Abstract
We compared the cost of continuous renal replacement therapy (CRRT) in critically ill patients using two different anticoagulation strategies: regional citrate and low-dose systemic heparin in a single-centre, prospective observational study in an adult Australian tertiary intensive care unit (ICU). All patients receiving CRRT between October 2015 and May 2016 were included in the study. Costs were modelled using the number of filter sets, number of dialysis bags, amount of citrate, heparin and calcium replacement required, and cost of monitoring the anticoagulation. The primary outcome was cost associated with CRRT per patient per day. The secondary outcome was efficacy of CRRT. In total, 66 patients were commenced on dialysis that required anticoagulation. Twenty-four patients were commenced on regional citrate anticoagulation and 42 patients commenced on systemic low-dose heparin anticoagulation. Median filter life, though not statistically significant, was longer in the citrate group by 7.7 hours (
Keywords
The success of continuous renal replacement therapy (CRRT) depends, in part, on the continuous delivery of the therapy. Circuit failure due to clotting is a frequent problem in CRRT and as a result anticoagulation of the circuit, either systemically or regionally, is recommended to maintain filter life and improve dialysis efficiency.1
Traditionally, heparin has been the main anticoagulant used, with >80% of Australian and New Zealand intensive care units (ICUs) using unfractionated heparin as their first-line agent. 2 Heparin has the benefits of low cost, easy monitoring, easy reversibility and clinical familiarity. However, it is not without limitations and there is concern regarding the safety of heparin in critically ill patients, highlighting the need for alternative anticoagulation agents. Such limitations include altered pharmacokinetics due to heparin resistance and critical illness–induced heparin binding, bleeding, heparin-induced altered inflammatory effects and heparin-induced thrombocytopenia. 3
Citrate regional anticoagulation has become an accepted alternative, with a recent randomised controlled trial (RCT) and meta-analysis evidence suggesting it prolongs filter life and reduces the risk of bleeding compared with regional heparin.4,6–10 This may translate into improved efficiency, improved patient outcomes, decreased nursing workload, decreased consumption of consumables and decreased cost. Citrate anticoagulation has been made easier with commercially available protocols and consumables that help offset the known risks of acid–base and electrolyte imbalances. It is contraindicated in those with hepatic failure.3–6
While citrate has been proven to prolong filter life, it is not clear whether this translates to improved clinical outcomes, with several RCTs and meta-analyses showing no difference in patient mortality or ICU length of stay (LOS) when compared with regional or systemic heparin.7–10 While one of the key considerations in choosing anticoagulation for renal replacement therapy is based on extending filter life and reducing consumable use, a thorough cost comparison of the two modalities has not previously been done. To address this question we conducted a single-centre prospective observational study comparing the costs for each of the anticoagulation modalities.
Methods
Design and setting
This study is a prospective observational study conducted in a 23-bed tertiary general ICU with cardiothoracic, liver transplant and neurosurgical patients. Both anticoagulation modalities had well-established protocols for use. The project was given institutional approval by our hospital’s quality assurance program.
Eligibility criteria
All patients receiving CRRT between October 2015 and May 2016 who needed regional citrate or low-dose systemic heparin anticoagulation were included in the study. Patients who crossed over from heparin to citrate anticoagulation or vice versa were excluded from data analysis. The treating intensivist determined the choice of anticoagulation. Recruitment was conducted for a total of eight months and, in total, 66 patients were recruited.
Interventions
Citrate or heparin anticoagulation was conducted according to the department protocols (see Appendix 1 on
The need for dialysis, choice of anticoagulation, need for blood transfusion and decision to stop therapy was determined by the treating intensive care specialist. CRRT was delivered by trained ICU nursing staff. The protocol for heparin anticoagulation is a low-dose continuous heparin infusion of 200–500 units/h with daily monitoring of coagulation (activated partial thromboplastin time only), electrolytes, urea (Ur) and creatinine (Cr). Citrate anticoagulation was administered with strict adherence to the departmental protocol, with careful calcium replacement and electrolyte monitoring. The protocol stipulates a minimum 4-hourly ionised calcium monitoring. Citrate anticoagulation has been in use in the department since 2014. The protocol used is based on the Royal Brisbane and Women’s Hospital Intensive Care Unit 2014 Work Unit Guideline of citrate anticoagulation for CRRT using the Prismaflex circuit.
