Abstract
The present study assessed age- and sex-specific patterns of migraine prevalence in a US population of 40,892 men, women, and children who participated in the 2003 National Health Interview Survey. Gaussian mixture models characterised the relationship between migraine, age, and sex. Migraine prevalence was 8.6% (males), 17.5% (females), and 13.2% (overall) and showed a bimodal distribution in both sexes (peaking in the late teens and 20s and around 50 years of age). Rate of change in migraine prevalence for both sexes increased the fastest from age 3 years to the mid-20s. Beyond the age of 10 years, females had a higher prevalence of migraine than males. The prevalence ratio for females versus males was highest during the female reproductive/child-bearing years, consistent with a relationship between menstruation and migraine. After age 42 years, the prevalence ratio was approximately 2-fold higher in women.
Introduction
Migraine is common and disabling, and attacks are usually frequent (e.g. 77% of migraineurs have ≥1 attack per month) (1). The condition negatively affects family relationships and productivity (2). Studies evaluating migraine prevalence help elucidate the impact of migraine and identify the true burden of disease in children (3,4). adolescents, and adults (1,5–7). Several epidemiology studies evaluated a narrow age range (1,4,7–15) with fewer assessing prevalence across the life span (1,7,16).
Overall migraine prevalence is ≥2-fold higher in adolescent girls and women than in adolescent boys and men (1,5,7,8,14,16). Although peak migraine prevalence occurs at similar ages in women and men, it has a more gradual rise and decline in the male population (1,16). In females, migraine prevalence increases during adolescence, peaks in the 30s, and declines most markedly after menopause (1,7,16). In women of child-bearing age who are predisposed to migraine, decreasing oestrogen levels around the time of menses is often a key initiator of migraine attacks (i.e. menstrual migraine between days –2 and +3 of menses) (17–20). Estimates suggest that 50–60% of female migraineurs report having menstrual migraine (21).
Epidemiology studies have evaluated short-term (<6 months (4,10)) or long-term (>1 year (1,22)) prevalence rates or have determined the prevalence by country, within a narrow age range, or over the life-time (8,22–25). However, changes in migraine prevalence from childhood through adolescence and into adulthood have not been previously assessed in a single data set. The current analysis evaluated the age- and sex-related migraine prevalence in the US, with emphasis on evaluating the rate of change (acceleration/deceleration) of migraine prevalence by age and sex.
Subjects and methods
Study design
Data were collected from the 2003 National Health Interview Survey (NHIS) conducted by the Centers for Disease Control and Prevention. It includes a Family Core, Sample Adult Core, and Sample Child Core (completed by parent/guardian for children aged <18 years). Non-institutionalised US residents were selected using a 3-stage cluster probability sampling that oversamples blacks and Hispanics (26).
Respondents
Interviewers administered the NHIS in person to one randomly selected adult and child (if applicable) per household. The Sample Adult Core contains health-related questions, including whether ‘a doctor has diagnosed migraine within the past 90 days’. For the Sample Child Core, the parent was asked: ‘During the past 12 months has the child had frequent or severe headaches, including migraine?’ For this analysis, the age range was 3 to 85 + years (those >85 years were classified as 85-years-old).
Statistical analysis
Descriptive statistics summarised population demographics. Migraine prevalence was weighted to arrive at the national estimate using sampling weights from the NHIS. These sampling weights reflect the probability of selection and adjustments for non-response, and were calibrated to the 2000 US Census totals for sex, age, and race/ethnicity. This method of calibration allowed us to generalise the results to the entire US population. Based on the suggestion that sex-specific age prevalence was bimodal in distribution, we applied Gaussian mixture modelling (GMM (27) to the prevalence curves. GMM allows for flexible estimation of the parameters that describe the densities of the populations, namely the mixture parameters (π) the mean distributions (µ), as well as the SD (σ). These component functions are combined to provide a multimodal density. Furthermore, GMM estimated a posterior probability distribution for k component densities, the number of components, given the data and the model. The fit of the GMM was assessed using a maximum likelihood approach. The number of components for each of the mixture models was selected by visually inspecting non-parametric loss curves of the sex-specific age/migraine curves. The curves suggested both distributions were bimodal and approximately normally distributed. The salient characteristics of the GMMs were evaluated by computing and examining the first and second derivatives, corresponding to the velocity and the acceleration of prevalence over the age continuum. Finally, the ratios of the models, expressed as both densities and prevalences, were computed to examine relative risk and absolute risk, respectively. The first and second derivatives of the derived functions allowed computation of the velocity (change/time) and acceleration of prevalence across all ages for each sex. Age group categories were based on the rate of change of the fitted functions using comparisons of the ratio of density to migraine prevalence across ages. Prevalence ratios and likelihood ratios of migraine were determined (see equations below).

