Abstract
In this study we aimed to assess the brain distribution of 5-HT1A receptors in migraine patients without aura. Ten female migraine patients and 24 female healthy volunteers underwent magnetic resonance imaging and positron emission tomography using a radioligand antagonist of 5-HT1A receptors [4-(2'-methoxyphenyl)-1-[2'-(N-2-pirydynyl)-p-fluorobenzamido]-ethylpiperazine (18F-MPPF)]. A simplified reference tissue model was used to generate parametric images of 5-HT1A receptor binding potential (BP) values. Statistical Parametrical Mapping (SPM) analysis showed increased MPPF BP in posterior cortical areas and hippocampi bilaterally in patients compared with controls. Region of interest (ROI) analysis showed a non-significant trend in favour of a BP increase patients in cortical regions identified by the SPM analysis except in hippocampi, left parietal areas and raphe nuclei. During the interictal period of migraine patients without aura, the increase of MPPF BP in posterior cortical and limbic areas could reflect an increase in receptor density or a decrease of endogenous serotonin, which could explain their altered cortical excitability.
Introduction
Migraine is a common and chronic disorder, characterized by attacks of moderate to severe headache, frequently unilateral, often associated with nausea, vomiting, photophobia or phonophobia (1) and presenting a high prevalence of depression and generalized anxiety disorder (2, 3). The involvement of serotonin (5-HT) in the pathophysiology of migraine has been documented for over 30 years.
Pharmacological agents that cause 5 HT release, such as reserpine, are able to induce a migraine attack or to increase the incidence of migraine-like headaches in migrainous patients (4, 5). Many of the pharmacological agents used to treat migraine attacks interact with serotonin and its receptors (6, 7). In particular, a recent animal study has suggested that triptans and ergot derivatives, known as agonist of 5-HT1B/D, could also interact with 5-HT1A receptors (8). Similarly, the preventive treatments methysergide and pizotifen, antagonists of 5-HT2 receptors, also have a weak affinity for 5-HT1A receptors (6, 7); in consequence, 5-HT1A receptors may also contribute to the efficacy of some drugs used in migraine prophylaxis (9). 5-HT1A receptor hypersensitivity is the most likely explanation for the increased prolactine response to buspirone, a 5-HT1A receptor agonist, in migrainous patients (10). Finally, an increase of serotonin synthesis during migraine attacks has been demonstrated in vivo in patients suffering from migraine without aura using positron emission tomography (PET) and α-[11C]methyl-L-tryptophan (AMT) (11) and could reflect an alteration of 5-HT synthesis via regulation of 5-HT1A raphe autoreceptors.
5-HT1A receptors are mostly neuronal. They are either heteroreceptors, when located in target regions of 5HT neurons with a particularly high concentration in limbic areas, or autoreceptors on the soma and dendrites of 5HT neurons in raphe nuclei, where they exert a negative feedback on serotoninergic neuron firing activity (12, 13).
The 4-(2′-methoxyphenyl)-1-[2′-(N-2-pirydynyl)-p-fluorobenzamido]-ethylpiperazine (18F-MPPF) is a radiotracer antagonist of 5-HT1A receptors that has an affinity more similar to that of 5-HT for 5-HT1A receptors (Ki = 3.3 nM) (14) compared with other 5-HT1A radioligands, such as 11C-WAY100635. Its affinity allows its own displacement by endogenous 5-HT (15, 16). In the present study we used 18F-MPPF for PET imaging of brain 5HT1A receptors. The aim was to assess the interictal 5-HT1A receptor's availability, which reflects the 5HT endogenous level, in patients suffering from migraine without aura.
Methods
Patients
Data concerning patient's histories and clinical features are summarized in Table 1.
Patients' clinical data

R, right; L, left; PET, positron emission tomography; GHQ, General Health Questionnaire.
Twelve (right-handed) women with a diagnosis of migraine headache without aura according to the International Headache Society (1), and having suffered from migraine attacks for over 5 years, were recruited from migraine out-patients followed in the Functional Neurology Department of the Neurological Hospital Pierre Wertheimer (Lyon, France).
On the day of the PET scanning, depression and migraine symptoms were evaluated using the General Health Questionnaire (GHQ-28) and Migraine Disability Assessment Scale (MIDAS, (17)) (level 1–4), respectively.