Solutions
The citrate haemofiltration solutions used were Prismocitrate 18/0 (18 mmol/L citrate) pre–blood pump, Prism0cal B22 dialysate fluid, Hemosol B0 replacement fluid, and CaCl2 10% neat infusion. Heparin haemofiltration solution used was Hemosol B0 (all solutions manufactured by Baxter Healthcare, Toongabbie, New South Wales).
Data collection
We collected baseline data regarding age, sex, severity of illness (Acute Physiology and Chronic Health Evaluation (APACHE) II score) and indication for dialysis. Data collected related to efficacy included hours of CRRT received and Ur and Cr reduction per day. Data regarding CRRT consumables included quantity of haemofiltration bags, filters/circuits, CaCl2 vials, arterial blood gases (ABGs) and venous blood gases, coagulation tests, and red blood cell transfusions.
We also noted the reason for CRRT circuit change: filter clotted, access issues or elective circuit change. Clotted filter was defined as transmembrane pressure greater than 250 mmHg or visible clot within the circuit limiting blood flow. Access issues were defined as persistent access pressure less than –50 mmHg. Elective change was defined as circuits stopped for necessary patient care (i.e. transfers, physiotherapy) or the mandatory change as per manufacturer instruction at 72 h of therapy or 780 L of blood flow through the filter.
Consumables and associated costs
Table 1 describes the costs of CRRT-related consumables used in the cost analysis. Costs were modelled using the current costs as per the Medicare Benefits Schedule, 11 Pharmaceutical Benefits Schedule, 12 National Blood Authority Australia, 13 and Sir Charles Gairdner Hospital ICU purchasing invoices (note that the hospital-affiliated pathology company charges hospital inpatients 90% of Medicare Benefits Schedule pricing).
Cost of consumables for continuous renal replacement therapy.
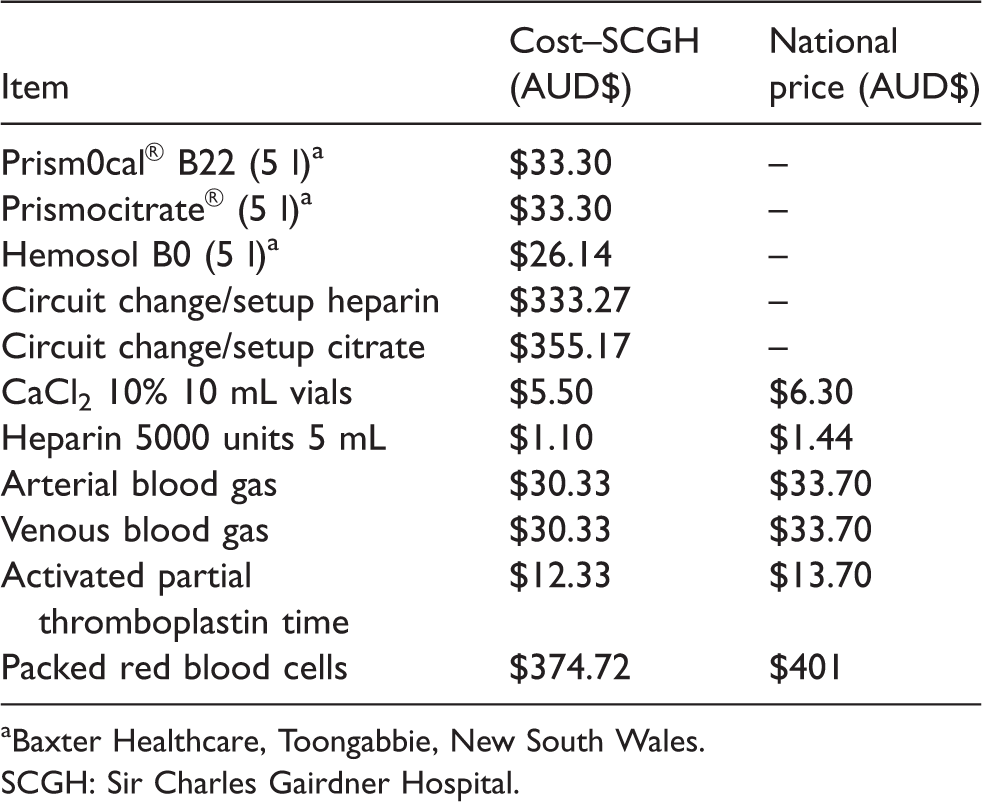
aBaxter Healthcare, Toongabbie, New South Wales.
