Abstract
Live animal models can be used to train anaesthetists to perform emergency front-of-neck-access. Cannula cricothyroidotomy success reported in previous wet lab studies contradicts human clinical data. This prospective, observational study reports success of a cannula-first ‘can't intubate, can't oxygenate’ algorithm for impalpable anatomy during high fidelity team simulations using live, anaesthetised pigs.
Forty-two trained anaesthesia teams were instructed to follow the Royal Perth Hospital can't intubate, can't oxygenate algorithm to re-oxygenate a desaturating pig with impalpable neck anatomy (mean (standard deviation, SD) 16.2 (3.5) kg); mean (SD) tracheal internal diameter 11 (1.4) mm. Teams were informed that failure would prompt veterinary-led euthanasia.
All teams performed percutaneous cannula cricothyroidotomy as the initial technique, with a median (interquartile range, IQR (range)) start time of 42 (35–50 (24–93)) s. First-pass percutaneous cannula success was 29% to both insufflate tracheal oxygen and re-oxygenate. Insufflation success improved with repeated percutaneous attempts (up to three), but prolonged hypoxia time increasingly necessitated euthanasia (insufflation 57%; re-oxygenation 48%). First, second and third percutaneous attempts achieved insufflation at median (IQR (range)) 74 (64–91 (46–110)) s, 111 (95–136 (79–150)) s and 141 (127–159 (122–179)) s, respectively. Eighteen teams failed with percutaneous cannulae and performed scalpel techniques, predominantly dissection cannulation (n = 17) which achieved insufflation in all cases (insufflation 100%; re-oxygenation 47%). Scalpel attempts were started at median (IQR (range)) 142 (133–218 (97–293)) s and achieved insufflation at 232 (205–303 (152–344)) s.
While percutaneous cannula cricothyroidotomy could rapidly re-oxygenate, the success rate was low and teams repeated attempts beyond the recommended 60 s time frame, delaying transition to the more successful dissection cannula technique. We recommend this ‘cannula-first’ can't intubate, can't oxygenate algorithm adopts a ‘single best effort’ strategy for percutaneous cannula, with failure prompting a scalpel technique.
Keywords
Introduction
