Abstract
At the Royal Perth Hospital, we have been developing and teaching a can’t intubate, can’t oxygenate (CICO) rescue algorithm for over 19 years, based on live animal simulation. The algorithm involves a ‘cannula-first’ approach, with jet oxygenation and progression to scalpel techniques if required in a stepwise fashion. There is little reported experience of this approach to the CICO scenario in humans. We present eight cases in which a cannula-first Royal Perth Hospital approach was successfully implemented during an airway crisis. We recommend that institutions teach and practice this approach; we believe it is effective, safe and minimally invasive when undertaken by clinicians who have been trained in it and have immediate access to the requisite equipment. The equipment is low cost, comprising a 14G Insyte cannula, saline, 5 ml syringe and a Rapid-O2. Training can be provided using low-fidelity manikins or part-task trainers.
Introduction
‘Can’t intubate, can’t oxygenate’ (CICO) rescue techniques involve oxygen delivery via front-of-neck access, a process that can be described as percutaneous emergency oxygenation (PEO). 1 Methods for PEO can be divided into two groups. In one group the neck and airway are opened with a scalpel to facilitate the insertion of a tracheotomy tube. In the other group the airway is punctured with a needle or cannula to permit rescue oxygenation, prior to further airway management. In the absence of robust clinical data, the question of which method is better remains unanswered. 2
To capture the strengths and mitigate the weaknesses of the two available PEO methods, a ‘cannula-first’ approach to the CICO scenario was developed by Heard and colleagues in the Royal Perth Hospital (RPH) Animal Training Facility, allowing progression to more invasive scalpel techniques if required in a stepwise fashion. 3 The approach is summarised in Figure 1. If a cannula is successfully placed, a Seldinger conversion to a Melker 5.0 mm cuffed emergency cricothyrotomy catheter (Cook Medical Inc., Bloomington, IN, USA) is advised, unless PEO via the cannula allows the patient to be safely woken or the trachea to be intubated by the oral or nasal route. This approach has been refined to maximise success, based on experience from over 10,000 cannula and scalpel PEO attempts by senior anaesthetists on anaesthetised sheep, in a high-fidelity model that provides real-time bleeding, stress and absolute endpoints of end-tidal carbon dioxide (CO2) and peripheral oxygen saturation (SpO2).
