Abstract
Repetitive low-frequency electrical stimulation (LFS) induces pain inhibition in healthy volunteers and in animals, but it is unknown whether it has an analgesic effect in patients with headache. The aim of this study was to investigate if LFS could induce prolonged pain inhibition, called long-term depression (LTD), in patients with chronic tension-type headache (CTTH). Twenty CTTH patients and 20 healthy volunteers were exposed to 20 min LFS (1 Hz) to the forehead. LTD was measured as a decrease in pain response to electrical stimulation in a 1-h post-LFS period following LFS. The LFS induced a significant and stable inhibition of pain (LTD) both in patients with CTTH (post-LFS average decrease in pain rating: 19.6 ± 3.9%, all P < 0.005, Holm–Sidak) and in healthy controls (30.1 ± 5.0%, all P < 0.001, Holm–Sidak). During the LFS period, the pain ratings decreased consistently in both groups. In conclusion, a significant and stable pain inhibition (LTD) can be induced in CTTH patients by LFS.
Keywords
Introduction
Tension-type headache (TTH) is the most common kind of headache. Approximately 30–40% of the population experience frequent episodic TTH, while 3% has chronic tension-type headache (CTTH), i.e. daily or nearly daily headache (1,2). Thus, this disorder is responsible for a considerable loss of quality of life and has a large socio-economic impact. Because of the limited knowledge of the mechanisms leading to CTTH, attempts to develop an efficient treatment with few side effects have so far been unsuccessful (3,4).
Several studies have documented that CTTH patients have altered pain perception. They experience stimuli more intense than healthy people, reflected as lower pain thresholds and higher pain ratings to suprathreshold stimuli (5). This has been demonstrated as increased sensitivity to many different stimuli delivered at various locations and tissues – pressure, thermal, electrical, intramuscular infusion of hypertonic saline and intake of glycerol trinitrate (5–9). This difference is most likely caused by increased excitability of central pain pathways, so called central sensitisation (3,10,11). However, deficient descending pain inhibition may also play a role in CTTH (12,13).
Low frequency stimulation (LFS) has proven robust at inducing a prolonged state of inhibition, known as long-term depression (LTD), in animals and healthy subjects (14,15). Suprathreshold, low-frequency electrical stimulation of the orofacial area in healthy humans induces a long-lasting reduction of pain scores and suppression of the blink reflex (16,17), the masseter inhibitory reflex (18) and somatosensory evoked potentials (19). The most efficient stimulation frequency to induce LTD has been found to be 1 Hz (20). The effect of LFS on pain rating and brainstem reflexes has, to our knowledge, never been investigated in chronic pain patients. Studies addressing the effect of LTD on clinical analgesia have been requested (21).
The primary aim of this study was to investigate whether LTD can be induced in patients with CTTH. Secondary aims were to investigate the effect of LFS on the nociceptive blink reflex as a measure of nociceptive modulation at the brainstem level and to compare the effect of LTD between patients with CTTH and healthy volunteers.
Subjects and methods
Participants
The study cohort was 20 patients with a diagnosis of CTTH and 20 age and sex matched healthy volunteers. The patients were recruited from an out-patient headache clinic (Danish Headache Centre, Glostrup, Denmark) and had completed a 4-week headache diary. CTTH was diagnosed according to the International Classification of Headache Disorders (ICHD-II) (2). Participants were recruited between July 2008 and September 2008. Patients included in the study were aged between 18 and 65 years. Exclusion criteria for patients were more than one attack of migraine per month, pregnancy/nursing, serious somatic or psychiatric illnesses, medication-overuse according to ICHD-II, daily intake of medicine to prevent headache and/or antidepressant medicine and intake of analgesics on the day of examination, or 12 h before. Healthy volunteers had an inclusion criterion of between 18–65 years of age, and they had the same exclusion criteria but were not included if they were employed at the neurological department at Glostrup Hospital or had a history of more than 12 headache days per year. Demographic data such as age, sex, hand dominance, weight, height blood pressure and years with CTTH were recorded. All patients underwent a physical and neurological examination. All participants gave written informed consent to participate in the study, which was conducted in accordance with the Declaration of Helsinki and approved by the local ethics Committee.
