Abstract
A ‘can’t intubate, can’t oxygenate’ (CICO) situation is an uncommon and time-critical emergency. Many institutions have adopted a ‘scalpel–bougie–endotracheal tube (ETT)’ technique based on evidence produced by the 4th National Audit Project of the Royal College of Anaesthetists and 2015 Difficult Airway Society guidelines. We made a modification to the traditional ‘scalpel–bougie–ETT’ technique, using a shortened bougie and replacing the ETT with a cuffed Melker airway in a preassembled device (called ‘Secure Airway for Front-of-neck Emergencies’ (SAFE airway device)), which we felt might reduce cognitive load on a single operator in an emergency CICO situation. We then performed a simulation crossover study using 20 volunteer anaesthetic doctors to compare this modification with the traditional technique. After a standardised pre-recorded video demonstration, participants performed simulated tube insertion using both the standard ‘scalpel–bougie–ETT’ technique and the SAFE airway device in randomised order. Participants were filmed for accurate timestamping. The primary outcome was time to successful tube insertion while secondary outcomes included number of attempts and ease of insertion. Overall time to cuff inflation was shorter, statistically and practically, with the SAFE airway device compared with the ETT (median 30 vs. 52 seconds, P < 0.001). Twenty-five percent of participants required multiple attempts using the ETT method versus 5% using the SAFE airway device, which was also rated as being easier to use. This study demonstrates that the SAFE airway device was fast, effective, easy to use and acceptable to airway practitioners in a simulated manikin environment. These findings indicate that further studies of the SAFE airway device are warranted.
Introduction
A ‘can’t intubate, can’t oxygenate’ (CICO) situation is an uncommon and time-critical emergency occurring when failure to successfully oxygenate a patient via a facemask, supraglottic airway or endotracheal tube (ETT) necessitates emergency front-of-neck access (FONA). Emergency FONA cricothyroidotomies are an essential skill for anaesthetists and can be classified as scalpel- or cannula-based, whilst other techniques, such as emergency tracheostomy, are more frequently performed by a surgeon with specialist equipment and training. 1
Worldwide, many institutions have adopted a ‘scalpel–bougie–ETT’ technique based on evidence produced by the 4th National Audit Project of the Royal College of Anaesthetists (NAP4) and the 2015 Difficult Airway Society (DAS) guidelines.1 –3 The NAP4 study found that surgical airway techniques, when performed by surgeons, emergency physicians and anaesthetists, were consistently successful at achieving airway access, albeit with variable patient outcomes. 2 Conversely, the same audit identified that the failure rate of cannula cricothyroidotomy approached 60% in the cases studied. 2 The Australian and New Zealand College of Anaesthetists (ANZCA) supports either a cannula or scalpel technique, depending upon the choice and skill of the operator. 4 Both techniques are taught as part of ANZCA-endorsed continuing professional development emergency response workshops. 5
The traditional ‘scalpel–bougie–ETT’ technique, as described in the 2015 DAS guidelines, uses a transverse scalpel incision through the cricothyroid membrane to allow passage of a Frova intubating bougie (Cook Medical, Bloomington, USA), which then allows railroading of a size 6.0 ETT into the trachea. 3 This size of ETT, readily available in most critical care settings, is a compromise between ease of insertion and airflow resistance. However, the ETT’s shape may be suboptimal for FONA, as the advancing tip can catch on soft tissue during insertion.6,7 This has been noted before by Marimuthu et al., who proposed using a smaller size ETT or an airway with a tapered tip (such as the Melker cuffed emergency cricothyrotomy catheter (Cook Medical, Bloomington, USA)) to circumvent this issue. 7
In the authors’ experience, the 70 cm length of the Frova intubating bougie can cause delays in oxygenation during unassisted FONA, owing to the difficulty of loading the ETT with one hand, whilst also stabilising the bougie at the neck insertion site. 6 This difficulty results from the physical distance between the free bougie and the operator, the narrow calibre of the ETT, and the tendency of the bougie to catch on the internal surface of the ETT’s 15 mm proximal connector. This interruption may increase the risk of accidental dislodgment as a single operator may resort to releasing the bougie at the skin incision site in order to advance the ETT. The Melker cuffed emergency cricothyrotomy catheter is 110 mm long with an internal diameter of 5 mm. It is part of a percutaneous cricothyrotomy kit, using a wire-guided Seldinger technique to convert a cannula into an ETT. As shown by Marimuthu et al., the Melker catheter fits more closely over the Frova bougie (compared with size 6.0 ETT; Figure 1) and allows easy passage through the neck with minimal tissue hold-up. 7 Similar to the ETT, the cuffed Melker catheter allows for satisfactory airway protection and lung ventilation via a self-inflating bag or ventilator circuit.