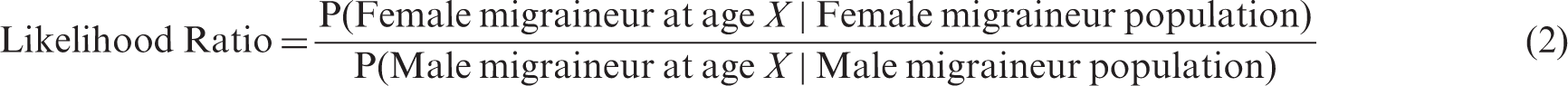
For likelihood ratios, the numerator corresponds to the migraine density at age X for women and the denominator provides migraine density at age X for men. In contrast, the prevalence ratio (females:males) determined migraine prevalence at age X for women divided by the migraine prevalence at age X for men. The likelihood ratio (density) and prevalence ratio provide distinct and unique information on what is happening within the population. Stata® v.10.0 (StataCorp LP, College Station, TX, USA) and R v.2.7 (R Foundation for Statistical Computing, Vienna, Austria) were used for the GMM analysis; the results from the two algorithms were compared. SE was estimated using Taylor series linearisation because of the clustered sampling design.
Results
Demographics
The NHIS included 35,921 households that contained 92,148 individuals in 36,573 families. Interviewers administered the questionnaires to 43,101 individuals (30,852 adults, 12,249 children). Our analysis includes data from 40,892 respondents (excluding missing values or non-evaluable responses). Given the large sample, migraineurs were statistically older than non-migraineurs (0.9 years; 95% CI, 0.2–1.5; P = 0.007; t = 2.7); however, the mean (SE) ages of the migraineur (men, 36.2 [0.5] years; women, 38.2 [0.6] years) and non-migraineur (men, 36.4 [0.2] years; women, 38.3 [0.22] years) populations were similar.
Prevalence of migraine
The weighted migraine prevalence was 8.6% in males (95% CI, 8.1–9.2), 17.5% in females (95% CI, 16.9–18.2), and 13.2% overall (95% CI, 12.8–13.7; Figure 1). Women showed a higher migraine prevalence (Figure 2A), especially between 20–40 years old (1.5- to 2.9-fold higher vs men). From the age of 10 years to the end of life, females had a greater prevalence of migraine than males (Figure 2B). The greatest difference in prevalence ratio occurred at 30.2 years; beyond 42 years of age, the prevalence ratio was approximately 2-fold higher in women than in men. For both sexes, migraine prevalence was highest in the peak reproductive years.
One-year period prevalence (95% CIs) of self-reported, physician-diagnosed migraine in the US population. (A) Age-specific, 1-year period prevalence of self-reported, physician-diagnosed migraine. (B) Prevalence ratios. For each age, prevalence for women was divided by the prevalence for men.

The broad range of ages surveyed in this sample, coupled with our modelling strategy, revealed a bimodal distribution of migraine in both sexes. Two separate populations of migraineurs with peak prevalences that differed by sex were apparent (Figure 3A,B). The SD for the younger population was smaller in both sexes than in the second (older) population. For women, the peak periods for migraine risk were at a mean ± SD age of 25 ± 8.6 years and 50 ± 15.8 years (Figure 3A). Approximately one-third of the total female migraineur population was contained within the first population, and there was an overall higher density probability for migraine risk than in the second population at its peak. For men, the peak risk periods (18.7 ± 7.4 years and 47.6 ± 16.8 years) were earlier than in women, and both peaks were approximately of the same magnitude (Figure 3B).
