Abstract
Olfactory hypersensitivity (OHS) may occur during migraine attacks and seems to be very specific to this form of headache. OHS is also observed during migraine-free periods and is associated with the presence of odour-triggered attacks. Yet the pathophysiology of OHS remains unknown. The aim of our study was to evaluate olfactory processing in migraineurs with OHS and to investigate whether regional cerebral blood flow (rCBF) associated with olfactory stimulation is modified in these patients compared with controls. Eleven migraineurs with OHS and 12 controls participated in a H215O-positron emission tomography study, including three scans in which odours were delivered and three scans where only odourless air was delivered. rCBF during olfactory condition was compared with that for the odourless baseline condition. Between-group analyses were performed using voxel-based and region-of-interest analyses. During both olfactory and non-olfactory conditions, we observed higher rCBF in the left piriform cortex and antero-superior temporal gyrus in migraineurs compared with controls. During odour stimulation, migraineurs also showed significantly higher activation than controls in the left temporal pole and significantly lower activation in the frontal (left inferior as well as left and right middle frontal gyri) and temporo-parietal (left and right angular, and right posterior superior temporal gyri) regions, posterior cingulate gyrus and right locus coeruleus. These results could reflect a particular role of both the piriform cortex and antero-superior temporal gyrus in OHS and odour-triggered migraine. Whether these rCBF changes are the cause or a consequence of odour-triggered migraines and interictal OHS remains unknown. Further comparisons between migraineurs with and without OHS are warranted to address this issue. The abnormal cerebral activation patterns during olfactory stimulation might reflect altered cerebrovascular response to olfactory stimulation due to the migraine disease, or an abnormal top-down regulation process related to OHS.
Introduction
Olfaction has been poorly explored in migraineurs, even though olfactory hypersensitivity (OHS) may occur during migraine attacks and has been found to be very specific to this form of headache (1, 2). OHS is also observed between migraine attacks (3) and has recently been significantly associated with the presence of odour-triggered migraines, migraine attack frequency and an altered hedonicity judgement of odours (4).
The pathophysiology of interictal olfactory disturbances in migraineurs remains unknown. Studies on olfactory thresholds reported in migraineurs show seemingly discordant results. Whereas one study has reported a decreased threshold for vanillin, suggestive of interictal hyperacuity to odours (5), others have shown an increased detection threshold for pyridine (6) or unmodified thresholds for acetone (5). Nevertheless, in studies on migraine, the term OHS does not necessarily refer to a decreased olfactory detection threshold and is rather used in a broader sense, reflecting the discomfort perceived by the patient (4, 7). Interictal OHS may thus reflect an alteration of brain excitability, a hypothesis based on studies of visual, auditory and somatosensory evoked potentials, and findings from magnetic resonance spectroscopy and magneto-encephalography (8–12). In olfaction, smaller event-related potential amplitudes have been reported in migraineurs, suggestive of a dysfunction in central olfactory processing (13).
Knowledge of olfactory neural networks has recently been increasing through the use of cerebral imaging (14–18). For example, it has been shown that perceptual processes such as intensity and hedonicity judgments are associated with an involvement of specific neural networks (19–22). A recent positron emission tomography (PET) study of patients suffering from multiple chemical sensitivity, a syndrome that encompasses OHS, has reported increased olfactory-triggered activation of the anterior cingulate and cuneus (23). In the field of migraine, functional imaging studies during spontaneous and triggered migraine attacks have revealed activation in areas related to pain processing, such as the insula, thalamus and cingulate cortex, but also activation of the brainstem (24–30). To our knowledge, no imaging study has specifically explored interictal OHS in migraineurs. The aim of our study was to investigate whether activation patterns associated with olfactory stimulation are altered in migraineurs suffering from OHS during migraine-free intervals when compared with controls.