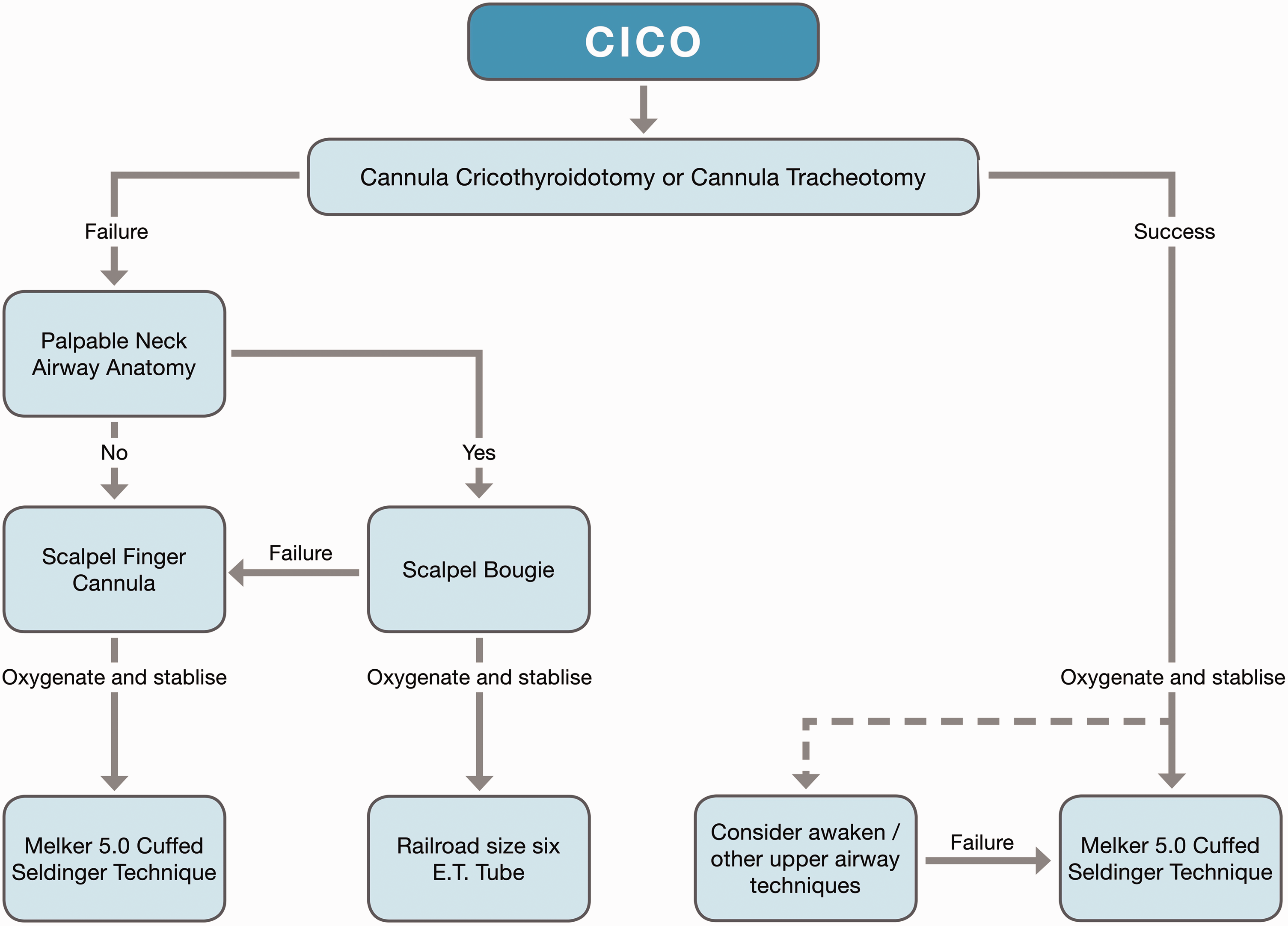
The Royal Perth Hospital cannula-first algorithm for managing the can’t’ intubate, can’t oxygenate (CICO) scenario. Melker5.0 (Cook Medical Inc., Bloomington, IN, USA).
Despite the high fidelity of the RPH anaesthetised sheep model, there are important anatomical and physiological differences to human patients in an airway crisis. Furthermore, in the absence of appropriate training and equipment, cannula-first techniques in patients have been associated with low success and high complication rates. 4 It has therefore been suggested that clinical evidence to support the RPH cannula-first approach is needed before widespread adoption can be recommended. 5 We report a series of eight cases in which a cannula-first technique consistent with RPH teaching was deployed during an airway crisis. These represent all such cannula-first cases over the period of which we are aware.
Materials and methods
Clinicians who have attended the RPH Wet Lab or its affiliated courses are asked to alert our group to CICO cases they have been involved in since attending the training. A list of these cases is maintained by the senior author (AH). In February 2018, we collated 10 years’ worth of these cases. During this period there were approximately 450 clinician attendances at the RPH Wet Lab, a majority of whom were anaesthetic trainees and consultants, but with a significant minority of other airway practitioners (emergency physicians, intensive care physicians, GP anaesthetists and paramedics). In any given year, nearly all senior trainees in our anaesthetic department attend the Wet Lab once; consultants tend to attend every 3 to 5 years. To the best of our knowledge all CICO cases that occurred at RPH during this period are included in the analysis; however, most clinicians who attended the RPH Wet Lab over this period were not permanent staff and may have encountered CICO scenarios elsewhere and not reported them. In addition, approximately 150 clinicians attended the All Wales Airway Group courses where the same algorithm, techniques and equipment choices are taught in a standardised way. 6 Reports of CICO cases from these clinicians were included in the analysis.
