Abstract
Background: Among serotonin receptors, 5-HT1A receptors are implicated in the regulation of central serotoninergic tone and could be involved in the abnormal brain 5-HT turnover suspected in migraineurs. The aim of this study was to investigate 5-HT1A receptors’ availability during migraine attacks.
Methods: Ten patients suffering from odor-triggered migraine attacks and 10 control subjects were investigated using positron emission tomography (PET) and [18F]MPPF PET tracer, a selective 5-HT1A antagonist. All subjects underwent calibrated olfactory stimulations prior to the PET study.
Results: Four patients developed a migraine attack during the PET study. In these patients, statistical parametrical mapping and region of interest analyses showed an increased [18F]MPPF binding potential (BPND) in the pontine raphe when compared to headache-free migraineurs and control subjects. This ictal change was confirmed at the individual level in each of the four affected patients. In comparison with the headache-free migraineurs, patients with a migraine attack also showed significantly increased [18F]MPPF BPND in the left orbitofrontal cortex, precentral gyrus and temporal pole. No significant change in [18F]MPPF BPND was observed between headache-free migraineurs and controls.
Conclusions: Our results emphasize the role of 5HT1A receptors in the pontine raphe nuclei during the early stage of migraine attacks.
Introduction
The serotoninergic system plays an important role in the endogenous pain control system and possibly in migraine pathophysiology (1). Reduced systemic serotonin (5-HT) concentration between attacks was the first observation of a serotoninergic dysfunction in migraine (2), though its relation to central nervous system 5-HT remains unclear. Other results support the possibility of an abnormal central serotonin turnover in migraineurs. The interictal habituation deficit of evoked cortical responses observed in migraineurs is thought to reflect a low 5-HT neurotransmission (3,4). Increased brainstem serotonin transporter (SERT) availability has been reported in migraineurs interictally (5), while during attacks [15O]H2O positron emission tomography (PET) studies have shown specific brainstem activation (6–10), suggesting the involvement of the locus coeruleus and/or the raphe nuclei. PET study using alpha-[11C]methyl-L-tryptophan (AMT) also suggests that changes in the rate of brain serotonin synthesis occur during migraine attacks (11).
5-HT1A receptors play a major role in the regulation of the central serotoninergic tone, in particular those located in the raphe nuclei, which act as autoreceptors to provide a negative feedback on serotoninergic neuron firing activity. The other forms of 5-HT1A receptors are heteroreceptors, which are widely distributed over the cortex, with greatest concentration in limbic areas (12,13). Thus, 5-HT1A receptors could be involved in the abnormal turnover of central 5-HT suspected in migraineurs. Several studies have demonstrated an increased prolactin response in migraine patients to various 5-HT1A receptor agonists, suggesting a hypersensitivity of these receptors between attacks (14–16). Furthermore, our group has recently reported an increased binding potential of 5-HT1A receptors in the posterior cortical areas of migraineurs without aura using [18F]MPPF-PET (17).
To further investigate the role of the central serotoninergic system during migraine attacks, we have undertaken a [18F]MPPF-PET study in migraineurs suffering from interictal olfactory hypersensitivity (OHS) and odor-triggered migraine attacks, a situation offering the possibility to elicit the patient’s usual migraine attack immediately prior to the PET study through olfactory stimulations.
Material and methods
Subjects
Ten migraineurs (eight right-handers, two left-handers, mean age ± standard deviation [SD] = 36.2 ± 10.7) and 10 age-matched control subjects (nine right-handers and one left-hander, mean age ± SD = 39.7 ± 12) were included in the study. Patients fulfilled the International Headache Society criteria (18) for episodic migraine without aura and suffered from interictal OHS and odor-triggered attacks. The presence of OHS during migraine-free periods was evaluated by asking patients whether they regarded themselves as clearly hypersensitive to odors between attacks, and by using a validated chemical odor intolerance index (19). Exclusion criteria for all subjects included chronic daily headache, known morphological brain abnormality, current substance abuse, anosmia, rhinal disorders, migraine preventive medication and significant negative affect as evaluated by the General Health Questionnaire (GHQ-28). None of the subjects took analgesic or anti-migraine drugs during the three days before the PET study. Control subjects did not suffer from any type of headache or from OHS. In women of childbearing age (8 controls and 8 patients), the PET study was performed during the first 10 days of their cycle, ensuring a comparable sex-hormone status in these women. Two control subjects and two patients were menopausal. All subjects provided written informed consent, and the procedure was approved by the local ethical committee (CCP, Centre Léon Bérard, Lyon, France).