Material and methods
Subjects
Eleven female migraineurs (nine right-handers, two left-handers, age 24–53 years, mean 35 years) and 12 matched healthy female controls (10 right-handers and two left-handers, age 24–60 years, mean 38 years) were included in the study. Patients had to fulfil International Headache Society criteria for episodic migraine (Headache Classification Committee of The International Headache Society, 2004), to suffer from odour-triggered attacks and interictal OHS. The presence of interictal OHS was determined by asking patients whether they regarded themselves as clearly hypersensitive to odours between attacks. Only one patient suffered from migraine with aura, though the majority of her attacks were without aura. None of the patients reported a clear-cut side-predominance of migraine attacks and none was taking migraine preventive medication. Other medications included oral contraceptives in eight patients and thyroxin in one. Controls suffered from neither any type of headache, nor OHS. Exclusion criteria for all subjects (migraineurs and controls) included chronic daily headache with or without drug overuse, possible brain damage, current substance abuse, known anosmia and rhinal disorders (cold, active allergies, history of nasal-sinus surgery). Ten of the 12 control subjects and nine of the 11 patients were women of childbearing age for whom the PET study was necessarily performed during the first 10 days of the cycle to exclude the risk of pregnancy and fetal exposure to radiation. In addition, all women underwent a pregnancy test before PET imaging. The remaining control subjects (n = 2) and patients (n = 2) were menopauses. All subjects provided written informed consent, and the procedure was approved by the local ethics committee (CCP, Centre Léon Bérard, Lyon) in accordance with the Declaration of Helsinki and local laws.
Quantitative evaluation of OHS
OHS was further evaluated in patients and controls using a chemical odour intolerance index previously validated in patients with multiple chemical sensitivity (31). This index is a five-item visual rating scale used to judge intolerance to five types of odorants: pesticide, paint, perfume, new carpet and car exhaust. Each scale ranges from 1 (almost never experiencing symptoms) to 5 (almost always). A global value is obtained by summing the values of the five-item rating scales, ranging from 5 (no odour intolerance) to 25 (maximal odour intolerance).
Odorous stimuli