Electrical stimulation and blink reflex recording
Each electrical stimulus had duration of 1 ms and was delivered by the Aδ-specific concentric electrode (22), on the non-dominant side of the forehead. The electric pain threshold (Ip ) was determined according to the method of limits (23). A painful stimulus was defined as a sharp/pricking painful sensation. The intensity used to elicit the blink reflex and during LFS was 4 times the Ip , called IS . Thus, the IS was used as a test stimuli to rate pain, to elicit the blink reflex and to induce LTD.
Pre- and post-LFS, the stimuli were given as a series of 10 stimuli with an inter-stimulus interval of 9–11 s, to evaluate the sensitivity to electrical pain and to record the blink reflex. A series was started every eighth minute and participants were asked to rate the average pain of all 10 stimuli after each series. Perception of pain to electrical stimuli was assessed on a verbal rating scale (VRS) where 0 corresponded to no pain and 100 to the worst imaginable pain.
No immediate warning was given before each stimulus but, 5–10 s before start of a series, it was announced. Participants were asked to rest with closed eyes during recordings.
LFS was given for 20 min with the same electrode, stimulation site and intensity of stimulation as described above, but with a frequency of 1 Hz. The participants were asked to rate the pain from the electrical stimuli every minute during LFS. The BR was not recorded during LFS.
Keypoint Workstation (Alpine Biomed Corp., USA) was used both to stimulate and to record the EMG response of the orbicular oculi muscles. Data were recorded by the software Keypoint.Net (Alpine Biomed Corp.). Upon completion of the study, Keypoint.Net was also used to read the latency, duration and integral of the R2 components of the blink reflex. The reading of the reflex was done blinded, both to time of recording and to subject.
Sensitivity to pressure
Total tenderness score (TTS) of the pericranial myofascial tissues was recorded manually with rotating palpation according to the Total Tenderness Scoring system, which has previously proved to be reliable (24). Eight pairs of muscles and tendon insertions (masseter, coronoid, temporal, frontal, mastoid, sternocleidomastoid, neck and trapezius muscle insertions) were palpated. Tenderness was scored on a 4-point (0–3) scale bilaterally at each location and was summated. Pressure pain threshold (PPT) was recorded on the dorsum of the second middle phalanx (PPTfinger) and at the temporal region (PPTtemple), three measurements for each time point, at each place. An electronic pressure algometer (Somedic AB, Stockholm, Sweden) was used to measure pressure pain threshold. A circular stimulation probe (0.5 cm2) and a pressure-loading rate of 10 kPa/s were used. The algometer and the method used are described in detail elsewhere (6). Local tenderness (LTS) was scored on a 0–100 verbal rating scale after a prespecified pressure of 180 arbitrary units by means of a palpometer (25) by small rotating movements of observer’s second and third fingers onto the masseter muscle. The LTS pressure was applied 2 cm caudal from the middle of the line between the mandibular angle and the external canthus.
Procedure
TTS was recorded bilaterally, while participants were sitting. PPT and LTS were recorded on the non-dominant side, while the participants were in the supine position on an examination couch in a quiet room. Dominant side was decided according to handedness. The recordings were performed by the same observer (KL).
The electrode was fixed to the skin over the supra-orbital nerve on the non-dominant side. The electrode was placed where the participant had the strongest sensation of the stimuli (to ensure close proximity to the supra-orbital nerve). The forehead and peri-orbital skin were cleaned with a paste of glycerol and pumice stone (Elektrodepasta; Hospital Pharmacy, Denmark). The recording surface electrodes were placed bilaterally, lateral to the eyes and infra-orbital. A ground electrode was placed between the eyes.
