Abstract
Background: The causal relationship between experimental headache and vasodilatation has not been fully clarified. In the present study, we combined headache and vascular data from eight experimental studies and conducted detailed statistical analyses. Given that substances used in all these experiments were vasodilators we examined a possible correlation between headache scores and increases in arterial diameter.
Methods: We identified nine studies and retrieved raw data in 89 healthy subjects (46 females, 43 males), mean age 27 years (range 18–59 years). The following variables were collected: maximal median headache intensity scores on a verbal rating scale (VRS) during immediate headache (0–120 minutes); the mean velocity of blood flow in the middle cerebral artery (VmeanMCA); and the diameter of the frontal branch of the superficial temporal artery (STA) during the maximal median headache intensity.
Results: The scatter plots show no relationship between maximal headache score and the relative changes in VmeanMCA and diameter of the STA. The main analyses of covariance showed a significant effect only of heart rate on headache (p = .014). The interaction tests were insignificant for all variables.
Conclusions: The major outcome is a finding of no linear relationship between experimental immediate headache and dilatation of the MCA or STA.
Introduction
In the second century, Galen suggested that throbbing pain during headache originated from blood vessels. In 1672, Thomas Willis proposed the first vascular hypothesis of migraine. More than 70 years ago, Graham and Wolff (1) provided the first observations in humans demonstrating that focal head pain may be elicited from both extra- and intracranial vessels. More recently it was reported that focal headache may be induced by balloon dilatation of cerebral arteries (2). All substances that can induce headache are vasodilators and most of them induce head pain in healthy subjects simultaneous with dilatation of extra- and intracerebral arteries (3–9). The causal relationship between experimental headache and vasodilatation has not, however, been fully clarified. Most experimental studies focusing on the headache-eliciting effect of vasodilating substances have used small sample size (usually between 10 and 12 subjects) (3–9). The power was therefore small to test a possible correlation between headache intensity and arterial dilatation. In the present study, we combine headache and vascular data from eight experimental studies and thus are able to conduct detailed statistical analyses. Given that substances used in all these experiments are vasodilators, we examined a possible correlation between headache scores and increases in arterial diameter. We hypothesized that there is a linear relationship between maximal median headache intensity and dilatation of the middle cerebral artery (MCA) and superficial temporal artery (STA) in pharmacologically induced immediate headache in healthy subjects. In addition, we aimed to examine a possible relationship between headache and vasodilatation within each vasoactive substance.
Methods
We identified nine studies (4–7;10–14) and were able to retrieve raw data in 89 healthy subjects (46 females, 43 males), mean age 27 years (range 18–59 years). The following variables were collected: maximal median headache intensity scores on a verbal rating scale (VRS) during immediate headache (0–120 minutes); the mean velocity of blood flow in the middle cerebral artery (VmeanMCA); and the diameter of the frontal branch of the superficial temporal artery (STA) during the maximal median headache intensity.
Exclusion criteria were a history of migraine or any other type of headache (except infrequent episodic tension-type headache [>1/month]); any daily medication apart from oral contraceptives; serious somatic or psychiatric diseases. All studies were approved by the Ethics Committee of the County of Copenhagen and were undertaken in accordance with the Helsinki Declaration of 1964, as revised in Edinburgh in 2000.
Headache intensity
Headache intensity was recorded on a VRS from 0 to 10 (0 = no headache; 1 = very mild headache (including a feeling of pressing or throbbing); 5 = moderate headache; 10 = worst imaginable headache) (9).
The middle cerebral artery
VmeanMCA was recorded bilaterally by transcranial Doppler (TCD) with hand-held 2-MHz probes (Multidop X, DWL, Sipplingen, Germany), as previously described (15). To correct VMCA measurements for possible significant changes in end-tidal PCO2 (PetCO2), changes in PetCO2 were recorded simultaneously to the TCD measurements using an open mask that caused no respiratory resistance (ProPac Encore®, Welch Allyn Protocol, Beaverton, OR, USA).
The superficial temporal artery
The diameter of the frontal branch of the STA was measured by a high-resolution ultrasonography unit (Dermascan C, Cortex Technology, Hadsund, Denmark: 20 MHz, bandwidth 15 MHz), as previously described (16).
Vital signs
Heart rate and blood pressure were measured with an auto-inflatable cuff (ProPac Encore®, Welch Allyn Protocol, Beaverton, OR, USA). ECG (Cardiofax V, Nihon-Kohden, Shinjuku-ku, Tokyo, Japan) was monitored on an LCD screen and recorded on paper.
