Abstract
Objective: Our study was conducted to describe prescription refill patterns among patients newly treated with triptans.
Background: Although triptans are efficacious in treating migraine headache, the persistency of triptan use among newly initiated users has not been well described.
Methods: From a US pharmacy claims database, we identified patients receiving new triptan monotherapy prescriptions from 2001 to 2005. Prescription refill information was gathered for two years for each patient. Persistency was defined as sustained refills of the index triptan prescription, regardless of duration between refills.
Results: Of 40,892 patients receiving a new triptan prescription, 53.8% (N = 22031) did not persistently refill their index triptan. Of these, 25.5% discontinued prescription migraine therapy, 7.4% switched to a different triptan, and 67.1% switched to a non-triptan migraine medication at the time of their first refill. Only 46.2% of patients received at least one persistent refill.
Conclusions: Migraine patients were more likely to discontinue their triptan after their index prescription than at any other time in their prescription refill history. The majority of patients did not persistently refill triptans, but filled prescriptions for non-specific migraine therapies such as opioids and non-steroidal anti-inflammatory drugs. Reasons for triptan discontinuation warrant further investigation.
Introduction
Migraine headache is a common neurological disorder, affecting approximately 12% of the US population, and incurring substantial economic and social cost to individuals during their most productive life years (1–3). Migraine frequently afflicts individuals for years at a time, and is currently managed by most sufferers with appropriate treatments used at the onset of attack (4). Because of the long-term chronicity of migraine, it has been recommended that patients utilize the correct treatment and persist with that treatment unless attacks improve or subside (5–7). A variety of medication classes is available for the acute management of migraine headache. Guidelines recommend non-steroidal anti-inflammatory drugs (NSAIDs) as first-line therapy for migraine pain, but specific agents, such as triptans and ergotamines, are frequently prescribed to patients whose migraines respond poorly to NSAIDs or combination analgesics (8). Triptans in particular have been shown to be efficacious acute migraine-specific treatments in several large placebo-controlled trials (9), and their early use is recommended for moderate to severe migraine attacks (10).
Despite the safety and efficacy of triptan medication, pharmacy claims data show paradoxically low retention of triptan refills over time (11,12). An Israeli health maintenance organization (HMO) study of 1498 patients receiving new triptan prescriptions found that 56% of patients purchased triptans only once during the 6–18 month follow-up period (13). Several pharmacy claims analyses conducted across Europe show similar results, with single-time triptan fill rates of 38%–56% (11,14–16). The utilization and retention of triptans has been examined in managed care settings in the USA as well (17,18). Etemad and colleagues (2005) used a US managed care population to examine the utilization of triptans and found that 39% of 5294 new triptan users received only one claim (refill of the index prescription) during a 12-month follow-up period (17).
Other literature has endeavored to compare triptan refill rates with the refilling of other migraine-specific medications within the same dataset. Of 2343 migraineurs from 18 community pharmacies in the Netherlands, 42% received only one anti-migraine prescription in a 12-month period whereas 12.5% (N = 292) were 'multiple-drug users’, who received either more than one specific anti-migraine drug, or different dosage forms/strengths of their index drug (15). A UK-based study found that 55% of 3196 patients established on sumatriptan received a single prescription, 36% continued, and 9% switched from sumatriptan to another triptan within a 15-month period (11).
Although it is clear that triptan prescriptions filled by users during a prespecified time interval is low in many populations, little light has been shed thus far on the pattern of persistency to triptans across refills or across time. Persistency can be defined as the renewal rate of prescriptions, or the duration of continuous treatment with one's initially prescribed medication (19). The analysis of triptan persistency, however, poses several challenges in a claims database. Because there is wide variation in the interpatient frequency of migraine attacks and the rate at which patients take triptan tablets, establishing baseline criteria for persistent versus non-persistent use requires the observation of sufficiently long time intervals to estimate one’s baseline pattern of attacks. Even then, prescription refill patterns may not accurately reflect migraine frequency patterns. Medication persistency in other chronic-episodic conditions, such as asthma, has been described in terms of continuous refills, taking into account the timeliness and consistency of refilling at each point in the timeline (20,21). Similarly, at each point in a patient's refill history, the placement of switching patterns may be identified by comparing lapses in sustained medication use against observable uptakes of alternative treatments. This is particularly relevant for migraine because migraineurs prescribed triptans may switch between triptans and other acute therapy options, co-medicate with migraine-specific as well as non-specific prescription drugs or go through periods of treatment discontinuation when attacks are not experienced.
Population-based migraine medication usage data suggests that migraineurs use opioids, barbituates, analgesic salicylates and NSAIDs both in addition and as alternatives to triptans (22), yet available claims studies have not identified potential switches or possible co-medication with non-triptan migraine medications. It is also not clear at which point in a patient's prescription history this type of non-triptan medication use is most likely to occur.
To address these questions, we conducted a US-based pharmacy claims data analysis using a contemporary cohort of new triptan users and followed their prescription refill patterns over two years of observation. In the current study, we aim to describe (i) the persistency of index triptan use among a large cohort of new users and (ii) triptan and non-triptan use and discontinuation patterns at the time of each prescription refill.