Sensitivity to pressure was determined. A series of blink reflex recordings was started after threshold detection. After three stable blink reflex recordings, LFS was started. Upon completing the LFS period, 8 series of blink reflex recordings were made and pain of the electrical stimuli was rated during the 1-h post-LFS period. After blink reflex recordings, electrical thresholds were detected and sensitivity to pressure determined (Figure 1).
Study design. Three stable blink reflex (BR) series were recorded (Pre) before starting the 20-min 1-Hz low-frequency stimulation (LFS) period, with stimulation intensity of 4 times the pain threshold. After a 5-min resting period, the first of 8 (Post) BR series was started. There were 10 stimuli in a BR series with an inter-stimuli interval of 9–11 s. BR series every 8th min.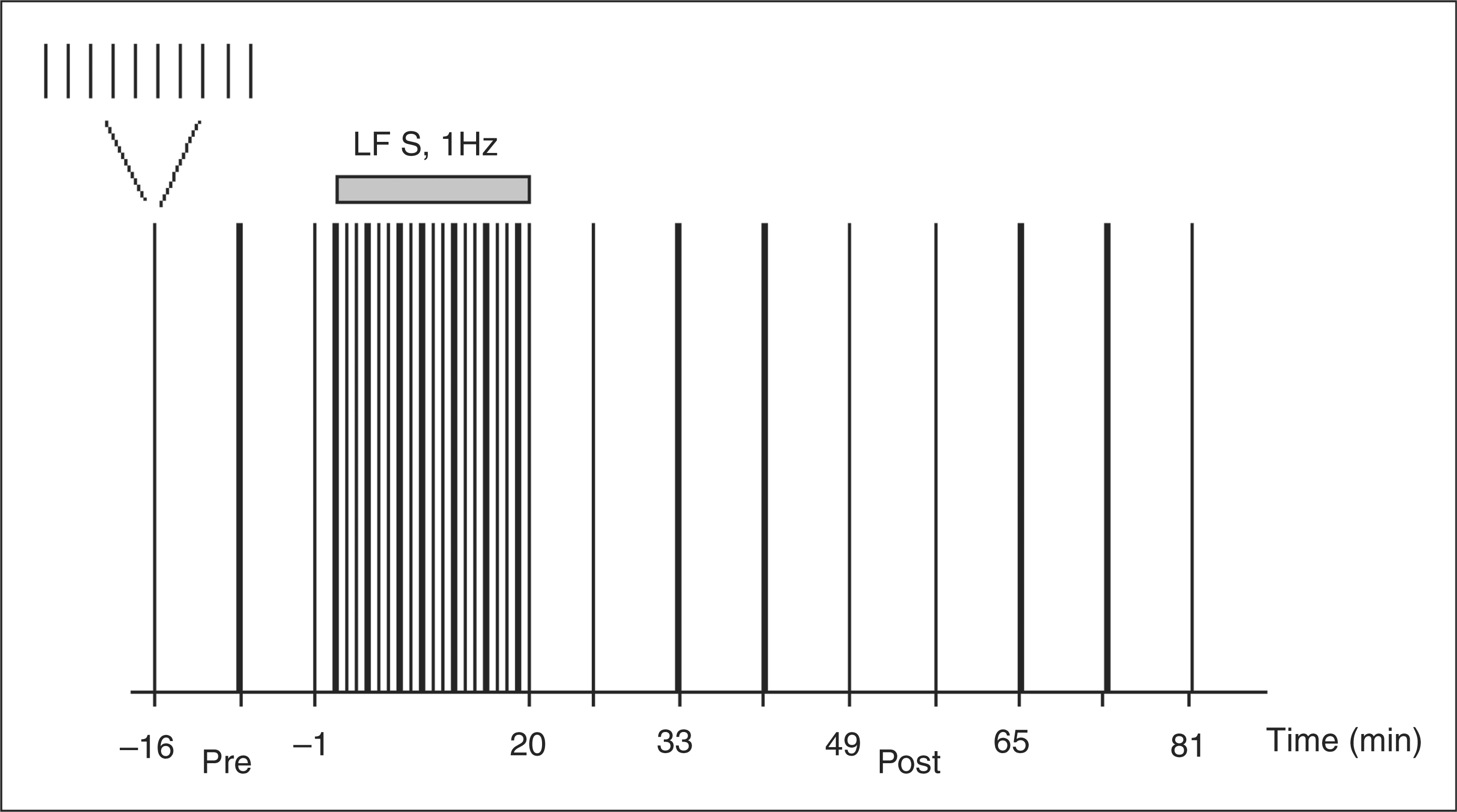
Statistical analysis
The primary end-point was the percentage change in pain score to suprathreshold electrical stimuli (overall rating of the 10 sweeps in a series) from baseline to the post-LFS period in patients with CTTH. Pain score at baseline and in the post-LFS period were calculated as average of the 3- and 8-series, respectively. Secondary end-points were difference in pain scores from baseline to post-LFS period in healthy volunteers, difference in pain reduction between groups and the post-LFS reductions in blink reflex integral (percentage change from baseline) within and between groups. Baseline data for the pain rating were calculated as the average of the pain score of the 3 stable series before the LFS.
This was tested with two-way repeated measurement ANOVA (subject, participants; factor, CTTH, HV; factor time, time –1, 25, 33, 41, 49, 57, 65, 73, 81 min). Further secondary end-points were the percentage change from baseline in blink reflex duration and latency within and between groups. These parameters were tested as described for primary end-point. PPT, TTS, LTS, Ip , VRSstim and IS were tested as explanatory end-points with independent or paired t-test or Mann–Whitney test; this was done within groups and between groups. Parametric or non-parametric statistics were used if the distribution of data was normal. Statistical analyses were performed with SigmaStat v.3.5 software (Systat Software Inc., USA) or SPSS v.15.0 software (SPSS Inc., USA). A significance level of 5% was chosen.
