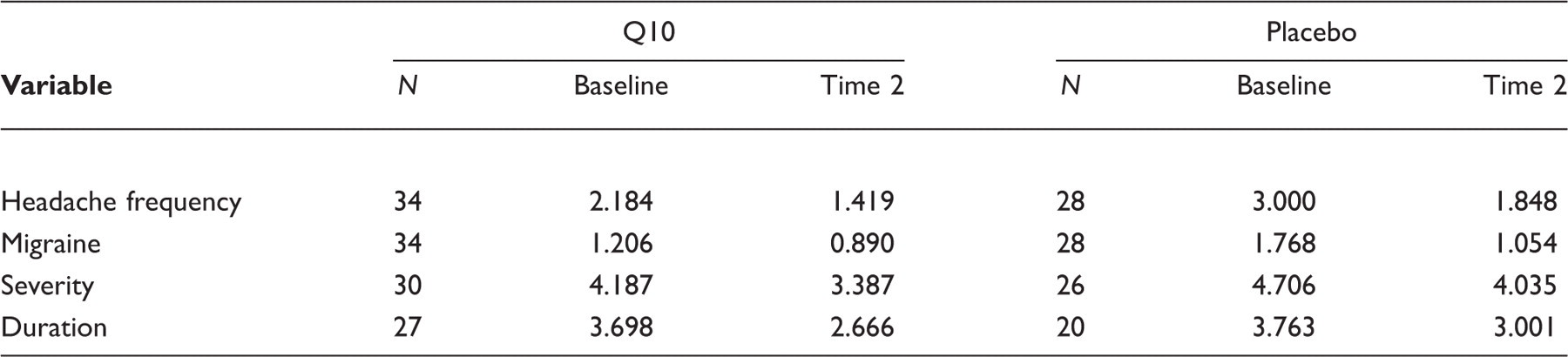
Headache duration
Means for baseline (weeks 1–4 average) and time 2 (weeks 13–16) for the full data sample
Discussion
Nutraceuticals, including vitamins and supplements, are commonly used by patients for health promotion and disease prevention/treatment. CoQ10 is one such supplement for migraine. It has potential pathophysiological modes of action in migraine including both as an anti-oxidant that can manage the inflammatory nature of migraine and as a mitochondrial component involved in the electron transport chain and energy production. CoQ10 has been demonstrated to be deficient in a significant number of migraine patients with an open-label response to treatment in a pediatric sample (13). Adult studies including an open-label (5) and a blinded study (6) have also demonstrated CoQ10’s potential effectiveness. In the randomized, controlled study, 42 adults were treated with either 300 mg of CoQ10 (100 mg for three doses per day) compared with placebo, with a significant difference in the 50% responder rate.
Results of the present study demonstrate that children and adolescents with episodic and chronic migraine improved over time with multidisciplinary, standardized treatment including both CoQ10 supplementation and placebo. Participants randomized to the CoQ10 and placebo groups reported decreased headache frequency, duration and severity. Those participants randomized to the CoQ10 group did show a greater initial reduction in headache frequency from baseline to week 1–4 in the chronic migraine group, but did not show any difference in duration or severity compared with participants in the placebo group. No differences between groups were found in headache-related disability over time.
The finding of a significant difference between the CoQ10 and placebo in the first treatment period, with a trend consistent with a difference in effectiveness of CoQ10 and placebo through the entire first stage (weeks 1–4) and equalization after the crossover (Figure 2) suggests that the effects of CoQ10 are very rapid for headache frequency response in chronic migraine. This was not demonstrated by a significant difference in the rate of change. The rate of change was measured by comparison of baseline with weeks 1–4 for each group. The results were complicated by a difference in baseline frequency in the chronic migraine group, thus limiting the interpretation of weeks 1–4 for the chronic migraine group.
Although for the episodic migraine this initial differential effect was not observed, the crossover response with worsening in the CoQ10 to placebo arm, in contrast to improvement in the placebo to CoQ10 in the first 4 weeks after crossover, suggests that this same effect of early response occurs for episodic migraine. This timing and pattern of improvement suggests that the mechanism by which CoQ10 helps to prevent headaches is a relatively rapid process. This may be due to the early effect of both mechanisms of CoQ10 – counteracting the negative oxidative effects of inflammation and the low energy states during migraine with the CoQ10 providing an energy shift.
In this study, the CoQ10 or placebo was added on to the full multidisciplinary treatment previously described (14,15). The observation of significant improvement throughout the study confirms the overall effectiveness of a multidisciplinary approach and thus represents a ‘real world’ approach that can be used clinically. This approach has a negative impact on the difference between the two treatment arms, because the improvement of both arms was greater than the difference between either arm. It is therefore unknown what the isolated effect of CoQ10 would be if it were the only treatment. This limitation was implemented because of the potential unknown safety concerns, pharmacokinetics, and pharmacodynamics of CoQ10 in the age group studied, as well as to provide a real world experience. With the superior safety profile demonstrated in this study and the trend toward overall improvement, a large study that focuses on a single preventative treatment may demonstrate CoQ10’s overall effectiveness. Conversely, CoQ10 may provide a synergistic effectiveness with existing preventative medication.
Compared with the randomized, controlled study in adults, the dose of CoQ10 used in this study was lower. This was due to the limited number of dosing studies at the initiation of this study with the only dosing parameter being 1–3 mg/kg in adult cardiac patients (17) and the daily nutritional intake and bioavailability (18). Subsequent studies have demonstrated the effectiveness of higher doses in neurological diseases (19) in addition to the adult migraine study. With the trend of improvement seen in the doses used in this study, higher doses can be suspected to be more effective, but a dose-response study is needed to confirm this hypothesis.
The number of subjects who did not return for timely follow-up and were considered drop-outs limited statistical analyses. Multivariate analyses were not done because of limited statistical power. It is notable that in the first stage of the study, there was a higher drop-out rate in the placebo arm (n = 26) compared with the CoQ10 arm (n = 22), predominantly because of the episodic migraine subjects, although the difference was not significant. This drop-out occurred after a significant improvement in frequency, especially after week 8 seen in the patients that continued. These patients may not have returned because this rapid degree of improvement augmented by the CoQ10 may have resulted in a response level at which the subjects and their parents no longer felt treatment was necessary. This pattern has been demonstrated in a long-term analysis of pediatric and adolescent migraine patients who had not returned for treatment and following contact were found to have a similar degree of improvement to those who continued with treatment (15).
In summary, this study demonstrated that CoQ10 at 100 mg was well tolerated and was effective with significant early differences in response compared with placebo using a multidisciplinary treatment paradigm. This raises the likelihood of CoQ10 functioning as an effective prophylactic agent – either independently or synergistically with existing preventative medications. It also raises the need for further higher dose studies and larger scale studies to define the overall effectiveness.
Footnotes
Funding
This study was sponsored by a grant from Tishcon, Inc.