Methods
Study design
This study was designed as a retrospective analysis of the i3 InVision Data Mart (Ingenix, Eden Prairie, MN, USA); a large US claims database which includes records for more than 36 million patients over a span of six years. Claims for pharmacy services are submitted electronically by the pharmacy at the time prescriptions are filled. Pharmacy claims data in the InVision DataMart include drug name, dosage form, drug strength, fill date, days of supply, patient out-of-pocket fees, and de-identified patient and prescriber codes, allowing for longitudinal tracking of medication refill patterns and changes in medications. Pharmacy records for each patient between 1 January 2000 and 31 December 2007 were utilized for the purposes of this analysis.
Study sample
Patients in this cohort were migraineurs newly treated with triptans during a five-year index period (1 January 2001–31 December 2005). To be considered new to treatment, patients must have received no prescription of any migraine-specific drugs (triptans or ergotamines) in the year prior to the index triptan prescription, or have received no prescription of any non-specific migraine drug(s) within 15 days following a migraine diagnosis during the one year pre-index period. The date of the first triptan monotherapy prescription, satisfying the above criteria, was the index date. Figure 1 illustrates the criteria used to define patient cohort selection. All patients were followed for a period of two years.
Study design. The figure illustrates criteria used to define patient cohort selection. All patients needed to be continuously enrolled for a minimum of 3 years, and could not have received a triptan prescription for a minimum of 1 year prior to the index date (i.e. pre-index period). Patients were followed for a period of 2 years following the index triptan date (i.e. observation period).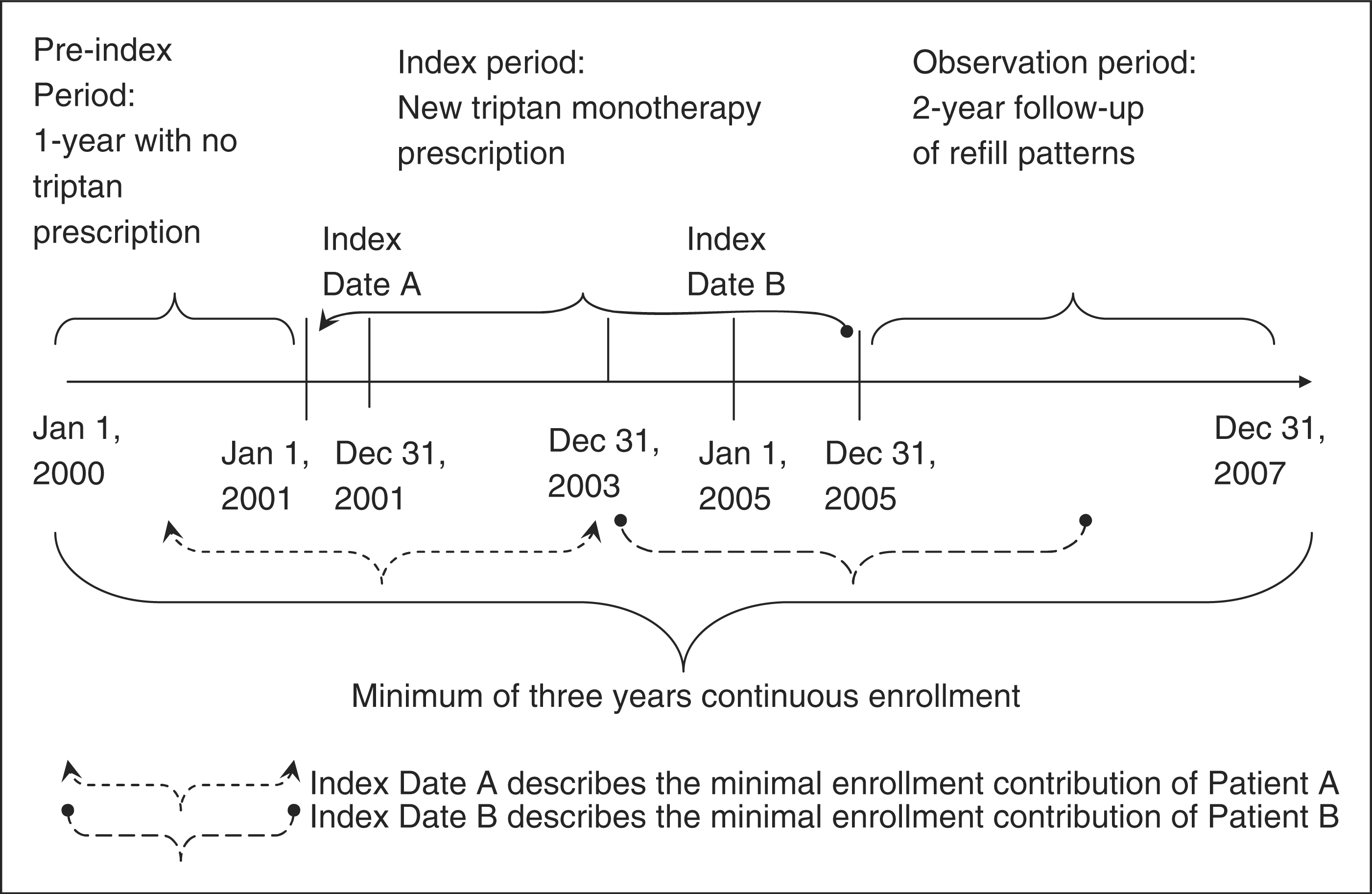
Migraine-specific medications tracked in the pre-index period for the purposes of selecting this cohort of newly treated triptan users included sumatriptan, rizatriptan, eletriptan, almotriptan, naratriptan, frovatriptan, zolmitriptan and ergotamine products. To determine non-specific migraine medication use in the pre-index period, migraine diagnoses were identified by the International Classification of Diseases (ICD-9 codes 346.00, 346.01, 346.10, 346.11, 346.20, 346.21, 346.80, 346.81, 346.90 and 346.91). Non-specific migraine medications occurring within 15 days following the appearance of a migraine diagnosis code were classified as NSAIDs (e.g. ibuprofen, diclofenac sodium); opioids (e.g. codeine, hydrocodone bitartrate, propoxyphene HCL); barbituates (e.g. butalbital, butalbital-containing combinations); salicylates (e.g. acetaminophen, aspirin and caffeine combinations); and other non-specific drugs, including select anti-emetics and antihistamines. All associated prescription National Drug Codes (NDCs) were grouped by drug class irrespective of dosage, strength or formulation, and refills were also drug-specific. Refill formulations included pills, injectables, dissolving tablets and nasal sprays.