Sixty odorants were used during the PET study. They were divided into three sets of 20 odorants each (Table 1). Each set was composed of odorants with different intensity, hedonicity and familiarity scores as evaluated by normal subjects in a previous study (32). No significant difference was found between the three sets for intensity (F2,38 = 0.963, NS), hedonicity (F2,38 = 1.40, NS) or familiarity (F2,38 = 2.54, NS). In each set, the presentation order of the stimuli was pseudorandomized but identical for all subjects. Odorants were diluted to a concentration of 10% using mineral oil (Sigma Aldrich, Lyon, France), except those with very high potency that were diluted down to 1%. Five millilitres of this solution was absorbed into compressed polypropylene filaments inside 100-ml white polyethylene squeeze bottles with a dropper (Fisher Scientific, Illkirch, France).
Lists of odorants (sets 1, 2 and 3) used for the three positron emission tomography scans in the O condition and respective means (±
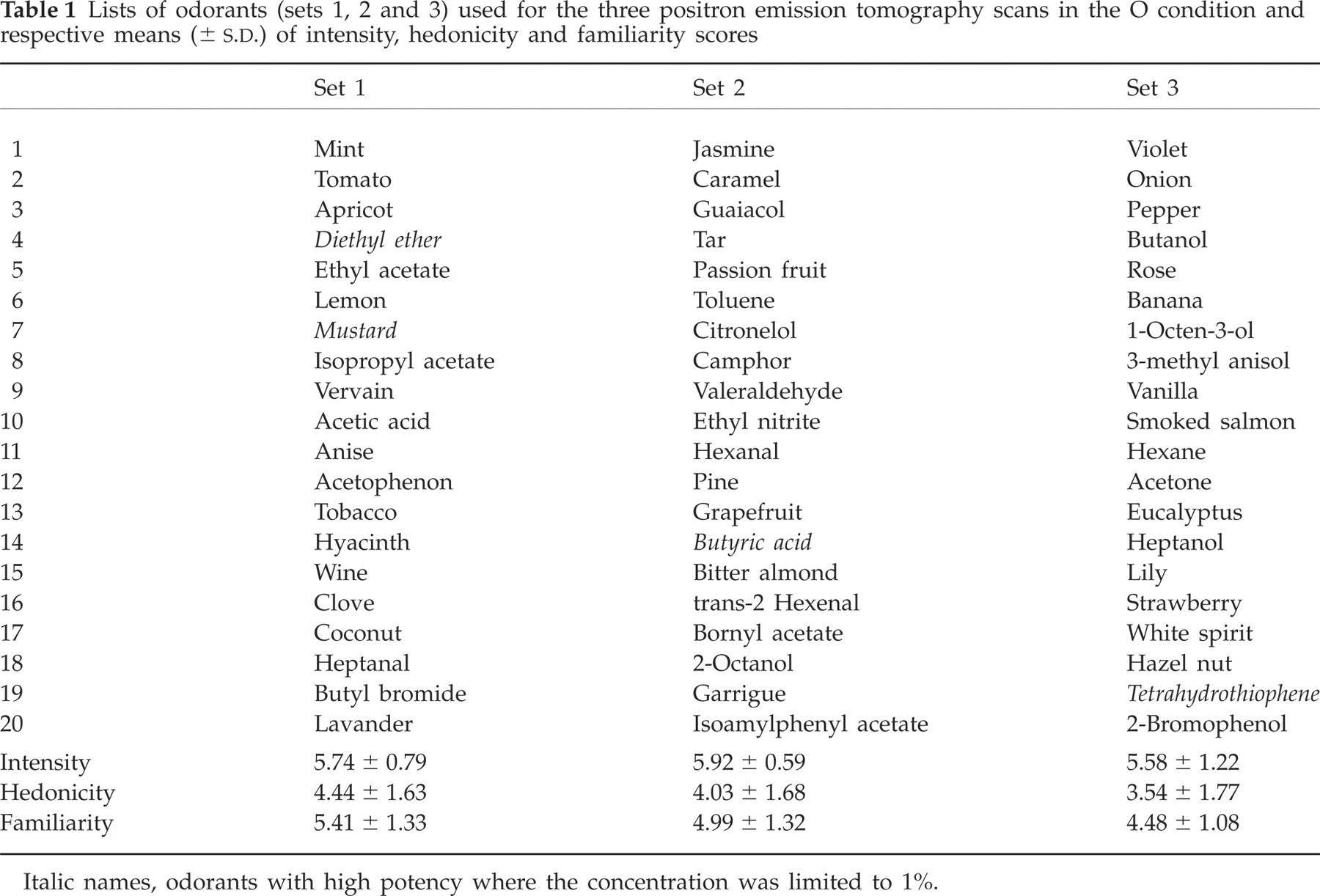
Italic names, odorants with high potency where the concentration was limited to 1%.
Stimulating material
Odours were presented using an airflow olfactometer, which allowed the stimulation to be synchronized with breathing (33). Briefly, vector air was pumped with a compressor and treated with a charcoal filter before flowing into an air-dilution olfactometer. At the end of the expiration, one stimulus was injected into the olfactometer by squeezing the bottle into the injection head, so that the odour (or odourless air) was carried to the subject through a standard mask (Respiron; Europe Medical, Bourg en Bresse, France). Breathing was recorded with a thermal probe close to the right nostril, and controlled by stereo headphones. Subjects were stimulated during each breathing cycle. For a mean respiratory cycle of about 3–4 s, about 15 stimulations were performed per scan.
Experimental procedure
During scanning, each subject underwent a total of six PET scans, alternatively including three scans in which odours were delivered and three in which odourless pure air was delivered. A scan was performed approximately every 10 min and lasted 1 min. During scans with odours, the three sets of odorants were used, on the basis of one set per scan. The order of sets of odorants was different between participants and followed a square Latin design. To avoid surprising the subjects and to optimize data acquisition, the stimulations started 20 s before the beginning of the scan. In patients, the PET study was always performed at least 72 h after the last migraine attack.
General instructions were provided at the beginning of the experiment. Subjects knew that half of the scans were associated with odour stimuli and the other half without. They were asked to breathe similarly for both conditions. During olfactory conditions, the subjects were asked to concentrate only on the odours. Scans were performed while the subjects kept their eyes closed and the room lighting was dimmed. Prior to scanning, subjects underwent a training session in which they were trained to breathe regularly without sniffing, and to smell odours during inspiration. Before and after each scan, patients were asked to report any headache, to rate the intensity and to describe the nature and location of the headache.