The probability density curves and migraine prevalence across the life span in (A) females and (B) males.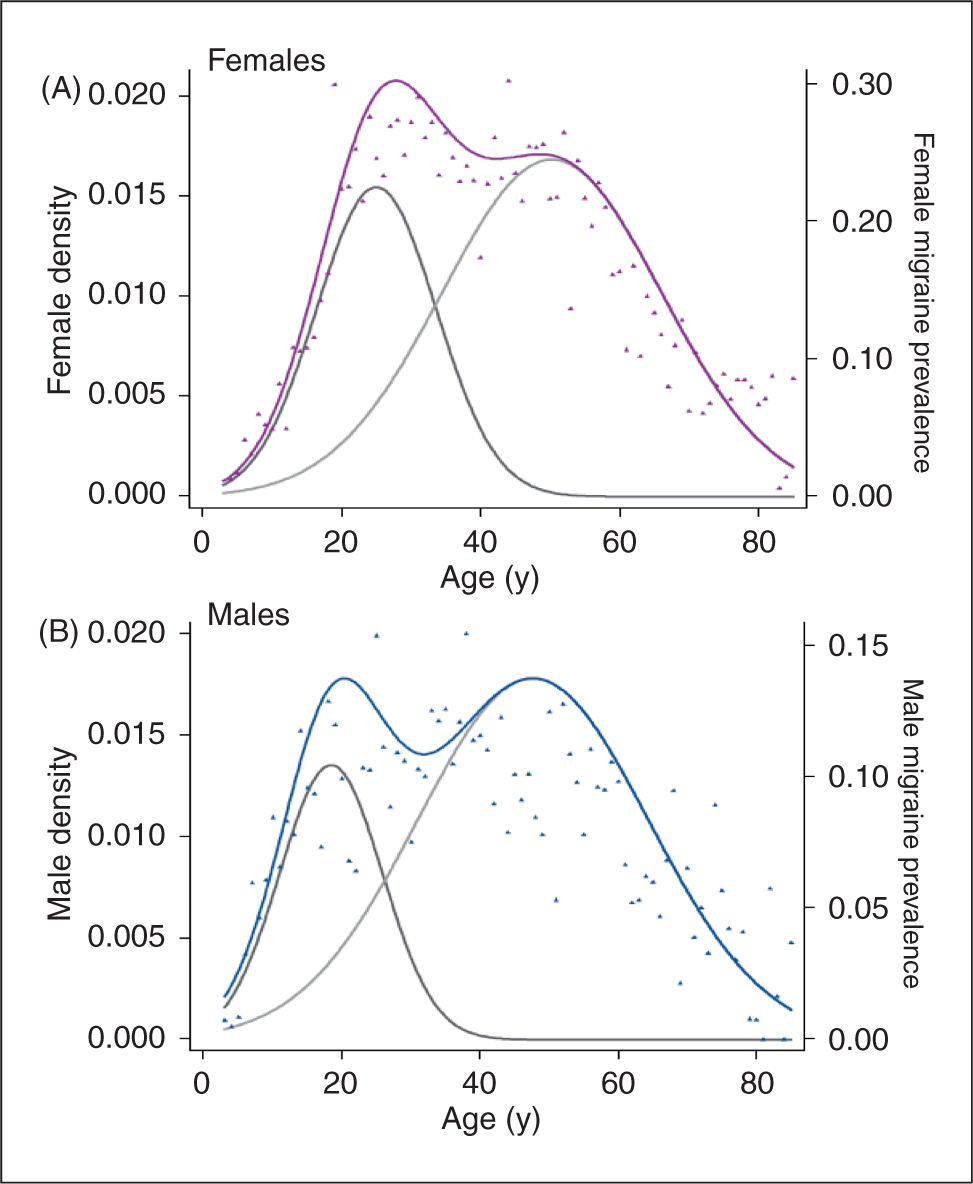
Analysis of the rate of change (first derivative) in migraine prevalence (Figure 4A,B) showed that the fastest increase (velocity) in prevalence occurred from 3 years old to the early 20s (men) or mid-20s (women). In women (Figure 4A), the rate accelerated up to approximately 20 years of age, when it slowed its acceleration through the mid-20s. By the mid-30s, the rate decelerated and became slower than at any other time point previously. At around 40 years old (i.e. perimenopausal period), there was an acceleration of the rate again, but it still stayed below the rate of change seen from childhood through adolescence. Although the relative changes in the first derivatives were similar between sexes, the ages in which the rate changes occurred were different. In men (Figure 4B), the rapid decline in the acceleration occurred during the mid-teens, and by 20 years old the rate was decelerating, earlier than in women. However, during the late 30s to early 40s, migraine prevalence in men showed a much more profound acceleration than migraine prevalence in women within the same age range. In both sexes, in the mid-60s the rate of migraine continued to fall at an increasing rate.