Morphological brain imaging
All subjects underwent a brain MRI the same week of the PET study, except for two patients who only had a computed tomography (CT) scan. No abnormality was detected in any subject.
Experimental procedure
The [18F]MPPF-PET study reported here is the second part of a larger PET investigation of olfactory hypersensitivity in migraineurs. The first part of the procedure consisted in a [15O]H2O PET study investigating whether olfactory stimulation produced different activation patterns in migraineurs with OHS as compared to control subjects (20). The [18F]MPPF-PET study started 20 minutes after the last olfactory stimulation and 10 minutes after the end of [15O]H2O PET data acquisition. During the procedure, four patients developed a typical migraine attack without aura. In one patient, the attack started during the [15O]H2O PET study, while in three patients, the attack developed between the two PET studies. In all four patients, the migraine progressively worsened, and lasted until the end of the [18F]MPPF PET data acquisition. None of the other six patients developed a migraine attack during the study.
[18F]MPPF-PET data acquisition
2′-Methoxyphenyl-(N-2′-pyridinyl)-p-(18)F-fluoro-benzamidoethylpiperazine [18F]MPPF is a specific antagonist of 5-HT1A receptors, which affinity allows its displacement by endogenous 5-HT (21–23). Its synthesis has been previously described (24) and provided a specific activity of 32–76 GBq/µmol. The PET sessions were performed during the afternoon on a CTI-SIEMENS HR+ (Siemens, Knoxville, TN, USA) camera. A 10-minute transmission scan was first performed using three 68Ge rod sources. After intravenous injection of a bolus of 186 ± 30 MBq, frames of varying duration (20 s to 5 min) were acquired in 3D mode over a 60-minute period postinjection, with a field of view covering the entire brain and brainstem.
PET data preprocessing
PET images were corrected for scatter and attenuation and reconstructed using a filtered back projection Hanning filter with a cut-off of 0.5 cycles/pixel, providing a volume of 63 slices (2.42 mm thick), with 128 × 128 voxels in plane (2.06 × 2.06 mm). Binding potential (BPND) was calculated using the simplified reference tissue model (SRTM) (25) previously validated for [18F]MPPF in our laboratory (26). This model uses the cerebellum white matter as a reference region devoid of 5-HT1A receptor, and provides BPND, k2 and R1 = K1ref/K1ROI parametric images. These images were then spatially normalized with SPM2 into the space defined by the International Consortium for Brain Mapping (27) on a home-made [18F]MPPF template (26), and smoothed with an isotropic 3D-Gaussian filter of 8 mm full-width at half-maximum.
Analysis of PET data
All SPM analyses were carried out on the normalized smoothed parametric BPND images using an analysis of variance (ANOVA) and a “no-global normalization” procedure. Statistical parametric mapping (SPM) of the t statistic was first plotted with a threshold of p < .001 uncorrected at the voxel level, whereas only voxels or clusters that showed a p < .05 after correction for multiple comparisons, based on the Gaussian field theory (Family Wise Error, FWE, SPM2) (28), were considered as significant.
SPM analysis of the whole brain and brainstem
All these analyses were performed with an explicit mask that includes the entire brain and brainstem and an implicit mask that ignores zeros. We first compared the entire group of OHS migraineurs with the controls. We then split the group of migraineurs into two subgroups of patients, with and without a migraine attack during the PET study, and compared each of these two subgroups with each other and with the controls. This analysis took into account the fact that patients who suffered from a migraine attack during the PET study were younger (mean age: 27.3) than the headache-free migraineurs (mean age of 42.1) and the controls (39.7), a putative bias that could affect our conclusions because [18F]MPPF-BPND varies with age (26). To address this issue, we first included the three groups of subjects in the same analysis with age considered as a covariate of no-interest. We then select a subgroup of six control subjects whose mean age was comparable to that of the four patients who suffered from a migraine attack during the PET study (28 vs. 27.3 years old), and compared these two populations. We further tested the potential impact of age by performing a SPM analysis looking at the correlation between age and [18F]MPPF-BPND.