Airway design. Demonstrating the (1) cuffed Melker airway’s tapered tip compared with (2) standard size 6.0 endotracheal tube.
We made a modification to the traditional ‘scalpel–bougie–ETT’ technique, using a shortened bougie and replacing the endotracheal tube with a pre-loaded Melker cuffed catheter. A standard Rapi-Fit connector (Cook Medical, Bloomington, USA) was attached to the end of the bougie to prevent the Melker catheter from separating during insertion (Figure 2). We felt that this pre-assembled device, called the ‘Secure Airway for Front-of-neck Emergencies’ (SAFE), has several potential advantages. First, the shorter 30 cm Frova intubating bougie is easier to hold and manipulate (Figure 2). Second, the pre-loaded airway device eliminates the issue of the plastic ETT connector catching on the bougie, potentially delaying advancement. Third, the Melker catheter’s tapered distal end fits closely over the bougie and seems ideally shaped to minimise hold-up on pre-tracheal soft tissue. 7 Fourth, the Melker airway can only be inserted to a depth of 90 mm, minimising the risk of accidental endobronchial intubation, something previously identified as a potential issue with over-insertion of a standard size 6.0 ETT. 6 Similarly, the shortened Frova bougie will allow only a maximum of 180 mm to be inserted into the trachea, theoretically reducing the risk of bronchial tree trauma. 6 Finally, the Melker catheter side flanges make it is easier to secure after insertion than a standard size 6.0 ETT, potentially reducing the chance of accidental dislodgment.
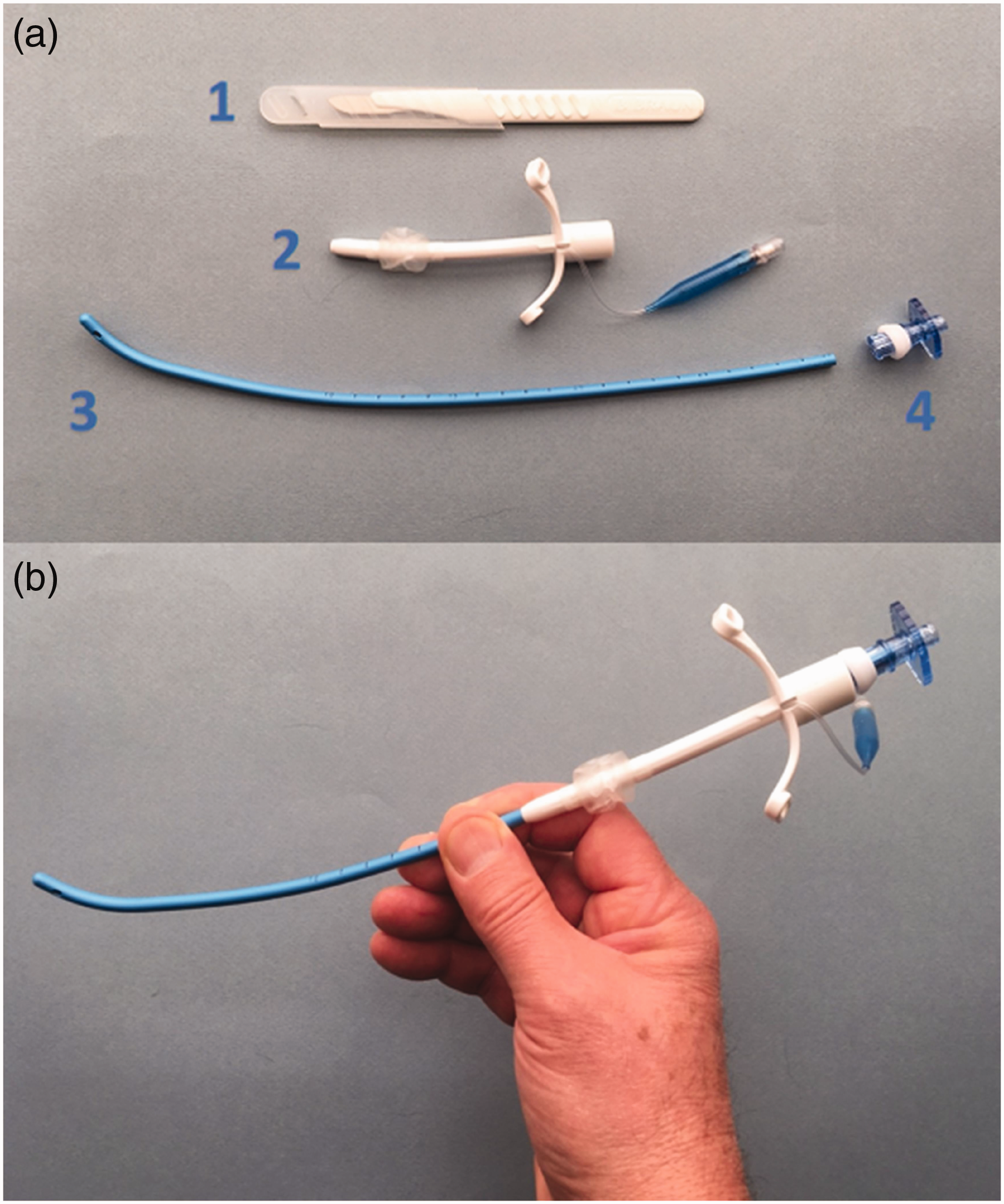