SCGH: Sir Charles Gairdner Hospital.
Outcomes
The primary outcome was costs associated with CRRT per patient per day. Data was collected prospectively and the cost analysis was done using this data retrospectively. Cost assessment included the number of filter sets used, the number of dialysis and replacement bags, citrate anticoagulation cost (Prismocitrate), heparin cost, calcium replacement cost, cost of monitoring (ABG, venous blood gases, activated partial thromboplastin times) and cost of blood transfusions.
The secondary outcome was efficacy of CRRT. Efficacy was assessed by the number of filters used, reason for circuit change for each mode of anticoagulation, number of hours on CRRT and daily Ur and Cr reduction.
Whilst not an endpoint in our study, we have also provided data for the clinical endpoints ICU LOS, ICU mortality and number of blood transfusions.
Analysis
Summary statistics, including mean and standard deviation (SD), are provided for all continuous variables. Frequencies and percentages are provided for all categorical variables.
The average duration of filter life was calculated as the total hours of dialysis received by a patient, divided by the total sum of filters used by that patient.
Independent sample Student’s t-test was used to compare average duration of filter life, APACHE II score, age, ICU LOS and prescribed dose received (%) between treatment groups. For analysis purposes, ICU LOS and average duration of filter life were log-transformed and percentage of prescribed dose received was arcsine-transformed (excluding the two extreme values over 100%).
Wilcoxon ranked-sum test/Mann–Whitney