SPM and region of interest analyses of the raphe nuclei
The key role played by the raphe nuclei in the regulation of the central serotoninergic system provided a strong rationale for specifically exploring this structure in our study. However, the above analyses proved unsuitable for the detection of a significant cluster in the raphe due to its small size (∼190 voxels) as compared to the k threshold value associated with a significant p corrected cluster for our sample size (∼250 voxels in our analyses). To further evaluate [18F]MPPF binding in the raphe nuclei, we reprocessed the analyses with the same design (four patients with a migraine attack, six headache-free migraineurs and 10 controls) using an explicit mask of the brainstem. The contrasts “migraineurs with an attack versus headache-free migraineurs” and “migraineurs with an attack versus controls” were evaluated. In addition, we performed an individual analysis of each patient with a migraine attack during the PET study, as compared to (i) the ten normal subjects with age as a covariate of no-interest and (ii) the six age-matched controls. We finally reprocessed the correlation between age and [18F]MPPF-BPND using the brainstem mask.
We also performed a region of intereset (ROI)-based analysis of the raphe. The raphe nuclei cannot be anatomically delineated on MRI but represent the only detectable brainstem structure on [18F]MPPF PET images. We therefore traced a ROI encompassing this area on an average [18F]MPPF-PET image derived from an independent and previously published dataset of 50 controls (24), by setting the threshold of activity at 90% of the maximum localized in the brainstem. This ROI extended over 13 consecutive slices with a total volume of 1400 mm3. This approach has some limitations: (i) not all brainstem serotoninergic nuclei express levels of 5-HT1A autoreceptors detectable with [18F]MPPF PET, so that our ROI does not reflect the entire set of these nuclei; (ii) both partial volume and spillover effects alter the precise delineation of the raphe borders; (iii) the close proximity of the different raphe nuclei makes it difficult to precisely determine the borders between them. Overall, one must keep in mind that our raphe ROIs remains an approximation of the serotoninergic brainstem structures and that it is not possible to precisely ascribe our findings within this ROI to specific nuclei. Due to these limitations, we decided to subdivide the ROI into a superior mesencephalic raphe and a pontine raphe by a line running parallel to the junction between the midbrain and the pons without further specifying the underlying nuclei.
We compared the BPND values of the raphe nuclei of the two subgroups of patients and controls using nonparametric Kruskal-Wallis and exact tests. As for the SPM analyses, we also compared the four patients experiencing a migraine attack during the PET study and the six age-matched controls using nonparametric Mann-Whitney and exact tests. All these statistical analyses were performed using SPSS (version 12.0 for Windows; SPSS, Chicago, IL, USA) with a level of significance at p < .05 (bilateral for the Kruskal-Wallis test and unilateral for the Mann-Whitney, provided that this latter post-hoc test was based on a unidirectional hypothesis).
Results
Clinical findings
Mean chemical odor intolerance index ± SD was 16.5 ± 3.2 in migraineurs and 9 ± 3.5 in controls with no overlap between the two populations. Only one patient occasionally suffered from migraine with aura, though the majority of her attacks were without aura. None of the patients reported a clear-cut side-predominance of their migraine attacks. Mean duration of migraine was 18 ± 8 years. Mean migraine attack frequency per month was three (2–6).
During the PET study, four of the 10 migraine patients suffered from a typical migraine attack without aura (three on the right side, one on the left side).
PET findings
SPM analysis of the whole brain
The entire group of migraineurs did not show significant differences in [18F]MPPF BPND when compared to controls. In contrast, the four patients experiencing a migraine attack during the PET study showed significantly increased [18F]MPPF BPND in the left orbitofrontal cortex and precentral gyrus, with a similar trend in the left insula and second somatosensory (SII) area, when compared to headache-free migraineurs (Table 1, Figure 1). The comparison between the four patients with a migraine attack during the PET study and the 10 controls did not disclose a significant cluster, but detected a voxel in the pontine region associated with a Z-score of 5.1 and a significant p value after correction for multiple comparisons at the voxel level (p
FWE corrected = 0.014). Furthermore, when the same four patients were compared to the six age-matched controls, a significant cluster of increased [18F]MPPF BPND was found in the left temporal pole. Finally, we observed a significant negative correlation between age and [18F]MPPFBPND restricted to the mesial superior frontal cortex, that spared all the regions where differences were observed between groups.