The preassembled Secure Airway for Front-of-neck Emergencies (SAFE) airway device). (a) (1) Size 10 disposable scalpel, (2) 5.0 cuffed Melker airway, (3) shortened Frova intubating bougie, (4) Rapi-Fit connector and (b) Preassembled SAFE airway device.
The SAFE airway device is inserted in a similar manner to the ‘scalpel–bougie–ETT’ technique (following the 2015 DAS guidelines), using a transverse scalpel incision through the cricothyroid membrane to allow passage of the shortened bougie into the trachea. Once the bougie has been inserted into the trachea, the Melker catheter is advanced over the bougie into the airway. The bougie is then removed from the Melker catheter prior to endotracheal cuff inflation, which is followed by test ventilation and confirmation of appropriate placement.
The aim of this study was to assess the utility of using the SAFE airway device for emergency FONA when performed by experienced and skilled practitioners, when compared with the traditional ‘scalpel–bougie–ETT’ techniques. We hypothesised that, in a simulated environment, insertion of the SAFE device, compared with a ‘scalpel–bougie–ETT’ technique would be (1) faster, (2) preferred by operators, and (3) have fewer unsuccessful attempts.
Methods
To test our hypothesis, we performed a simulation crossover study using the VBM Crico Trainer – Frova (Sulz, Germany), a low-fidelity bench model, and standardised equipment set-ups (Figure 3). This standardisation meant that the only source of variability between attempts was the CICO airway device used by participants (SAFE airway device vs. ‘scalpel–bougie–ETT’), with all other available equipment remaining identical. Ethics approval was obtained from the University of Tasmania Human Research Ethics Committee (HREC ID H0026506).
