Abstract
Background: The purpose of this study was to determine whether transcranial direct current stimulation (tDCS) can be an effective prophylactic therapy for migraine and migraine-associated pain.
Method: This painless and non-invasive method was applied for 6 weeks over the visual cortex (V1), delivered three times per week. Thirty patients were assigned to cathodal or to sham stimulation, and 26 patients participated in the final analyses (cathodal: n = 13, sham: n = 13). During the first 3 weeks both groups received only placebo stimulation. Measures of attack frequency and duration, intensity of pain and number of migraine-related days were recorded 2 months before, during and 2 months post-treatment.
Results: Patients treated by cathodal tDCS showed a significant reduction in the duration of attacks, the intensity of pain and the number of migraine-related days post-treatment as compared to the baseline period, but not in the frequency of the attacks. However, compared to the sham group, only the intensity of the pain was significantly reduced post-stimulation. No patients experienced severe adverse effects.
Conclusion: Our results suggest that the application of cathodal stimulation over the V1 might be an effective prophylactic therapy in migraine, at least with regard to pain control.
Introduction
Worldwide at least 10–15% of the population suffer from migraine (1), resulting in several thousand days out of work per year. Prophylactic treatment is suggested for patients impaired by frequent or intense headache attacks. Beta-blockers or anticonvulsants have been shown to be effective in migraine prophylaxis. Nevertheless, the effectiveness of prophylactic therapy is still limited. The drug side-effects are frequently worrisome, and the frequency of headache and intensity of pain not significantly diminished. Therefore, the demand for non-pharmacological alternatives is high.
Recently the application of a non-invasive stimulation technique, transcranial magnetic stimulation (TMS), has become of significant interest in migraine treatment (2). The application of TMS in migraine is based on the belief that by exciting cortical neurons, it could prevent the advance of spreading depression that probably represents the pathological basis of the aura at the beginning of the attack (3). Indeed, in several studies applying repetitive TMS (rTMS) over the visual cortex (V1) or over the area involved in pain perception, a high percentage of patients reported that the stimulation had diminished or terminated headache (4,5). As a prophylactic treatment, high-frequency rTMS over the dorsolateral prefrontal cortex (DLPFC) was able to reduce the number of migraine attacks in chronic migraine (6). Also, a recent study using 1 Hz rTMS over the vertex demonstrated a significant decrease in attacks post-stimulation (7).
Similarly to rTMS, transcranial direct current stimulation (tDCS) can also induce reversible circumscribed and remote cortical excitability changes (8,9). However, whereas rTMS interrupts or excites neuronal firing, tDCS can modulate the spontaneous firing rates of neurons by changing resting membrane potential (10,11). Depending on the polarity of the stimulation, cathodal stimulation in animals reduced or completely inhibited spontaneous firing of cortical cells, while anodal stimulation increased them. In humans, it has been shown that cathodal tDCS can decrease and anodal tDCS can increase the excitability of the motor cortex (8,9). Results derived from studies on visual perception (12), somatosensory function (13) and cognitive actions (14) suggest that cathodal stimulation can decrease the excitability of visual, somatosensory and prefrontal cortices.
Migraineurs report abnormal visual discomfort when viewing striped patterns (15), and demonstrate differences in visual cortical processing when compared to non-headache controls in psychophysical [16–19] and electrophysiological studies (20). These differences between people with and without migraine on various visual measures have been attributed to abnormal visual cortical processing. It is generally suggested by visual psychophysical studies measuring task performance, reaction times or phosphene thresholds and by functional magnetic resonance (fMRI) studies detecting changes of metabolic activity that the excitability of the V1 is higher between attacks (6,16,21–24). As migraine patients are more sensitive to any kind of sensory overload than to a single sensory stimulus, the definitions of cortical hyperreactivity/hyperresponsivity are frequently applied in order to characterize the response of the migraineur’s brain to repeated stimulations (25).
The aim of the present study was to test whether repeated sessions of cathodal tDCS applied over the V1 in migraine patients result in a decrease in headache frequency, intensity and duration of the attacks by decreasing the cortical excitability. Furthermore, the efficacy of blinding within the conditions was tested by combining cathodal and sham stimulation in a patient group.