SPM analysis contrasting the four patients with a migraine attack during the PET study with the six headache-free migraineurs. Statistical analysis discloses two significant clusters of increased BPND in patients with a migraine attack as compared to the headache-free migraineurs, located in (A) the left precentral gyrus (−48, 2, 24) and (B) the left orbitofrontal cortex (−16, 16, −24). A nonsignificant trend toward greater [18F]MPPF BPND is also observed in (C) the left insula (−38, −28, 12) in patients with a migraine attack as compared to headache-free migraineurs. SPM analyses between each migraine subgroup (4 migraineurs with an attack and 6 migraineurs without) and each group of control subjects (10 control subjects and the subset of 6 age-matched controls) L = left. R = right. The cluster observed in the right parahippocampal gyrus is considered as an edge effect. This p value was found for one voxel. Values between brackets indicate the number of subjects.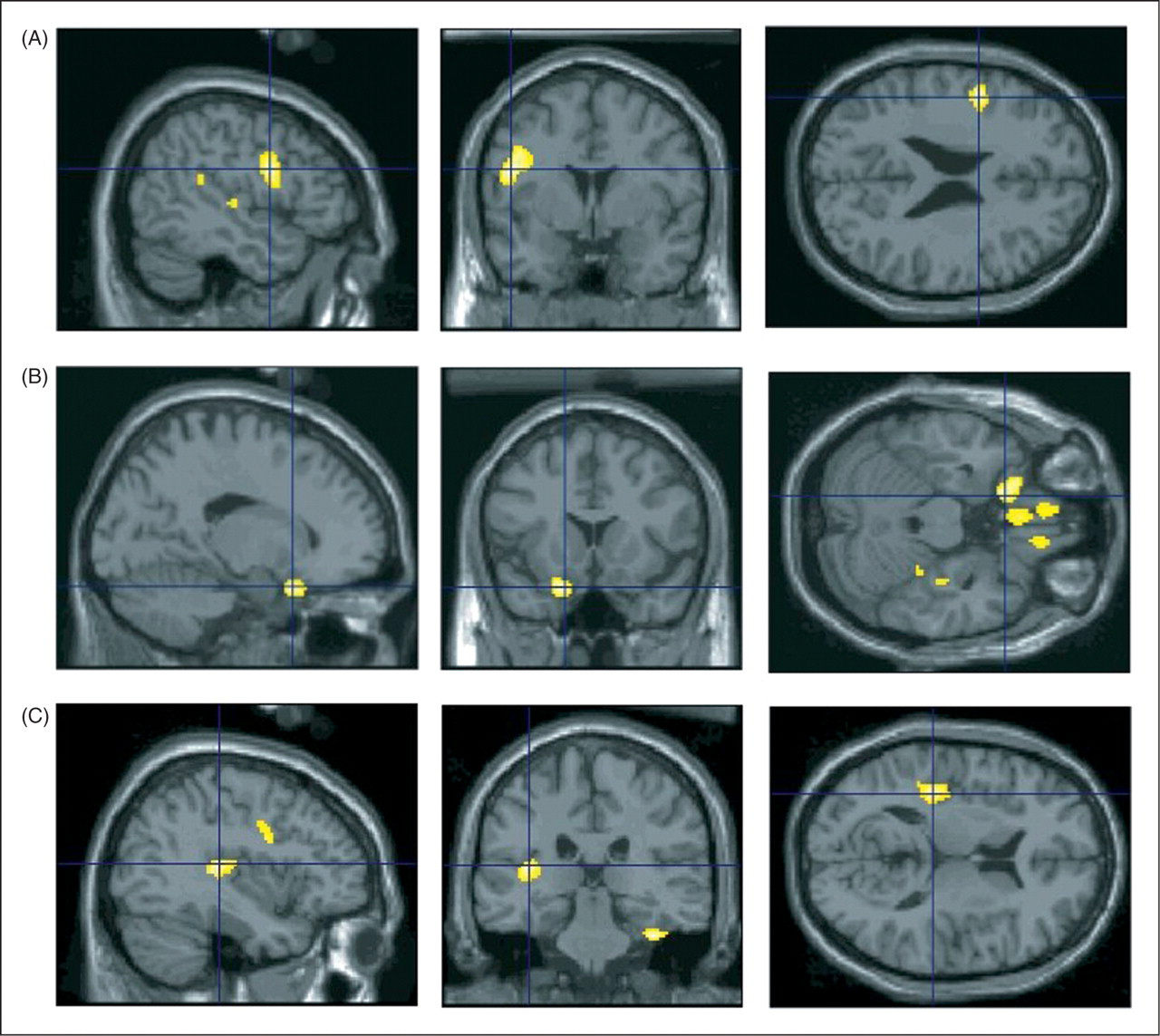

SPM and ROI analyses of the brainstem
When using an explicit mask of the brainstem, patients who suffered a migraine attack during the study showed significant clusters of increased [18F]MPPF BPND in the pons when compared to headache-free migraineurs, the entire group of 10 control subjects, or the subgroup of six age-matched controls (Table 1; Figure 2). One of these clusters overlapped with the pontine portion of the raphe ROI, whereas the other was located immediately anterior to them.
