Abstract
Acute kidney injury is a frequent complication in critically ill patients and often necessitates kidney replacement therapy. Optimising antimicrobial dosing in this population is essential yet challenging owing to altered pharmacokinetics in critical illness and the wide variability of prescriptions utilised in kidney replacement therapy. This case report investigates the pharmacokinetics of cefazolin in a critically ill patient receiving prolonged intermittent kidney replacement therapy, offering insights into dosing considerations in this clinical setting.
Keywords
Introduction
Septic shock causing acute kidney injury requiring kidney replacement therapy is associated with a 90-day mortality of 56%. 1 In the critically ill septic patient, multiple pharmacokinetic variables are altered, including an increased volume of distribution, alteration of protein binding capacity and organ dysfunction affecting clearance.2–5 Severe kidney dysfunction necessitating renal replacement therapy may further complicate antimicrobial dosing, especially when using prolonged intermittent kidney replacement therapy (PIKRT) with its wide variability of prescriptions, such as frequency, duration, blood and dialysate flow rates, all of which significantly impact clearance of highly dialysed antimicrobials such as cefazolin.4,6,7 Given the elevated mortality in this patient population and complexities of altered pharmacokinetics, there is a critical need to optimise antimicrobial dosing strategies to improve therapeutic efficacy and clinical outcomes.
Cefazolin is a first-generation cephalosporin which exhibits bactericidal action through bacterial cell wall synthesis inhibition.2,8 It is a front-line treatment option for methicillin-susceptible
Cefazolin has a molecular weight of 475 Da and is primarily renally excreted with 70–80% excreted as unchanged drug.8,13 In patients with end-stage kidney disease, the clearance is approximately 0.2 L/h. 6 Cefazolin is highly protein bound (80–90%) in healthy individuals, with a volume of distribution of 0.13 L/kg in patients with normal renal function, increasing to 0.16 L/kg in patients with end-stage kidney disease.5,6 This case report investigates cefazolin pharmacokinetics in a critically ill patient undergoing PIKRT.
Case report
A 77-year-old female (156 cm height, 49 kg weight) presented via the emergency department with disseminated MSSA bacteraemia associated with obstructive urosepsis of the left kidney, polyarticular septic arthritis, tricuspid valve endocarditis and septic pulmonary emboli. MSSA was isolated from multiple sets of blood cultures, bilateral knee and shoulder aspirates as well as urine and endotracheal specimens. The patient had a known penicillin hypersensitivity manifesting as nausea, vomiting and rash.
Operative management included insertion of left ureteric stent on day 1 of hospitalisation followed by bilateral arthroscopic knee and open right shoulder washouts on day 5. The patient was admitted to the intensive care unit (ICU) within 24 h of hospitalisation with septic shock and an established acute kidney injury (serum creatinine 215 µmol/L, estimated glomerular filtration rate (eGFR) 19 mL/min per 1.73 m2 from baseline serum creatinine of 75 µmol/L, eGFR 66 mL/min per 1.73 m2) which did not resolve with resolution of hydronephrosis, and necessitated kidney replacement therapy. During the sampling period, the patient remained oliguric with a urine output of 5–15 mL/h, with serum creatinine of 184 µmol/L and eGFR 22 mL/min per 1.73 m2. The first negative blood cultures occurred on day 7 of hospitalisation.
The patient was commenced on cefazolin from day 1 of the admission. During the sampling period, the patient was administered either intravenous (IV) cefazolin 1 g (~20 mg/kg) over 30 min every 12 h (while not undergoing PIKRT), or IV cefazolin 2 g (~40 mg/kg) over 30 min (while undergoing PIKRT).
PIKRT was prescribed in accordance with the ICU’s standard practices using the Fresenius 5008 with a 1.4 m2 filter (Fresenius, Sydney, Australia). On the sampling day, the dialysis session ran over 8 h with a blood flow rate at 200 mL/min; dialysate flow at 240 mL/min; predilutional substitution rate at 50 mL/min and a net total ultrafiltration of 2360 mL. This delivered a urea reduction ratio of 62.94% and a Kt/V (K = clearance, t = time, V = volume of distribution) of 1.31.
MSSA-directed therapy was continued for a total of 12 weeks owing to magnetic resonance imaging showing evidence of L2–L4 osteomyelitis/discitis. After 21 days of cefazolin treatment, the patient had several weeks of broader antimicrobial therapy including cefepime, meropenem and moxifloxacin owing to concerns of hospital acquired bacterial infections. Kidney recovery occurred approximately four weeks into admission and following approximately five months in hospital, including 17 days in ICU and over two months of rehabilitation, the patient was successfully discharged to a nursing home facility with stable kidney function (serum creatinine 78 µmol/L).