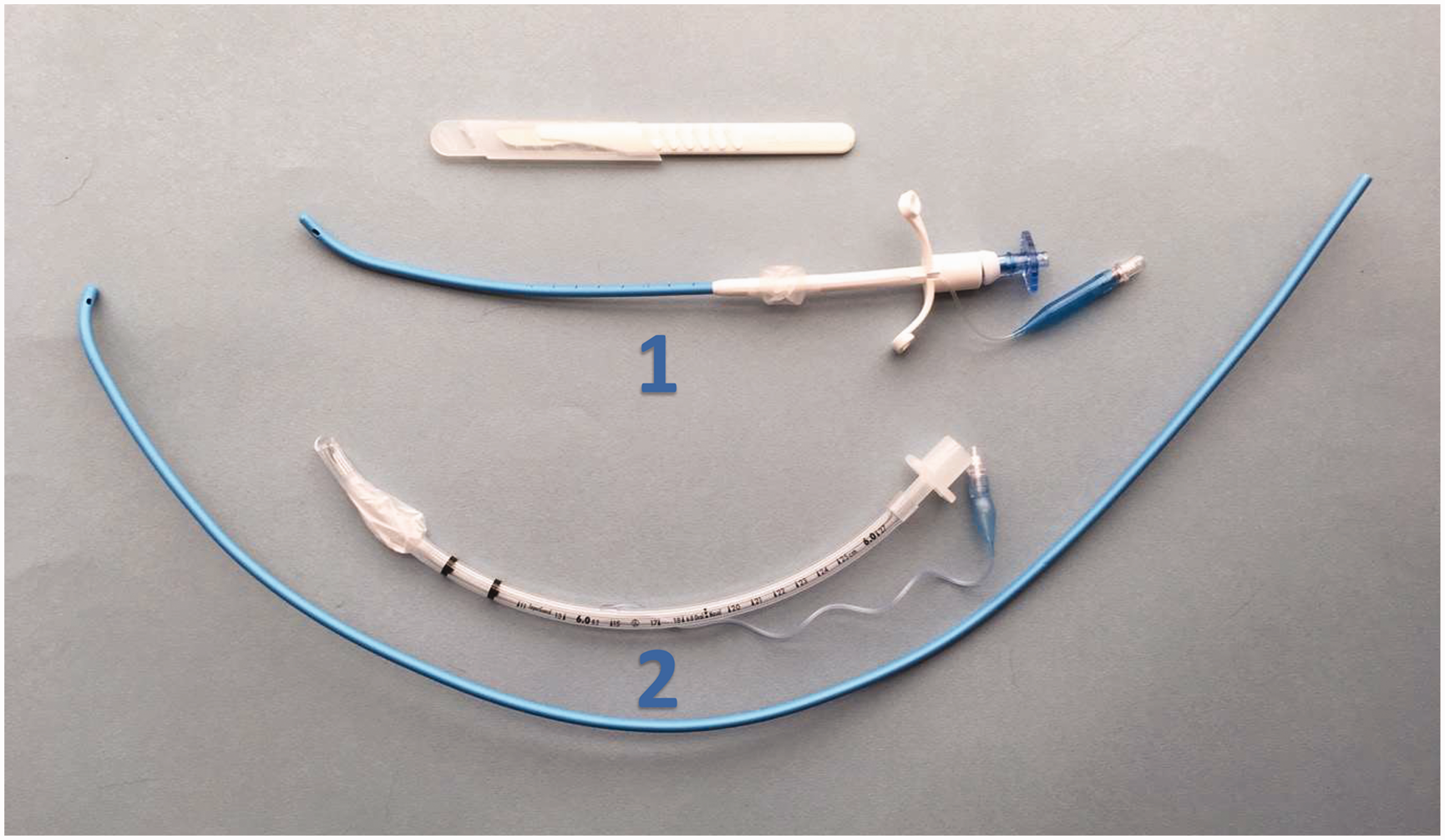
Study equipment. Size 10 disposable scalpel with (1) preassembled Secure Airway for Front-of-neck Emergencies airway device and (2) size 6.0 endotracheal tube and Frova intubating bougie.
To calculate sample size, we set power at 0.95, with the following a priori assumptions (based on prior experience): a mean (standard deviation (SD)) time difference of 10 (SD 10) s, using a one-tailed paired Wilcoxon test for an alpha of 0.05. A sample of 11 was required. The primary outcome was analysed using a one-tailed paired Wilcoxon (signed-rank) test using SPSS version 1.0.0.1347 (IBM, NY, USA). Secondary outcomes were analysed using descriptive statistics.