As only 20 patients received at least one packed red blood cell (PRBC) transfusion, this variable was dichotomised for all patients to indicate whether any PRBC transfusion was received (yes/no). Chi-square test analyses were used to compare any PRBC transfusion received, sex, and ICU mortality between treatment groups.
Average cost of treatment per day was calculated as the total cost of treatment divided by the total number of days on CRRT treatment. Univariate and multivariate linear regression was used to compare cost of treatment with respect to treatment groups. Variables considered in these analyses included age, sex, APACHE II score, treatment group and ICU outcome. Variable selection was conducted and those variables significant at the 5% level were retained in the final model. Cost of treatment was log-transformed for analysis purposes.
Results
Of the 66 patients enrolled, 24 patients were commenced on regional citrate anticoagulation and 42 patients commenced on systemic low-dose heparin anticoagulation. Table 2 outlines the summary statistics for all patient demographics and clinical variables. There was no significant difference in age (
Summary statistics for all demographic and clinical variables.
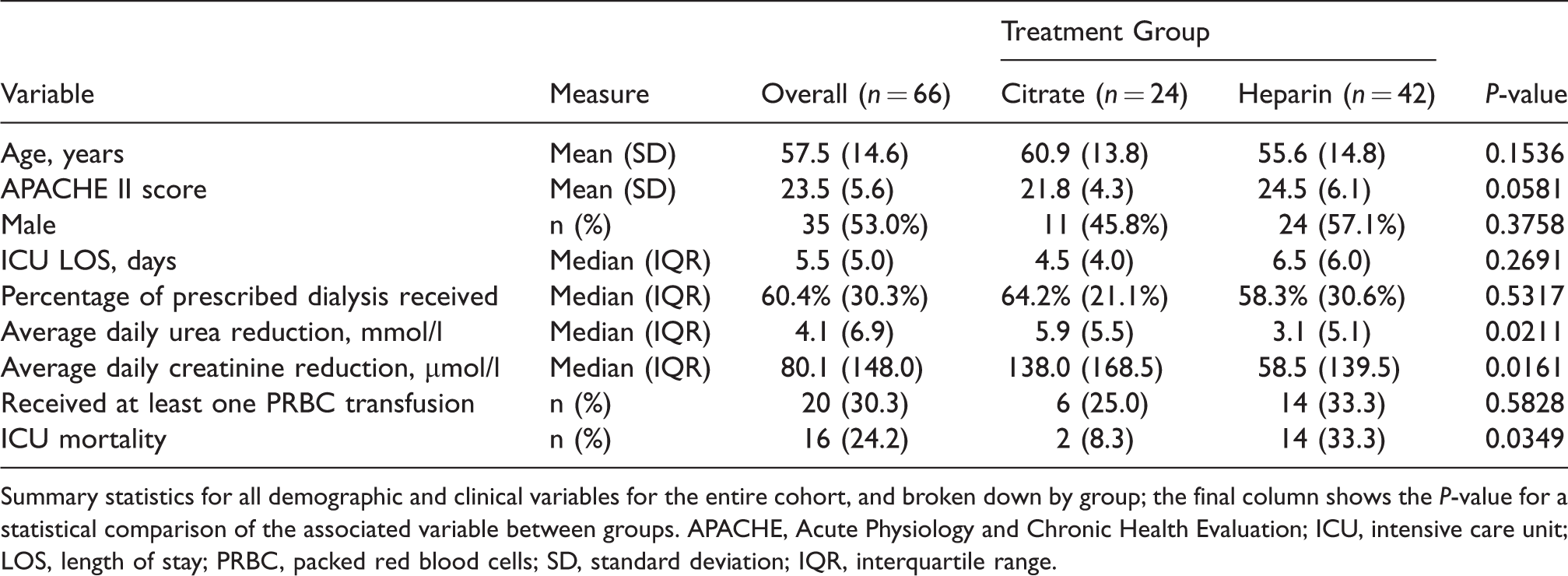
Summary statistics for all demographic and clinical variables for the entire cohort, and broken down by group; the final column shows the
Average daily Ur and Cr reduction was significantly different between treatment groups (
The median duration of time each patient spent on CRRT was 72 h in both groups, the interquartile range (IQR) was larger in the heparin group (IQR 72 h) compared with the citrate (IQR 48 h) group.