The ‘can’t intubate, can’t oxygenate’ (CICO) scenario is rare in anaesthesia but accounts for a large proportion of airway-related deaths. 1 Failure to oxygenate via the supraglottic route necessitates emergency front-of-neck access (eFONA).2–5 The need for anaesthetists to train for CICO events is widely acknowledged1,6,7 but there is a lack of evidence to guide best practice. 8 The 4th National Audit Project of the Royal College of Anaesthetists (NAP4) identified a low success rate (37%) of narrow-bore cannula cricothyroidotomy from reported airway crises, and recommended further research focused at identifying the success rates and optimal techniques for cannula cricothyroidotomy. 1 Following this report, international airway groups including the Canadian Airway Focus Group (CAFG) 5 and Difficult Airway Society (DAS) changed their recommendations on cannula cricothyroidotomy, with the latter removing the cannula step from eFONA management altogether and advocating a scalpel–bougie technique for all patients. 4
The Australian and New Zealand College of Anaesthetists (ANZCA) continues to support both cannula and scalpel techniques, 9 recognising the Royal Perth Hospital (RPH) CICO algorithm (2009) as suitable for infraglottic airway access by anaesthetists (Figure 1). 10 The stepwise progression from cannula to scalpel advocated by Heard and colleagues has heavily influenced CICO training in Australia and around the world. 11 A recent survey of Australian centres found that 59% base their CICO training on the RPH CICO algorithm, with 93% keeping cannula equipment (± scalpel) at point of care. 11 The RPH CICO algorithm has been adopted by centres in New Zealand 11 and the United Kingdom, 12 and also laid the foundations for the All-Wales Airway Group CICO algorithm 13 and the Oxytain CICO algorithm in North America.14,15 The RPH CICO group have pioneered the stepwise approach to CICO management, the use of cannula cricothyroidotomy as the initial eFONA technique for all patients and a focus on oxygen stabilisation over ventilation. The RPH CICO algorithm was developed in high fidelity ‘wet lab’ facilities using live anaesthetised sheep. Although animal models cannot perfectly imitate human anatomy, 16 live anaesthetised animal models offer the opportunity to simulate and research rare airway events by creating conditions otherwise unachievable using manikin or human cadaver models.10,17,18
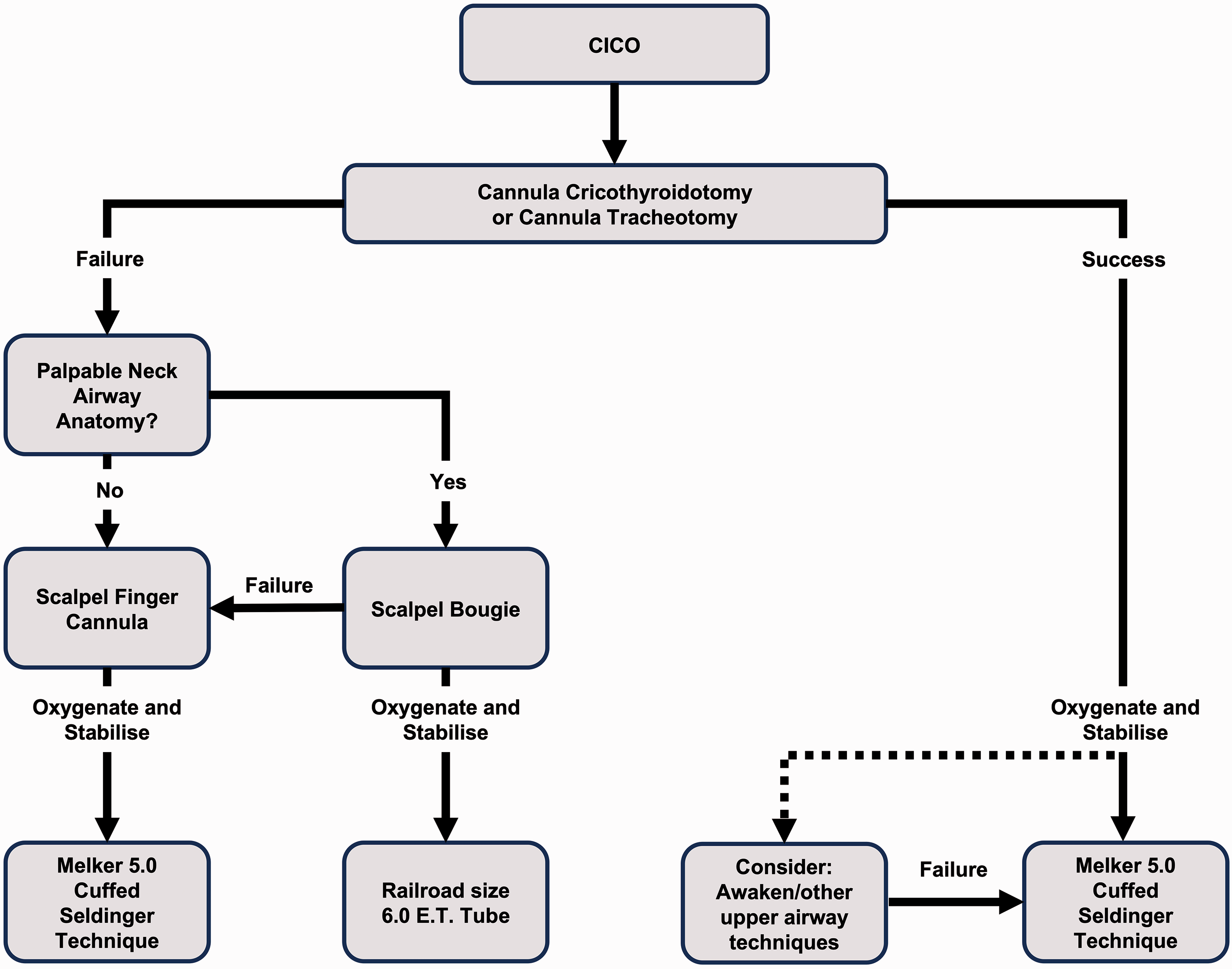
The Royal Perth Hospital ‘cannot intubate, cannot oxygenate’ algorithm (with permission).