The rate of change (first derivatives) in prevalence over the age continuum in (A) females and (B) males. *The horizontal line at 0 on the y-axis indicates no change (i.e. acceleration or deceleration) in the rate. †During childhood and early adolescence, the rate is accelerating quickly. ‡Rate begins to slow its acceleration. §Rate is decelerating.
The probability densities revealed differences in the risk of migraine at various ages for both sexes (Figure 5A). Among male migraineurs, there was a greater density of migraine at <22 years old than for female migraineurs of similar ages (i.e. leftward shift of the male probability density curve; Figure 5A). The likelihood ratio (i.e. female/male density ratio) also showed that from 22–42 years old, women had a greater likelihood of migraine than men (Figure 5B). At 22, 42, and 56 years of age, the densities were identical between sexes; after 42 years old, the likelihood ratio remained similar between sexes.
(A) Probability densities for men and women. (B) Female/male likelihood ratio (ratio of probability densities). *Densities are the same at these ages.
Discussion
Age-specific migraine prevalence appears quite dynamic as reflected by the large changes in the rate of acceleration with sex and by age. Because migraine prevalence is a function of incidence (i.e. new cases) and remission, migraine prevalence is increasing when incidence is greater than remission and decreasing when remission out-paces incidence. The rate of acceleration of the prevalence rate was highest during the teenage years in both boys and girls. These data are consistent with recently reported migraine incidence data: <25% (boys) and <15% (girls) of new-onset cases occur before 14 years old, with peak migraine incidence at 24.1 years (men) and 25.2 years (women) (28). Thus, both sexes showed a bimodal prevalence distribution, peaking in the mid-20s and around 50 years old for women (slightly earlier for men). Women had an overall ≥2-fold higher prevalence than men, which became apparent in the later teenage years and continued throughout life. Boys and male adolescents <22 years old had a higher density of migraineurs than did similarly aged girls; this was reversed after 22 years old. The reproductive/child-bearing years in women showed the highest prevalence rates and prevalence ratio, especially during the 20s and 30s. Men also showed the highest migraine prevalence during the reproductive years, although migraine prevalence was lower than in women at all postpubertal ages.
A strength of this analysis was that the NHIS contained a broad age range (3–85 years), which helped us identify a bimodal distribution in migraine prevalence in both sexes. The peaks occurred later in women than in men. When comparing shapes of the prevalence curves, the peak in women was higher in the first versus the second population; the broad and relatively flat second peak might validly be explained by the oestrogen fluctuations during the perimenopausal phase (mean menopause age, 53 years in the U (29)), especially because the second peak was more of a decrease in the rate of decline rather than an increase in prevalence. In men, however, both peaks have approximately equal size, although the second population was more broad, which suggests that male migraineurs are a heterogeneous group.
The reasons for the second peak in prevalence in men is uncertain, though research in this area is evolving. Emerging data suggest that there are age-related hormonal changes in men, which might contribute to the second prevalence peak. Between the ages of 40–80 years, total testosterone (the major substrate for oestradiol production) decreases slightly (30). Changes in total oestradiol levels are complex and vary with body mass index. Free oestradiol levels have been shown to decrease with age in men in middle life (31). The decline in bioactive oestradiol might contribute to the second prevalence peak in middle-aged men, though this is highly speculative.
Previous studies of migraine prevalence have not shown the bimodality reported here (1,5,7,16,24). Methodological differences might account for the lack of correspondence. Many of the previous studies examined age in 5-year categories; here, we fit curves to each year of age. Perhaps the larger age groupings removed variability in the data, decreasing sensitivity to bimodality. Second, the NHIS included a broad age range (3–85 + years), which reduces the bias associated with estimating prevalence when the age range is more limited. Other epidemiology studies limited the age ranges to 16–65 years, ≥15 years, and ≥12 years (1,7,16,32). Some epidemiologists (33) and other methodologists (34,35) have suggested that truncation of age range, especially when the disease affects younger individuals, may introduce serious bias in risk estimates for that disease. Limiting the age range may have attenuated the bimodality of the distributions. Support for these effects comes from our group, which used the same NHIS data plotted to a curved-fitted function with a truncated age range (18–85 years). Using this methodology, migraine prevalence peaked at 34 years old for both sexes and only displayed a single peak across the life span (36). In contrast, in the current analysis, when the full NHIS age range was used, without a curved-fitted function, a bimodal distribution in prevalence was seen.