Data acquisition H2 15O
Subjects were imaged in a whole-body tomography (Siemens Exact HR+) scanner in 3-D mode, with a transaxial resolution of 4.5 mm (full width half maximum). Sixty-three slices of 2.43 mm thickness with an axial field of view of 15.2 cm were acquired during each scan. Subjects were positioned using a gauze band wound around the head to reduce head movement. A 10-min transmission scan was first conducted to correct the effects of radiation self-attenuation, using an external positron-emitting isotope (68Ge). An intravenous bolus injection of 333 MBq H2 15O was injected into the antecubital vein of the left arm at the beginning of each scan. The acquisition of data began when the radioactive counts exceeded the background activity by 200% and lasted 60 s. The images were attenuation-corrected and reconstructed with filtered back-projection using a Hanning filter.
Image analysis
Statistical Parametric Mapping (SPM2) (Wellcome Department of Cognitive Neurology, London, UK) was used for image processing and analysis. The steps included interscan realignment, spatial normalization (default 7 × 8 × 7 basis function, 12 non-linear iterations) to stereotactic space defined by the Montreal Neurological Institute's (MNI) reference brain, and smoothing of the images using a 10-mm isotropic Gaussian kernel. The localization of activation clusters was given with reference to the MNI magnetic resonance imaging template and the anatomical atlases produced by Mai et al. (34) and Duvernoy (35). Random-effect analyses were performed on the whole brain using the ‘single-subject: conditions and covariates’ PET model to average experimental conditions into a single contrast image. One contrast was performed by comparing the brain's response in the olfactory condition with that obtained in the odourless condition [odour vs. no odour (O vs. NO)]. In a second step, contrast images were then analysed in separate ‘basic model’ one-way analysis of variance (
The random-effect analysis described above compared activation patterns from a double contrast (O vs. NO and migraineurs vs. controls). The analysis could thus miss between-group differences if the amplitude of activation differences between olfactory conditions were similar for both groups. In particular, we hypothesized that migraine patients with OHS might demonstrate abnormal baseline regional cerebral blood flow (rCBF) in brain regions known or suspected to be involved in olfaction (19, 21, 36) or migraine attacks (30). Previous H2
15O-PET studies have demonstrated that patient populations, such as those with schizophrenia, can disclose abnormal rCBF in baseline condition compared with normal subjects (37). However, because such abnormalities cannot be quantitatively measured as absolute blood flow changes, they are referred as ‘relative’ hyper- or hypoperfusion. Regions of interest (ROIs) involved in olfaction are the piriform cortex (PC), the amygdala, the left anterior ventral insula (−36, 18, 0) and the right/left orbitofrontal cortex (±30, 30, −15). ROIs reported to be activated during migraine attacks are the anterior (4, 30, 19) and posterior (0, −20, 38 and 0, −5, 34) cingulate, the insula (−40, 14, 3), the antero-superior temporal gyri (−42, 12, −24 and 34, 18, −18), the thalami (−8, −4, 14 and 10, −6, 8) and the dorsal pons (−4, −28, −20). Since the locus coeruleus is a region of the dorsolateral pons thought to be involved in migraine processing and known to send projections onto the olfactory bulb and PC (38), we also examined its mean activation level on both sides by selecting coordinates (−6, −36, −24 and 6, −36, −24) from a sleep PET study (39). The mean activation levels within these ROIs were extracted for each participant and conditions, using the MarsBar SPM toolbox (http://marsbar.sourceforge.net/).
Results
Clinical findings
The chemical odour intolerance index ranged from 14 to 24 (median 16.5) in migraineurs and was about 9 (range 5–12) in controls. Among the 11 migraineurs, one patient (no. 3) developed a typical migraine attack without aura during the PET study. She was thus excluded from analyses, since haemodynamic changes observed during a migraine attack could interfere with the olfactory activation pattern. Three patients (patients 1, 6 and 7) developed a migraine attack during the hour following the H2O-PET, lateralized on the right side (patients 1 and 7) or on the left side (patient 6). One patient developed an attack the day after the study.
Since one stimulation (odour or odourless air) was performed per breathing cycle, it was possible to compute the breathing frequency from the number of stimulations delivered per scan. Results showed that respiratory frequency was decreased during odour stimulation compared with odourless air (F1,20 = 17.890, P = 0.0004), but no significant difference in breathing patterns was found between either group of subjects (F1,20 = 2.224, P = 0.1515).
PET findings
Significant rCBF differences between migraineurs and controls groups (migraineurs minus controls, and controls minus migraineurs) for the O–NO contrast were extracted and are listed in Table 2.