Statistical methods
The headache scores were analyzed by analysis of covariance (ANCOVA) (i.e. regression analyses) with an additive effect of “study” and the relevant regressor variable. This effectively amounts to combining the within-study regression coefficients to a common value and testing its significance. We also performed tests of interaction to see whether this pooling of regression coefficients is permissible. Tests of interaction were conducted using standard linear-model methodology, by introducing an interaction term between study and the regressor variable and testing its significance using an F-test. Similarly, the study effect was tested in the additive model using an F test for significance of the study term. The headache response variable is highly discretized, but the methods are reasonably robust against deviations from the normal distribution.
All analyses were performed with R, version 2.11.1. (http://www.R-project.org) and SPSS Statistics software 17.0.0 (Chicago, IL, USA). Five percent (p < .05) was chosen as the level of significance.
Results
We retrieved raw data in 89 healthy subjects and plotted headache and vascular variables in scatter plots (Figure 1). The plots show no relationship between maximal headache score and the relative changes in VmeanMCA and diameter of the STA.
Scatter plots show the degree of correlation between maximal headache score during immediate phase and relative changes in vascular variables in 89 healthy volunteers. All plots include studies using the following provoking substances: vasoactive intestinal peptide (VIP) (study 1), carbachol (study 2), prostaglandin I2 (PGI2) (study 3), prostaglandin E2 (PGE2) (study 4), calcitonin gene-related peptide (CGRP) (study 5), glyceryl trinitrate (0.5 µg/kg GTN) (study 6), (0.25 µg/kg GTN) (Study 7) and prostaglandin D2 (PDE2) (study 8). (A) Headache and the relative changes in the mean velocity of blood flow in the middle cerebral artery (VmeanMCA). (B) Headache and the relative changes in diameter of superficial temporal artery (STA). (C) Headache and the relative changes in heart rate. The coefficient of determination (R2) showed trend to linear regression for heart rate (p = .032). The plots with VmeanMCA and STA data set show no relationship between headache and vascular variables.
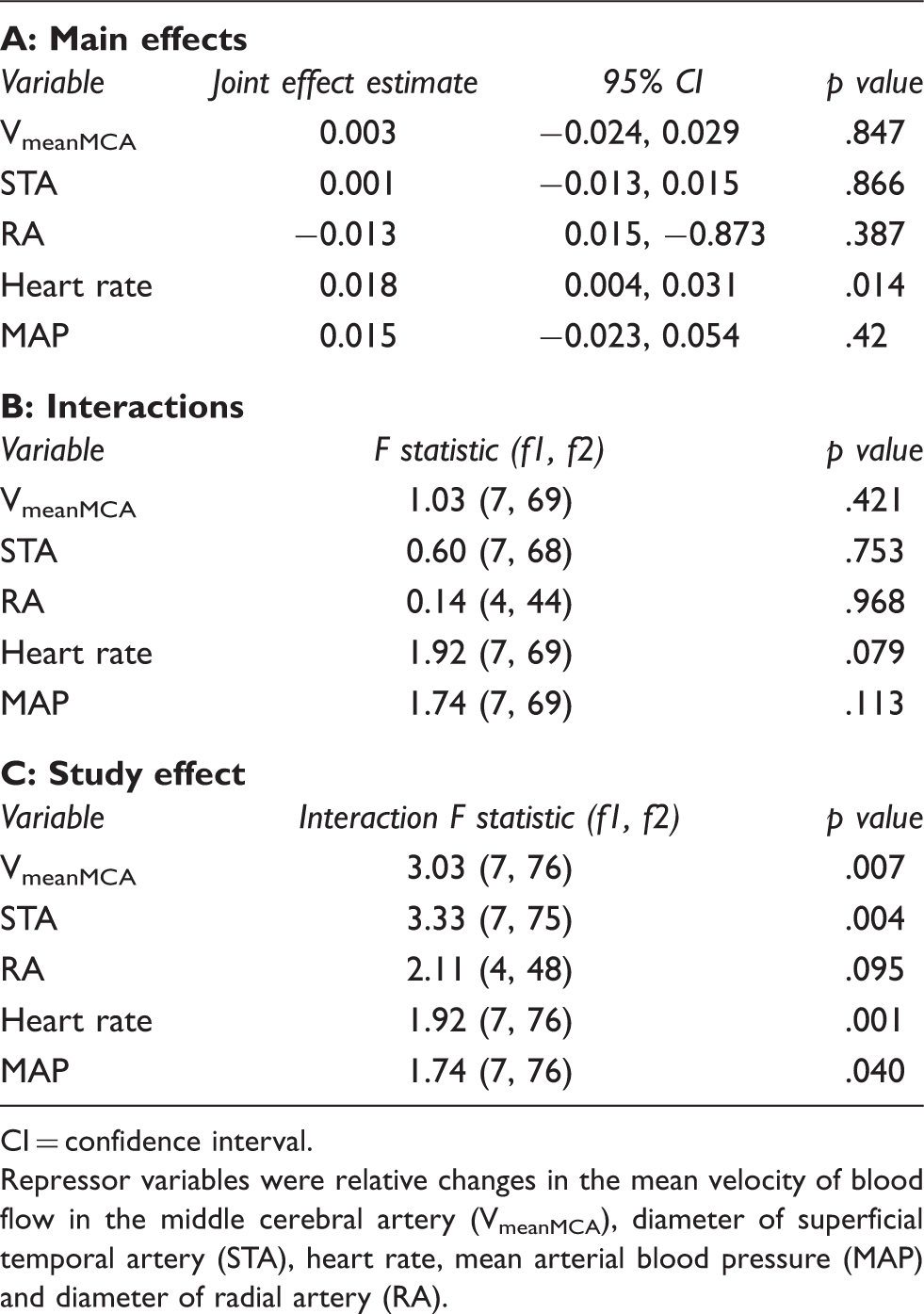
The main ANCOVA analyses are given in Table 1. They showed a significant effect only of heart rate on headache (p = .014). The interaction tests were insignificant for all variables. It should be noted that the only individual study with a significant effect of heart rate is the PGE2 study (study 4) (p < .017). Forest plots illustrating the relative strength of effects are shown in Figure 2.