Results
Demographic data of chronic tension-type headache patients (CTTH) and healthy volunteers (HV)

BMI, body mass index; TTH, tension-type headache.
Pain parameters
The LFS induced a significant relative pain reduction in the post-LFS period both in CTTH patients (19.6 ± 3.9% [mean ± SEM], P < 0.005, Holm–Sidak) and the healthy volunteers (30.1 ± 5.0%, P < 0.001, Holm–Sidak) with no difference between the groups (n.s., Holm–Sidak). Between the groups, the post-LFS relative pain reduction was significantly larger in the healthy volunteers than in the CTTH at 3 of the 8 recordings (time: 33, 41, 81 min), but not overall (Figure 2A). In absolute values, the average reduction in pain score from baseline ratings to post-LFS in CTTH patients was: 58.0 ± 3.0% to 46.9 ± 3.6% (all P < 0.001, Holm–Sidak) and in healthy volunteers 39.9 ± 3.4% to 26.9 ± 2.9% (all P < 0.001, Holm–Sidak). The absolute pain rating was significantly higher in CTTH patients than the healthy volunteers at each time point (P < 0.001, Holm–Sidak; Figure 2B).
(A) Pain score (percentage of baseline) of the electrical forehead stimulation over time. Pain was rated as average of the 10 stimuli in a series after each series was completed. Time 0–20 min was during low-frequency stimulation. (B) Pain score, absolute values. Tested with 2-way ANOVA, Holm–Sidak; statistical significance (P < 0.05) are marked with asterisks. VRS, verbal rating score; n.s., not significant; CTTH (n = 20), chronic tension-type headache; HV (n = 20), healthy volunteers.
During the 20-min LFS period, the pain rating decreased consistently and significantly in the CTTH patients (time 1, 65.3 ± 4.0%; time 20 min, 43.6 ± 5.4%; P < 0.001, Holm–Sidak) as it did in the healthy volunteers (46.5 ± 4.0% to 25.6 ± 4.0%; P < 0.001, Holm–Sidak; Figure 3).