Exclusion criteria included any other neurological disease than migraine, and a history of analgesic abuse. At the time of the PET study, none of the patients was taking prophylactic migraine treatment. All of them had been attack-free for at least 3 days preceding the PET study.
All patients gave their informed consent to the protocol, which was approved by the local ethics committee (CPP, Centre Léon Bérard, Lyon) in accordance with the Declaration of Helsinki and French regulations on Biomedical Research.
Healthy subjects
All 24 female healthy subjects (19–67 years old, mean 43.1 ± 13.3 years) from our normative database (18) were considered. As previously described, control subjects had been selected on the basis of (i) no sign or history of neurological, psychiatric, cardiovascular, pleuro-pulmonary or haematological illness, (ii) no ongoing neuroleptic, antiparkinsonian methyl-dopa, β-blocker, monoamine oxidase inhibitor-A or -B, tricyclic antidepressant, thymoregulator or antimigraine treatment, (iii) no hormone replacement therapy, and (iv) a score below the threshold for depression (< 7) for the GHQ-28.
Magnetic resonance imaging acquisition
All patients and control subjects underwent a 3-D anatomical T1-weighted sequence on a 1.5-T Siemens Magnetom scanner (Siemens AG, Erlangen, Germany). Data were acquired in the sagittal plane. The anatomical volume, consisting of 130–170 slices with 1-mm3 voxels, covered the whole brain. Magnetic resonance images (MRIs) were visually analysed by a neurologist to ensure that no brain lesion or malformation was present.
PET data acquisition and pre-processing
The methodology of tracer production, scan acquisition and data pre-processing was the same as previously described (19).
To summarize, 18F-MPPF was acquired with a CTI Exact HR+ scanner for 60 min after the injection of 151.6 MBq of 18F-MPPF (mean ±
Parametric images of binding potential (BP) values as defined by (20) were obtained using a simplified reference tissue model (SRTM) introduced by (21) and validated by our group for 18F-MPPF studies (18). BP is a quantitative index combining the local density receptor and apparent in vivo affinity modulated by the extracellular endogenous serotonin concentration. Our previous study (18) confirmed that BP is a good index of local receptor concentration in normal subjects.
Using Statistical Parametrical Mapping (SPM2; Wellcome Trust Centre for Neuroimaging, UCL, London, UK) parametric images of BP were transformed into a standard space [Montreal Neurological Institute template of the ICBM Project (22)]. Normalized BP images were then smoothed using an 8 × 8 × 8-mm full-width at half-maximum isotropic Gaussian kernel to take into account interindividual anatomy variability and to improve the sensitivity of the statistical analysis (23).
BP global analysis
The BP global values were extracted for all patients and subjects using SPM. BP global values in patients were compared with BP global values in controls with Student's t-test. The significance threshold was set at P < 0.05.
Voxel-based analysis of PET data
We explored possible statistical differences between BP images of patients and controls using the linear model at each and every voxel (24). SPM2 was used on the normalized smoothed parametric BP images of patients and control subjects.
The statistical differences between patients and controls were estimated taking the age and global BP into account as covariable of no interest and using an
Region of interest analysis
This analysis was carried out using an existing interactive 3-D atlas of the human brain created at the Montreal Neurological Institute (26). This atlas was obtained from a T1 MRI volume acquired from a single subject, spatially normalized into Talairach space and segmented into 90 identified structures. A set of 65 regions of interest (ROIs), including all cortical, limbic and thalamic areas, was selected from the 90 available. For the raphe nuclei, which are difficult to delineate on MRI, the contours were drawn on a MPPF-PET template by thresholding the activity at 80% of the local maximum in the brainstem (average of 50 normalized MPPF scans from our control database). The ROI was then displayed on the MRI template to verify its proper location in the periaqueductal grey matter of the brainstem.
For each patient, all ROIs were then applied on normalized and smoothed parametric BP images to extract a mean BP value for each region.
For each ROI, BP values in patients were compared with those in controls using a general linear model (SAS® software; SAS Inc., Cary, NC, USA). The group effect (patients vs. controls) was tested after adjustment for age (confounding effect). The significance threshold was set at P < 0.001 to correct for multiple comparisons.