Twenty participants were randomly selected from a group of anaesthetists (registrars and consultants) who had volunteered to participate in the study following distribution of advertising material. All participants had previous experience in simulated CICO FONA. All introductory briefs were carried out by the same investigator to ensure standardisation between participants. Each participant was asked to fill in a pre-attempt questionnaire (Appendix 1) and then view a standardised pre-recorded video demonstration of the SAFE airway device and ‘scalpel–bougie–ETT’ techniques. The videos, demonstrating the DAS scalpel cricothyroidotomy (palpable anatomy) technique for each assembly, were shown once only. The SAFE airway device insertion video was identical to the DAS ‘scalpel–bougie–ETT’ technique up until the point of successful access to the trachea using a size 10 scalpel. At this point, rather than inserting a standard 70 cm Frova bougie, the shortened Frova bougie included as part of the SAFE airway device is inserted instead, followed by advancement of the Melker airway pre-mounted onto the bougie. As in the DAS ‘scalpel–bougie–ETT’ technique, removal of bougie is followed by cuff inflation and ventilation.
Participants then went on to perform both the SAFE device and ‘scalpel–bougie–ETT’ techniques. The order in which participants performed the interventions was block randomised. For the ETT attempt, participants were presented with a standardised kit with all packaging removed (simulating the SET stage in the VORTEX Priming Status algorithm where the selected proceduralist prepares CICO equipment and identifies front of neck anatomy 8 ). In this kit, the Frova bougie was 70 cm long. For the SAFE attempt, participants were presented with a pre-assembled SAFE device, also free of packaging. This consisted of a 5.0 mm cuffed Melker endotracheal tube mounted on a shortened 30 cm Frova bougie (preserving the Coudé tip), with a standard 15 mm Rapi-Fit adaptor in-situ on the proximal end of the device.
Each participant was filmed performing the technique to facilitate accurate timestamping. Additionally, all attempts were analysed by two blinded reviewers who graded subjective ease of device insertion (from 0 = very difficult to 6 = very easy). Although these reviewers were part of the study team, strategies to prevent individual participant identification were made, including encouraging the use of gloves and filming only their hands. Although the trial was conducted as a low-fidelity simulation, participants were encouraged to engage with the manikin as a real clinical situation, being thorough, precise and not aiming for the fastest completion possible. If an insertion attempt using either the ETT or SAFE airway techniques was unsuccessful, such as incorrect positioning of the airway device outside the trachea, the timer was continued until either successful insertion was performed or the participant had three consecutive failed attempts. Post completion of both techniques, participants were asked to complete another questionnaire ranking their perceived ease of insertion for each device (Appendix 1).
The primary outcome was time to successful insertion, measured from the moment the participant first touched the bougie to the point of cuff inflation in the simulated trachea. Time taken to palpate the manikin’s airway anatomy and perform the initial surgical incision were not included in the cumulative measured time as these were assumed to be similar between using the SAFE device and the ‘scalpel–bougie–ETT’ technique (and therefore would not impact the data being assessed). Secondary outcome measures were number of attempts, number of incorrect (paratracheal) placements, observer and participant ease of insertion scores (X-point Likert-type scale from X ‘Very Easy’ to Y ‘Very Difficult’), frequency of bougie ‘catching’ inside the ETT and skin ‘hold-up’ on advancement of the endotracheal tube. We also analysed participants’ free text survey responses (see Appendix 1).
Results
Twenty participants performed a total of 40 FONA interventions. Participants reported a median 12 years of anaesthesia experience (interquartile range 7–19.5). Quantitative results are listed in Table 1.
Comparison of data. Data are presented as median, range and percentage.