Methods
Patients
Clinical and demographic characteristics of the patients with regard to the different stimulation conditions

Experimental design
The study had three phases: (i) baseline evaluation, consisting of an 8-week period registering the frequency of the migraine attacks, the intensity, the onset and duration of the pain, the number of migraine-related days (the number of days on which the patients had migraine-related symptoms) and the type of analgesics in case of a migraine attack; (ii) 6-week treatment, consisting of treatment sessions with sham and active tDCS (15 min) for 3 days/week; and (iii) a follow-up period of 8 weeks. During the first 3 weeks of the treatment period all of the patients received only sham stimulation, during the second period half of them sham, half of them cathodal stimulation (Figure 1). Using the order of entrance into the study, every second patient was cathodally stimulated. Randomization was controlled by one of the authors, who did not have any contact with the patients during the stimulation and follow-up period. Patients had to keep a headache diary during the entire period ((i) to (iii)).
The study had three phases: an 8-week period registering the frequency of the migraine attacks, the intensity, the onset and duration of the pain, the number of headache-days; a 6-week treatment period consisting of treatment sessions with sham and active tDCS (15 min stimulation for 3 days/week) and a follow-up period of 8 weeks. During the first 3 weeks of the treatment period all of the patients received only sham stimulation; during the second period half of them sham, half of them cathodal stimulation.
Transcranial direct current stimulation
Patients were seated in a comfortable reclining chair with a mounted headrest throughout the experiments. The stimulations with regard to one given patient were always done by the same investigator. Direct current was transferred by a saline-soaked pair of surface sponge electrodes (5 × 7 cm) and delivered by a specially developed, battery-driven constant current stimulator (NeuroConn, Ilmenau, Germany). The cathode was placed over the Oz and the anode over the Cz electrode positions according to the 10–20 EEG system. A constant current of 1 mA intensity was applied for 15 min. Patients felt the current as an itching sensation at both electrodes at the beginning of the stimulation. For sham stimulation, the electrodes were placed in the same positions as for cathodal stimulation, but the stimulator was turned off automatically after 30 s of stimulation. This meant that all patients felt the initial itching sensation. The patients were blinded with regard to the type of tDCS and they were aware of the fact that they might receive sham or real stimulation.
