Abstract
2,4-Dinitrophenol (DNP), previously used as a weight loss drug, was banned owing to its narrow therapeutic index and severe side effects. DNP disrupts mitochondrial oxidative phosphorylation and inhibits adenosine triphosphate synthesis, causing heat production, increased basal metabolic rate and enhanced glycolysis. Despite bans, unregulated online and darknet sales have led to a rise in DNP toxicity cases. We report a case of a young man who survived a deliberate, highly lethal DNP overdose through coordinated team-based care including early aggressive cooling and high-volume mechanical ventilation, crucial in maintaining normothermia and normocapnia. This case adds valuable insights to the literature on DNP toxicity management.
Introduction
2,4-Dinitrophenol (HOC6H3(NO2)2) (DNP) was previously marketed as a weight loss drug. It works by disrupting mitochondrial oxidative phosphorylation and inhibiting aerobic adenosine triphosphate synthesis, thereby generating heat, leading to a higher basal metabolic rate and increased glycolysis. 1 This facilitates weight loss. DNP has a narrow therapeutic index and significant adverse effects such as hyperthermia, tachycardia, diaphoresis and tachypnoea. Fatal toxicities usually present with acute renal failure, rhabdomyolysis, cerebral oedema, hepatic and pancreatic injury, haematopenia, multi-organ failure and patients ultimately succumb from cardiac arrest.1–3 These factors alongside complications of hyperthermia and uncontrolled carbon dioxide (CO2) production led to DNP being banned in Britain, and subsequently by the US Food and Drug Administration (FDA) in 1938.1–4 However, there has been a recent increase in the incidence of DNP toxicity, attributed to unregulated online and darknet sales in the UK, USA and Australia, mainly targeting body builders. 3 Between 2001 and 2010, 456 cases of DNP exposure from deliberate overdoses and a few accidental toxicities were reported to the World Health Organization Poison Control Centre. This showed an 11.9% fatality rate, which is likely an underestimate as some countries were unable to provide mortality data and follow-up was not always done.1,5 We report a case of a young man who survived a deliberate overdose of DNP. On literature review, we found that this is the first reported case of survival following a lethal DNP overdose managed by prompt aggressive cooling and high-volume mechanical ventilation. Consent for this case report was obtained from the patient’s next-of-kin while he was mechanically ventilated.
Case report
First 24 h
A 20-year-old male patient (weight 80 kg) presented to the emergency department after a staggered DNP intentional overdose with suicidal intent. He had consumed a total of 6 g over 2 days (powder formulation, 1 g every 2 h, five times on day 1, then 1 g on day 2; equating to ~75 mg/kg per 24 h). Apart from ephedrine 50 mg for three days before the admission, he had not consumed any other prescription or non-prescription medications. He presented to the emergency department 7 h after his last ingestion. On arrival, his initial vital signs showed normothermia (37.1°C, tympanic), heart rate 120 beats/min, blood pressure 145/64 mmHg, respiratory rate 24 breaths/min and oxygen saturation 97% on room air. Following consultation with a toxicologist, the team identified this as a highly lethal overdose with expected severe hyperthermia and its associated complications, alongside rapid CO2 production. In anticipation of severe hyperthermia, efforts to cool him were initiated immediately even while he was still normothermic. Despite receiving 1 l of cold 0.9% sodium chloride solution, his temperature climbed to 37.9°C within an hour. He was extremely diaphoretic and tachypnoeic with a respiratory rate of 28 breaths/min but remained conscious, Glasgow Coma Scale 15 and normotensive. Sepsis was initially considered; however, this was later deemed unlikely given no prior infective symptoms and unremarkable inflammatory markers.