We contacted all lead clinicians involved in the cases, and retrospectively examined case notes, to identify themes and learning points. Cases meeting the following criteria were included: the lead clinician managing the case responded to our requests for details concerning management of the airway crisis; a cannula-first RPH rescue approach was used during the airway crisis; the lead clinician managing the case had attended the RPH Wet Lab course, or the All Wales Airway Group CICO course; the clinician had access to equipment required for the RPH CICO rescue approach; and the key clinical details in the clinician’s reports could be verified with the patient notes or another team member that witnessed the case. A ‘cannula-first RPH CICO rescue approach’ was defined as one involving the percutaneous use of a 14G Insyte cannula (Becton Dickinson, Sandy, UT, USA), including a jet oxygenation strategy described by Heard and colleagues at the time of the incident.1,3
Minimum data extracted from source documents included year, country, in-hospital location, clinical situation, clinician qualifications, prior training, equipment availability, cannula used, PEO technique details, and the final airway management and patient outcomes. Patient written consent or a waiver of consent (provided in writing by the RPH Human Research Ethics Committee if the patient had died since the case occurred) was obtained for each case. Completion of the manuscript was delayed when the senior author suffered a catastrophic cervical spine injury in September 2018.
Results
Twelve cases were reported to the senior author, eight of which satisfied the inclusion criteria. These are described chronologically below and are summarised in Table 1. Of the excluded cases, one followed the RPH CICO rescue approach but had already been published as a single case report. 7 One case used the RPH jet oxygenation strategy during a failed extubation following a neck dissection, but the oxygen was delivered via a Portex Mini-trach (Portex, Smiths Medical, Minneapolis, MN, USA) inserted pre-emptively by the surgeon before the end of the case. One case utilised the RPH cannula insertion technique prior to Melker conversion, in the context of tenuous oxygenation via a supraglottic airway device and without any jet oxygenation, which is a situation considered in our teaching but is not itself a CICO scenario. Finally, one case described the use of the RPH ‘scalpel-finger’ technique 1 to identify the trachea in a patient with impalpable anterior neck anatomy, but then proceeded directly to Melker conversion without jet oxygenation due to a lack of available equipment. The reporters of these cases were notified of the exclusion and were encouraged to publish their cases independently.