Adverse effects of tDCS
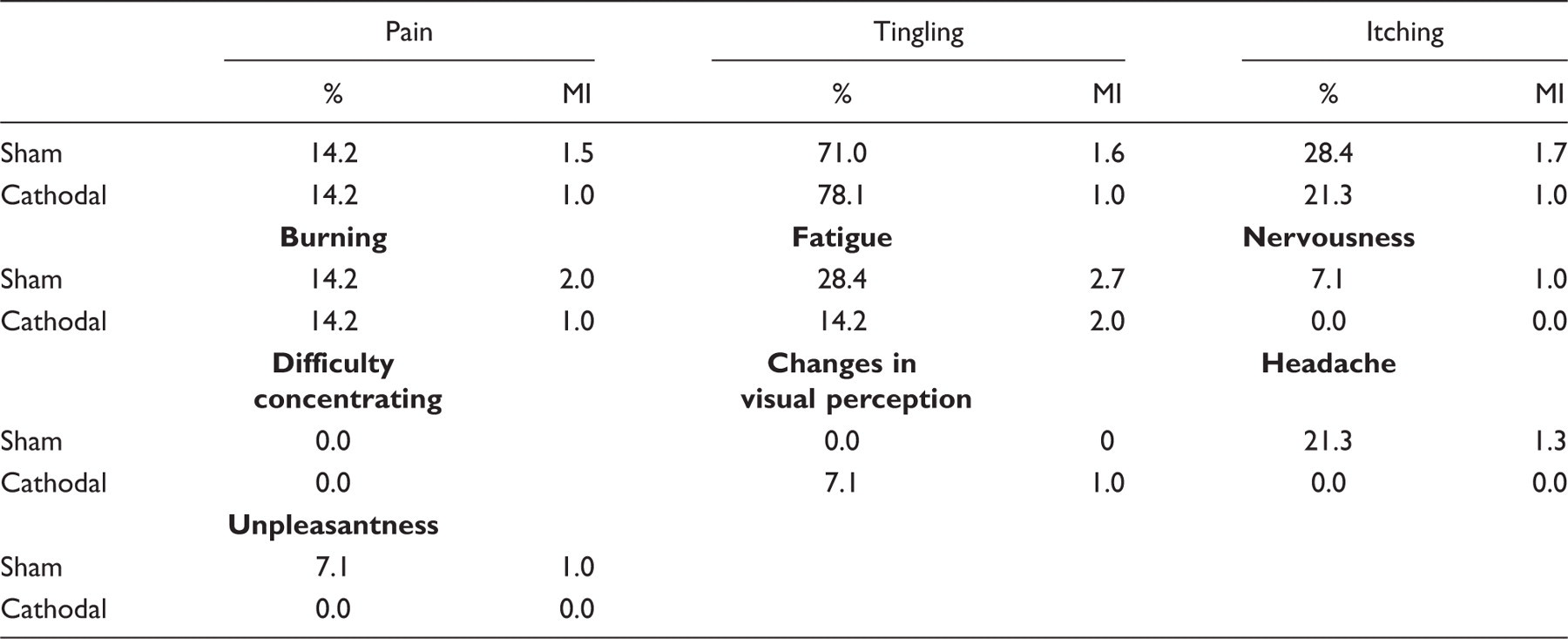
The cathodal current was applied continuously for 15 minutes/day 3 times per week in this protocol. Because the potential adverse effects of this technique are as yet unknown, patients completed a questionnaire (23) after the treatment period. The questionnaire contained rating scales for the presence and severity of headache, difficulties in concentrating, acute mood changes, visual perceptual changes, fatigue and discomforting sensations like pain, tingling, itching or burning under the electrodes during and after tDCS.
Statistical analyses
A per protocol analysis was done because four patients left the study without providing us their data for an intention-to-treat analysis. For all outcome measures, the number of migraine attacks, the number of migraine-related days and hours and the intensity of the pain were calculated for each patient before, during and after tDCS. Because we had a small number of patients in both groups and the non-normal distribution of many of the documented parameters within this study did not allow using ANOVA based on a normality assumption, we used the Mann–Whitney U-test to assess the differences between the placebo and verum groups. To compare the effects of the study treatment within the placebo or verum group, respectively, the Wilcoxon signed rank test was performed. Significance level was set at p ≤ 0.05.
The incidences of side-effects were coded in a binary system (no = 0, yes = 1) and the severities of the side-effects were rated on a numerical analogue scale (NAS) from 1 to 5, 1 being very mild and 5 being an extremely strong intensity of any given side-effect. The incidences and severities of the adverse effects were separately calculated during and after stimulation (27).
Results
Before the stimulation there was no significant difference between groups with regard to the number of migraine attacks (p = 0.77), the number of headache-related days (p = 0.14), the duration of the attacks (p = 0.44) and the intensity of the pain during an attack (p = 0.84).
Frequency of migraine attacks
Within the cathodal and sham groups, the number of migraine attacks was not significantly reduced when the baseline values were compared to the post-stimulation values (cathodal: p = 0.38; sham: p = 0.2) (Figure 2). Comparing the effects between the placebo and verum groups after stimulation, no significance differences could be observed (p = 0.88).