Brain regions with significant regional cerebral blood flow differences when comparing the contrast images of both groups (‘Migraineurs vs. Controls’ contrasts)
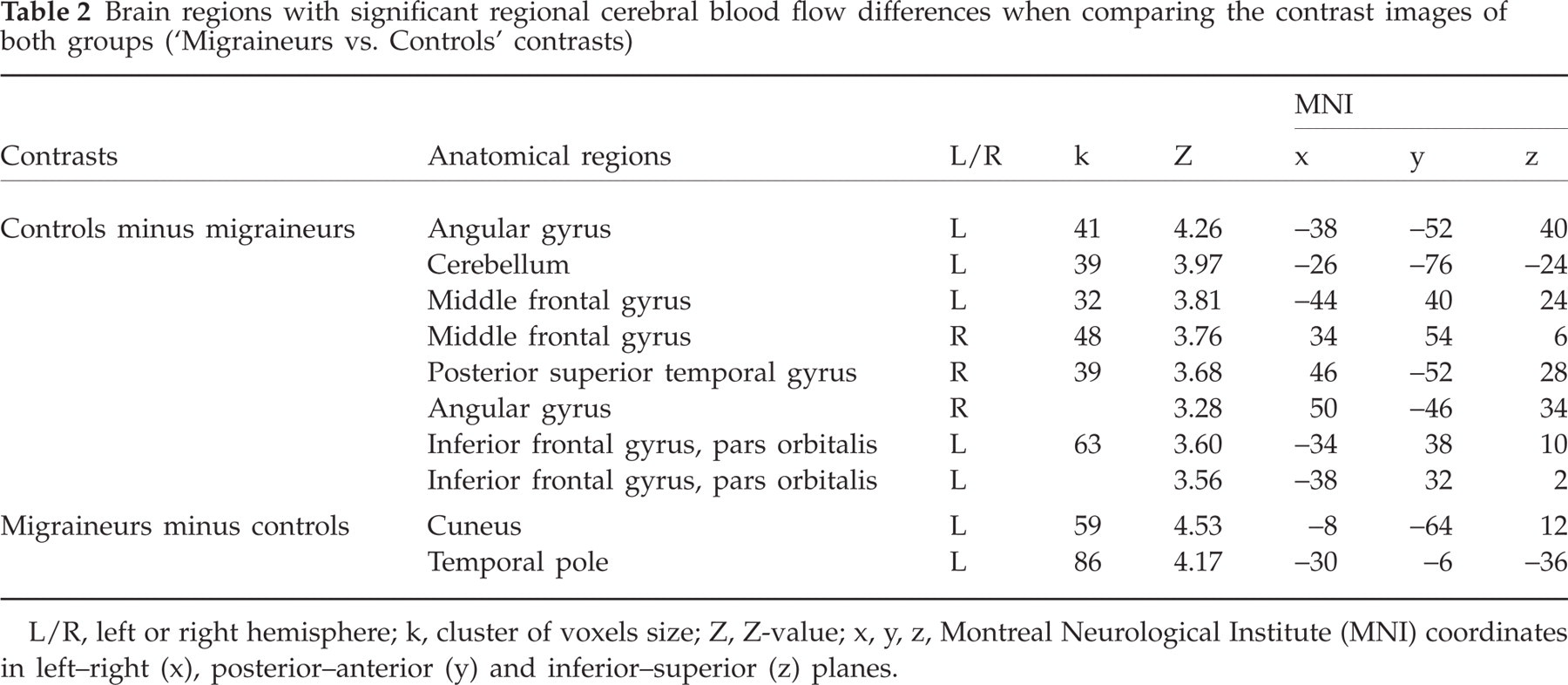
L/R, left or right hemisphere; k, cluster of voxels size; Z, Z-value; x, y, z, Montreal Neurological Institute (MNI) coordinates in left–right (x), posterior–anterior (y) and inferior–superior (z) planes.
Controls minus migraineurs contrast
Significant rCBF differences were observed in the left angular gyrus (−38, −52, 40), right and left middle frontal gyrus (34, 54, 6; −44, 40, 24), right postero-superior temporal gyrus (46, −52, 28), pars orbitalis of the left inferior frontal gyrus (−34, 38, 10) and the cerebellum (−26, −76, −24) (see Fig. 1). Results from statistical analyses performed on cluster activation levels as a function of group and odour conditions are reported in Table 3A.