The Department of Anaesthesia at Fiona Stanley Hospital, Perth, has developed a comprehensive eFONA course in collaboration with the School of Veterinary Medicine at Murdoch University. 19 While the course runs independently of the RPH group, CICO training is based on the RPH CICO algorithm. The course consists of interactive lectures, manikin training, fresh pig cadaveric skills training and high-fidelity simulation using live anaesthetised pigs. Uniquely, senior anaesthetists are taught in tandem with trained anaesthetic technicians (assistants), with each member of the anaesthesia team acting within their normal clinical role. This blended format utilises both team training, proved to improve performance in emergency resuscitative scenarios, and simulation, an effective modality to deliver such training. 20 The immersive learning environment allows participants to act in both the learner and observer roles within the context of simulation. 21 The course is accredited by ANZCA for CICO emergency response training. 22 The pigs used in sessions are small (10–25 kg) with tracheas that are narrow, easily compressible and lying deep to thick fascia and neck muscles. In the supine position, pigs typically have impalpable front-of-neck anatomy, making percutaneous cannulation of the trachea difficult. Provisional course audit data identified a percutaneous cannula cricothyroidotomy success rate of 35% from 204 attempts in cadaver and live pigs. 23 These results are in contrast to the high success rate (97%) of cannula cricothyroidotomy previously reported in a live anaesthetised sheep model with palpable anatomy,10,18 but closer to the 30%–50% success rate from human clinical data.1,24,25
Despite the central role of the RPH CICO algorithm in the ‘cannula-first’ approach to CICO management, the algorithm has not changed significantly since its original publication in 2009. 10 To our knowledge, no study group outside of RPH has studied the algorithm as a whole in the simulation setting. The current study aimed to measure the success rates and times to success of the eFONA steps of the RPH CICO algorithm for impalpable neck anatomy (percutaneous cannulation and dissection cannulation), using anaesthesia teams and a live anaesthetised pig model in a CICO simulation. The combination of impalpable neck anatomy, inclusion of anaesthesia teams and live animal simulations run to the natural endpoint of re-oxygenation or euthanasia, offered a unique opportunity both to train perioperative teams and study a widely adopted CICO algorithm in a simulation environment. We believe that these high-fidelity conditions may be more representative of a real life CICO crisis, therefore we hypothesise that the success of percutaneous cannulation would be low, equivalent to that found in human clinical data.
Methods
This prospective, observational study included data from all 42 live pig simulations from 18 airway courses held between 2017 and 2018. Each simulation involved a single live, anaesthetised pig managed by a pre-allocated anaesthesia team. Animal care and ethics approval was provided by the Murdoch University Animal Ethics Committee (T2804/15), in accordance with the Animal Welfare Act, WA (2013). 26 The research protocol and recruitment of anaesthesia staff was approved by the South Metropolitan Health Service Human Research Ethics Committee on 27 March 2017 (2016-246). All anaesthetists and anaesthetic technicians who attended the airway training courses were recruited (84 participants) and all gave written, informed consent. Inclusion in the study was voluntary and not a requirement for completion of airway training. This manuscript adheres to the applicable animal research: reporting of in vivo experiments (ARRIVE) guidelines. 27 The study design was observational in nature as the only change made to the course teaching protocol for the study was that simulations were run only once per pig (rather than multiple times per pig).
Each airway training course was delivered to two or three anaesthesia teams taught in tandem. Anaesthesia teams consisted of a single senior anaesthetist (consultant or senior registrar) and a single trained anaesthetic assistant. All teams received structured airway training totalling six hours in the following order:
pre-course reading including links to RPH CICO educational videos (YouTube)28 (one hour—pre-course); interactive lectures based on the RPH CICO algorithm/techniques, DAS 2015 guidelines and Vortex cognitive aid (one hour);2,4,10 ‘dry lab’ stage allowing eFONA techniques to be practised on ‘Frova’ Crico-Trainer manikins (VBM Medical, Germany) (one hour); ‘wet lab’ stage in the veterinary facility using fresh pig cadavers prior to simulations, allowing for practise of eFONA techniques on the porcine airway (one hour); live animal simulation—one anaesthesia team at a time (one hour); formal team debrief session with faculty—post-simulations (one hour).