Results
Clinical data
All patients suffered from migraine without aura and were headache free during the PET investigation. Only 10 of the 12 patients, showing a score below the threshold for depression (< 7) for the GHQ-28 and a delay of attack-free preceding the PET study inferior at 3 days, were included in the study; GHQ-28 depression scores ranged between 0 and 2 (mean ±
Three patients suffered from migraine frequently lateralized on the right side, and seven did not show any predominant side for migraine (Table 1). On average, they had been suffering from migraine for 17.2 ± 6.01 years with a mean frequency of 3.1 ± 0.87 attacks per month. All patients had a MIDAS level of 3 of intensity and invalidity of their migraine. None of patients or control subjects had an MRI lesion.
BP global analysis
There was no difference in the mean BP global between patients (mean ±
SPM analysis
The contrast ‘patients–controls’ showed a significant increase of 18F-MPPF BP in seven different regions in patients compared with controls (Table 2). As illustrated in Fig. 1, these clusters were localized (i) for the left hemisphere, in the lateral parieto-occipital junction (Zmax = 6.33, k = 327, P < 0.03), the hippocampus (Zmax = 6.01, k = 1701, P < 0.001), the post-central gyrus (Zmax = 4.68, k = 957, P < 0.001) and the inferior parietal lobule (Zmax = 4.33, k = 354, P < 0.02), and (ii) for the right hemisphere, in the junction between superior temporal and middle temporal gyri (Zmax = 5.13, k = 2144, P < 0.001), the mesial aspect of the parieto-occipital junction (Zmax = 4.89, k = 1280, P < 0.001) and the hippocampus (Zmax = 4.74, k = 695, P < 0.002).
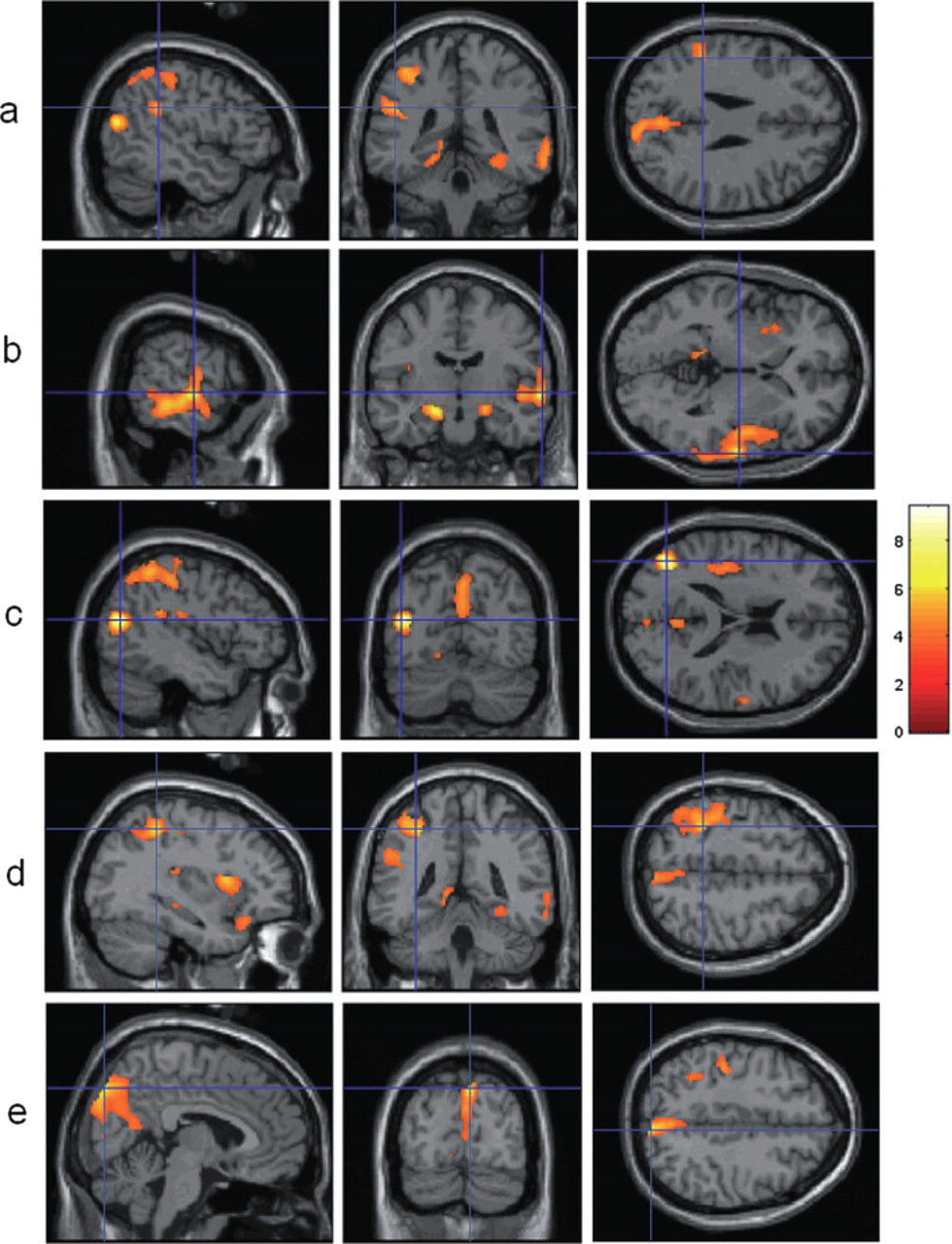
Statistical Parametrical Mapping results: regions showing an increase of 5-HT1A binding potential (BP) in patients compared with 24 controls. All displayed regions had a T score thresholded at P < 0.05 after correction for multiple comparisons. Colour scale: Z score. The blue crosses are centred on significant clusters (except for the hippocampi). Statistical analysis showed seven regions with a significant increase of 5-HT1A BP in patients in (a) left inferior parietal lobule, (b) left and right hippocampi and right junction between superior temporal and middle temporal gyri, (c) left lateral parieto-occipital junction, (d) left post-central gyrus, and (e) right internal parieto-occipital junction (cuneus and precuneus).
Contrast ‘patients–controls’ of the Statistical Parametrical Mapping analysis