Regional cerebral blood flow (rCBF) differences found in the odour–no odour (O–NO) contrast between controls (Con) and migraineurs (Mig) in (A) the angular gyrus (−38, −52, 40); (B) the right middle frontal gyrus (34, 54, 6); (C) the right superior temporal gyrus (46, −52, 28 and 50, −46, 34) and (D) the inferior frontal gyrus, pars orbitalis (−34, 38, 10 and −38, 32, 2). The Statistical Parametric Mapping (SPMs) are superimposed on axial sections of a T1-weighted scan (threshold set at P = 0.0001). Graphs: levels of activation in the O and NO conditions in these four areas in Con and Mig inferred from functional regions of interest. Vertical bars, standard errors; ∗P < 0.05; ∗∗P < 0.01; ∗∗∗P < 0.001.
For all analyses, free degrees is 1, 20. ROI, region of interest.
Detailed examination of data showed that the significant group × odour interaction resulted from an rCBF increase in controls and an rCBF decrease in migraineurs in the O condition, when compared with the NO condition, in the angular gyrus (P = 0.0262 and P < 0.0001, respectively), right middle frontal gyrus (P = 0.0048 and P = 0.0040), left middle frontal gyrus (34, 54, 6; P < 0.0001 and P = 0.5430), right posterior supero-temporal gyrus (P = 0.0014 and P = 0.0002) and inferior frontal gyrus pars orbitalis (P = 0.0015 and P = 0.0326). The significant interaction resulted from an rCBF increase in controls in the cerebellum (P < 0.0001) and left middle frontal gyrus (−44, 40, 24; P < 0.0001) without any change in rCBF during O condition in migraineurs.
Migraineurs minus controls contrast
Significant differences were observed in the left cuneus (−8, −64, 12) and temporal pole (−30, −6, −36) (Fig. 2). For the cuneus, the significant group × odour interaction resulted from a deactivation in controls, but not in migraineurs, in the O condition when compared with the NO condition (P < 0.0001). For the left temporal pole, the significant interaction was due to a combination of significant odour-triggered activation in patients (P < 0.0001) and deactivation in controls (P = 0.0436) in the O condition when compared with the NO condition.

Regional cerebral blood flow (rCBF) differences found in the odour–no odour (O–NO) contrast between controls (Con) and migraineurs (Mig) in the temporal pole (−30, −6, −36). See Fig. 1 for details.
ROI analysis
In olfactory ROIs, significant activation was observed during O condition in both migraineurs and controls in the PC and amygdala of both hemispheres, the left anterior ventral insula (−34, 18, 0) and right orbitofrontal cortex (Table 3B). For the left PC, we also found a significant effect of group factor resulting from a higher rCBF in migraineurs than controls for both O and NO conditions (Fig. 3A,C).

Regional cerebral blood flow (rCBF) differences found in the odour–no odour (O–NO) contrast between controls (Con) and migraineurs (Mig) in (A) the left piriform cortex (PC) and (B) the left anterior superior temporal gyrus (−42, 12, −24). (C) High CBF observed in the baseline condition with the Mig–Con contrast. See Fig. 1 for details.
In ROIs shown to be activated during spontaneous migraine attacks (Table 3C), we found a significant effect of group factor in the left antero-superior temporal gyrus (−42, 12, 24) due to higher rCBF in patients than controls in O and NO conditions (Fig. 3B). We also observed a significant effect of odour factor resulting from a higher rCBF in the O than NO condition for both groups in the dorsal pons, right anterior supero-temporal gyrus, left anterior ventral insula, and right and left locus coeruleus. Significant group × odour interactions were found in the posterior cingulate gyrus (0, −45, 34) and in the right locus coeruleus (Fig. 4). These were explained by significant activation in controls during olfactory stimulation when compared with the baseline condition (P = 0.0132 and P = 0.0002, respectively), not observed in migraineurs.

Regional cerebral blood flow (rCBF) differences found in the odour–no odour (O–NO) contrast between controls (Con) and migraineurs (Mig) in the right locus coeruleus (6, −36, −24). See Fig. 1 for details.
Discussion
The aim of this H2 15O-PET study was to investigate possible functional alteration of the olfactory neural networks in migraineurs with interictal OHS. Our results show significant differences in cerebral activation patterns, as well as baseline rCBF differences between migraineurs and controls in both olfactory and non-olfactory brain regions.