Patients were required to be enrolled for a minimum of 12 months (one year) preceding the index triptan prescription, and 24 months (two years) following the receipt of their index triptan prescription. The index triptan prescription was by necessity a monotherapy prescription; patients with triptan co-administrations (e.g. another triptan or non-triptan migraine prescription) occurring on the same date were excluded in order to preserve homogeneity within this cohort of newly treated triptan users.
Definitions
At the time of each migraine prescription refill, patients in the index triptan population were classified according to their refill pattern as described in Figure 2, which graphically illustrates the classification scheme for the cohort.
Refill patterns diagram. Patients were classified as in-class switchers, between-class switchers or discontinuers at the time of each subsequent migraine medication refill. Patients receiving an index triptan, receiving an NSAID at their next refill and receiving the index triptan for their second refill were considered “add-ons”, or co-medicating users at the time of the first and second refill, but persistent to the index triptan for all three prescriptions. Patients receiving another triptan at their next refill, regardless of whether this refill was followed by the index triptan, were considered to have switched “within-class” to an alternate triptan. Rx = prescription. Btw = between. NSAID = non-steroidal anti-inflammatory drug.
Patients were terminally classified at the time of each migraine prescription refill as within-class switchers (switching from the index triptan to another triptan), between-class switchers (switching from index triptan to a non-triptan migraine medication) or discontinuers, who had no further prescriptions of specific or non-specific migraine medications for the remainder of their two-year observation period. Although no minimum time criterion was set for migraine medication discontinuation, in order to better capture migraneurs with a low frequency of attacks, patients were censored if the end of the two-year observation period occurred before 90 days following the last day of possession of migraine medication. If the end of the two-year observation period occurred after 90 days following the last day of possession of migraine medication, that patient was classified as a discontinuer on the last day of medication possession.
Patients who continued to refill the index triptan were considered persistent to their index triptan. In this way, persistency was defined as consecutive refills of the initially prescribed triptan, regardless of duration between refills. Add-on medication users were a subcategory of persistent users receiving other migraine prescriptions while continuing to receive refills of the index triptan. Add-ons were either as co-administrators, who had several medications (including another non-index triptan) prescribed on the same date along with the index triptan; or co-medicators, who had sequential administration of non-specific migraine medication and the index triptan (not occurring on the same date). Whereas in the former case a single prescription would be sufficient to determine an add-on therapy, in the latter case, the next three consecutive prescriptions were examined. If a prescription for the index triptan was followed by a non-specific medication and then by an index triptan prescription, the second prescription was considered an add-on, but patients were ‘persistent’ over all the three prescriptions. If, on the other hand, the index triptan was followed by an index triptan refill, and the third prescription was for a non-specific medication, the patient was coded as a between-class switcher at the time of their second prescription.
Measures
Patient demographic and clinical characteristics, such as age, health plan and the presence of comorbidities were identified to describe the study sample at baseline. Pre-index co-medication or comorbidites were identified on the basis of applicable ICD-9 codes and/or NDC drug codes. Established cardiovascular disease was defined as the presence of any of the following during 12 months preceding the index date: acute myocardial infarction (AMI), evidence of old myocardial infarction (MI), angina pectoris, heart failure, ventricular arrhythmia, atrial fibrillation, coronary artery disease or prescriptions for anti-anginal drugs. Cardiovascular risk equivalent(s) at baseline were identified by the presence of cerebrovascular disease (CVD), diabetes, peripheral vascular disease (PVD) or prescriptions for anti-diabetic drugs during the one year pre-index period 1 . ICD-9 codes for depression or anxiety diagnosis included 300.4, 311 and 298 (dysthymic disorder, anxiety, depression, depression with anxiety, depressive reaction, neurotic depressive state, reactive depression, depressive disorders NOS, depressive-type psychosis, psychogenic depressive psychosis, psychotic reactive depression, reactive depressive psychosis). Corresponding NDC codes for citalopram, escitalopram, fluoxetine, fluvoxamine, paroxetine, sertraline, zimelidine, venlafaxine and desvenlafaxine were also included in assessment of use of selective serotonin reuptake inhibitors/selective norepinephrine reuptake inhibitors (SSRIs/SNRIs).