Summary of cases.
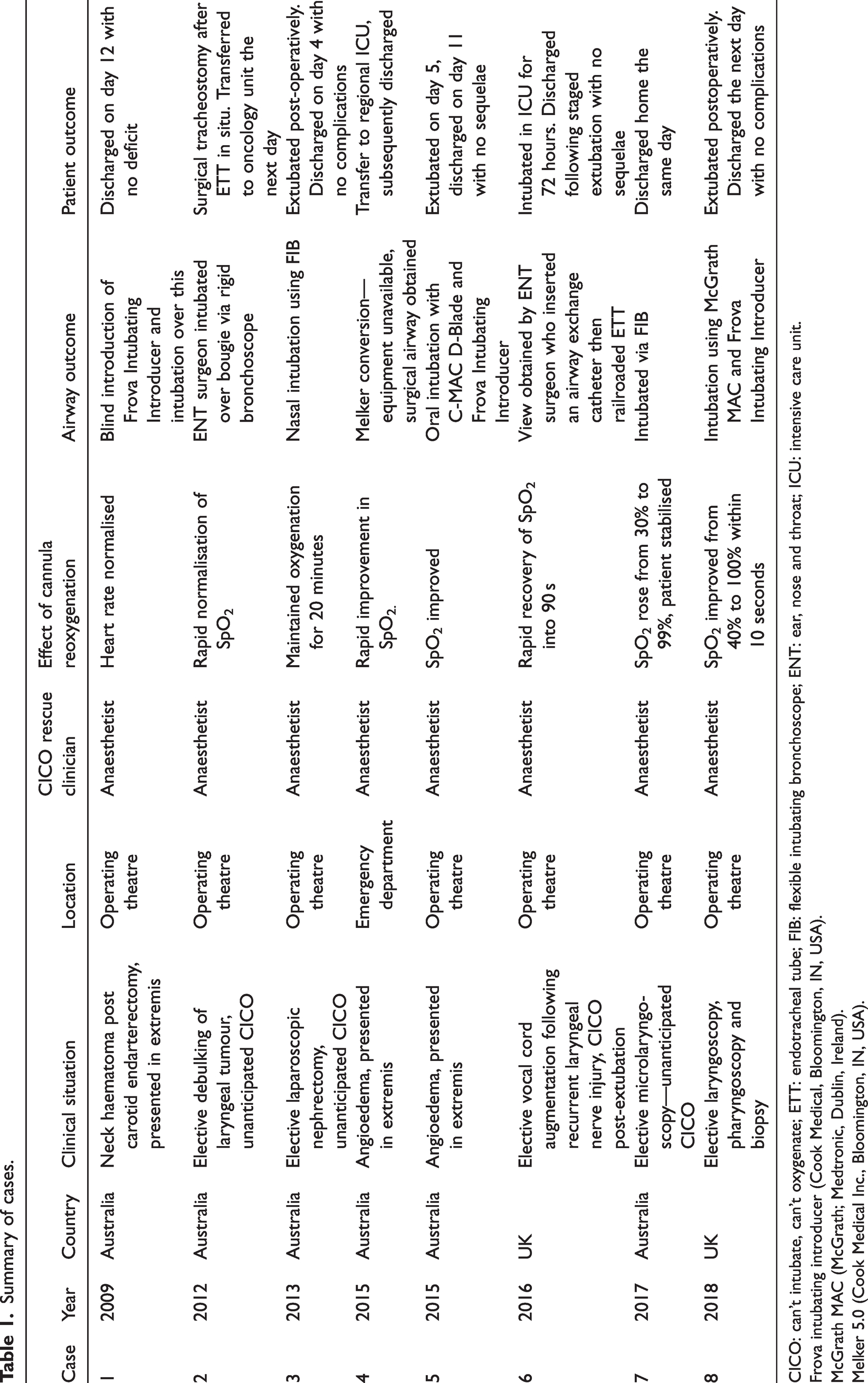
CICO: can’t intubate, can’t oxygenate; ETT: endotracheal tube; FIB: flexible intubating bronchoscope; ENT: ear, nose and throat; ICU: intensive care unit.
Frova intubating introducer (Cook Medical, Bloomington, IN, USA).
McGrath MAC (McGrath; Medtronic, Dublin, Ireland).
Melker 5.0 (Cook Medical Inc., Bloomington, IN, USA).
Case 1 (2009, Australia)
An 83-year-old man was transferred from a ward to the operating theatre in extremis, due to a large neck haematoma on day 2 after elective carotid endarterectomy. An attempt at pre-induction, pre-emptive percutaneous cannula insertion into the airway failed. Evacuation of the haematoma by the surgeon during attempted pre-oxygenation did not improve the ability of the patient to breathe. The patient became bradycardic. After induction, facemask ventilation failed, laryngoscopy revealed no discernible landmarks and a supraglottic airway device (SAD) was inserted but was not effective. Cannula cricothyroidotomy with a 14G Insyte cannula was successful allowing oxygenation via a Manujet (VBM GmbH, Sulz, Germany). It became apparent that breath stacking was occurring, and assisted expiration was required. The patient’s heart rate normalised although SpO2 was still unrecordable. A Frova Intubating Introducer (Cook Medical, Bloomington, IN, USA) was successfully passed blindly into the trachea and intubation was performed over this with a size 7.0 mm endotracheal tube (ETT). The patient remained intubated for 2 days and had a 4-day intensive care unit (ICU) stay. He had no neurological deficit and was discharged 12 days later.
Case 2 (2012, Australia)
A 63-year-old woman presented for debulking of a laryngeal tumour. After intravenous induction there was a large epiglottic tumour seen on videolaryngoscopy which bled profusely, resulting in a loss of view. Facemask ventilation became difficult and resulted in blood entering the lungs; SpO2 dropped and a decision to proceed to PEO was made. Cannula cricothyroidotomy with a 14G Insyte cannula was performed, followed by oxygenation via a Rapid-O2 (Meditech Systems Ltd., Dorset, UK), resulting in rapid normalisation of the SpO2. An ear, nose and throat (ENT) surgeon then used a rigid bronchoscope to intubate via a Frova intubating introducer. Once intubated a semi-elective tracheostomy was performed. The patient was woken up postoperatively and transferred to the oncology unit the next day.