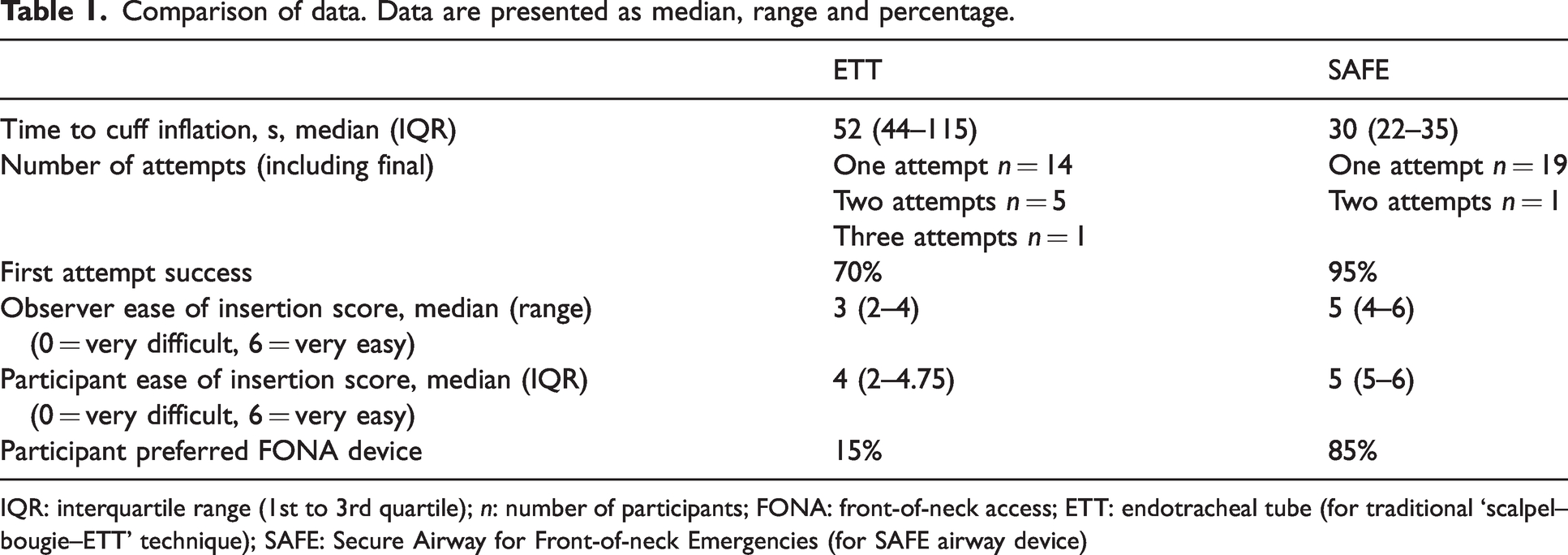
IQR: interquartile range (1st to 3rd quartile); n: number of participants; FONA: front-of-neck access; ETT: endotracheal tube (for traditional ‘scalpel–bougie–ETT’ technique); SAFE: Secure Airway for Front-of-neck Emergencies (for SAFE airway device)
Time to cuff inflation was shorter, both statistically and practically, with the SAFE device than the ETT (median 30 s vs. 52 s, P < 0.001) (Figure 4). All participants were ultimately successful in placing the tube in the trachea; however, the first pass insertion success was 19/20 in the SAFE device group (95%) versus 14/20 (70%) in the ETT group. Time to cuff inflation was positively skewed for the ETT method, but normally distributed for the SAFE airway device. Outliers with very long insertion times in the ETT group often required more than one attempt to achieve correct placement.