SPM analysis with a brainstem mask contrasting the four patients with a migraine attack during the PET study with the six age-matched controls. (A) Group analysis shows significant increased [18F]MPPF BPND in three clusters (6, −20, −24; p corrected < .001; Z-score = 4.42), (4, −26, −42; p corrected < .001; Z-score = 3.82) and (10, −32, −34; p corrected = .03; Z-score = 3.53). (B) Individual analyses show significant increased [18F]MPPF BPND in the pons of each of the four patients at the following location #1 (4, −14, −32), #2 (4, −16, −24), #3 (6, −16, −34 and 2, −26, 46) and #4 (10, −22, −30).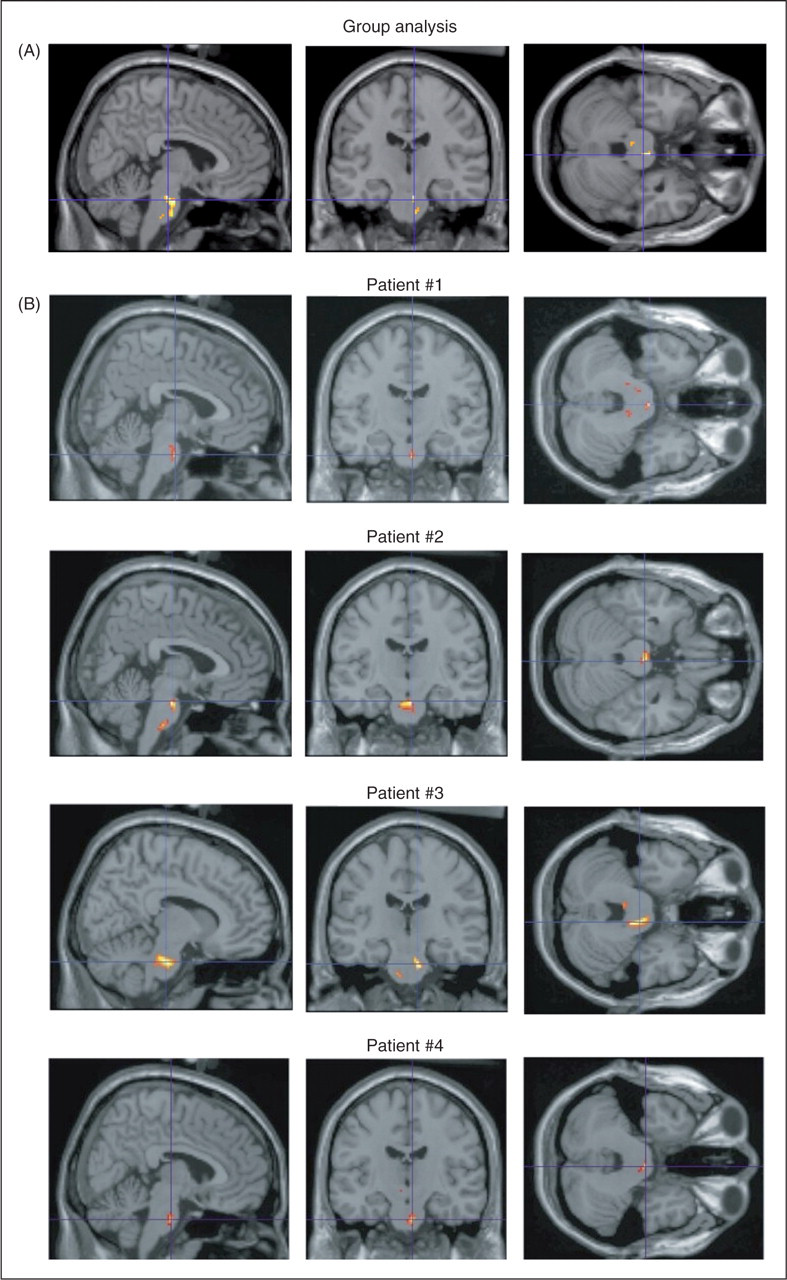
SPM analyses in the pons between each of the four migraine patients with a migraine attack and the six age-matched controls

Nonparametric analyses of the raphe nuclei between each group of migraineurs (with and without a migraine attack) and each group of control subjects (10 control subjects and 6 age-matched controls)
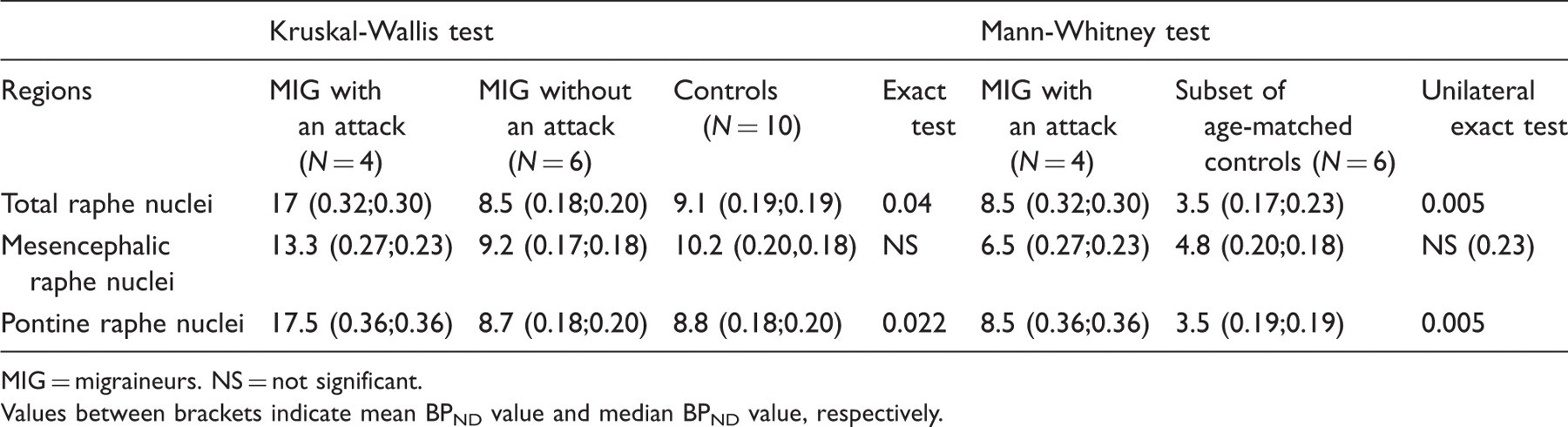
MIG = migraineurs. NS = not significant.
Values between brackets indicate mean BPND value and median BPND value, respectively.
Discussion
This study shows that 5-HT1A receptor availability is modified in migraineurs during their attacks. A [18F]MPPFBPND increase was observed in the brainstem, particularly in the pontine region in migraineurs with a migraine attack during the PET study when compared to controls and to headache-free migraineurs. Moreover, when compared to headache-free migraineurs, these patients exhibitedBPND increases located in the left frontal central gyrus and orbitofrontal cortex.
Methodological issues