The mean number of migraine attacks 8 weeks before and 8 weeks after tDCS for the cathodal and placebo groups. The stimulation had no significant effect on the frequency of attacks. Bars represent SEM.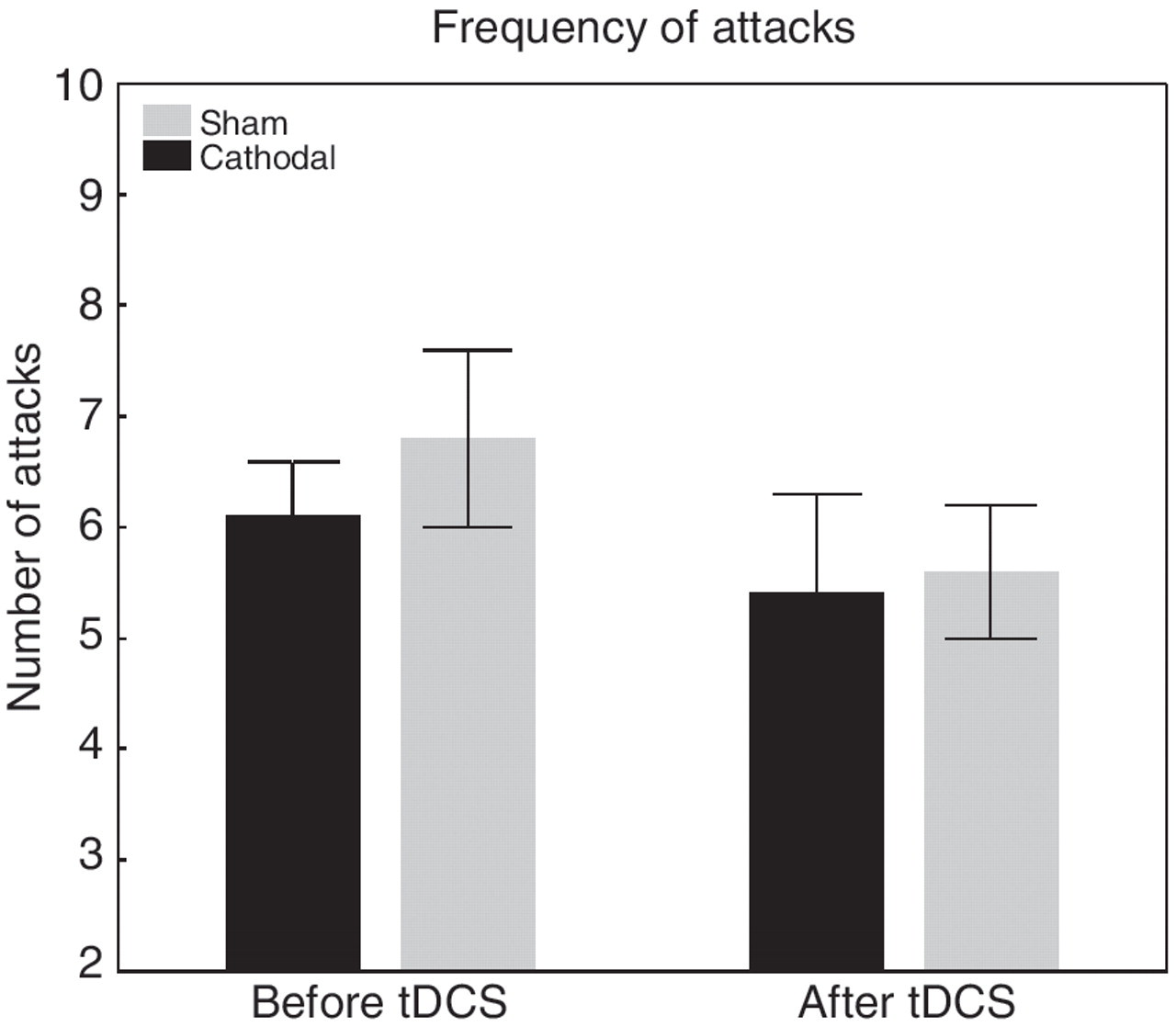
Migraine-related days
There was a significant reduction in migraine-related days after stimulation in the verum (from 16.2 ± 2.4 days to 9.31 ± 1.5 days; p = 0.004) but not in the sham group (from 12.8 ± 2.77 days to 11.0 ± 3.5 days; p = 0.17) (Figure 3). However, comparing the number of migraine-related days between the placebo and verum groups post-stimulation, no significant difference was seen (p = 0.61).
The mean number of migraine-related days 8 weeks before and 8 weeks after tDCS for the cathodal and placebo groups. *Statistical significance (p < 0.05). There was no statistical difference between the sham and verum groups. Bars represent SEM.
Duration of the migraine
There was a significant reduction in the mean duration of the migraine attacks after stimulation in the verum (from 8.95 ± 1.6 hours to 7.2 ± 1.6 hours; p = 0.05) but not in the sham group (from 9.9 ± 1.5 hours to 9.4 ± 1.8 hours; p = 0.55) (Figure 4). However, comparing the average duration of the attacks after stimulation between the placebo and verum groups, no significant difference was seen (p = 0.38).