Baseline opioid or other analgesic use was also extracted to assess non-specific migraine medication usage for pre-existing disease conditions in the pre-index period.
Data analysis
Based on refill classifications, the probability of the occurrence of each pattern was calculated based on the total number of patients exhibiting each pattern. The overall rates and percentages of persistency, discontinuation and switching at each refill were calculated. The probability of persistency at the kth refill was equal to the number of patients with k consecutive refills of the initial index triptan divided by the total number of patients in the index population. The percentage of discontinuation, between-class switching and within-class switching were calculated similarly. The probability of persistency between refills was computed by dividing the number of patients with k-1 refills by the number of patients with k consecutive refills. Kaplan-Meier (K-M) estimates were used for plotting survival curves over refills, and were estimated separately for each event of interest: discontinuation, switching or any change in persistency. The median number of refills to each event was computed for the index population. p values were considered significant at the p < .05 level. All statistical analyses were conducted using SAS version 9.1.2 (SAS, Carey, NC, USA).
Results
Baseline demographic and clinical characteristics for all patients (N = 40, 892)
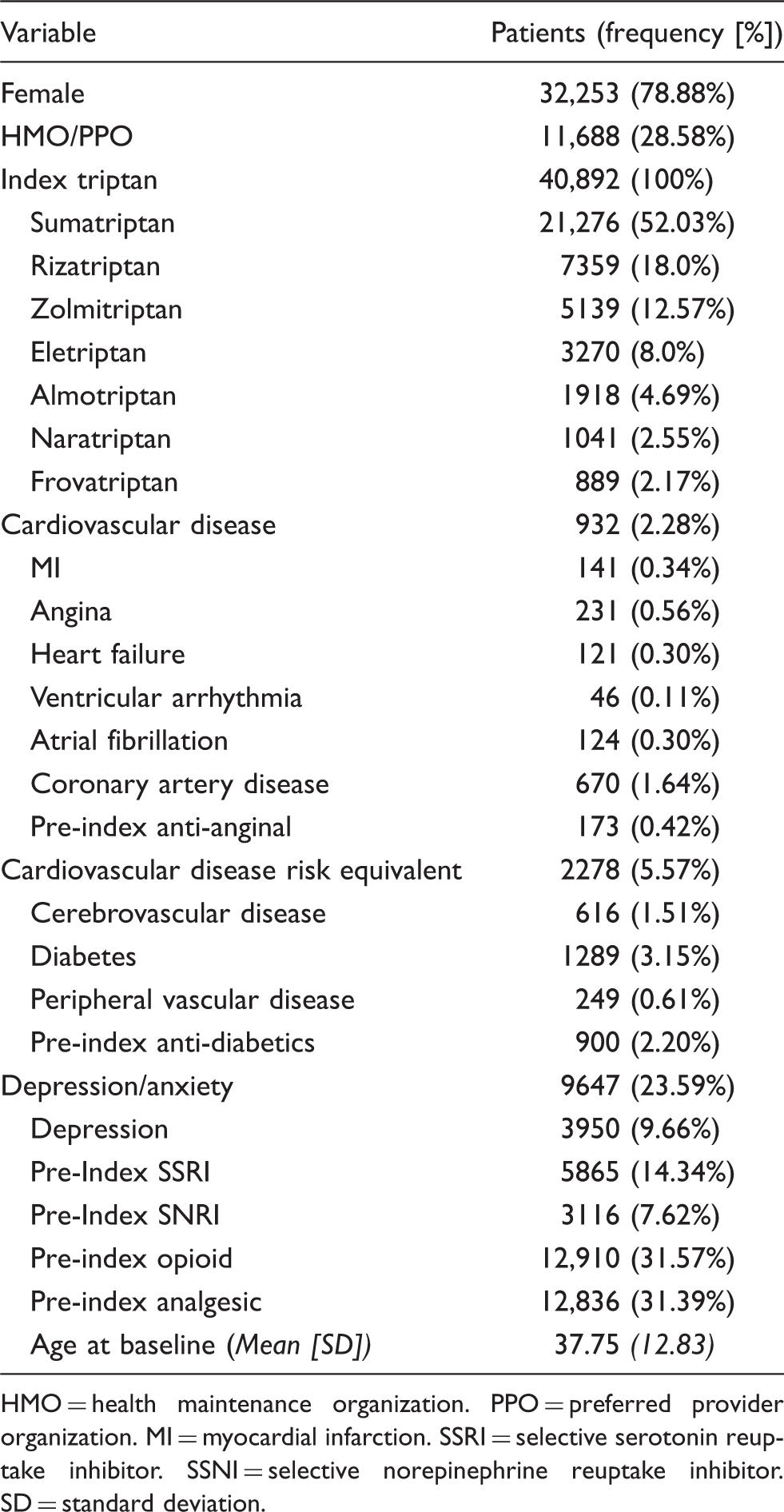
HMO = health maintenance organization. PPO = preferred provider organization. MI = myocardial infarction. SSRI = selective serotonin reuptake inhibitor. SSNI = selective norepinephrine reuptake inhibitor. SD = standard deviation.