An important issue that needs to be discussed concerns the age difference between the subgroups of patients and the controls. Indeed, a prior study performed in our laboratory demonstrated a close relationship between [18F]MPPF BPND and age (26). In the present study, our 10 patients and control subjects were selected to be age-matched (mean age: 36.2 and 39.7, respectively), but we could not control the mean age of the subset of patients who developed a migraine attack during the PET study. However, our statistical analyses strongly suggest that our main findings were not confounded by age. First, age was considered as a covariate of no-interest in all analyses, except those with age-matched subgroups. Second, the comparison between the patients experiencing a migraine attack and the controls could be reprocessed by selecting a subset of age-matched control subjects (mean age of 27.3 and 28 years, respectively). Finally, we did not observe any significant correlation between [18F]MPPF BPND and age in the raphe nuclei, the central gyrus or the orbitofrontal cortex, regions where we found significant differences between groups in the SPM analyses.
Another related issue is the relatively small number of subjects in each of the subgroups considered. It must be stressed, however, that our statistical analyses take this into account both by using nonparametric tests for the ROI analysis of the raphe nuclei, and by using stringent criteria of significance for SPM analyses, using correction for multiple comparisons at the cluster level. Despite these strict criteria, the majority of the differences observed between groups were associated with highly significant p values. We are thus confident that our findings are sound despite the small number of subjects.