There was no significant difference in the rate of blood transfusion between the citrate and heparin anticoagulation groups (
There was a significant difference in the number of deaths between the two treatment groups; specifically, the ICU mortality rate of patients in the heparin group was 33.3% (
Cost outcomes
Table 3 summarises the cost of treatment, using the national pricing figures, per patient per day, broken down by the categorical variables. Cost of treatment per day was calculated as the total cost of treatment, based on the national pricing, divided by the total number of days on CRRT treatment.
Summary statistics for cost of treatment.
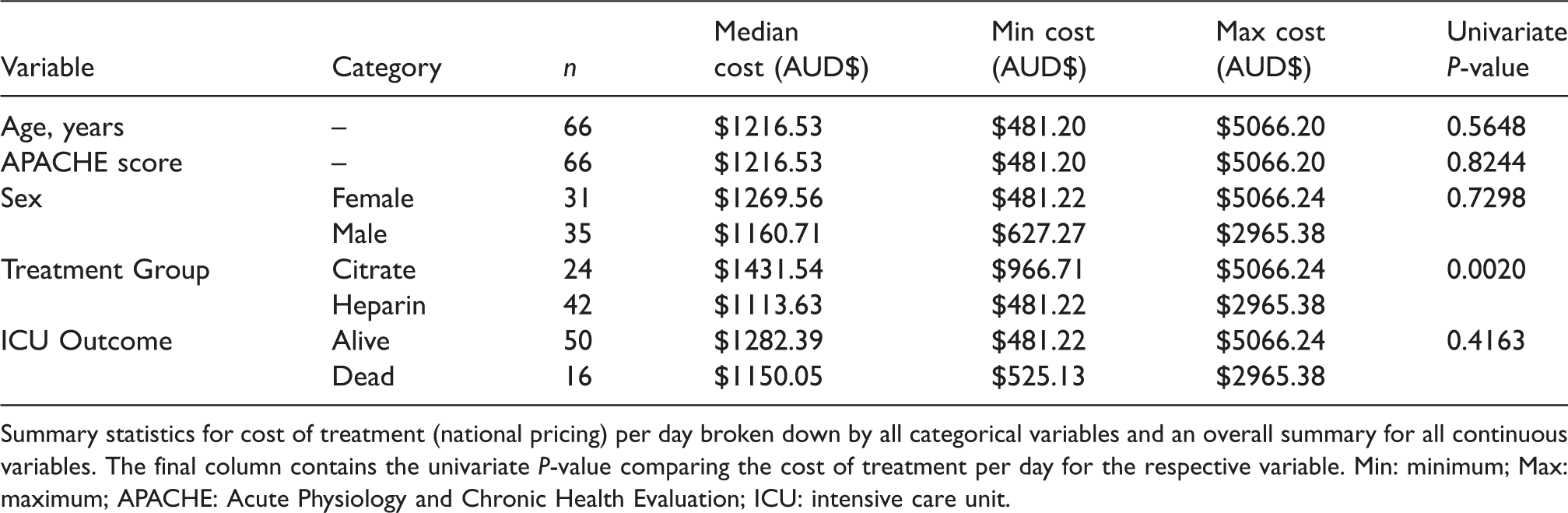
Summary statistics for cost of treatment (national pricing) per day broken down by all categorical variables and an overall summary for all continuous variables. The final column contains the univariate
Treatment group was found to be the only variable significantly associated with cost of treatment per day (
Given that most ICUs do not pay the national prices for consumables and laboratory tests, but rather discounted rates, we also conducted a cost comparison on the hospital’s ICU costing. This found citrate median cost of treatment per day to be AUD$262.58 higher than the heparin group.
Further cost analysis excluding the cost of PRBC transfusions was also performed, which found a median price difference of AUD$463.15 (