"Forest plots", similar to those used in meta analysis, are shown. The thinner lines show estimates and 95% confidence intervals for the (univariate) effect of a covariate, separately for each study. Assuming a common effect for all studies, the joint estimate is shown in the thicker bottom line. Not all covariates were measured in all studies. The mean velocity of blood flow in the middle cerebral artery (VmeanMCA), diameter of superficial temporal artery (STA), heart rate, mean arterial blood pressure (MAP) and diameter of radial artery (Radialis) are shown. Study codes as in Figure 1. (A) Analyses of covariance (regressor effects) and (B) analysis of interaction and study effect (C) in 89 healthy volunteers CI = confidence interval. Repressor variables were relative changes in the mean velocity of blood flow in the middle cerebral artery (VmeanMCA), diameter of superficial temporal artery (STA), heart rate, mean arterial blood pressure (MAP) and diameter of radial artery (RA).
Discussion
In the present study we examined for the first time the correlation between pharmacologically induced headache and vasodilatation in normal subjects. The major outcome is a finding of no linear relationship between experimental immediate headache and dilatation of the MCA or STA. However, we found a statistically significant relationship between headache score and heart rate.
Vasodilatation and pain in animal studies
Remarkably few animal studies have examined the effect of vasodilatation on C-fiber activation and sensitization. Kaube et al. (17) studied mechanical distention of the superior sagittal sinus with a small intraluminal device and recorded single units in the dorsolateral C2 spinal cord (i.e. at the central level). The authors concluded that distention with physiological frequencies and amplitudes of the superior sagittal sinus in the anesthetized cat does not suffice to stimulate single units in the spinal cord. Strassman et al. (18) tested for responses to intravascular infusion into the lumen of the sinus, using hypertonic sodium chloride as a stimulus. Trigeminal ganglion cells responded to both topical application and infusion, indicating that the neurons have receptive endings within or adjacent to the walls of the sinus. The authors did not test for sensitization (lowered mechanical threshold) following hypertonic saline. Interestingly, there was no discharge evoked by rapid infusion of normal physiological saline (0.9%) into the superior sagittal sinus sufficient to produce visible distention (Strassman, personal communication). The question is whether the sinus is a relevant structure to address in the study of headache and vasodilatation. Cerebral arteries and veins differ considerably in vessel-wall morphology (19). Furthermore, an experimentally applied force in a small segment of the sagittal sinus might not be equivalent to physiological dilatation produced by smooth muscle relaxation and the effectiveness of vasodilatation as a stimulus might be greatly altered by opening the cranium (20). If technically possible, it would be relevant to examine vasodilatation and activation/ sensitization of sensory afferents in a “closed cranium” preparation.
To clarify the relationship between nociception and vasodilatation, vasodilator agents have been used. Levy and Strassman (21) applied NO donor sodium nitroprusside on the dura. NO did not change the level of spontaneous activity but produced sensitization of meningeal nociceptors (21). Similarly, dural and systemic administration of the powerful dilator calcitonin gene-related peptide (CGRP) did not activate or sensitize meningeal nociceptors (22). In contrast, Cumberbatch et al. (23) reported that CGRP caused a meningeal vasodilatation that resulted in sensitization of central trigeminal neurones. The facilitation of facial sensory processing was blocked by activation of pre-synaptic 5-HT1B/1D receptors. These data suggest that the effects of NO and CGRP are complex, and multiple mechanisms should be required for robust headache eliciting effect of both agents in humans (3,8,9).