The eFONA techniques taught included percutaneous cannula cricothyroidotomy, oxygen insufflation strategies, scalpel–bougie technique, scalpel–finger–cannula technique (dissection cannulation) and conversion of a cannula to a Melker airway (Cook Critical Care, Bloomington, IN, USA) as per RPH CICO teaching.
Dedicated veterinary anaesthetists maintained the welfare and anaesthesia of the pigs at all times (Appendix 1). Live anaesthetised pigs were sourced from pre-existing veterinary student surgical training, adhering to ethical practice by adding value with no additional loss of animal life. The breed of pigs used was ‘large white’, including male and female pigs of approximately two to three months of age. After completion of veterinary surgical training, pigs were either euthanised for cadaveric airway training or kept anaesthetised for live airway simulations. Pigs deemed most physiologically stable following surgery were chosen by veterinary anaesthetists for live simulations. Prior to airway simulations, total intravenous anaesthesia (TIVA) was established and pigs were mechanically ventilated using 100% oxygen via an endotracheal tube (GE Aestiva 5 anaesthetic machines, Wauwatosa, WI, USA). Monitoring included peripheral oxygen saturations (SpO2), end-tidal carbon dioxide (EtCO2), electrocardiography (ECG) and invasive blood pressure (SurgiVet™ multiparameter monitor, Sound Veterinary Equipment Pty Ltd, Rowville, Victoria) (see Figure 2).
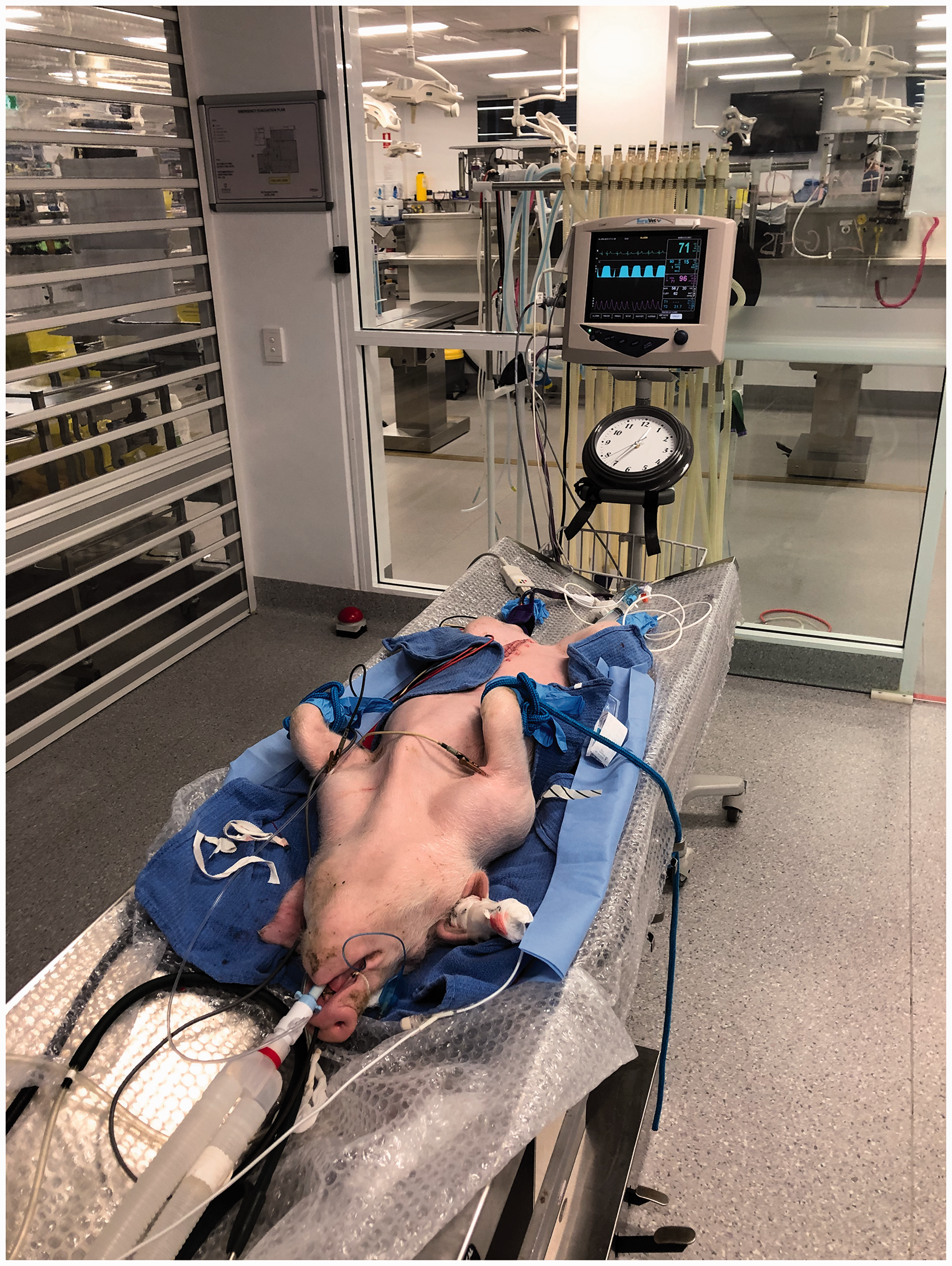
Live pig simulation. Live anaesthetised pigs are intubated and ventilated from above. Neck is exposed to allow performance of emergency front-of-neck access (eFONA) techniques, with monitoring and clock visible to anaesthesia teams.
Teams were briefed that they would encounter a simulated airway crisis whereby CICO had already been declared. Teams were instructed to perform eFONA following the RPH CICO algorithm assuming impalpable neck anatomy as taught in the dry and wet labs immediately prior to live simulations. This would involve a percutaneous cannula cricothyroidotomy (‘percutaneous cannulation’) as the initial eFONA technique, using a 14 gauge 45 mm intravenous cannula (BD Insyte™ Autoguard™ BC; Becton Dickinson, Franklin Lakes, NJ, USA). Successful aspiration of air from both the needle on initial cannulation and the cannula after feeding off the needle into the airway would confirm correct placement, prompting oxygen insufflation via a Rapid-O2 oxygen insufflation device (Meditech Systems Ltd., Shaftesbury, UK). Oxygen insufflation strategies were as per the RPH CICO algorithm (initial rescue four second oxygen insufflation at a flow rate of 15 l/min = 250 ml/s). The