Pain score, absolute values, of the 20-min low-frequency (1 Hz) electrical forehead stimulation. Pain was rated verbally every minute on a 0–100 scale. CTTH, chronic tension-type headache; HV, healthy volunteers.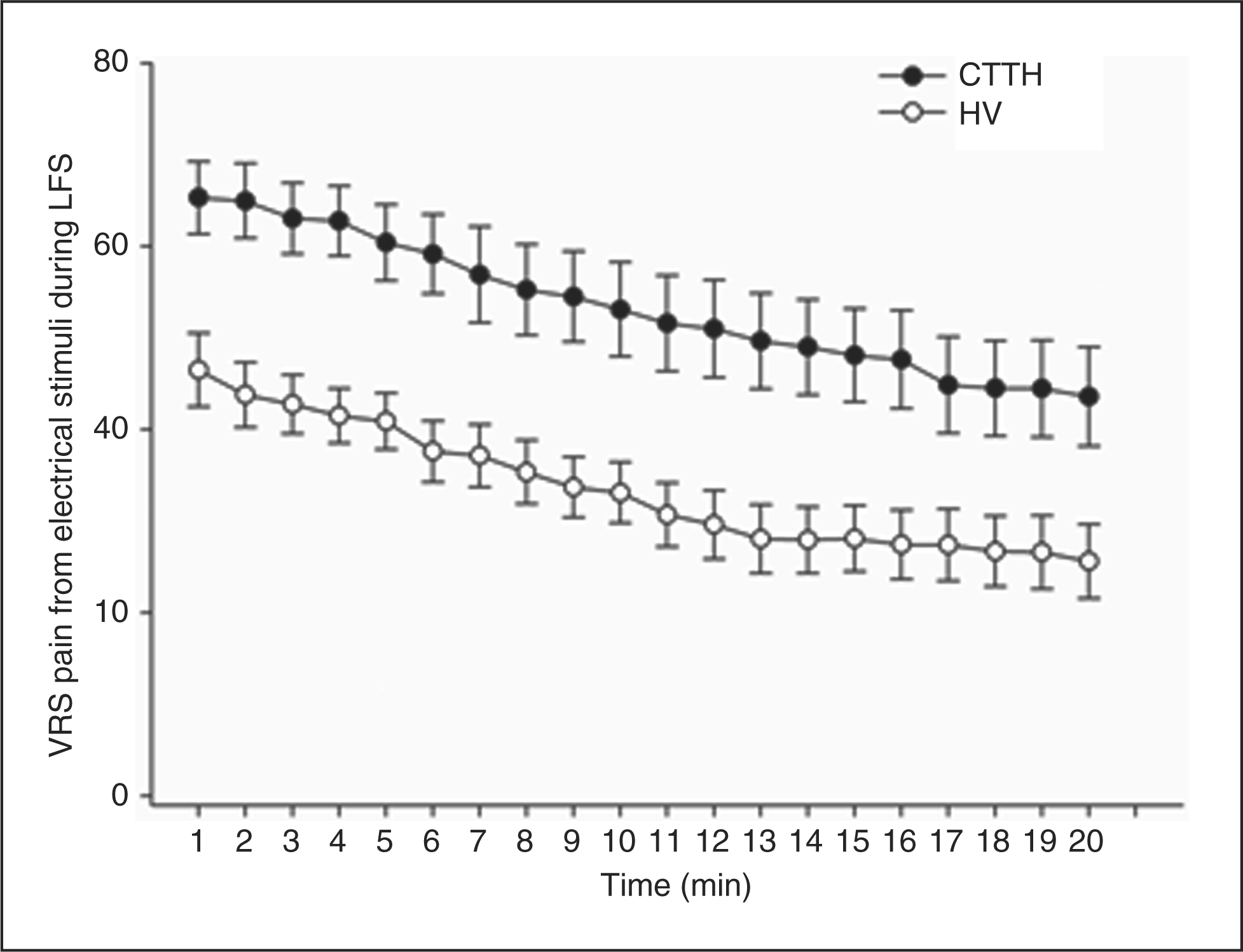
Nociceptive blink reflex
The LFS induced a significant blink reflex integral reduction in the post-LFS period in CTTH patients (ipsilateral, 25.4 ± 2.2%; contralateral, 18.5 ± 2.8%; both P < 0.01, Holm–Sidak) and in healthy volunteers (25.2 ± 1.8%; 28.6 ± 3.1%; both P < 0.001, Holm–Sidak). There was no significant difference in percentage change from baseline in blink reflex integral between CTTH and HV during the different time points on either side (Figure 4).