Informed consent was obtained to report this case.
Analytical methods
A total of eight blood samples were taken over a 24 h period which included one PIKRT session. These samples were centrifuged within 6 h at 1800 to 3000 rev/min for 10 to 15 min. Plasma specimens were stored at −80°C prior to assay. Total and unbound cefazolin concentrations were measured in plasma specimens by validated high performance liquid chromatography mass spectrometry at the University of Queensland Centre for Clinical Research, Brisbane, Australia. Analytes were validated from 1 to 500 mg/L for total cefazolin and 0.5 to 500 mg/L for unbound cefazolin, with an intra- and inter-run precision of <5% for total cefazolin and <10% for unbound cefazolin.
Compartmental modelling of unbound cefazolin concentrations was undertaken using NONMEM (v.7.5.1), 16 testing both one- and two-compartment models, with elimination from the central compartment and several residual error models (additive, proportional, combined). The model included assessment of two components of clearance to represent the endogenous (off-PIKRT) clearance and the PIKRT-related clearance. Given that no pre-dose concentration was available, and prior dosing included a period on continuous kidney replacement therapy, the pre-dose concentration was estimated as a part of the modelling procedure. Simulations were performed within NONMEM using the final model (detailed in Supplementary material supplement 1 online) to obtain time versus concentrations profile and percentage of drug removed during PIKRT of proposed dose regimens.