x, y, z are in Talairach coordinates, k is the cluster size (expressed in voxels).
The other contrast, ‘controls–patients’, revealed no significant decrease of 18F-MPPF BP in patients compared with controls.
ROI analysis
After adjustment for the age effect, no significant increase of mean BP in patients compared with controls was observed at P < 0.001 (Table 3). However, at an inferior P-value (P < 0.05), four ROIs showed a BP increase that was localized in the left medial occipito-temporal gyrus (P = 0.04), and right cuneus (P = 0.04), pre-cuneus (P = 0.01) and superior temporal gyrus (P = 0.03).
Region of interest (ROI) analysis

Mean BP values in ROIs for patients and controls, least square means in patients and controls and P-values obtained from the General Linear Model for the group effect (patients vs. controls) after adjustment for age.
L, left; R, right, lsmean, least square mean; BP, binding potential.
BP values in raphe nuclei were not significantly different in patients and controls. No decrease of BP was observed in any ROI in migraine patients.
Discussion
Voxel-based SPM analysis of MPPF BP shows that the brain serotoninergic system is modified between attacks in migraine without aura. This interictal change consists of a MPPF BP increase in parieto-occipital, temporal and limbic areas. The ROI analysis did not confirm this MPPF BP increase at a threshold set at 0.001 to correct for multiple comparisons. This result could be due to the use of a strict P-value, and, contrary to the voxel-to-voxel approach in the SPM analysis, to the use for statistics of an average BP value within the ROI. Nevertheless, the analysis showed a non-significant trend in favour of a BP increase in migraine patients in four posterior cortical ROIs.
This increase of MPPF BP for 5-HT1A receptors may reflect either an increase of 5-HT1A receptor density or a reduction of endogenous serotonin release. MPPF being displaceable by endogenous 5-HT, a reduction of endogenous serotonin synthesis and release can cause an increase of MPPF binding (15, 16). Either or both mechanisms could be involved in the MPPF BP increase that we observed.
A serotonin metabolism dysfunction has been demonstrated in migraine without aura, but data concerning free plasmatic 5HT are often contradictory (27–29). Between attacks, migrainous patients were shown to have a lower plasmatic 5-HT level than controls and patients with tension headache (30).