He was intubated promptly to prevent worsening hyperthermia, induced with fentanyl 50 µg, propofol 150 mg and rocuronium 100 mg (temperature 38°C). The brief pause in ventilation for rapid sequence intubation caused a rapid rise in the patient’s mixed venous CO2 partial pressure to 70 mmHg with a corresponding severe respiratory acidosis (pH 7.18, arterial CO2 partial pressure (PaCO2) 70 mmHg, base excess –4.4) as shown in Figure 1. He was manually hyperventilated at a rate of 30 breaths/min for a few minutes to lower his end-tidal CO2 partial pressure (PETCO2) from 114 mmHg to approximately 50 mmHg. Subsequently, he was paralysed and mechanically ventilated on a volume-controlled mode with high minute ventilation of 18–20 l/min aiming for a normal PaCO2. The patient was further cooled with an infusion of 2 l of ice-cold 0.9% sodium chloride solution, and fanning, yet his temperature still reached 39.9°C. On arrival to the intensive care unit (ICU), Arctic Sun™ (Medivance Inc., Louisville, CO, USA) cooling pads were applied, with the water temperature dropping down to 4°C to control his temperature. The patient’s biochemistry is presented in Table 1. High flux continuous veno-venous haemodiafiltration (CVVHDF) was initiated 3 h post presentation in further efforts to cool him when the temperature was 39.9°C (Figure 2), and to correct his hyperkalaemia (K+ 6.0 mmol/l). The dialysis circuit was packed with ice as well.
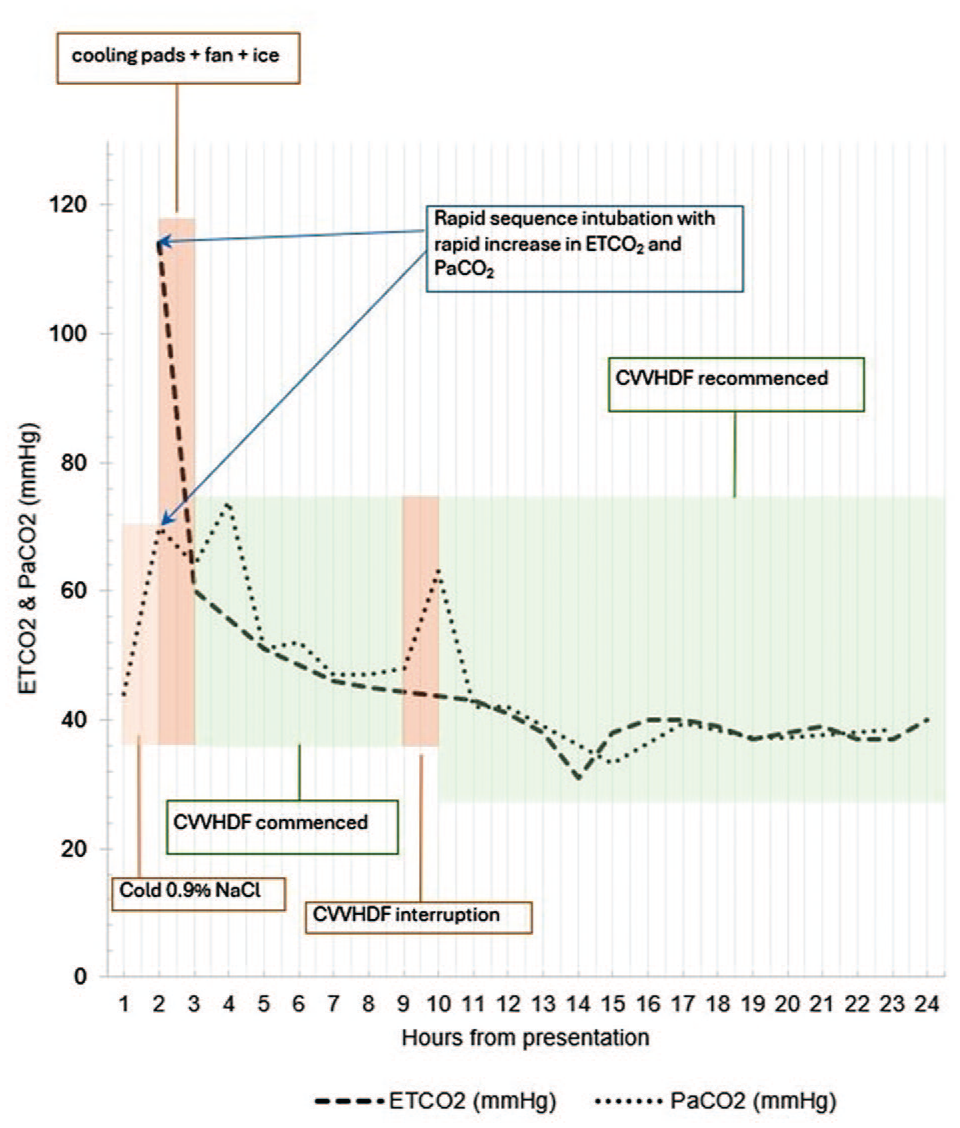
ETCO2 PaCO2 trend.