Involvement of the pontine raphe nuclei during a migraine attack
The raphe nuclei, interconnected with the periaqueductal gray (PAG) and the locus coeruleus in the brainstem, play an important role in the endogenous control of pain. The serotoninergic neuron clusters are organized into eight nuclei located at the midline of the brainstem. They have been allocated into two groups: the rostral group, confined to the mesencephalon and rostral pons (dorsal raphe, median raphe nuclei and caudal linear) and the caudal group, extending from the caudal pons to the caudal portion of the medulla oblongata (raphe magnus, raphe obscurus, raphe pallidus nuclei and parts of the adjacent lateral reticular formation) (29). The central modulation of noxious stimuli particularly involves the dorsal raphe and raphe magnus nuclei, interconnected by a descending projection (29). Regarding migraine pain, animal experiments studying the consequences of a noxious stimulation of the superior sagittal sinus suggest that pain modulation resulting from concomitant stimulation of the PAG is mediated by the raphe magnus (30–32). A recent study shows that the neuronal activity of the raphe magnus is altered by migraine triggering factors such as repetitive flash light, while stimulation of this nucleus suppresses responses of trigeminal neurons to noxious stimulation of the dura mater (33). In humans, a dysfunction of the pontine raphe has been suggested in a migraineur who presented right-sided attacks associated with a pontine cavernoma affecting the left raphe magnus (34). Although previous [15O] H2O PET studies performed during spontaneous or triggered migraine attacks consistently showed an increase of the regional blood flow in median/paramedian regions of the brainstem (6–10), the spatial resolution did not enable to identify which specific structures were involved. The present findings reveal an involvement of the pontine raphe at a very early stage of odor-triggered migraine attacks. Due to the lack of resolution of PET images, we are not able to precisely identify which specific nuclei of the pontine raphe were involved during migraine attacks. Similarly, it remains difficult to draw a firm conclusion on the anatomical correspondence of one of our two pontine SPM clusters located immediately anterior to the raphe nuclei depicted on [18F]MPPF PET images. Nevertheless, both SPM and ROI analyses confirmed the presence of migraine-related changes within the pontine raphe proper. Furthermore, the lack of detectable change in the mesencephalon argues against the involvement of the dorsal raphe nucleus, which extends from the level of the oculomotor nucleus to the middle of the pons (29). This negative finding might just reflect a lack of sensitivity of our study, but also points to the possibility that this major serotoninergic nucleus is less involved than those strictly located in the pons in the pathophysiology of migraine. We hypothesize that a transient dysfunction of the pontine raphe nuclei, including the raphe magnus, due to olfactory stimulation in our patients, may lead to disinhibition of the trigeminal nociceptive system in migraineurs and thus provoke a migraine attack.
The increase of [18F]MPPF BPND observed in the pons in our patients could also reflect a nonspecific increase of perfusion during the migraine attack. However, our methodological model provides independent parametric images of BPND and R1 (the ratio between perfusion level at any voxel of interest and mean perfusion level in the cerebellar reference region), for which post-hoc analysis did not disclose any significant difference between patients with a migraine attack and the other groups of subjects. Moreover, SPM and ROI showed an increase of [18F]MPPF BPND only in the raphe nuclei located at the level the pons, while previous H2O-PET studies reported larger migraine-induced brainstem activation (6–10). Hence, it is unlikely that the increase of [18F]MPPF BPND observed in the pons in our study primarily reflected an increase of perfusion in the brainstem concomitant with a migraine attack.
5-HT1A receptors binding in the brainstem during a migraine attack
The increase of [18F]MPPF BPND observed in the pontine raphe nuclei emphasizes the involvement of 5-HT1A receptors during migraine attacks, but remains of complex interpretation. [18F]MPPF and 5-HT demonstrate comparable affinities for 5-HT1A receptors, allowing for competition between the two molecules whereby a decrease in extracellular concentration of endogenous 5-HT will lead to an increased [18F]MPPF binding (22,23). Accordingly, a combined alpha-[11C]methyl-L-tryptophan (AMT) and [18F]MPPF PET study performed in healthy subjects showed a negative correlation between serotonin synthesis and 5-HT1A receptors binding potential (35). Based on these observations, an increase in [18F]MPPF BPND can reflect either an up-regulation of 5-HT1A receptors, a decrease in apparent Kd (increased affinity), a decreased extracellular concentration of endogenous 5-HT, or any combination of the above. Although our data do not allow to separate these different possibilities, several experimental and clinical arguments favour the hypothesis of a decreased extracellular concentration of endogenous 5-HT.