Case 3 (2013, Australia)
A 51-year-old 83 kg man presented for laparoscopic nephrectomy. Preoperative airway assessment showed an interdental distance of 4 cm with reduced jaw protrusion. After induction, facemask ventilation was unsuccessful despite adequate paralysis and use of an oropharyngeal airway and two-person technique. It proved impossible to insert a size 3 blade C-MAC (Karl Storz, Tuttlingen, Germany) videolaryngoscope (VL) into the mouth so a size 4 classic LMA (LMA-Classic Intavent Orthofix, Maidenhead, UK) was inserted. The device seal was poor, and oxygenation was inadequate. Passing a flexible intubating bronchoscope (FIB) into the glottis through the supraglottic airway device (SAD) proved impossible. A 14G Insyte cannula was placed through the cricothyroid membrane (CTM) to allow oxygenation via a Rapid-O2 prior to SAD removal to facilitate further FIB attempts. Oxygenation was maintained over 20 minutes; oral FIB passage via a Berman intubating airway (Vital Signs, Totowa, NJ, USA) was unsuccessful. Nasal FIB insertion was successful on the second attempt, allowing railroading of an ETT.
The cannula was left in situ for extubation at the end of the operation. There were no complications.
Case 4 (2015, Australia)
A 31-year-old man was admitted to an emergency department (ED) with stridor and airway swelling due to angioedema secondary to angiotensin-converting enzyme inhibitor use. After induction and paralysis, videolaryngoscopy with a C-MAC was attempted by two consultant anaesthetists with no identifiable anatomy. Facemask ventilation failed and hypoxia ensued. A 14G Insyte cannula was inserted through the CTM and oxygenation via Rapid-O2 led to a rapid improvement in SpO2. It was felt intubation via the upper airway would be near impossible and percutaneous cuffed airway insertion was necessary, so a Melker airway was called for but was found to be paediatric sized. An attempt at SAD insertion was made which was successful. A surgeon then performed a surgical airway which was difficult requiring both a horizontal and vertical incision. The patient was transferred to an ICU then subsequently discharged.
Case 5 (2015, Australia)
A 73-year-old man presented with angioedema of the tongue. He was distressed and uncooperative. An awake fibreoptic intubation was attempted but failed and the patient desaturated. He was induced and paralysed. An intubation attempt with a C-MAC failed. A 14G Insyte cannula was placed through the CTM with oxygenation via a Rapid-O2. A second attempt at endotracheal intubation via the upper airway was successful using a C-MAC D-Blade and Frova intubating introducer. He was discharged with no sequelae.
Case 6 (2016, UK)
A 74-year-old woman presented for unilateral vocal cord augmentation. She had a known previous Cormack and Lehane (C-L) grade 4 laryngoscopy, so an advanced airway management strategy involving jet ventilation was planned. A Hunsaker Mon-Jet tube (Xomed, Jacksonville, FL, USA) was placed using a C-MAC D-Blade. A SAD was placed at the end of the procedure but failed during lightening of anaesthesia with no facemask oxygenation possible and loss of the view with the D-Blade. The surgeon attempted rigid bronchoscopy but the light source failed and SpO2 dropped to 69% so a second anaesthetist made the decision to proceed to PEO. A 14G Insyte cannula was successfully placed and after a single jet with a Rapid-O2, SpO2 rose from the low 50s to the low 90s. This allowed time for re-intubation via the upper airway—the surgical equipment problem was rectified, and a view of the glottis re-established allowing insertion of an airway exchange catheter and railroading of an ETT. Due to significant airway oedema, the patient remained intubated for 72 hours in the ICU with the cricothyroid cannula in situ. After staged extubation the patient was discharged home a few days later without further complications.