The average duration of attacks 8 weeks before and 8 weeks after tDCS for the cathodal and placebo groups. *Statistical significance (p < 0.05). There was no statistical difference between the sham and verum groups. Bars represent SEM.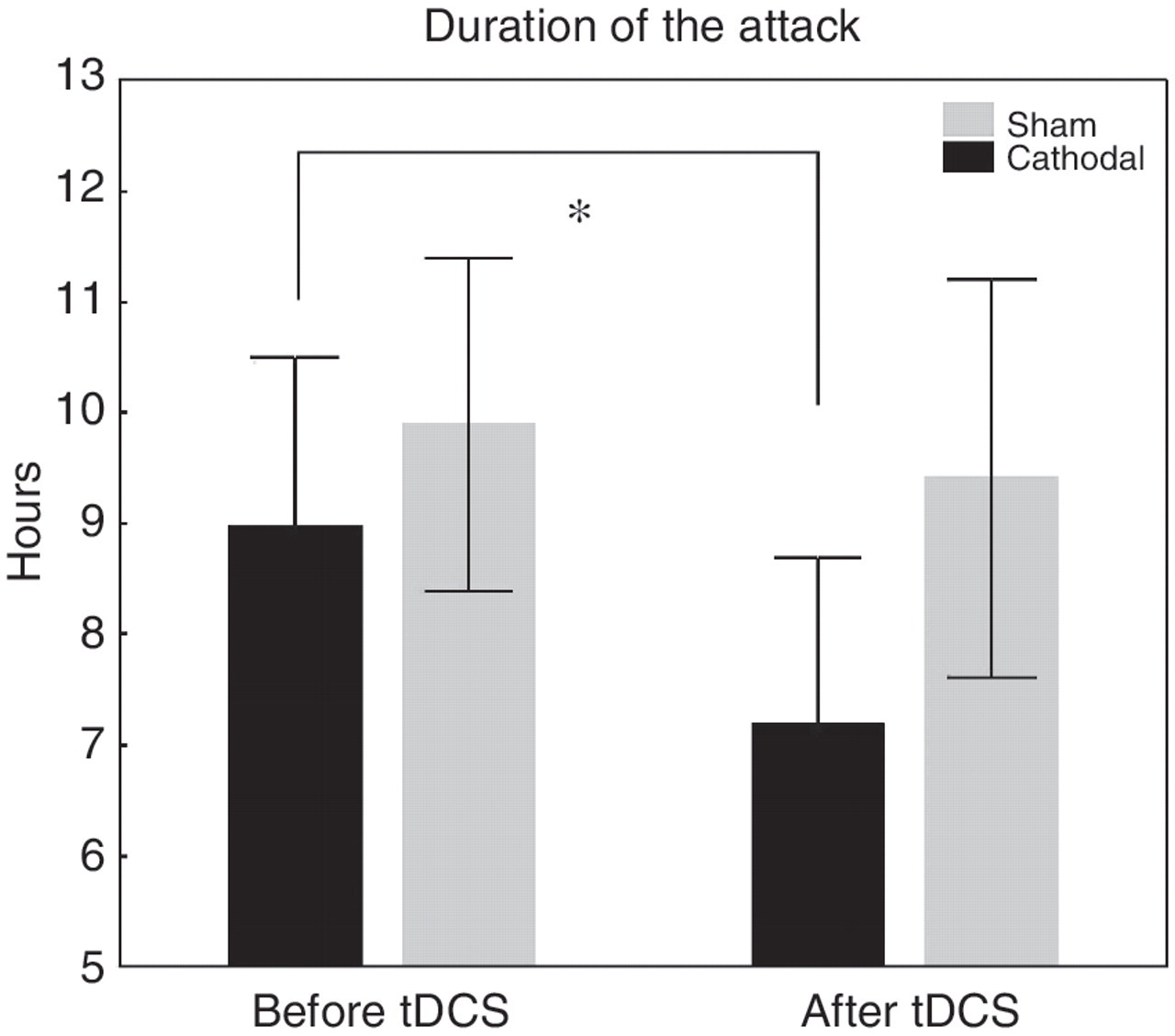
Intensity of pain
In the verum group the mean pain intensity decreased significantly when the baseline values (1.99 ± 0.1) were compared to the post-tDCS values (1.54 ± 0.2) (p = 0.02) (Figure 5). In the sham group there was no significant change (from 1.88 ± 0.1 to 1.92 ± 0.2; p = 0.7). Comparing the intensity of pain between the placebo and verum groups after stimulation, a significant difference was observed (p = 0.05).