At baseline, 24% of the sample had diagnosed depression or anxiety, and 29% had cardiovascular disease risk factors. Approximately 5.5% and 2.3% of the sample had cardiovascular risk–equivalent disease and established cardiovascular disease, respectively. About one-third of patients had used opioids or analgesics in the pre-index period for some non-migraine related condition(s). The distribution of patients persistent to the index triptan at the time of each refill is presented in Figure 3.
Triptan refills by proportion of patients (N = 40,892). The figure depicts the declining triptan refills across refill counts as a percentage of the total sample population. 22,031 patients (53.8%) received no consecutive refill of their index triptan, whereas 12.8% were persistent to their index triptan for exactly one additional refill.
Persistency, switching and discontinuation over refills (N = 40,892)

C = censored. DS = discontinued. BCS = between-class switch. WCS = within-class switch. Rx = prescription.
% C, % DS, % BCS, % WSC and % persistent were calculated by dividing the frequency of those exhibiting each pattern at the time of each refill by the total number of patients in the index sample (i.e. 40,892). Persistency between refills was calculated by dividing the frequency of those persistent (N) at each refill by the total persistent (N) at the prior refill.
Because 53.8% of patients received no consecutive refill of their index prescription, 46.2% were considered persistent to their triptan for at least one refill. At the time of the seventh refill, only 10% of the index population remained persistent to their index triptan. The between-refill persistency rate shows that patients were least likely to remain persistent between the index prescription and the first refill (persistency = 0.462). At higher refill counts (i.e. two or more refills), patients were increasingly more likely to remain persistent to the index tripan from one subsequent prescription to the next.
Figure 4 graphically depicts the medication switch and discontinuation pattern of those receiving no consecutive index triptan prescription at the time of the first medication refill (i.e. 53.8%). At the time of the first migraine medication refill, 7.4% of these patients (N = 1635) switched within-class to another triptan, 25.5% (N = 5626) discontinued migraine therapy (or received no prescription) for the remainder of the observation period, and most received a non-index medication refill and were considered to have switched “between-class”. Non-specific migraine medications most commonly filled following the index triptan included opioids and NSAIDs.
Depiction of prescription pattern at first refill (N = 22,031). 53.8% of patients received no consectutive refill of their index triptan. 25.5% of these patients appeared to have discontinued (specific or non-specific) migraine prescriptions for the remainder of their follow-up, 34% received an received a prescription for an opioid or opioid-containing medication, 23% received an NSAID prescription and 7.4% received a non-index triptan prescription. The remainder received prescriptions for barbituates and other non-specific migraine drugs.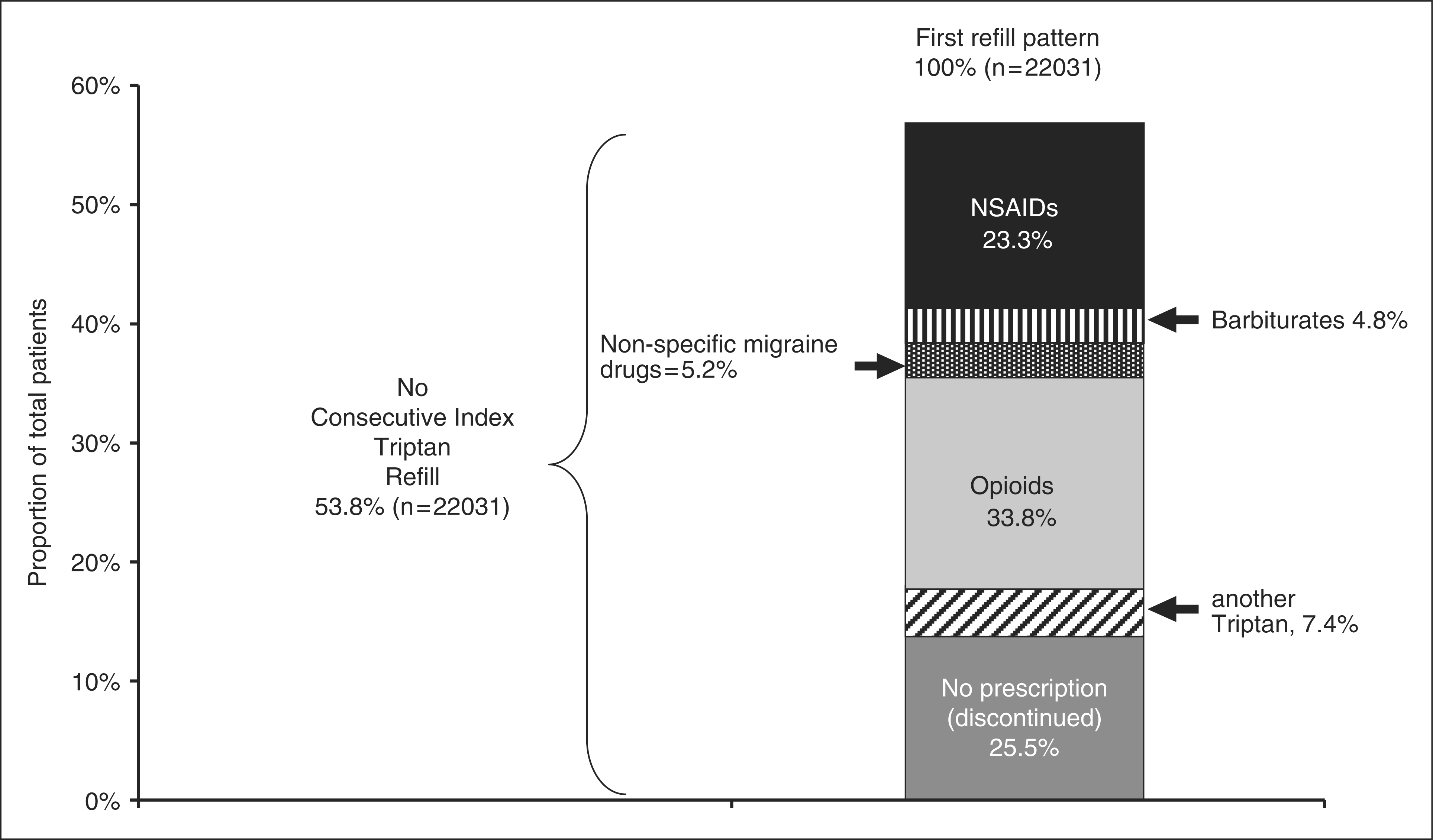
Prescription pattern among those switching between class at first refill* (N = 14,770)

Rx = prescription. NSAIDs = non-steroidal anti-inflammatory drugs.
Non-triptan switching categories are not mutually exclusive. They do not sum to 100% due to co-medication (receiving 2 or more non-triptan prescriptions on the same day of the between-class switch).
Kaplan-Meier estimates and their corresponding survival functions are shown in Figure 5. The median number of refills to between-class switch for the population was 3, the median number of refills to migraine medication discontinuation was 8, and the median refills to any change in persistency was 1. Median refills could not be estimated for within-class switching, as the 50th percentile of event data for the index population occurred after the 730-day (two-year) observation period. Events predominantly occurred at the time of the first refill, which reflects the rapidly declining proportion of those persistent past the index script. Of specific events, between-class switching occurred at the fastest rate for the population (median refills = 3), followed by discontinuation, and within-class switching occurred at the slowest rate (non-estimatable).