RPH CICO algorithm advocates up to three rapid attempts at percutaneous cannulation within a one minute time frame before progression to a scalpel-based technique. In the event of failed percutaneous cannulation with impalpable neck anatomy, the algorithm advocates transition to the scalpel–finger–cannula technique, also described by Heard as the ‘dissection cannula’ technique 29 (‘dissection cannulation’). This technique uses the same equipment as percutaneous cannulation, but is preceded by an 8–10 cm vertical scalpel incision in the midline and blunt ‘finger’ dissection to the airway. During simulations, the anaesthetist acted as the crisis team leader and primary operator to perform techniques. The anaesthetic technician acted as assistant, primarily preparing and handing airway equipment to the anaesthetist and aiding decision-making by verbal and physical prompts to the anaesthetist. A ‘CICO pack’ containing all necessary eFONA equipment in original packaging was made available on a trolley similar to our institutional practice (Appendix 2). A clock was visible throughout simulations for teams to time eFONA techniques and insufflation strategies. Teams were informed that there would be no intervention from teaching faculty until they had gained access to the airway and insufflated tracheal oxygen. Therefore, if teams deviated from the algorithm, the simulations were continued and the outcomes recorded, with faculty only intervening if participants’ actions risked injury to themselves (e.g. needlestick injury). As per ethical practice, the action or inaction of participants could not directly lead to the death of an animal. When participants repeatedly failed eFONA attempts, prolonged desaturation risked the development of critical hypoxia, which would eventually lead to cardiac arrest (a direct cause of death through inaction). Therefore, at the onset of critical hypoxia, euthanasia was administered at the discretion of veterinary anaesthetists to prevent hypoxic cardiac arrest. Specific clinical signs and characteristic ECG changes (Appendix 1) were recognised by veterinary anaesthetists at the onset of critical hypoxia, which prompted the administration of intravenous pentobarbital. Although participants were blinded to the administration of intravenous euthanasia, they were not blinded to the resultant effect on vital signs. Teams were therefore instructed to continue with eFONA techniques regardless of loss of monitored signs or apparent cardiac arrest. After participant preparation, pigs were rendered apnoeic by cessation of mechanical ventilation and TIVA was continued. The tracheal tube was withdrawn over a bougie through which EtCO2 was measured.