The primary olfactory cortex mainly comprises the PC, olfactory tubercle, lateral entorhinal and periamygdaloid cortices, and the anterior cortical amygdaloid nucleus (40). This primary cortex has direct and indirect connections with secondary olfactory areas, i.e. orbitofrontal and insular cortices. A peculiarity of the olfactory system is to present also an extremely rich supply of centrifugal fibres, including projections from the locus coeruleus and raphe nuclei (41, 42). In the present study, most areas pertaining to this olfactory neural network (bilateral PC, amygdala, right orbitofrontal cortex, left anterior ventral insula) were activated in migraineurs and controls during odour stimulation. This result suggests that odours presented at a supraliminar threshold were perceived and corollary neural information processed by both migraineurs and controls. However, several crucial differences were observed between the two groups regarding patterns of activation. During olfactory stimulation, in migraineurs when compared with controls, the absence of activation or alternatively deactivation was observed in frontal regions (left inferior frontal gyrus, left and right middle frontal gyri), parieto-temporal regions (left and right angular gyrus, right postero-superior temporal gyrus), posterior cingulate gyrus and right locus coeruleus, whereas activation was found in the left temporal pole. Furthermore, a higher rCBF was observed in the left PC and the antero-superior temporal gyrus in migraineurs than in controls during both O and NO conditions (Fig. 3A). These differences confirm that migraineurs with OHS exhibit an altered cerebral responsiveness to olfactory stimulation and suggest the presence of permanent dysfunction in some olfactory brain regions.
Pitfalls
Since it has previously been shown by some authors that sniffing alone can induce activation of the PC (43), a source of potential confusion may be related to the different breathing patterns across experimental conditions and groups. Respiratory flow was not recorded in the present study, but several data allowed us to conclude that activation patterns did not result from different breathing patterns between controls and migraineurs. First, a recent study has demonstrated opposite results to those reported by Sobel, concluding that sniffing in the absence of odorants does not activate the PC (44). Second, our subjects were trained to breathe regularly and not to sniff. In previous studies in which subjects were asked to breathe in the same way, no respiratory flow differences could be evidenced between experimental conditions (21, 45). Finally, we have shown in the present study that breathing frequency, indirectly calculated from the number of stimulations, did not differ between controls and migraineurs. Therefore, we believe that any difference in activation patterns observed in this study is unlikely to reflect different breathing patterns between groups.
A higher rCBF in the left PC and antero-superior temporal gyrus
The left PC and antero-superior temporal gyrus were the only two regions showing higher baseline rCBF in patients than in controls, an abnormality that persisted during odour stimulation. However, the relative hyperperfusion observed in the PC in our patients cannot be readily interpreted in terms of functional activity, since higher levels of both excitatory and inhibitory neurotransmission can result in elevated energy demand (46, 47). However, our findings are consistent with the view that hyperactivity of the PC could result in facilitated triggering of the trigeminovascular system in response to odours during the interictal or pre-ictal period.
Apart from the major role played by the PC in olfaction, other data indeed support its involvement in odour-triggered migraine. It is well know that most odorants possess trigeminal characteristics and therefore activate the trigeminal system (48), but reciprocally, it has also been shown that trigeminal stimuli as CO2 can activate the PC (49). Thus, there are several possible sites of interaction between olfactory and trigeminal information, not only at the peripheral level (olfactory epithelium and bulb), but also at the most central level including the PC (50). It is also hypothesized that odour-triggered migraine could result from activation by odours, and via the PC, of the parasympathetic descending pathway involving the preganglionic parasympathetic neurons in the superior salivatory nucleus. This nucleus in turn would activate postganglionic parasympathetic neurons in the sphenopalatine ganglion, resulting in vasodilation and local release of inflammatory molecules stimulating meningeal perivascular nociceptors (51). The left lateralization of the rCBF changes observed in the PC and antero-superior temporal gyrus in migraineurs is consistent with the predominantly left-sided organization of the parasympathetic pathways in the central nervous system (52–55). Interestingly, alteration of the matrix of cortical blood vessels and neurons has been observed within the PC after cortical spreading depression in the rat (56).