Our analysis has some potential limitations, including the cross-sectional nature of the data. Migraine prevalence at any given age is determined by incidence and duration of illness or remission. In a cross-sectional study, these factors are difficult to disentangle. Another limitation is that in the NHIS, parents reported migraine, or other severe headache, in their children; in younger children, evaluation of migraine may be more difficult than in older children who can accurately report their symptoms to parents and doctors. For adults, self-report of migraine was used (‘if a doctor had diagnosed migraine within the past 3 months’). Some researchers have used a migraine definition based on respondent self-reported physician diagnosis (32), whereas others have used an International Classification of Headache Disorders-II (ICHD) criterion (1,5,7,16). Self-report could introduce bias because of misdiagnosis or underdiagnosis. Misdiagnosis was unlikely because evidence indicates that 98% of patients with a diagnosis of migraine by primary care physicians actually had migraine (37). Underdiagnosis may be a limitation because those patients would be missed by the NHIS. In one study, underdiagnosis by primary care doctors occurred in one of four patients with ICHD-defined migraine (37). Furthermore, patients who never seek medical attention but have ICHD-defined migraine (38) or those who were diagnosed or had medical treatment for migraine before the 3-month window (in the NHIS) would be missed. Although it is possible that migraine may have been over-reported in our study, all the available data on this subject support under-reporting of migraine. Thus, the current estimation of migraine prevalence may be lower than actual prevalence because of the reliance on physician diagnosis in the NHIS. Nonetheless, our overall migraine prevalence of 17.5% for women and 8.6% for men is within the range reported by others (Lipton et al., (1) 17.1% [women] and 5.6% [men]; Jette et al., (32) 15.2% [women] and 6.1% [men]). Despite some differences between studies, women clearly experienced migraine at 2–3 times the rate of men, and their reproductive/child-bearing years were when they were most susceptible (1,32)
As noted, the questions for migraine were different for children (i.e. migraine within 1 year) and adults (migraine within 3 months), raising the issue of whether it is valid to combine the data. Because of the shorter time interval for the adult query, migraine may have been under-reported more in adults in comparison with the responses for the children (i.e. in those aged >18 years vs <18 years). However, a large difference in under-reporting would have been expected to result in the initial peak prevalence occurring at age 18 years, but this was not observed; indeed, the first peak occurred after age 20 years in both sexes (Figure 2A). Thus, the differences in methodology for children and adults appear unlikely to have had a large affect on the validity of our results.
In our study, the prevalence ratio of migraine increased from the age of 3 years to approximately 20 years in both boys and girls. Deceleration of the rate occurred after age 20 years in both sexes. Adolescent girls and women had a higher prevalence of migraine from 10 years old onward (vs males). These data are not surprising, given that there is a substantial body of evidence linking menstrual cycling and migraine in females (17–19) and that the average age of menarche in girls in the US is 12–13 years old (39).
The data in children are consistent with other studies. A study in Turkey in 2002 found that migraine prevalence increased yearly in boys and girls from 7 to >10 years old (3) . A German study in 7–14-year-olds found that headache prevalence increased from childhood (39%) through early adolescence (63%; P < 0.001), and migraine appeared to start at a younger age (7.1 years) than tension headaches (7.9 years; P < 0.001) (4). Moreover, although pubescent girls showed a more substantial increase in headache frequency by 13 and 14 years old versus boys (P < 0.001), boys actually experienced headaches at a slightly earlier age (7.3 years) than girls (7.8 years; P < 0.001) (4). Another study showed that although 12-year-old boys and girls had similar migraine prevalence, by 13, 14, and 15 years old, girls showed higher migraine prevalence (increasing yearly) (10). These international studies are consistent with the NHIS (i.e. density curve was shifted left in boys <22 years old vs girls of similar age). This shift indicates that, among migraineurs, boys had a greater likelihood for migraine at earlier ages than similarly aged girls. Our data support similar findings that migraine prevalence in adolescent girls accelerates quickly (4). Similarly, migraine incidence data shows that <25% (boys) and <15% (girls) of new-onset cases occur before 14 years old, with peak migraine incidence at 24.1 years (men) and 25.2 years (women) (28).