Case 7 (2017, Australia)
A 54-year-old man presented for elective microlaryngoscopy in the setting of laryngeal cancer and recent radiotherapy. After induction, it was not possible to facemask ventilate, a grade 4 C-L laryngoscopic view was obtained with distorted anatomy, a SAD was inserted but failed and SpO2 fell to 30%. A decision to perform PEO was made. There were no discernible landmarks; however, location of the trachea with a 14G Insyte cannula was successful. A Rapid-O2 was used to insufflate oxygen; the chest was noted to rise on insufflation and then fall, with a rapid return of SpO2 to 99%.
The patient was subsequently successfully intubated with an ETT via an FIB. The cannula was withdrawn with no apparent bleeding. The patient was extubated with no sequelae and discharged the same day.
Case 8 (2018, UK)
A 74-year-old 80 kg man presented for elective laryngoscopy, pharyngoscopy and biopsy. He was pre-oxygenated using high-flow humidified nasal oxygenation (Optiflow Fisher and Paykel Healthcare Limited, Auckland, New Zealand) at 30 l/min then general anaesthesia was induced and nasal oxygen flow rate increased to 70 l/min. Laryngoscopy with a McGrath MAC (McGrath; Medtronic, Dublin, Ireland) VL revealed a partially visible, anatomically distorted epiglottis. The glottis was not visible. Pooled secretions were suctioned eliciting a small amount of contact bleeding. In view of the distorted anatomy and bleeding, blind intubation was deemed unwise and intubation with an Ambu aScope 3 (Ambu A/S, Ballerup, Denmark) FIB was attempted. Before visualisation of the glottis, the patient rapidly desaturated. Facemask ventilation was attempted but was unsuccessful. A CICO situation was declared. Equipment packs for both cannula and scalpel cricothyroidotomy were immediately available. The anaesthetist proceeded with PEO using a 14G Insyte cannula. A single four-second insufflation was performed using a Rapid-O2, and SpO2 increased from 40% to 100% within 10 seconds. A Melker emergency cricothyrotomy catheter set was opened, and the guidewire passed easily through the cannula. SpO2 remained stable for several minutes without further insufflation. A brief team discussion ensued, in view of the potential trauma involved in converting to a definitive front-of-neck airway, a further attempt at oral intubation was made. This was successful using McGrath MAC VL and a Frova intubating introducer. Postoperatively the patient was woken with the cricothyroid guidewire in situ. He was transferred to the post-anaesthesia care unit, where observations remained stable and the airway was well maintained. The guidewire was then removed. He had an overnight ward admission for observation and was discharged the following morning without sequelae.
Discussion
Cannula-first CICO rescue
In our series, a 14G Insyte cannula was successfully placed during a CICO scenario in all eight cases, in which appropriate equipment and personnel trained in the technique were immediately available. This illustrates that with the right equipment and focused training, cannula cricothyroidotomy or tracheotomy can be successfully performed under the most stressful conditions.
In case 1, two tracheal cannulation attempts were required. This is consistent with our live animal experience, which shows that failure with the cannula does not have an adverse effect on repeat cannulation attempts.8,9 Failed cannulation can be immediately identified (by failure to aspirate air) before attempted oxygen administration, causing minimal tissue destruction, and allowing multiple attempts in a short time.
In addition, cannula attempts do not preclude subsequent scalpel techniques 8 ,9—as illustrated by case 4. This allows the operator to attempt both cannula and scalpel tracheotomy, if required, maximising the potential for success. Conversely, our experience in the animal model is that failed scalpel attempts are often associated with delayed recognition (for example, only after attempted oxygen administration), bleeding into the trachea and/or significant tissue destruction. These make both subsequent cannula and subsequent scalpel attempts prone to failure.8,9
Case 7 illustrates that cannula tracheotomy can be successful even when the airway anatomy is impalpable, allowing rapid reoxygenation with minimal trauma. In a live animal model with simulated impalpable anterior neck anatomy, participants achieve a 44% success rate at trans-tracheal cannula placement (increasing to 83% when ultrasound is used). 10 If cannula tracheotomy is unsuccessful, we advise opening the skin and subcutaneous tissues with a midline longitudinal scalpel incision plus blunt dissection to locate the trachea, followed by cannula tracheotomy and jet oxygenation.1,8 Other authors have promoted a modification of our advice when the airway is impalpable, in which they recommend a midline incision and blunt dissection followed by a scalpel-bougie technique. 5 However, in our live animal model of impalpable airway anatomy, this results in delayed and less successful oxygen delivery. 8
Standardised technique, equipment and training
In this small series, the cannula-first approach was not associated with barotrauma or other serious complications. In contrast, other authors describe poor outcomes from cannula tracheotomy and jet insufflation: in a systematic review of the use of transtracheal jet ventilation, device failure was reported in 42% of CICO cases, and barotrauma in 32%. 4 However, the insertion technique, equipment, and oxygenation strategies captured in these publications are widely varied.