Time to changes in triptan persistency. Survival curves for any change in persistency, between-class switch and discontinuation are shown. The median number of refills to between-class switch for the population was three, to migraine medication discontinuation was eight and to any change in persistency was one. Median refills could not be estimated for within-class switching, as the 50th percentile of event data for the index population occurred after the 2-year observation period.
Discussion
This study found that within a large, insured, US managed-care population, triptan persistency was low during a two year follow-up period. Nearly 54% of patients did not refill their initial tripan after just one prescription. In fact, migraineurs were more likely to discontinue their triptan after their index prescription than at any other time in their prescription refill history. We found that the percentage of patients not persistent past their index triptan was nearly 54%; as compared to the 39% single-time use of triptans among those not on prophylactics found within a similar US-based population in the Etemad et al. study (17). Our triptan persistency results are closer to the single-time triptan utilization results in ex-US populations, which range from 42% to 56% (11–13,15), and also do not differentiate between those on concomitant prophylactic therapies. Furthermore, patients in this study population are representative of US-based triptan users and migraineurs in general with respect to gender, age and comorbid disorders. In line with previous pharmacy studies (17,18), the majority of triptan users were middle-aged females (79%, mean age 38), and observed rates of depression and cardiovascular disease found among this sample rival US population–based averages among migraineurs (24% vs.17%–30% US, 2.28% vs. 2.2%–4% US (23), respectively); enhancing the generalizability of our findings to triptan users at large.
Unlike previous studies, however, the five-year index period defined for the purposes of this research generated a significantly larger sample of newly treated triptan users than any other claims study to date (N = 40,892). Our findings are likely not entirely reflective of the single-time use of triptans among new triptan users, given the terminal behavior classifications assigned at each refill which did not allow for “lapsed” triptan use. However, given the similarity of our triptan persistency results (53.8%) with past triptan utilization results (39%–56%) (13,17), one’s pattern of behavior at the time of their first two migraine refills seems a fairly good proxy for their overall triptan use during the two-year period. Our results, at the very least, support the notion that the majority of newly initiated triptan users refill another acute migraine medication consecutively after initiation of triptan therapy, regardless of whether they return to their index triptan at some later time.
Several reasons have been proposed for low triptan-refilling rates in claims data sources. After finding that less than 15% of patients received more than one triptan product in two years, a recent Italian-based study proposed that the high cost and potential side effects of triptans may play a role, and concluded that prescription data did not appear to “support optimal patient satisfaction with triptan therapies” (14). A small, pharmacy-based study in the Netherlands of single-time users of sumatriptan found that reasons for terminating treatment after only one prescription were lack of efficacy, side effects, uncertain diagnosis of headache and diminished headache frequency (16). Lack of index triptan efficacy, in particular, has been cited as a reason for switching from lower-dose triptans to alternative triptan formulations or other triptan types. The Ifergane study found that “single-time users” of triptans were significantly more prevalent among those prescribed sumatriptan 50 mg than among those using the 100 mg strength (13). Furthermore, our study found that co-medication was a fairly common occurrence (∼17%) among otherwise persistent triptan users, indicating that migraineurs with triptan prescriptions have and may use more than one migraine medication option concurrently during attacks to achieve relief.