The mean pain intensity judged on a rating scale from 0 (no pain) to 3 (maximal pain) 8 weeks before and 8 weeks after tDCS in the cathodal and placebo groups. There was a significant decrease in pain intensity in the verum group compared to the placebo group (p = 0.05). Bars represent SEM.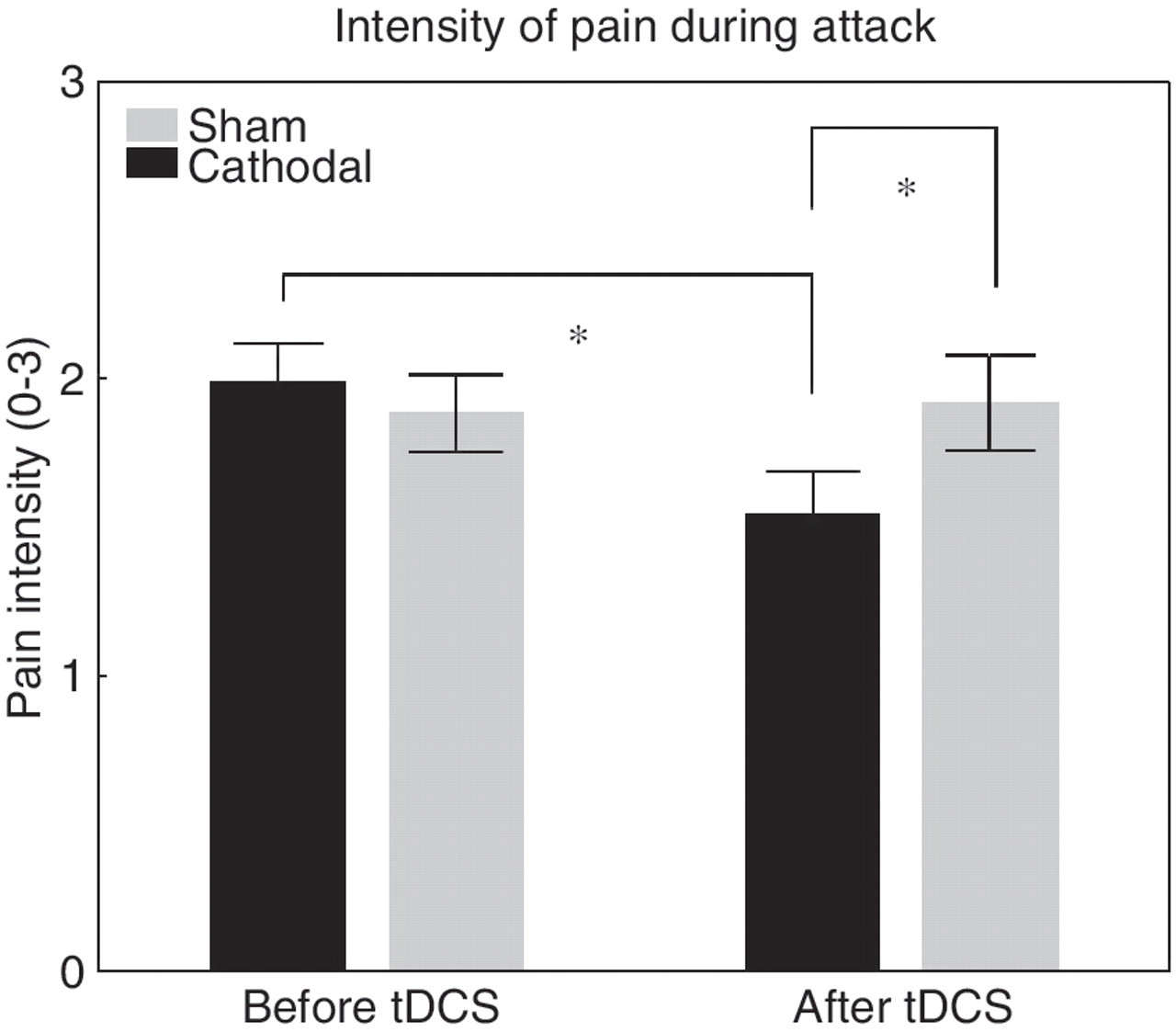
Comparing MwA and MwoA patients
The mean frequency, duration and intensity of migraine attacks with regard to the presence or absence of visual aura (± SEM)

Adverse effects of tDCS
A. Percentage of patients reporting adverse effects of tDCS during stimulation. MI: mean intensity using a scale from 0 to 5
Discussion
Given that migraine is associated with abnormal neuronal excitability between attacks, we hypothesized that inhibitory tDCS over the V1 might be effective in migraine prophylaxis by diminishing cortical excitability between attacks, and thus have therapeutic effects. However, the primary outcome of this study was negative: no effects were evident for the reduction in the number of attacks. Nevertheless, we could observe a significant reduction by verum treatment with regard to the duration of migraine, the number of migraine-related days and pain intensity.