Having run training sessions in the RPH Wet Lab for over 19 years, in which live, anaesthetised animals are used in a simulated CICO emergency scenario, our group has been able extensively to test equipment and techniques for managing the CICO scenario 1 and standardise how they are taught. 6 For cannula tracheotomy we recommend a 14G Insyte cannula, a 5 ml Luer-slip syringe and an ampoule of saline. Jet oxygenation is achieved using the Rapid-O2. We have found that when used according to the techniques we have developed, this equipment allows anaesthetists in the Wet Lab reliably to cannulate the trachea and deliver safe jet oxygenation.1,3
Much of the concern regarding a cannula approach to CICO management focuses on complications associated with high pressure source ventilation via the cannula. 4 We agree and hence advocate jet oxygenation rather than ventilation, in which oxygen is delivered in such a way as to re-expand the lungs and correct critical hypoxia only, rather than achieve a normal minute volume. We recommend an initial single oxygen jet of 4 seconds, equating to one litre of oxygen delivery. 1 This allows rapid reoxygenation from critical hypoxia, while reducing the frequency of insufflation, greatly reducing the risk of barotrauma. 11 Subsequently jetting according to saturation response—waiting for a drop in saturations following the peak that occurs after insufflation—ensures that the intrathoracic pressure is reducing before the next jet is delivered. 1 This approach was used in the cases in our series, and we believe was crucial in avoiding barotrauma. A recent in silico study in which a CICO scenario was modelled in 50 virtual subjects similarly found that using minimal minute ventilation, rapid reoxygenation could be achieved, while avoiding dynamic hyperinflation. 12
The choice of equipment is paramount in ensuring safety. Our group has developed a jet insufflation T-piece available commercially as the Rapid-O2 (no commercial interest). The Rapid-O2 performs comparably with the Manujet in terms of its potential for reoxygenation, 13 but has safety advantages. It allows expiratory flow and pressure release by providing a route of escape for gases. 13 This mitigates the risk of inadvertent lung hyperinflation and barotrauma. It also provides tactile feedback when an obstruction to flow is encountered, alerting the anaesthetist to the presence of a problem. Case 1 is the earliest case in this series and the only case using a Manujet (before widespread availability of the Rapid-O2), and of note active expiration was needed in this case. None of the cases involving the Rapid-O2 resulted in any complications or needed any treatment secondary to barotrauma.
Lack of training has also been proposed as a contributory factor in the low success rate of cannula cricothyroidotomy seen in NAP4. 14 We agree and recommend team training in a well-rehearsed approach that incorporates cannula and scalpel techniques in a stepwise progression, such as the RPH CICO rescue approach.1,3 The CICO rescue clinician in all cases in our series had been trained in this approach. It is taught widely across Australia but does not necessarily need to be provided using live animal simulation. The lead clinician in case 8, for example, had attended RPH CICO rescue approach manikin training in Wales, rather than live animal simulation training in Australia. This form of training can be provided on an annual basis. We believe it is feasible for clinicians in other countries to be similarly trained.
To facilitate this, our group has produced a comprehensive instructional guide 1 as well as demonstration videos that are freely available online, 15 and can be used in association with manikins or part-task trainers. In addition, we have produced resources for instructors: CICO instructor checklists, which ensure a systematic and consistent approach to teaching the RPH CICO rescue approach, 6 and regular courses for CICO instructors. These instructor courses have been held several times per year since 2014, in locations including Perth, Adelaide, London, Cardiff and Taiwan. Groups such as the Association of Anaesthetists and the All Wales Airway Group have been involved in their delivery.