A major contributor to the cost difference between the two groups was the cost of the blood gases necessary for calcium monitoring, and the cost of calcium replacement. The average cost of CaCl2 replacement per patient on citrate anticoagulation was AUD$263.54; the average cost of ABGs/venous blood gases per patient on citrate was AUD$613.93 compared with AUD$408.54 per patient on heparin. The departmental citrate protocol stipulates at a minimum, 4-hourly patient ionised calcium monitoring, and the average patient usage of CaCl2 vials for the duration of CRRT was 46 vials. Comparatively the haemofiltration solutions are also more expensive for citrate CRRT.
Filter outcomes
Post hoc analysis was also conducted to examine duration of filter life and anticoagulation modality. This was calculated as the total hours of dialysis received, divided by the total number of filters used. The median lifespan of a heparin filter was 18.8 h compared with 26.5 h for a citrate filter. Whilst the citrate filter survival was better than heparin, this was not statistically significant (
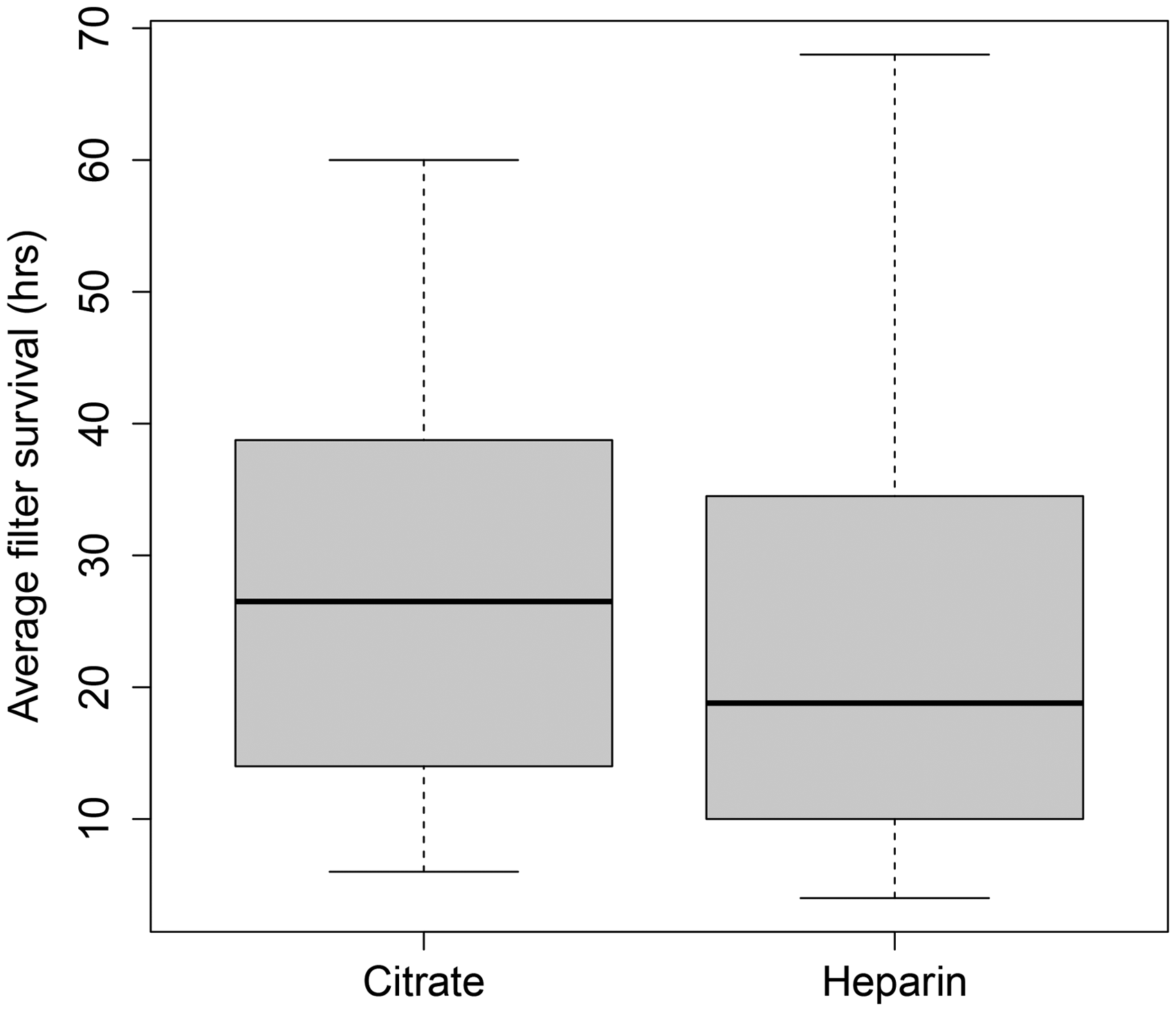
Box plot of average filter life, citrate versus heparin.
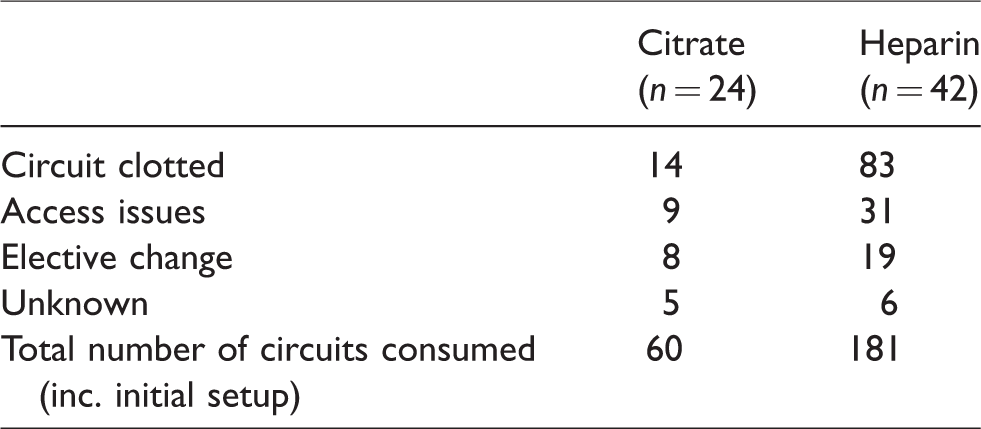
We also reviewed the reasons for circuit changes within the two groups (Table 4). Within the citrate group, 24 patients consumed a total of 60 circuits (including the initial setup circuit at the commencement of CRRT) of which only 14 circuit changes (23%) were due to circuit clotting. The heparin group of 42 patients consumed a total of 181 circuits, of which 83 changes (45%) were due to circuit clotting. The heparin group also had a higher fraction of circuit changes due to access issues compared with the citrate group (31/181 (17%) versus 9/60 (15%)).
Reasons for circuit change.