As far as we know, this is the first study using tDCS as a prophylactic treatment in migraine, so a comparison with previous results is not possible. With regard to transcranial stimulation methods, so far only two studies have examined the effects of rTMS in the prophylactic treatment of migraine (6,7). In the first study patients with chronic migraine received high-frequency rTMS treatment of the left DLPFC for 12 sessions on alternating days. Each rTMS session consisted of 10 trains of 2 s duration, separated by a 30 s pause, given at 20 Hz frequency and 90% motor threshold (MT) intensity. The authors reported significant reduction in headache attacks, the mean number of abortive pills used and the headache index after stimulation in comparison with sham stimulation. However, only 11 patients were included in this study (6 verum treatments). In the second study 27 migraineurs participated (14 verum) (7). They were treated with low-frequency rTMS using two trains of 500 pulses with a frequency of 1 Hz on 5 consecutive days over the vertex. A significant decrease in migraine attacks was observed in the verum group. Nevertheless, when comparing the effect of stimulation between the verum and placebo groups, no significant difference was evident. The same was true with regard to the migraine-related days and total hours with migraine. No effects were observed concerning the intensity of pain and use of analgesics.
According to recent results, the application of single or only a few TMS pulses is more successful in treating acute migraine attacks. Clarke et al. (4) treated 42 patients with migraine, using two stimulus pulses 5 s apart over the area of perceived pain or over the area of the brain generating the aura at the beginning of the attack. When assessed 20 minutes later, an improvement was observed in 69% of patients. The mean reduction in pain intensity was 25%. In another study a similar improvement was demonstrated (5), however, a high incidence of improvement in the control group (48%) treated with a placebo device was also reported. A recent study incorporated more patients (267 patients) (2) and used hand-held devices operated by the patients and not those used by healthcare professionals in the previous studies. The stimulator was placed over the visual areas. For the control stimulation patients were given similar-looking devices that did not provide magnetic pulses. The authors reported that the pain-free response rates after 2 h were significantly higher in the verum group (39%) than in the placebo-treated group (22%). Furthermore, sustained pain-free response rates significantly favoured verum treatment at 24 and 48 h after stimulation.
In our study there was a significant reduction in migraine-related days after stimulation in the verum but not in the sham group. However, comparing the number of migraine-related days post-stimulation between the placebo and verum groups, no significant difference was seen. The reason for this might be the higher number of migraine-related days at baseline in the cathodal (16.2) than in the sham group (12.8). Nevertheless, this difference in migraine-related days before treatment between the two groups was not statistically different.