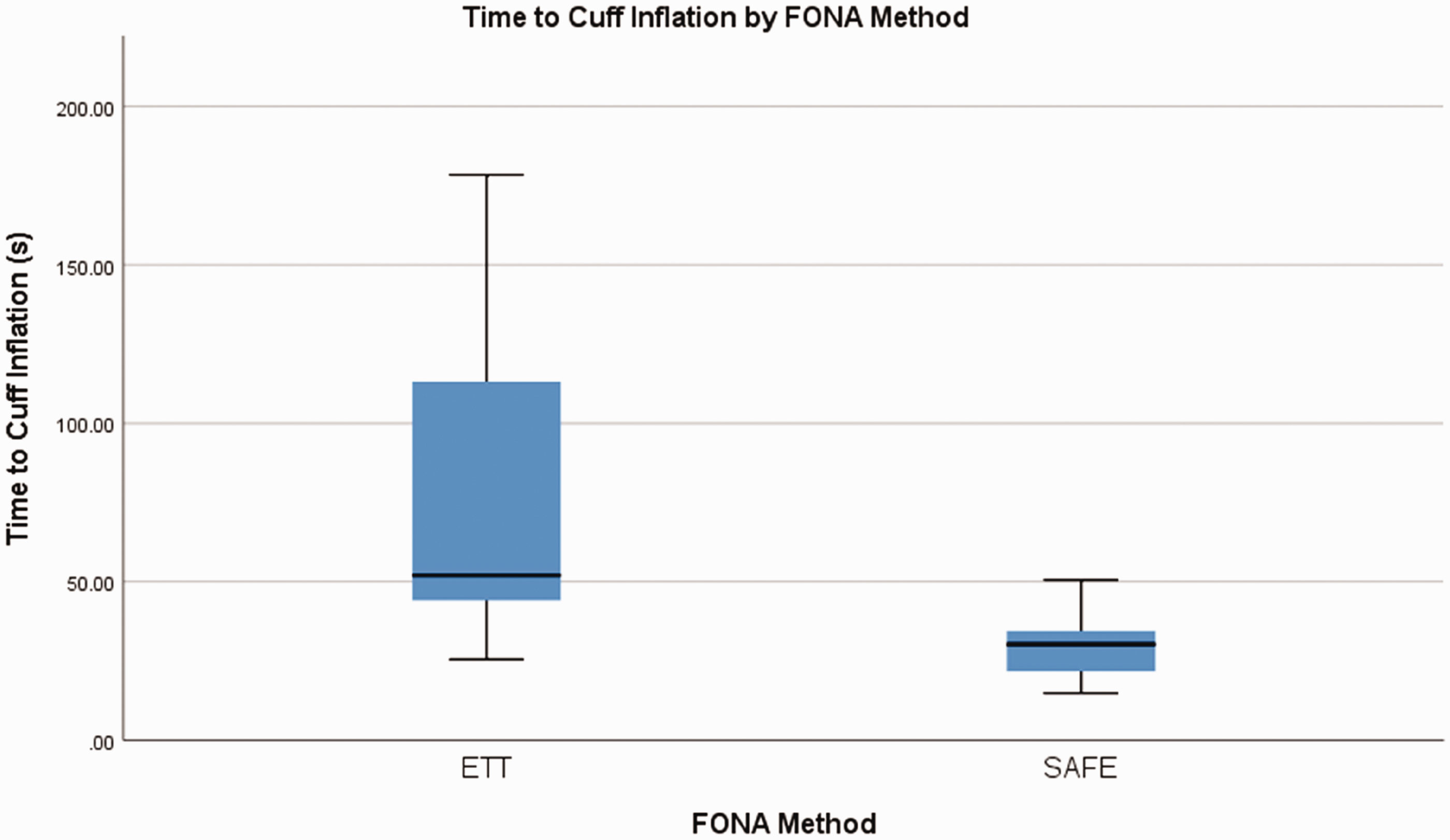
Box plots showing time to cuff inflation (in seconds) by participants for endotracheal tube (ETT) and preassembled Secure Airway for Front-of-neck Emergencies (SAFE) airway device. Demonstrates shorter time to successful insertion with SAFE airway device, P < 0.001. FONA: front-of-neck-access.
As cohorts, both the blinded reviewers and the participants rated the SAFE method as being easier to use than the ETT method. A Wilcoxon signed-ranks test indicated that SAFE ‘ease of use’ ranks were higher than ETT ranks (observers Z = –3.36, P < 0.001; participants Z = –3.65, P < 0.001). Analysis of paired ‘ease of use’ ranks showed that there was no significant association between perceived difficulty in using either technique; most participants who experienced difficulty with the ETT approach found the SAFE airway device easier.
Free text survey responses were manually coded. Perceived benefits were that the Melker airway was easier to pass through tissue (n = 5), that it was more secure when inserted (n = 6), that pre-assembly saved time (n = 5), that there was a lower risk of endobronchial intubation (n = 2) and that the SAFE approach was easier to use as sole operator (n = 2). Only two participants reported concerns with the SAFE airway device, both citing lack of familiarity with equipment. Overall, 85% of participants stated that the SAFE airway device would be their preferred technique for FONA, in comparison with 15% who chose ‘scalpel–bougie–ETT’.