Many female migraineurs (50–60%) experience menstrual migraine (21). Women with pure menstrual migraine experience migraines only between days –2 and +3 (perimenstrual period), whereas women with menstrually-related migraine regularly experience migraine during the perimenstrual window, as well as at other times of the cycle; the term menstrual migraine encompasses both pure menstrual migraine and menstrually-related migraine. Our analysis of the NHIS data indirectly supports that the differences seen in migraine prevalence between men and women during the child-bearing/reproductive years (puberty to approximately 50 years old) might be accounted for by the increased incidence of menstrual migraine attacks during this period. For example, if the NHIS migraine prevalence rate for female migraineurs was reduced by 60% (i.e. by removing women with menstrual migraine), the 2–3-fold difference seen between females and males in migraine prevalence would be reduced to 0.8–1.2-fold. Menstrual migraine is an important health issue because menstrual migraine attacks may be more severe, longer in duration, and more difficult to treat than non-menstrual migraine attacks (17,18,40). Women report that menstrual migraine attacks affect productivity more than non-menstrual migraine attacks (41). Female migraineurs in their prime working years, which roughly corresponds to the reproductive/child-bearing years (when menstrual migraine is most common), experience more days in which their activity was restricted when compared with men (16). Because of the pain, symptoms, and other effects (e.g. decreased productivity), migraine can have profound effects on the psychological well-being of women (42). Unfortunately, migraine and menstrual migraine continue to be underdiagnosed (1,38) Therefore, better identification of migraine in women is needed so that they receive effective therapeutic options.
Conclusions
This study supports the contention that migraine has breadth across multiple age groups. We found a bimodal distribution of migraine prevalence, suggesting that there may be two different groups of men and women, based on age, who differ in some aetiological or mechanistic factor that increases migraine risk. The data showed that migraine prevalence accelerated rapidly from childhood through adolescence in both sexes; a decelerating rate occurred in the early (men) or late (women) 20s. Both sexes showed a transient increase in the prevalence rate during their 40s; in women, this second peak was lower than the peak in the 20s. This study confirms the results of other studies that showed that women had the highest migraine prevalence during their reproductive/childbearing years and that overall prevalence was 2-fold higher in women versus men. A unique aspect of this analysis was the identification of a bimodal distribution in migraine prevalence for both sexes and that the migraine prevalence ratio varied substantially across the life span. Finally, the results reported here are likely generalisable to the US population because the NHIS was conducted by the Centers for Disease Control and Prevention with the goal of being representative of the US population.
Footnotes
Acknowledgements
This research and the funding for manuscript development was supported by Endo Pharmaceuticals Inc., Chadds Ford, Pennsylvania, USA.
TWV was employed by Endo Pharmaceuticals Inc. at the time this research was conducted. XH and JCC are current employees of Endo Pharmaceuticals Inc. DCB has collaborated with or worked for the following companies as a research consultant: Endo Pharmaceuticals Inc.; Allergan Pharmaceuticals; Merck & Co., Inc.; and Ortho McNeil Neurologics. RBL has worked with the following companies (currently or in the past) on advisory boards, speakers bureaux, and research or educational grants: Advanced Bionics; Allergan Pharmaceuticals; AstraZeneca; Boehringer-Ingelheim; Bristol-Myers Squibb; Cierra; Endo Pharmaceuticals Inc.; GlaxoSmithKline; Minster; Merck & Co., Inc.; Neuralieve; Novartis; OrthoMcNeil Neurologics Inc.; Pfizer Inc; Pozen Inc.; ProEthics, Ltd; and St Jude Children’s Research Hospital.
The authors would like to acknowledge manuscript preparation assistance provided by Kristine W. Schuler, MS, and the editorial support provided by Kevin Ryder, PhD, of Complete Healthcare Communications, Inc., Chadds Ford, PA.