Results
The unbound cefazolin pharmacokinetic data is contained in Table 1, the fitted pharmacokinetic curve is plotted along with the observed concentrations in Figure 1. Modelling proceeded with two compartments and a proportional error model. There was a significantly better fit as compared with a one-compartment model (change in objective function value –34.795,
The influence of prolonged intermittent kidney replacement therapy (PIKRT) on unbound cefazolin pharmacokinetics (PK).
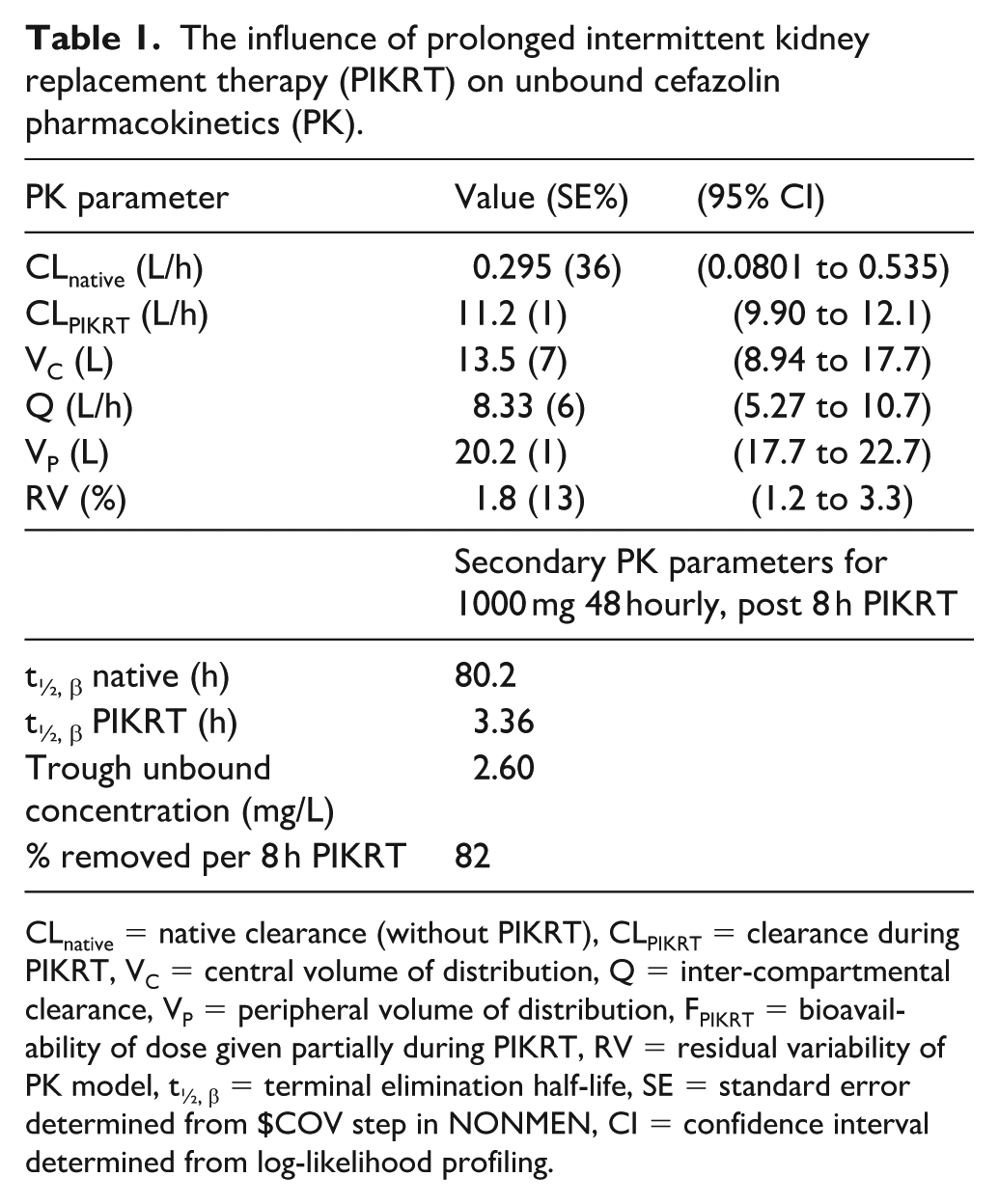
CLnative = native clearance (without PIKRT), CLPIKRT = clearance during PIKRT, VC = central volume of distribution, Q = inter-compartmental clearance, VP = peripheral volume of distribution, FPIKRT = bioavailability of dose given partially during PIKRT, RV = residual variability of PK model, t½, β = terminal elimination half-life, SE = standard error determined from $COV step in NONMEN, CI = confidence interval determined from log-likelihood profiling.
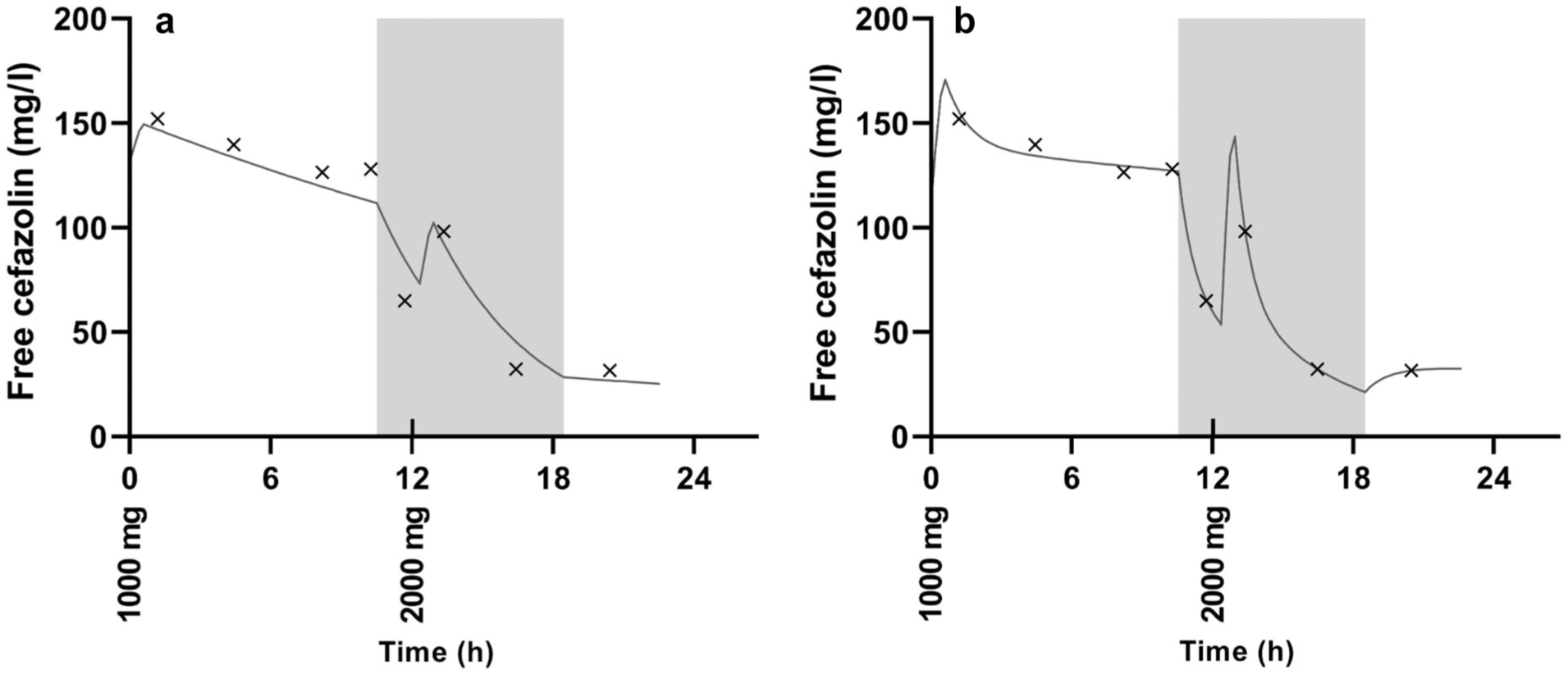
Time concentration profile of unbound cefazolin in a patient undergoing prolonged intermittent kidney replacement therapy (PIKRT). Time scale (in hours) is relative to the dose prior to the first sample. Solid line represents the time concentration profile based on the PK model parameters for a) 1-compartment model and b) 2-compartment model, crosses represent observed concentrations and shaded area represent times PIKRT was applied. Dose amounts (in milligrams) are included along the time axis at times doses were given.