Discussion
This study was designed to test the feasibility and acceptability of this novel FONA approach in a simulated setting. We hypothesised that the SAFE airway device would be faster, easier and would result in fewer unsuccessful attempts than the traditional ‘scalpel–bougie–ETT’ technique. The results support these hypotheses. Given that participants were not aware of the SAFE airway device prior to enrolment, the high rate of successful and timely insertion showed that even after a single pre-recorded video demonstration, candidates who identified they were familiar with CICO techniques were able to utilise the device effectively without prior exposure. Ease of use is also supported by the significantly smaller spread in insertion times for the SAFE airway device compared with the ETT method.
The pre-assembled nature of the device (in a single packet) was seen as a potential advantage by several participants if they were faced with a ‘real-life’ CICO situation. Indeed, the addition of a size 10 scalpel is all that is required for this emergency airway kit. This might save significant time during a period of high cognitive load, for example where a single operator is required to transition from failed supraglottic airway management into an emergency FONA technique. The study confirmed previous observations that the Melker airway device slid easily over the bougie and its tapered shape allowed minimal hold-up, even on insertion into the synthetic CICO trainers. This varied significantly to attempts made with the size 6.0 ETT, where hold-up was frequently noted both when mounting the ETT onto the bougie and at the point of skin insertion.
Malposition was less frequent with the SAFE airway device. There were seven unexpected paratracheal ETT placements, compared with only one SAFE airway device misplacement. Reviewers also noted several endobronchial bougie and size 6.0 ETT insertions that may have posed a hazard in a real-life clinical situation. Using the 70 cm Frova bougie, as described and taught in the DAS technique, could lead to bronchial tree injury, even if inserted less than 50% of its length. In contrast, the SAFE airway device was constructed to avoid this safety issue. The chosen 30 cm length seemed to be the optimal balance between weight, stability and ease of insertion while still minimising the risk of accidental dislodgment or airway trauma.
Although there does seem to be obvious potential benefits of this novel CICO SAFE airway device, we have identified a few potential risks. Some participants expressed concern that the length of the Melker device may be insufficient if used in a morbidly obese patient, potentially highlighting the need for different lengths of the SAFE airway device. However, in the setting of morbid obesity and an impalpable cricothyroid membrane, an alternative technique using midline vertical incision followed by blunt dissection would be used, as advocated by the DAS guidelines. This would allow the standard SAFE airway device to be used, although there remains a risk of the Melker airway’s side flanges abutting the surrounding soft tissue, causing difficulty with insertion or risk of dislodgment. Another consideration is availability of equipment—a size 6.0 ETT is available in most settings where anaesthesia is performed, whereas the Melker percutaneous cricothyrotomy kit is most frequently utilised in critical care environments. Therefore, training would be required with both standard DAS ‘scalpel–bougie–ETT’ equipment and the SAFE airway device in order to ensure technique familiarity for emergency situations. As both the Frova bougie and cuffed Melker airway are manufactured by Cook Medical, the authors believe that the creation of a preassembled kit in a single package should be technically straightforward.
This study demonstrates that the SAFE device is fast, effective, easy to use and preferred by airway practitioners in a simulated patient manikin environment. Time to insertion and malposition rates are favourable when compared with traditional ETT techniques, despite participants’ lack of previous exposure to the SAFE airway device. The authors’ particular belief is that the pre-assembled nature of the SAFE airway device will be particularly valuable in reducing the cognitive load on a single operator in an emergency CICO situation. It must be noted that it is unclear which factor contributes most towards the SAFE airway device’s perceived benefits as this was not studied independently, although it is likely that the pre-assembled nature of the device and the Melker airway’s tapered shape both contribute.
The authors believe that these preliminary findings justify further research to determine whether the pre-assembled SAFE airway device performs equally well in the hands of non-anaesthetists and in other settings, such as cadaveric and live tissue simulation. Such projects will help to ensure that clinicians responsible for managing high-acuity low-occurrence events such as a CICO situation can draw upon equipment and techniques that are simple, robust and effective.
Footnotes
Author Contribution(s)
Author note
Please note that this study was initially envisioned to include a non-anaesthetic doctor recruitment arm, but we were unable to recruit sufficient participants.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