The increased MPPF BP may reflect a low endogenous 5-HT during the attack-free period. 5-HT is considered to be an inhibitory neurotransmitter in the brain and could play a role in the central modulation of trigeminal pain. The cortical serotoninergic projections of raphe nuclei are numerous. They mainly target limbic areas, particularly the amygdala, the hippocampus and the entorhinal cortex (31), as well as parietal, occipital and frontal cortices (32, 33). 5-HT release or 5-HT-receptor-mediated transmission may contribute to the tonic suppression of nociceptive transmission (34). Recently, a PET study of 5-HT1A receptors using the 11C-WAY100635 in 11 healthy male subjects showed a negative correlation between BP and the intensity of the cold pressor pain in multiple brain areas of the pain matrix including prefrontal, cingulate and insular cortices (35). However, this study was performed only on male subjects, and we cannot exclude that the pain perception could be gender-dependent or slightly different in women subjects. Nevertheless, the localization of our MPPF BP changes, except for the hippocampi, is different from those involved in regulation of pain-related responses of this previous study, and brain areas involved in the pain matrix, in particular operculo-insular and cingulate cortex, do not show any interictal 5HT1A receptors changes. Interictal modifications of MPPF BP are thus unlikely to reflect functional modifications of pain processing in migraine. More probably, they are linked to the interictal changes in cortical excitability reported in migrainous patients. Indeed, a decrease of serotonin synthesis may cause an alteration of cortical excitability mediated via 5-HT1A receptors that have an inhibitory neuronal effect (36). The modifications of MPPF BP observed in posterior cortical areas in migraine patients could thus be linked to the lack of habituation of cortical responses to repetitive sensory stimulations during interictal state in migrainous patients, as demonstrated by visual and auditory-evoked potentials (AEP) studies ((37), see for a review (38, 39)). In migraine patients, the amplitudes of AEP with rising intensity show an increase that is higher than that observed in normals, with a steeper slope of the AEP vs. stimulus amplitude correlation (intensity dependence) (40–42). This intensity dependence being a valuable indicator of brain serotoninergic activity (43), this increased cortical excitability is considered to reflect a decrease in serotonin transmission. Thus, the hypothesis that the increased cortical MPPF BP that we observed in raphe cortical projection areas reflects reduced levels of endogenous 5HT would explain the interictal sensory stimulus hypersensitivity of migrainous patients.
In our study, no MPPF BP difference was observed in the raphe between migraine and control patients. Indeed, involvement of the raphe was expected in our patients. Evidence of raphe nucleus involvement in migraine is supported by lesion studies (44–46), by PET studies showing increased blood flow in the reticular or dorsal pons during the migraine attack and persistent after relief by sumatriptan (47–50), and, more recently, by serotonin transporter availability study (51).
The hypothesis that the cortical MPPF BP changes observed in our study were a consequence of reduced 5HT release at the raphe nuclei cortical terminals would have been supported by the finding of a change in raphe nuclei MPPF BP; this change would be compatible with an increased density of raphe nuclei 5HT1A autoreceptors, known to mediate inhibitory feedback effect on 5HT synthesis (12, 13). However, we did not observe any MPPF BP change in the raphe of migrainous patients. This may be due to the small size of this structure that entails a strong partial volume effect. In our patients, we did not perform any partial volume effect correction (PVC), as partial volume effects remain the same for all subjects when regions with similar volumes are compared. The absence of PVC cannot therefore explain our results. In addition, the absence of any MPPF BP modification in raphe suggests that increased cortical and limbic MPPF BP may reflect more an increase 5-HT1A receptor density than an increase of 5-HT1A receptor availability.
In conclusion, this PET study has shown an increase of MPPF BP in posterior parieto-occipital and temporal cortical areas and limbic areas during the interictal period in migraine patients and has provided an argument in favour of a low brain serotonin level in migraineurs without aura between attacks. However, to rule out the possibility that our results reflect overexpression of 5-HT1A cortical receptors between migraine attacks, it would be interesting to investigate migraine patients with PET using the 11C-WAY100635 radioligand, which is insensible to endogenous serotonin changes, due to its high binding affinity with 5-HT1A receptor.
Acknowledgements
We thank Didier Le Bars for 18F-MPPF radiosynthesis, and the CERMEP medical team for assistance.