Oxygenate and stabilise
In cases 2–8, cannula cricothyroidotomy was immediately followed by jet oxygenation using the Rapid-O2. In all these cases jet oxygenation was associated with rapid improvement in oxygen saturation. In cases 6 and 8 the anaesthetists noted that this occurred after a single jet. A similar rapid improvement in oxygen saturations was reported in a series of 23 patients in a CICO situation, in whom cannula cricothyroidotomy was performed. 16 The same saturation response is seen in our live sheep model 1 and is consistent with in silico modelling. 12
CICO is often defined not by a complete inability to intubate, but the inability to intubate with the human and technical resources available prior to the onset of critical hypoxaemia. A common theme in our case series is that although intubation was initially impossible in the context of rapidly developing critical hypoxaemia, cannula tracheotomy and rapid oxygenation created a ‘stop and think’ 5 or ‘Green Zone’ 17 moment, with stability and time to consider how best to secure the airway safely. In case 4 this involved creation of a surgical airway, and in all other cases the trachea was subsequently intubated using an oral or nasal route, thus involving minimal trauma.
Limitations
We did not attempt to poll all attendees systematically at our courses to identify cases, merely to report all cases of CICO that attendees at our courses had voluntarily informed us about. While no failures of the CICO rescue approach were reported to us, it is likely that there is selection and outcome bias. There is no ‘denominator’, and on the basis of this small case series we are not able to determine the incidence of successful and safe management of a CICO scenario using the RPH CICO rescue approach. Given the retrospective nature of the study, recall and information bias are also likely but were mitigated by review of the original notes. Also, we have made no clinical case comparisons with a scalpel-first approach, and thus are not able to determine whether a scalpel-first approach would be as or more successful.
Conclusion
The CICO scenario remains a rare but devastating event that all clinicians involved in airway management must be prepared to address rapidly and safely. Transition to PEO must be appropriately timed to avoid devastating hypoxaemia. Clinicians should determine their preferred technique prior to a CICO event, and ensure they undergo regular training and that appropriate equipment is readily available in their hospitals. Equipment must be simple, safe, effective and intuitive enough to use in high stress situations, and cheap enough to permit availability at the point of care and for training purposes. The technique(s) chosen should minimise trauma, allow rapid oxygenation and stabilisation, facilitate further airway management, aid recovery and be appropriate across a range of CICO scenarios. The eight cases we present illustrate that it is possible for a cannula-first approach to satisfy these criteria in humans. We feel this study is another important step to justify continued use of the approach in centres with appropriate equipment and training. Further studies will be required to establish the relative benefits compared with other approaches.
Footnotes
Author Contribution(s)
Acknowledgements
The authors would like to thank the following, who provided care to the study patients and provided initial reports of cases:
Dr K Tarrant, Christchurch Hospital, Christchurch, New Zealand
Dr J Dinsmore, Portsmouth Hospitals University NHS Trust, Portsmouth, UK
Dr N De Silva, Royal Perth Hospital, Perth, Australia
Dr G Mula, Betsi Cadwaladr University Health Board, Bangor, UK
Dr C Fuller, Royal Perth Hospital, Perth, Australia
Dr P Moran, Maidstone and Tunbridge Wells NHS Trust, Tunbridge Wells, UK
Dr D-W Van Riet, Rockingham General Hospital, Perth, Australia
Dr C Grobler. Royal Perth Hospital, Perth, Australia
Declaration of conflicting interests
All authors have organised CICO training sessions based on the RPH approach, which charge delegates a fee to attend. None of the authors benefits financially from these fees. All authors have been funded by the Taiwan Society of Anesthesiologists to deliver CICO training sessions in Taiwan. SGD has received funding from the Sydney Skills and Simulation Centre for teaching on the
Funding
The authors received no external financial support for the research, authorship, and/or publication of this article.