Blood test results on days 1–4 and before discharge from hospital.
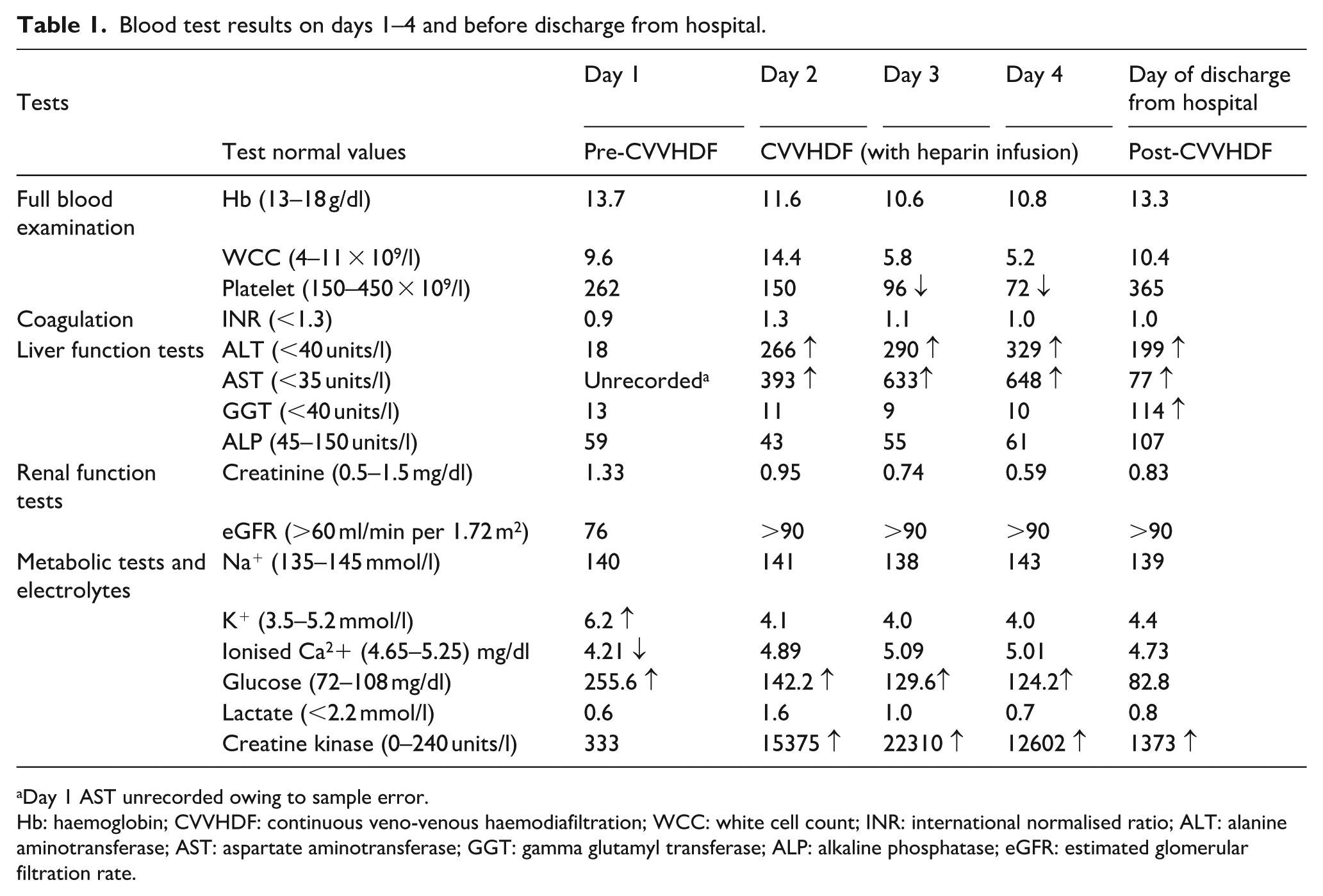
Day 1 AST unrecorded owing to sample error.