Blink reflex integral (percentage of baseline) of the R2
component over time. Ipsilateral side, recording on same side as stimulation electrode (non-dominant hand). Bilaterally in both groups the post-LFS recordings (time 25–81 min) was significantly smaller than the baseline recordings (time –16 to 1), marked with asterisks, tested with 2-way ANOVA, Holm–Sidak. CTTH, chronic tension-type headache; HV, healthy volunteers.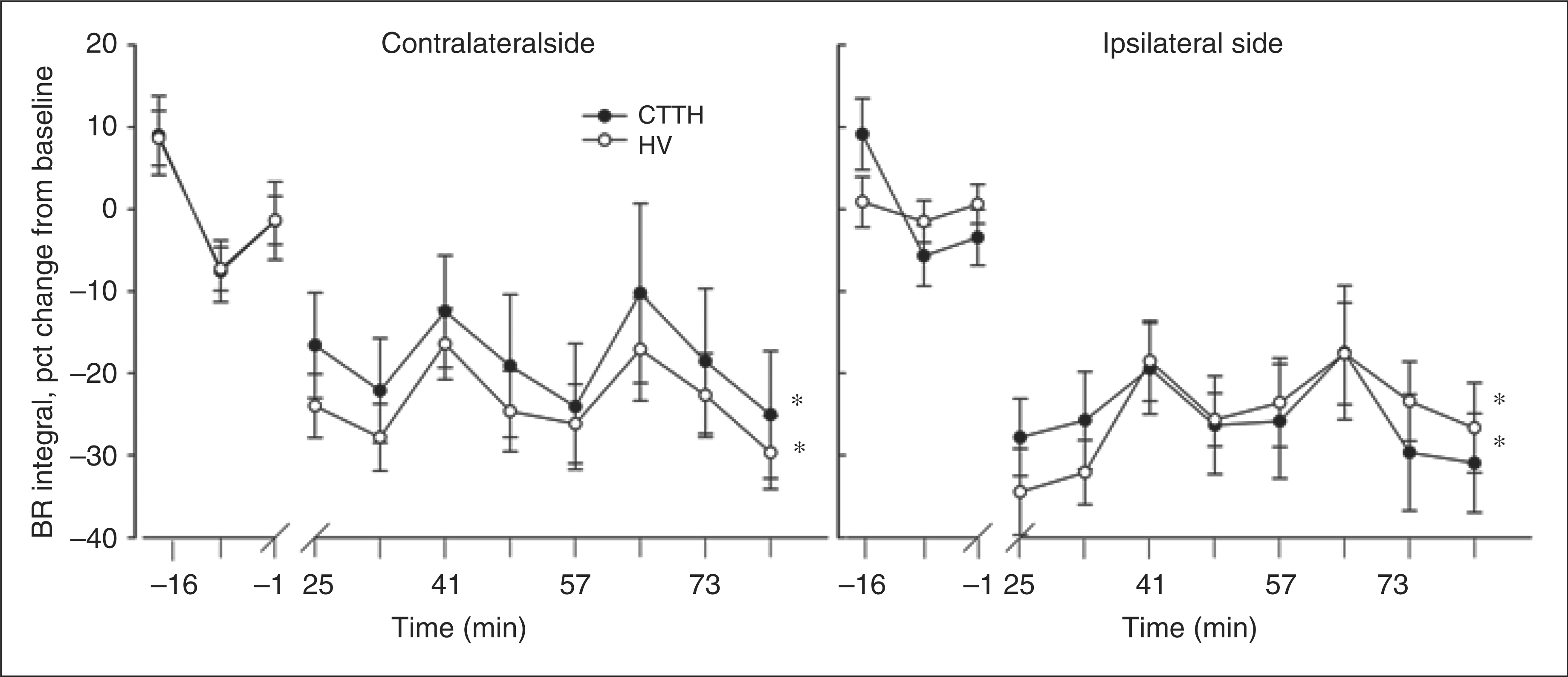
There was no overall significant difference in the percentage change from baseline of the latency or duration within groups or between CTTH patients and healthy volunteers on either side.