The reduction of brain 5-HT synthesis using tryptophan depletion intensifies the development of migrainous symptoms such as headache, photophobia and nausea (36,37). In a rat model, 5-HT depletion induced by administration of parachlorophenylalanine, a tryptophan hydroxylase inhibitor, enhances cortical spreading depression (CSD) and the trigeminal response to CSD (38).
Furthermore, the decreased extracellular concentration of endogenous 5-HT suspected in our patients might be associated with an upregulation of somatodendritic 5-HT1A autoreceptors, which play a key role in the regulation of central serotoninergic tone (39). The increased binding potential of 5-HT1A receptors observed in the pontine raphe nuclei could also reflect changes in Kd (radioligand equilibrium dissociation constant) (40). Indeed, the involvement of the pontine raphe nuclei per se and/or the neurotransmitter release at the onset of a migraine attack could influence receptor-ligand interaction. Overall, we believe that the increased [18F]MPPF BPND observed in the brainstem of patients with OHS scanned during a migraine attack is likely to reflect a combination of decreased extracellular concentration of 5-HT and upregulated 5-HT1A receptors at the very early stage of the attack.
This view remains difficult to reconcile with the increased brain 5-HT synthesis previously reported during migraine attacks in a [11C]AMT PET study (11). These seemingly discordant results might reflect differences in the precise timing of the PET study with regards to the development of the migraine attack. The increased brain 5-HT synthesis was observed at a stage of the migraine attack (three to six hours after the onset) which is later than that of the changes in [18F]MPPF binding we observed during the first hour of the migraine.
5-HT1A receptor binding in the cortex during a migraine attack
Migraineurs experiencing an attack after olfactory stimulation also showed increased [18F]MPPF BPND in various cortical areas. This was observed in the left temporal pole when compared to age-matched controls, as well as in the left precentral gyrus, orbitofrontal cortex and possibly the left posterior insula when compared to triggered headache-free migraineurs, all of which represent targets of serotoninergic projections from the raphe nuclei (41,42) involved in pain and/or olfactory processing.
Interestingly, the [15O]H2O PET study performed on the same subjects immediately prior to [18F]MPPF injection showed in migraineurs left temporal hyperperfusion at baseline and in response to odors. This hyperperfusion primarily affected the left piriform cortex, the anterosuperior temporal gyrus and the left temporal pole (20). The involvement of the superior temporal gyrus during a migraine attack has been reported in previous PET studies (6–9), as well as during migraine-associated visual aura explored by magnetoencephalography (43). The left temporal pole was on the other hand found to be involved in olfactory emotional processing (44) and aversive olfactory learning (45). An involvement of the posterior insula has been observed in nearly all functional pain imaging studies (46), including migraine attacks (6–10), and is thought to be related to the sensory-discriminative aspects of pain processing (47). The orbitofrontal cortex also appears to be involved during pain and olfactory processing (48,49). Finally, chronic electrical stimulation of the precentral cortex has demonstrated antinociceptive effect (50). We hypothesize that the increase of cortical [18F]MPPF BPND observed in patients with odor-triggered migraine is related to both unspecific pain/olfactory processing and specific mechanisms of migraine attack development.
Conclusion
In conclusion, the present [18F]MPPF PET study showed that 5-HT1A receptor availability is increased in the pontine raphe of migraineurs early during odor-triggered migraine attacks, emphasizing the role of these nuclei in the development migraine. Whether these findings are specific to migraineurs with OHS or could be extended to the general population of migraineurs would merit further study.
Footnotes
Acknowledgements
We wish to thank the control subjects and patients who participated in this experiment, the members of Centre d’Explorations et de Recherches Médicales par Emission de Positons (CERMEP) for their valuable assistance, and are greatly indebted to Dr P. Giraud and Dr A. Revol for assistance in recruiting patients. We are grateful to perfume and/or aroma companies (Givaudan, Lenoir, Perlarom) for supplying the odorants used in this study. This work was supported by the Hospices Civils de Lyon and the Programme Hospitalier de Recherche Clinique (PHRC 2004 of the Rhône-Alpes region).