Hb: haemoglobin; CVVHDF: continuous veno-venous haemodiafiltration; WCC: white cell count; INR: international normalised ratio; ALT: alanine aminotransferase; AST: aspartate aminotransferase; GGT: gamma glutamyl transferase; ALP: alkaline phosphatase; eGFR: estimated glomerular filtration rate.
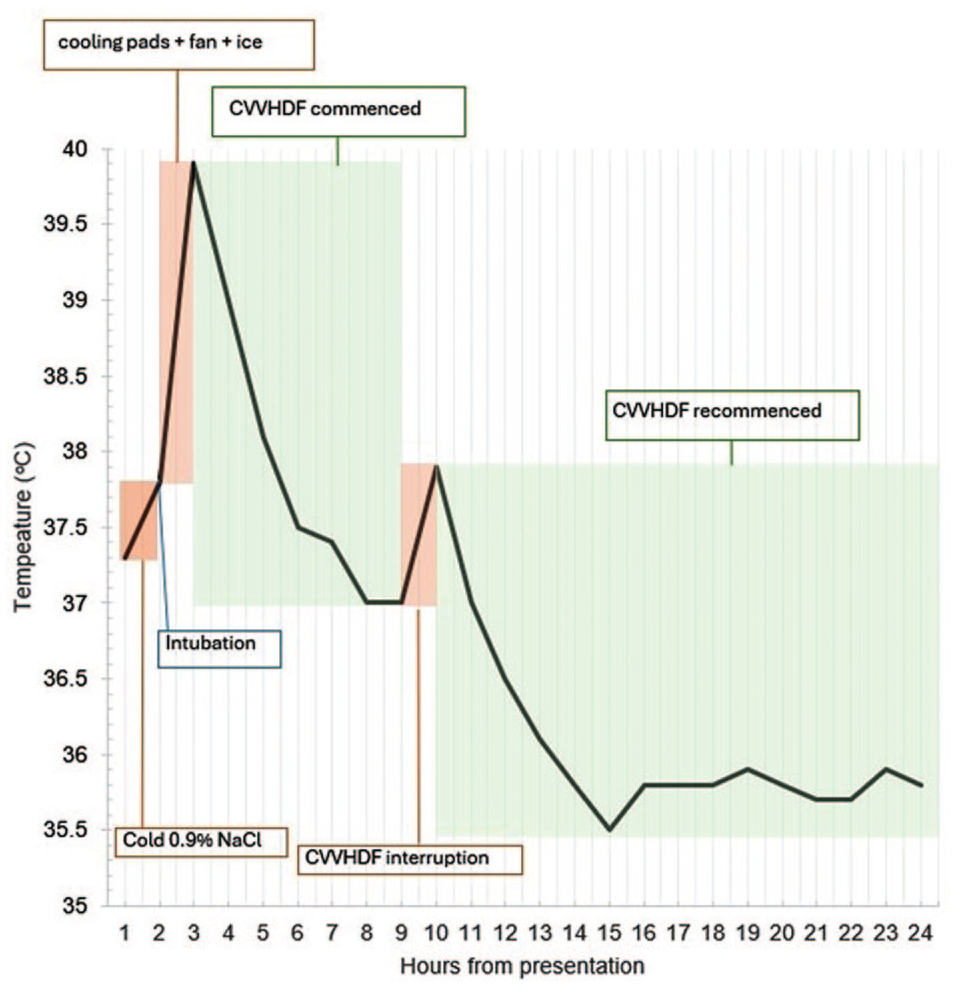
Trend of temperature chart.