Additional end-points
The habitual tension-type headache of CTTH patients did not change during the study. At the beginning of the study, the VRS median (quartiles) was 32.5 (17.5–55.0) and at the end it was 37.5 (17.5–52.5), (n.s., Mann–Whitney).
There was no significant difference between absolute stimulation intensity between CTTH (4.1 ± 0.3 mA) and healthy volunteers (4.8 ± 0.4 mA), (n.s., –1.8 to 0.3, t-test), but the pain rating of suprathreshold electrical stimuli was significantly higher in CTTH patients (54 ± 3.9 mA) than in healthy volunteers (37 ± 4.0 mA; P < 0.005; CI, 5–28, t-test). The electrical pain threshold (Ip ) increased significantly from start of study to post-LFS within CTTH 0.83 (0.55–1.27) to 1.38 (1.00–2.02); healthy volunteers 0.88 (0.59–1.03) to 1.90 (1.30–2.65), both, P < 0.05, Mann–Whitney. There was no significant difference between CTTH patients and healthy volunteers in Ip (n.s., Mann–Whitney).
Pressure pain threshold (PPT) of the finger is significantly smaller at end of study than at the start of the study in both chronic tension-type headache (CTTH) patients and in healthy volunteers (HV), but not in the temple. The PPT finger and temple were significantly higher in healthy volunteers than in CTTH patients, both at the start and at the end of the study. The electrical pain threshold (Ip ) increased significantly from start to end of study in both groups, but there was no difference between groups

Data presented as mean ± SEM or median (quartiles). kPa, kilopascal; mA, milliampere; It-test, independent; Pt-test, paired t-test; MW, Mann–Whitney; CI, confidence interval.
Discussion
The major finding of this study was that LTD could be induced in CTTH patients. Pain ratings decreased consistently during the 20-min LFS period (CTTH, from 65.3 ± 4.0 to 43.6 ± 5.4; healthy volunteers, from 46.5 ± 4.0 to 25.6 ± 4.0). The inhibition of pain was stably maintained compared to baseline during the 1-h post-LFS recording (CTTH, 19.6 ± 3.9%; healthy volunteers, 30.1 ± 5.0%), akin to the reduction in the R2 component of the nociceptive blink reflex integral. There was a tendency towards lower degree of pain inhibition in CTTH patients than in healthy subjects, but this was mainly due to higher baseline ratings in patients. The significant higher baseline pain ratings in CTTH patients indicate they are elsewhere on the stimulus–response curve and, therefore, data interpretation must be cautious. The decrease in absolute pain values was similar in the two groups (Figure 2B). The higher sensitivity to pressure, in both temple and finger, indicates that CTTH patients are centrally sensitised in keeping with previous findings (6).
The present data do not exclude the possibility that the central sensitisation in CTTH patients partly counteracts LTD, but the tendency to a smaller relative pain reduction in CTTH patients could be explained by differences in baseline ratings. We conclude that a significant degree of LTD can be induced even in patients with long-lasting chronic pain.