Similar to the 9%–14% triptan-to-triptan switching rates found in the literature (11), we also found that within-class switching to non-index triptans occurred infrequently (7.4%) at the time of first refill and throughout the observation period. Our slightly lower percentage may be reflective of our definition of within-class switching, which was dependent on triptan type regardless of dosing or formulation changes. Within-class switching similarly occurred at the slowest rate when compared to “between-class” switching or discontinuation, which seems to indicate a loyal triptan market (2). In other words, if a patient continues to use triptans consecutively past their first prescription, our data support the notion that they are most likely to stay on the triptan initially prescribed to them.
This study has several limitations in both definition and design which deserve mention. Primarily, there is strong potential for the misclassification of non-triptan medications used for migraine, which heightened the sizable proportion of “between-class switching” found among newly initiated triptan users in this study. Because of the various pain-related comorbidities of migraine headache, those migraineurs filling non-specific migraine medications such as opioids and NSAIDs at the time of their first refill were likely doing so for an underlying (non-migraine) condition. In this way, our definition of non-triptan “switching” was sensitive to the presence of a comorbid disorder warranting prolonged opioid or non-triptan use.
Because no minimum criterion was set for migraine medication discontinuation, it became difficult to distinguish those censored from those having discontinued any migraine medication. By design, censoring and discontinuation need to be defined in a dataset given a fixed observation period. If the end of the two-year observation period fell prior to 90 days (the days supplied) following an index triptan refill, that patient was censored at the last refill rather than categorized as discontinuing. This may have resulted in an underestimate of actual migraine medication discontinuation occurring at the end of one's observation period, and at higher refill counts. This point, however, does not change our primary study findings regarding the observed persistency of triptan use. Because it is unknown whether discontinuation may be higher than that recorded here given a longer observation period, it is difficult to estimate how much of a limitation our conservative estimate of migraine medication discontinuation in this two-year period was.
Our sample also did not include those receiving multiple migraine therapies on the index date; these patients would have comprised 7.5% of the larger cohort of triptan users. Triptan persistency may be higher among patients receiving more aggressive initial therapy co-administered with triptans, such that the exclusion of these patients may have biased our sample toward those with mild migraines who are less likely to remain persistent. It similarly can be argued, however, that those prescribed multiple migraine therapies along with triptans at onset are just as likely to opt for non-triptan options at next refill, having already been exposed to a wider range of non-specific, and perhaps less costly, migraine medications. In either case, the observed triptan persistency among this sample at best moderately underestimates actual persistency when including co-administration of migraine medication at baseline; but omitting these patients ensured the sample adequately reflected persistency patterns for the majority of patients who are prescribed triptans as monotherapy.
A major strength of the current study was that no minimum criterion was set for persistency, and we had the ability to capture migraineurs with infrequent or frequent attacks (i.e. those who received many vs. few prescriptions.) A patient could receive the index triptan and receive a refill up to two years later and still be considered persistent to index script, as long as no switching happened in between. In this same vein, however, triptan persistency may have served less as a measure of adherence, and more as a surrogate for the frequency of migraine attack among otherwise “healthy” migraineurs not necessitating opioid, NSAID or analgesic medications throughout follow-up. Certainly, patients receiving only one refill of the index triptan (12.8% of the sample) may not have needed an additional triptan prescription if they experienced infrequent or cyclical attacks only. Similarly, because other triptan formulations such as nasal sprays and injections were included in our analysis, it is arguable that alternate formulations may be packaged with more product than the standard eight pills per triptan refill.
The application of medication adherence analyses using prescription claims data sources for migraine attacks has various drawbacks in general. Popular measures of persistency for other chronic disease, such as the medication possession ratio (MPR) are inapplicable for acute, episodic disorders such as migraine which do not necessitate daily treatment but are only to be treated upon attack occurrence. Etemad and colleagues (17) provided an alternate measure of triptan persistency in their 2005 study describing the utilization pattern of new triptan users within the same claims database. In order to differentiate triptan discontinuation from low migraine attack frequency, the authors correlated early patterns in triptan use within the data set to triptan use for the duration of the observation period. It was found that 39% of new users received only one triptan prescription, but it is unclear whether the four-month interval used can predict a migraineur's future attack frequency. Other medication persistency literature has noted that observation periods longer than one year may be necessary to accurately differentiate treatment gaps from medication discontinuation (20).
The analysis conducted here more closely reflects that of Menckeberg et al.’s study (20), which distinguished patterns in the long-term medication use for asthma using a significantly longer (10-year) follow-up period, and estimated the transitional probabilities of refilling at least one inhalation corticosteroid (ICS) prescription for each year of follow-up and over the total study period (20). The authors differentiated states of use from “continuous” to “irregular” and found that new users of ICS had a lower probability of continuous use and more variability in refill patterns than did previous users; similar to the refill pattern exhibited by new triptan users in this cohort.