Discussion
Our cost comparison study shows that regional citrate anticoagulation is more expensive than low-dose systemic heparin for CRRT in the Australian setting.
Recent RCT and meta-analysis evidence supports citrate as the preferred anticoagulation regimen due to improved filter life, and, as a result, improved CRRT efficacy.4–10 We did observe similar increases in filter life as other studies—the median life of the citrate filter was 7.7 h longer than the heparin group (albeit not statistically significant). Median Ur and Cr reduction was greater in the citrate group, and there were significantly fewer circuit changes due to clotting in the citrate group.
Some authors have suggested that increased filter life may equate to improved cost-effectiveness.5–7 Therefore, it is important that our observational data shows that citrate confers an increase in overall cost, largely as a result of increased need for ABG testing, calcium replacement and citrate. Consumables for citrate are priced higher than those of heparin; the cost of calcium replacement and the cost of laboratory monitoring are the two major factors that contributed to the increased cost in our department. While the IQR for duration of therapy differed between groups, the median duration of therapy was similar. As our modelling is based on the average daily cost incurred per patient, the costs are standardised between groups, thereby accounting for any differences in duration of therapy and use of consumables between the two groups.
The number of filters consumed did not offset the above costs incurred. The heparin group consumed 1.7 times more filters per patient than the citrate group; however, the overall cost incurred was found to be less. The increased consumption of filters in the heparin group was attributable primarily to circuit clotting, which will no doubt be reflective of the anticoagulation modality but also the sicker patients in the heparin group, the latter of which is controlled for in the cost modelling. Another significant contributor to the increased circuit consumption in the heparin group was circuit failure due to access issues; this is less likely to be due to the mode of anticoagulation but rather the line position, patients’ volume status and degree of haemodynamic instability.
There were several limitations in this study. This was primarily a small, prospective observational study and, as such, randomisation, allocation concealment and blinding of the intervention were not possible. This introduces a significant component of bias that cannot be readily adjusted for. Nevertheless, given that prior studies have not shown a patient outcome benefit from citrate versus heparin anticoagulation, cost should be a consideration for clinicians choosing an anticoagulation option. The two groups were not evenly matched in number, this is likely due to the fact that heparin has been the more familiar anticoagulation modality in our department, and the significant number of patients with hepatic failure admitted.
Importantly, this study was not designed or powered to detect differences in patient-centred outcomes and we note the difference in mortality between the two groups. All deaths were reviewed and determined to be attributable to underlying disease and no deaths were related to the method of anticoagulation. Whilst there is a possibility that our data may be suggestive of a causative relationship between method of anticoagulation and mortality, this has been disproven in multiple trials designed specifically to investigate patient-centred outcomes.7–10 Another potential contributing factor to the difference in mortality was that the patients in the heparin group were sicker than those in the citrate group as evidenced by the higher APACHE II scores. This is likely due to clinicians avoiding citrate anticoagulation in sicker patients with liver dysfunction. Our cost comparison did not take into account nursing workload or time in changing filters, as for our ICU, nursing costs are fixed with one nurse to be allocated to one patient when they are receiving CRRT. We also did not account for the environmental costs of disposal of consumables. Finally, as a single-centre study this reflects local practices; other sites may be able to achieve better cost results with even fewer blood tests and better contracts for consumables.
Ultimately the choice of anticoagulation for CRRT will need to consider several factors: patient bleeding risk, presence of hepatic dysfunction, treatment efficacy, estimated duration of therapy, departmental familiarity and cost. One suggestion is that for patients likely to require only a short duration of CRRT, heparin is the most cost-effective option, whereas for patients needing increased clearance and prolonged filter life, citrate may be the better choice.
Conclusion
Regional citrate anticoagulation is associated with an increase in cost of AUD$317.91 per patient per day of therapy compared with low-dose systemic heparin anticoagulation for CRRT.
Supplemental Material
Supplemental material for A cost comparison of regional citrate versus low-dose systemic heparin anticoagulation in continuous renal replacement therapy
Supplemental Material for A cost comparison of regional citrate versus low-dose systemic heparin anticoagulation in continuous renal replacement therapy by Chathuri U Dissanayake, Chrianna I Bharat, Brigit L Roberts and Matthew HR Anstey in Anaesthesia and Intensive Care
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