In summary, more animal studies are needed to clarify whether mechanical distention of arteries leads to activation or sensitization of sensory afferents. Furthermore, studies are needed to understand mechanisms underlying the headache or migraine-eliciting effect of vasoactive substances.
Vasodilatation and head pain in humans
In 1940, Ray and Wolff (24) reported that stimulation or distention of the large cranial arteries and dural arteries evoked head pain associated with the feeling of nausea or sickness. The similarity between referred pain locations following stimulation of large cerebral arteries and headache patterns in migraine provide the strongest support yet for the involvement of vascular nociceptors in migraine pain. It is doubtful, however, whether this involvement can explain pharmacologically induced immediate headache in healthy volunteers. This question has been extensively studied using long-term (seven-day) exposure to NO. Thus, Christiansen et al. (25,26) reported a close temporal association between the disappearance of headache and the attenuation of the 5-isosorbide-mononitrate (5-ISMN)–induced dilatation of the STA, but no relation to decreasing MCA dilatation. These data suggested that dilatation of extracerebral arteries may be the mechanism of NO-triggered head pain. In support, Iversen et al. (27) reported that the time-effect curves for the effect of glyceryl trinitrate (GTN) on headache and on dilatation of MCA differed markedly. The authors suggested that MCA was most likely not the primary source of head pain in the GTN model (27). However, GTN is highly soluble lipid and easily penetrates membranes, including the blood-brain barrier (BBB). NO can therefore act both peripherally and centrally (i.e. multiple sites of action cannot be excluded).
Maximal absolute (median headache score) and percent changes (other variables)

VmeanMCA = blood flow velocity of the middle cerebral artery. STA = diameter of superficial temporal artery. HR = heart rate. MAP = mean arterial pressure. RA = diameter of radial artery. rCBF = regional cerebral blood flow. ND = not done. NS = not significant. tmin = time of maximal response.
The median maximal score was 0 and in these studies we examined a linear relationship between maximal dilatation of the middle cerebral artery (MCA) and superficial temporal artery (STA) and headache intensity. The following substances were infused: vasoactive intestinal peptide (VIP), carbachol, prostaglandin I2 (PGI2), prostaglandin E2 (PGE2), calcitonin gene-related peptide (CGRP), glyceryl trinitrate (GTN) and prostaglandin D2 (PDE2).
Note: Table shows maximal responses of each variable.
Taken together, these data suggest that vasodilatation per se cannot explain pharmacologically induced immediate headache, but suggest that it may play a role in combination with other effects.
Implications for migraine pathophysiology
For more than 100 years, and particularly after the first report by Graham and Wolff of extracranial vasodilatation during migraine (1), there has been an intense debate about the vascular hypothesis of migraine. In 1991, Friberg et al. (35) reported that migraine headache was associated with MCA dilatation (20%) on the headache side and that the MCA velocity on the headache side returned to normal after treatment with sumatriptan and recovery. Thomsen et al. (36) showed decreased velocity in the MCA on the pain side of half sided migraine attacks, and Iversen et al. (37) found the diameter of the STA larger than on the pain-free side during attack. Together, these studies thus show a mild intra- and extracranial dilatory influence. It was concluded that the dilatation was not enough to physically cause pain but rather reflected leakage of dilator substances in the perivascular space or parasympathetic effects. However, these findings have been challenged by Zwetsloot et al. (38) in a Doppler study and later by Schoonman et al. (39) in an MRI study. The latter confirmed previous observations in migraineurs that GTN-induced immediate vasodilatation and immediate headache (15). However, during delayed migraine attacks no vasodilatation was detected. It may be doubted whether these studies had the power to detect changes as small as those observed by Thomsen et al. (36) and Iversen et al. (37). Based on the fact that all migraine- triggering substances are strong vasodilators, that dilatation has been shown in some studies and that not all vessels have been measured, we believe that vascular mechanisms cannot be dismissed in the pathogenesis of migraine attacks. In the future, it would be interesting to investigate if headache or migraine can be provoked by substances without known vasodilatory effect and if the delayed pharmacologically (e.g. triggered by CGRP or pituitary adenylate cyclase-activating polypeptide) induced delayed migraine is associated with dilatation of the MCA or/and the MMA.
Footnotes
Acknowledgments
This work was supported by the Lundbeck Foundation as part of the Lundbeck Foundation Center for Neurovascular Signalling (LUCENS). We would like to thank Dr Jakob Møller Hansen, Dr Henrik Schytz, Dr Troels Wienecke and Dr Jesper Tvedskov for sharing raw data included in this manuscript.