An interesting finding of this study was that triptan persistency was at its lowest immediately following the index prescription; the highest loss in refills occurred at this time. This sharp decline in triptan refilling indicates that migraineurs are more likely to discontinue triptans after their initial prescription than at any other time. Lack of efficacy, which is one possible explanation, may be overcome by properly educating patients on the use of triptans early on. Prompt follow-up by the prescribing physician is therefore crucial to maintaining sustained triptan adherence, improving outcomes or identifying alternative treatment options.
Another interesting finding was the rate of “between-class switching” to opioids/NSAIDs at the time of the first refill. In fact, nearly 80% of patients filling a non-triptan prescription following the receipt of their index triptan had filled opioid or NSAID prescription(s) for the subsequent refill. Considering that one-third of migraineurs received opioids or analgesics in the pre-index period for some underlying (and possibly, non-migraineous) condition, this finding is not altogether surprising. The baseline presence of opioid and analgesic prescriptions, coupled with the apparent lack of continued triptan use in the post-index period, suggests that there is strong potential for migraineurs to at least co-medicate with, if not exclusively use, non-specific medications during migraine attacks.
A significant number of migraineurs suffer from comorbid conditions, namely cardiovascular illnesses, stroke, obesity, epilepsy, ulcers, gastroesophageal reflux and psychiatric disorders (24–29,30). It would certainly seem more safe, rational and cost-efficient to take non-specific medications to avert the risk of potential drug interactions. Particularly in the case of cardiovascular and psychiatric diagnoses or associated drug use, there is good reason not to persist on triptans, which pose a cardiovascular risk among those with ischemic heart disease and uncontrolled hypertension (31) and are warned against among those taking SSRIs/SNRIs to treat depression, anxiety and other mood disorders (32). We found that a combined 24% of sample had either diagnosed depression/anxiety at baseline or had received a prescription for an SSRI/SNRI, and a total of 8% had established cardiovascular disease (2.2%) or disease equivalent(s) (5.7%) in the pre-index period. Although this study did not control for the effects of SSRI use, cardiovascular co-morbidities or co-medications to isolate potential predictors of triptan persistency or non-triptan switching, it is possible that some of these patients discontinued triptans due to potential concerns regarding serotonin syndrome or other SSRI/SNRI drug interactions. It is certainly plausible that among a subpopulation of higher-risk migraine patients in this cohort, non-triptan prescription medications are, by necessity, used as a substitution for triptans to treat migraine pain. Given the limited applicability of triptans, situations such as these seem to indicate a shortcoming in current migraine-specific treatments, particularly among those with comorbid disorders.
As mentioned, because pharmacy claims records were not linked to electronic medical records, it is not clear whether NSAIDs, analgesics, butalbital-containing compounds or opioids were prescribed for the explicit purpose of migraine treatment during the observation period. However, given what is known about the rates of opioid use among severely disabled migraineurs and the addictive potential of these drugs (33–35), these data clearly demonstrate that diagnosed migraineurs prescribed triptans receive refills of prescription opioids and NSAIDs following their triptan prescription for treatment of some pain-related issue they currently experience or continue to experience following triptan initiation. The significant opioid refill rates in this study may also indicate that migraine patients are accustomed to non-specific therapies to help alleviate acute migraine pain. Given high baseline opioid and analgesic rates (∼32%) in this large and fairly representative US cohort, there is substantial potential for patients to have developed an increased tolerance for non-triptan pain medications when seeking new solutions to acute migraine pain. Unfortunately, past and recurrent opioid and analgesic use can contribute to the development of chronic daily headache among migraine sufferers (33), and can potentially lower triptan response among those who are accustomed to opioid therapies (36). This, in addition to the substantial co-medication rates (∼17%) observed among otherwise persistent triptan users, may indicate an unmet need for new migraine-specific therapy options among migraine patients, even among those newly initiated on triptans.
To our knowledge, this is the first migraine study to prospectively evaluate triptan persistency patterns within a prescription claims database. While the continuity of triptan refilling was low, the use of non-triptan therapies was high relative to migraine medication discontinuation rates during the observation period among this cohort.
Assuming that those who discontinue triptans continue to experience attacks, the data here suggests that opioid and analgesic prescriptions are both readily available and may be used by migraine patients in the place of migraine-specific treatments. Our results also suggest that new triptan users may opt for non-specific drugs to alleviate migraine pain at the expense of triptan therapy, as seen in their subsequent refill pattern. The identification of underlying reasons for low triptan persistency warrants further research among linked data sources, including claims data, medical records and self-reported patient surveys.
Footnotes
1
Applicable diagnostic and NDC codes can be provided upon request.
Funding
Financial support for this study was provided by Merck & Co., Inc.
Conflict of interest statement
Božena J. Katić is a contractual employee at Merck & Co. Srini Rajagopalan has worked for Merck, Schering-Plough (now Merck) and Wyeth Labs (now Pfizer) as a consultant. Ya-Ting Chen and Henry X. Hu are employees of Merck, in the Global Health Outcomes department, and Tony W. Ho is an employee of Merck & Co, in the Clinical Research department.