Furthermore, the PC plays a major role in the olfactory habituation process (57–61). OHS in migraineurs could reflect a dysfunction of this process, all the more so as comparable abnormalities have been demonstrated in migraineurs for other sensory modalities (8, 10, 62–65). According to this hypothesis, the increased rCBF (in the baseline and odour stimulation conditions) observed in the left PC in our migraineurs could be interpreted as a lack of normal habituation in response to permanent environmental olfactory stimulation.
The involvement of the temporal gyrus during migraine attacks was reported in previous PET studies (24, 30) and during a migraine visual aura explored by magnetoencephalography (66). In our study, an abnormally high rCBF observed in the left antero-superior temporal gyrus (−42, 12, −24) in migraineurs is likely to reflect the close anatomical relation between this structure and the most anterior part of the frontal PC (−40, 12, −20). Accordingly, a significant correlation was observed between rCBF values measured within these two regions in the baseline condition (r = 0.670, t = 3.412, P = 0.0341).
Dysfunction of a neural network in migraineurs during olfactory stimulation
Significant activation in the anterior temporal pole (−30, −6, −36) was observed in migraineurs, but not in controls during odour stimulation. The left temporal pole has also been found to be involved in olfactory emotional processing (67), and after aversive olfactory learning (68). Activation of this region in migraineurs with OHS could thus reflect a particular emotional status and partly explain the alteration in olfactory hedonic judgments previously observed in these patients (4).
By contrast, several brain regions (left inferior frontal gyrus, left and right middle frontal gyri, left and right angular gyri, right postero-superior temporal and angular gyri, posterior cingulate gyrus), as well as the right locus coeruleus and cerebellum, were not activated, or even deactivated by odours in migraineurs with OHS, whereas they were significantly activated in controls. In olfaction, involvement of the left inferior frontal gyrus has been demonstrated in semantic processing (15, 19, 32), whereas the other regions have been found to be activated during olfactory stimulation (69–71), although not classically considered playing an important role in olfaction processing. The decreased odour-triggered brain activation in our patients is consistent with the results recently reported in patients with multiple chemical sensitivity, although not in the same brain regions (23). As previously suggested, it could reflect a top–down regulation process that aims to compensate for OHS or the higher activation in the PC. Alternatively, the impaired cortical response to odours observed in various cortical brain regions could reflect abnormal processing related to migraine disease. Indeed, migraineurs have shown decreased blood flow velocity in response to visual stimulation (72, 73).
PET studies in migraineurs have shown activation in brainstem areas only during the ictal period (24, 29, 30, 74). In our study, we observed lack of activation in the right locus coeruleus during olfactory stimulation. The locus coeruleus is involved in arousal and stress responsiveness, but also in pain control (see for review (75, 76)). It is also known to play a role in olfactory modulation through its centrifugal connections towards the PC (38, 77) and could be involved in cranial parasympathetic outflow (78). Anatomical projections from the locus coeruleus towards the olfactory bulb and PC are reported to be ipsilateral (38), but contralateral inhibitory influence exerted by the locus coeruleus (79) could explain the effects observed in the present study in the left hemisphere. Whether the lack of activation in the locus coeruleus in response to olfactory stimulation could result in OHS or odour-triggered migraine remains an open issue.
Overall, we have found that migraineurs with OHS displayed complex rCBF abnormalities, both at baseline and during odour stimulation. These abnormalities include rCBF changes in different brain and brainstem regions involved in olfactory processing and migraine. Although some of these changes are consistent with the hypothesis of a hyperactive olfactory system in migraineurs with OHS, it remains difficult to explain all of our findings within a single framework. It is also possible that some of our results are specific to OHS, whereas some others would reflect the pathophysiology of migraine. To address this issue, further investigations comparing migraineurs with and without OHS are warranted.
Acknowledgements
We wish to thank the control subjects and patients who participated in this experiment, and the members of CERMEP (especially N. Costes and F. Lavenne) for their valuable assistance, and are greatly indebted to Dr P. Giraud and Dr A. Revol for assistance in recruiting patients. We are grateful to societies of perfume and/or aroma (Givaudan, Lenoir, Perlarom) for supplying the odorants used in this study. This work was supported by the Hospices Civils de Lyon and the Programme Hospitalier de Recherche Clinique (PHRC 2004 of the Rhone-Alpes Region).