With regard to the overall non-significant results between the placebo and verum groups in our study, we have to consider that cathodal stimulation over V1 might not be able to ameliorate cortical hyperexcitability in migraine as observed in an rTMS study (7). However, a clear tendency in the verum group can be seen concerning the reduction in attack duration and intensity of pain, therefore it is possible that in our study the intensity, the frequency and/or the duration of the stimulation was not long and strong enough to affect the frequency of migraine attacks. Compared to some previous studies that used anodal tDCS over the primary motor cortex for the treatment of chronic pain, the intensity of our stimulation is somewhat lower. For example, Fregni et al. (28) stimulated patients with chronic back pain using 2 mA intensity on 5 consecutive days reaching 58% in pain relief after stimulation. However, Antal et al. (29) applied only 1 mA intensity but using a smaller stimulation electrode in a heterogeneous patient group with chronic pain and observed that the average pain release was 38%. The reduced tDCS intensity is better suited for blinding. Furubayashi et al. (30) reported that an intensity of 3 mA is already painful, therefore 1 mA has a higher chance of going undifferentiated compared to placebo conditions than 2 mA. Because there was no difference with regard to the occurrence of itching/tingling/burning sensations between patients after sham and verum stimulation, and fatigue was experienced by an even higher percentage of sham patients compared to cathodally stimulated patients, we are relatively sure of having provided an optimal placebo condition. However, future studies should consider increasing the stimulation duration and/or frequency.
The higher incidence of headache and fatigue during and after tDCS compared in the sham group is surprising and raises the question of whether the cathodal stimulation improved fatigue in migraine patients. A higher proportion of migraineurs (55.6%) reported headache after tDCS compared to healthy subjects (7.8%) in a previous safety study (27). Furthermore, in another study, in which patients with chronic pain were treated with tDCS, 35–39% of the subjects described headache after stimulation (29). Nevertheless, we have not observed any serious complications such as seizures in connection with the application of tDCS. There are no data in the literature reporting epileptic jerks elicited by tDCS; it appears that cathodal tDCS has an anticonvulsant effect at least in the animal model (31). According to the histological analysis of rat brain tissue after tDCS, no cortical oedema, necrosis, or any sign of cell death (karyopyknosis, karyolysis and karyohexis) was observed. As tDCS is currently tested for clinical applications in phase II and III studies, the impact of consecutive sessions of cortical stimulations in different patient populations is not yet fully known. Our data concerning the adverse effects of the cathodal stimulation could be interpreted as safety evidence with regard to tDCS in migraine.
The limitations of this study should also be discussed. First, this clinical trial was an exploratory study; our sample size might not have been large enough to detect some characteristics associated with a positive effect of tDCS. Due to the exploratory nature of the study, the statistical analysis used also implemented exploratory methods. Second, the patient group was heterogeneous with regard to the type of migraine. Third, most of our patients favoured alternative, non-pharmacological migraine treatments and therefore they might have a positive anticipation for tDCS treatment. Thus, it cannot be ruled out that the observed improvements by tDCS were partly due to a placebo effect. However, this is probably true for many other patient populations in different studies in which transcranial stimulation methods are used. Future studies should include more patients and direct physiological measures of cortical excitability to prove whether the reduction in pain induced by cathodal tDCS is correlated with a reduction in cortical excitability. It is an interesting question, whether applying anodal tDCS should lead to opposite effects or no change at all. Nevertheless, anodal stimulation has to be applied carefully, because it may induce a migraine attack.
In summary, cathodal tDCS over the V1 was effective for migraine prophylaxis with regard to the number of migraine duration and mean pain intensity but not in the number of migraine attacks, when compared with sham stimulation. Therefore, our data are encouraging for further research in bigger patient populations, and also separating MwA patients from MwoA patients in order to clarify if these groups of migraine patients differ in neuronal functionality.
Footnotes
Funding
This study was supported by the German Ministry for Research and Education (BMBF-01EM 0513).
Conflict of interest
B. Percentage of patients reporting adverse effects of tDCS after (B) stimulation. MI: mean intensity using a scale from 0 to 5
| Pain |
Tingling |
Itching |
||||
|---|---|---|---|---|---|---|
| % | MI | % | MI | % | MI | |
| Sham | 7.1 | 1.0 | 7.1 | 1.0 | 14.2 | 1.0 |
| Cathodal | 0.0 | 0.0 | 7.1 | 1.0 | 0.0 | 0.0 |
|
|
|
|
||||
| Sham | 14.2 | 1.0 | 42.6 | 2.2 | 0.0 | 0.0 |
| Cathodal | 0.0 | 0.0 | 7.1 | 2.0 | 0.0 | 0.0 |
|
|
|
|
||||
| Sham | 0.0 | 0.0 | 0 | 35.5 | 2.0 | |
| Cathodal | 0.0 | 0.0 | 0.0 | 21.3 | 2.3 | |