The underlying mechanisms of LTD have been investigated in human and in animal experiments. High-frequency stimulation induces long-term potentiation, change similar to central sensitisation, which can be reverted by LFS; (26) thus, the objective of this study was to investigate the effect of LFS to chronic human pain patients who are known to be sensitised. Recently, the effect of LTD was demonstrated to be homotopic in man (27), and suggested to be primarily due to changes at the first nociceptive synapse (28). Nevertheless, the LTD effect of high-frequency in rats disappeared in spinalised animals (29) indicating descending pathways were also involved. LFS in perinatal rodent hippocampal neurons has recently been demonstrated to induce NMDA-dependent structural changes of the neurons, both pre- and postsynaptic, and is suspected to decrease the number of working synapses (30–32). These LTD effects have also been suggested to be mediated by calcium influx through the NMDA-receptor activating coupled kinase and phosphatase switches (33). Whether these findings in perinatal rodent hippocampal neurons are comparable to the neurons in chronic pain patients is as yet unknown, but some involvement of the NMDA-receptor system in the regulation of headache intensity in CTTH patients has been demonstrated (34).
The reduction in post-LFS pain and the reduction in blink reflex integral were of similar magnitude indicating that blink reflex may be an objective surrogate marker for pain reduction (16). The concomitant reduction of blink reflex indicates that at least part of the pain inhibition induced by the LFS takes place at the level of the brain stem. However, it cannot be concluded from our data whether this antinociception is caused by local mechanisms in the brain stem or by descending or ascending inhibitory pain mechanisms. The involvement of supraspinal brain structures after high-frequency stimulation has been investigated by means of positron emission tomography in rodents demonstrating changes in primary sensory cortex, amygdala, peri-aqueductal gray and rostral ventromedial medulla (35). Future human functional brain imaging studies could possibly elucidate the mechanisms involved in LTD in chronic pain patients.
With the present LFS design (1 Hz, 20 min, intensity of 4 times the pain threshold), we were able to induce LTD seen as a reduction of pain score to the electrical stimuli and as a decrease in blink reflex integral, while LFS did not affect headache or pain thresholds consistently. It is possible that the pain inhibition would have been more pronounced with a longer LFS period since the pain was reduced consistently throughout the present observation period as seen from Figure 3. Moreover, it would have been interesting to have a longer post-LFS period to observe the entire duration of the pain inhibitory effect. It is possible that the inhibition of pain could be more pronounced with longer LFS periods, with repetitive LFS periods (36) (e.g. several times per day or daily for some weeks) or with stimulation at several sites bilaterally at the same time. This should be examined in future studies. In future studies, the difference in baseline pain ratings and corresponding differences on the stimulus–response curve could be addressed by adjusting the stimulus intensity according to pain rating, instead of the pain threshold.
Electrical stimulation in the treatment of chronic pain is still controversial (37,38). It is mostly via the so-called transcutaneous electrical nerve stimulation (TENS). Whether TENS and LTD are reducing pain through the same mechanisms is doubtful (36), both mechanisms are largely unknown. TENS has been suggested to be a masking of the nociceptive inputs (38), LTD has been demonstrated to induce structural changes in the dendrite synapses (30,31). To our knowledge, there are no studies on the effect of LTD in chronic pain patients, and no blinded studies have demonstrated an effect of electrical stimuli in CTTH patients (39,40).
Conclusions
A significant and stable pain inhibition (LTD) can be induced in CTTH patients by low-frequency electrical stimulation. The corresponding decrease in the nociceptive brainstem reflex indicates that pain was modulated at the level of the brainstem; however, a peripheral effect cannot be excluded (17). Management of chronic pain patients is, in general, difficult and low-frequency electrical stimulation may be an attractive future non-pharmacological treatment option.
Footnotes
Acknowledgement
The authors thank Hanne Andresen for technical assistance.