Extracorporeal membrane oxygenation (ECMO) was initially considered for cooling, enhanced CO2 removal and potentially cardiac support. However, given that our hospital is not an ECMO centre, this patient would have needed to be transferred to an ECMO hospital. Logistical complexity precluded transportation owing to the inability to cool him during the transport with either ECMO, dialysis or cooling pads. The medical team, including ICU specialists, a toxicologist and paramedics, decided to continue with CVVHDF and existing treatments without ECMO. Within 4 h of starting CVVHDF and cooling pads, the patient’s temperature stabilised at 37.1°C. Six hours into CVVHDF, there was a 30-min interruption to haemodialysis when the filter ceased functioning, during which the patient’s temperature rose rapidly from 37.0°C to 37.9°C. Cooling effectively resumed when CVVHDF recommenced. Normothermia was achieved 8 h into admission. He required four boluses of 0.5 mg metaraminol initially postintubation, which was transitioned to noradrenaline. The mean arterial pressure was 65 mmHg on 15 µg/min of noradrenaline, and the bedside echocardiogram confirmed normal heart function without any contractility impairment. His arterial haemoglobin oxygen saturation (SpO2) was 98% on an inspired oxygen fraction (FiO2) of 0.3, PETCO2 40 mmHg and PaCO2 45 mmHg, with set respiratory rate of 26 breaths/min while paralysed. Fifty grams of activated charcoal was administered down a nasogastric tube for decontamination an hour after commencement of CVVHDF.
Day 2 to discharge
CVVHDF was maintained for 48 h. Vasopressors were weaned off by day 2 and the patient remained normothermic at 36°C, vital signs were stable with blood pressure 135/70 mmHg, set respiratory rate of 24 breaths/min, PETCO2 33 mmHg, SpO2 99% on FiO2 0.21. He no longer required a high minute ventilation of 18 l/min to maintain a normal PETCO2 and PaCO2 during continuous renal replacement therapy (CRRT) by around 48 h. This was interpreted to signal the end of metabolic uncoupling from DNP. The Arctic SunTM water temperature was 35°C when it was turned off on day 3, but pads were still left in situ in case the device had to be turned back on. The pads were removed when the CVVHDF was discontinued.
The patient was extubated on day 5. He was delirious for three days post-extubation but later had a good recollection of events leading to his presentation. He was transferred to a medical ward on day 8 and discharged home on day 10 with continued follow-up with the mental health team for further management of potential body dysmorphia and disordered behaviours. Unfortunately, we were not able to measure the drug levels of DNP.
Discussion
Though we were unable to get laboratory confirmation of DNP owing to lack of samples, the patient’s history and clinical presentation were consistent with DNP overdose.
Case reports have documented very short durations of 7–8 h from DNP ingestion and onset of severe symptoms, including death.1,5 The lowest recorded lethal dose was 0.6 mg/kg per day staggered over 41 days. 5 The highest reported dose of DNP associated with survival was 2.4 g in 1934, which, to our knowledge, is the only reported case of survival post-intentional lethal overdose. 5 Though staggered, our patient had consumed almost two and a half times this dose and survived.
DNP-associated fatalities are due to complications of severe hyperthermia and its associated complications as well as uncontrolled CO2 production leading to metabolic derangements, disseminated intravascular coagulation and multi-organ failure. A study that analysed 204 patients with DNP toxicity over 12 years in the UK and USA found that tachycardia, hyperpyrexia, acidosis and agitation or confusion were independent predictors of overdose death. 6 Our patient manifested most of these features, indicating his initial hypermetabolic state which prompted the rapid escalation of aggressive supportive treatment and monitoring. The ability to wean these supports and normalisation of PaCO2 levels on day 2 suggested the resolution of oxidative phosphorylation uncoupling and elimination of systemic DNP to at least sub-lethal levels.
Diagnosis, risk assessment and the urgency of cooling
In previous case reports, active cooling was not started before patients suffered cardiac arrest and eventually died.6,7 Where cooling was instituted, they primarily utilised principles of convection and conduction via iced towels, fanning and sponging 8 and pharmacologically trialled agents such as dantrolene and paracetamol.8,9
In our patient, once we recognised the high lethality and potential rapid progression of DNP toxicity, our immediate priority was to cool the patient with a theoretical aim to decrease the uncoupling of oxidative phosphorylation. Animal data has shown that therapeutic hypothermia has been associated with mitigating the uncoupling of oxidative phosphorylation and mitochondrial cell death. 10 We began active cooling despite the patient still being normothermic and escalated cooling aggressively. He was managed with cold intravenous fluids, ice packs, fanning, paralysis and cooling pads and started on CVVHDF (with tubing packed in ice) all within 3 h of his presentation. We believe this contributed significantly to his survival.