Discussion
Cefazolin is removed by both intermittent haemodialysis and continuous kidney replacement therapy compared with poor native clearance in severe kidney impairment.3,5–7,17 Although cefazolin clearance is variable based on individualised dialysis parameters, generally a 15–20 mg/kg dose (or 1–2 g) dose is recommended after dialysis for intermittent haemodialysis, though these studies were not performed in critically ill patients with acute kidney impairment.6,7 A higher dose of cefazolin ranging from 1 g to 2 g twice daily is suggested for patients undergoing continuous kidney replacement therapy.3,5 In this patient, PIKRT enhanced cefazolin clearance (11.15 L/h) compared with when the patient was off PIKRT (0.295 L/h), which is consistent with other forms of kidney replacement therapy in patients with end-stage kidney disease.
6
Pharmacokinetic modelling using our patient data demonstrates the
We observed a high unbound fraction in the plasma samples for this patient (mean of 56% unbound fraction while not on PIKRT), which could be due to reduced protein binding due to hypoalbuminemia or the competitive binding of excess urea.2,6 Only the unbound fraction of cefazolin is expected to be cleared by dialysis, therefore, a higher unbound fraction might result in greater clearance. 2 Volume of distribution of unbound cefazolin determined by modelling was 33.7 L (0.69 L/kg), which is higher than in healthy patients with normal or impaired kidney function. Volume of distribution of hydrophilic antimicrobials, such as cefazolin, are likely increased in sepsis owing to administration of large volumes of fluids required for resuscitation and treatment as well as greater distribution into the interstitial space due to reduced protein binding, which can increase tissue penetration.4,6 Though higher concentrations might be seen in tissues, the peak plasma concentration will likely be reduced and the half-life will be prolonged, thus also leading to lower clearance by dialysis. 2 These dynamic and often competing pharmacokinetic alterations highlight the complexity of antimicrobial dosing in critically ill patients.
Supratherapeutic dosing resulting in elevated cephalosporin plasma concentrations has been associated with increased risk of central nervous system adverse drug reactions including encephalopathy, seizures, confusion and coma.18,19 Other risk factors for cephalosporin induced neurotoxicity include renal impairment, older age and pre-existing central nervous system disease.18,19 The balance between ensuring adequate plasma and tissue concentrations while avoiding supratherapeutic exposure should also be considered, particularly in patients with risk factors for neurotoxicity.
Limitations of this case report include the absence of tissue concentration measurements, which would reflect tissue penetration of cefazolin. Additionally, the pharmacokinetic model represents a point in time of this patient’s ICU course; given the rapidly evolving physiology in critical illness, ongoing reassessment of antimicrobial dosing is essential as patient specific variables change.
Supplemental Material
sj-txt-1-aic-10.1177_0310057X251396229 – Supplemental material for Cefazolin pharmacokinetics during prolonged intermittent kidney replacement therapy
Supplemental material, sj-txt-1-aic-10.1177_0310057X251396229 for Cefazolin pharmacokinetics during prolonged intermittent kidney replacement therapy by Jessica H Xu, Sam Salman, David JR Morgan, Hayoung Won, Steven C Wallis, John Dyer, Jason A Roberts and Matthew DM Rawlins in Anaesthesia and Intensive Care
Footnotes
Author contribution(s)
Declaration of conflicting interests
The authors have no conflicts of interest to declare.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplementary material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