Our patient’s peak temperature was 39.9°C just before commencing CVVHDF and rapidly fell to 38.1°C within 2 h, thus avoiding significant complications of hyperthermia other than rhabdomyolysis and liver dysfunction. He did not sustain any irreversible organ dysfunction as his liver function tests remained relatively stable with serum transaminase levels below 650 units/l, peaking on day 4 from presentation and normalising close to discharge. Renal function tests were stable while being supported by CVVHDF. He had preserved cardiac function with troponin levels within normal limits throughout his admission (0.002 ng/ml) and coagulation tests also remained normal.
Options for cooling
In our case, while the Arctic SunTM, paralysis and other cooling techniques might have slowed the progression to severe hyperthermia beyond 40°C we found that CVVHDF (with ice packed around the tubing) was the most effective at lowering the patient’s core body temperature and maintaining normothermia, evidenced by our patient becoming normothermic within 4 h of commencing CVVHDF. This was further supported by the fact that the patient’s temperature rose by nearly 1°C within a 30-min time frame when the CVVHDF function was interrupted.
In studies of patients cooled for neuroprotection, CVVHDF showed a cooling induction rate of 4°C to 6°C/h, temperature control pads via Arctic Sun™ have shown 1.5°C to 2°C/h, while ice packs and cooling fans only approximately 1°C/h. 11
We considered starting our patient on ECMO for further cooling and also for the added benefit of extracorporeal CO2 removal (ECCO2R) and potential requirement for cardiac support but decided against it for logistical reasons. Although there is no reported evidence of ECMO efficacy in cooling being superior to CVVHDF, we anticipate that ECMO would cool faster by virtue of larger extracorporeal blood volume and flow and would have started our patient on ECMO if his hyperthermia worsened. 9
Hypercapnia management
One of the initial challenges in managing this patient involved managing his rapidly worsening hypercapnia and its resultant acid-base derangement from excessive CO2 production due to the uncoupling of oxidative phosphorylation. A brief pause in ventilation for rapid sequence intubation led to an expeditious rise in PETCO2 that matched that of the PaCO2. McGillis et al 12 reported a patient post-DNP overdose whose PaCO2 was as high as 130 mmHg after intubation and was ventilated at 10 ml/kg. However, this patient developed muscle rigidity making ventilation impossible, and progressed to pulseless electrical activity arrest and died. In our case, we overcame the high CO2 production by increasing the minute ventilation, first by bag-mask hand ventilation with rates up to 30–40 breaths/min and setting his ventilator for a minute ventilation of 18–20 l/min. In our otherwise healthy patient without any lung pathology, we tolerated non-lung protective ventilation for a short period to prioritise the management of acidosis and hypercapnia. Four hours post-intubation, we were able to stabilise our patient’s PaCO2 at around 50 mmHg. We further titrated the minute ventilation based on the PaCO2.
Aside from ECMO, we also considered ECCO2R but were unable to set up the circuit owing to technical difficulties with our device. Although ECCO2R has been shown to improve hypercapnic acidosis while reducing the burden of aggressive ventilation within 4 h of initiation, 13 ECCO2R as a modality to control CO2 in the poisoned setting remains largely without evidence.
Resolution of toxicity
DNP elimination is based on a two-compartment open model, with serum terminal half-lives in the range 7–10 h. However, this can extend up to 76 h owing to tissue binding observed in animal models. 5 Reduction in external interventions needed to control our patient’s temperature and maintain normocapnia indicated to us that uncoupling of oxidative phosphorylation and thus DNP toxicity was resolving.
Conclusion
We report a rare case of survival in deliberate, high-lethality DNP overdose. We believe that the coordinated team-based approach between paramedics, the toxicology service, emergency physicians and ICU teams allowed prompt initiation of early aggressive cooling, efficient transfer to the ICU for CVVHDF and high-volume mechanical ventilation to control hypercapnia, which saved this patient’s life. In DNP overdoses, there is a high likelihood of severe symptoms, therefore aggressive cooling therapies should be commenced early. The most effective cooling method in this case was CVVHDF, which could be considered for management of hyperthermia in future cases of DNP toxicity.
Footnotes
Author contributions
Declaration of conflicting interests
The authors have no conflicts of interest to declare.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