The pigs used in simulations desaturated quickly after apnoea due to a combination of recent surgery, prolonged anaesthesia and the supine position. Simulations were started when SpO2 fell to 92%, a level chosen based on preliminary work with the small live pig model. 19 This SpO2 level was found to result in rapid oxygen desaturation for simulation purposes yet allowed enough time for re-oxygenation if eFONA techniques were successful. At this point, the pre-allocated anaesthesia team was called into the simulated operating theatre, CICO was ‘declared’ and timing started. An attempt at percutaneous cannulation was defined as a single skin puncture, starting at the time of initial needle-to-skin contact. Any subsequent skin puncture with the same or new cannula was recorded as an additional (second or third) percutaneous attempt. Scalpel-based techniques included either dissection cannulation or scalpel–bougie technique, starting at the time of initial knife-to-skin contact. The primary outcome was the ability to successfully insufflate tracheal oxygen (‘insufflation’), resulting in a visible chest rise and confirmed by a measured rise in EtCO2. The secondary outcome was the ability to successfully re-oxygenate the hypoxic pig, defined as a return of SpO2 of 90% or greater (‘re-oxygenation’), after which simulations were ended and pigs were euthanased.
Success of individual eFONA steps of the algorithm was expressed as a proportion of attempts made by all teams (% attempts). Cumulative success with progression through the algorithm was expressed as a proportion of teams achieving success by that step (% teams). Times to the start of attempts, insufflation and re-oxygenation were recorded in seconds and expressed as medians (interquartile range, IQR (range)), as these times were not normally distributed. Administration of euthanasia and complications arising from airway techniques or oxygen delivery were recorded. Additional measurements included pig weight (kg) and pig tracheal internal diameter measured by dissection after euthanasia (mm), both expressed as mean (standard deviation, SD). Forty-two team simulations were available for inclusion in the study and represent a convenience sample from all airway courses run during the study period. The Wilson score interval method was used to generate 95% confidence intervals (CI) for proportions. 30
Results
Anaesthesia teams included a senior anaesthetist (total 17 consultant anaesthetists, 25 senior trainee anaesthetists) and a trained anaesthetic assistant (total 42). A flow chart of attempts made by all 42 teams with success rates to insufflation and re-oxygenation for each step of the RPH CICO algorithm is included in Figure 3. All 42 teams undertook a ‘cannula first’ approach, performing percutaneous cannulation as the initial eFONA technique. Insufflation success (primary outcome) of percutaneous cannulation was 29% (12/42) at first pass (95% CI 0.17 to 0.44). Subsequent eFONA attempts of teams unsuccessful with the first attempt are listed in Figure 3. When second and third percutaneous cannulation attempts were performed, insufflation success for these attempts was 28% (8/29) and 24% (4/17), respectively. Accounting for the cumulative effect of multiple punctures (up to three attempts), 57% (24/42) of teams were able to achieve insufflation by percutaneous cannulation attempts alone (95% CI 0.42 to 0.71). Delay performing techniques or repeated failure led to prolonged hypoxia time, which increased the risk of critical hypoxia and the need to administer euthanasia. Therefore, successful insufflation led to re-oxygenation (secondary outcome) in 100% (12/12), 75% (6/8) and 50% (2/4) of first, second and third percutaneous cannulation attempts, respectively. The cumulative re-oxygenation success with percutaneous cannulation alone was 48% (20/42) (95% CI 0.33 to 0.62) across all teams.
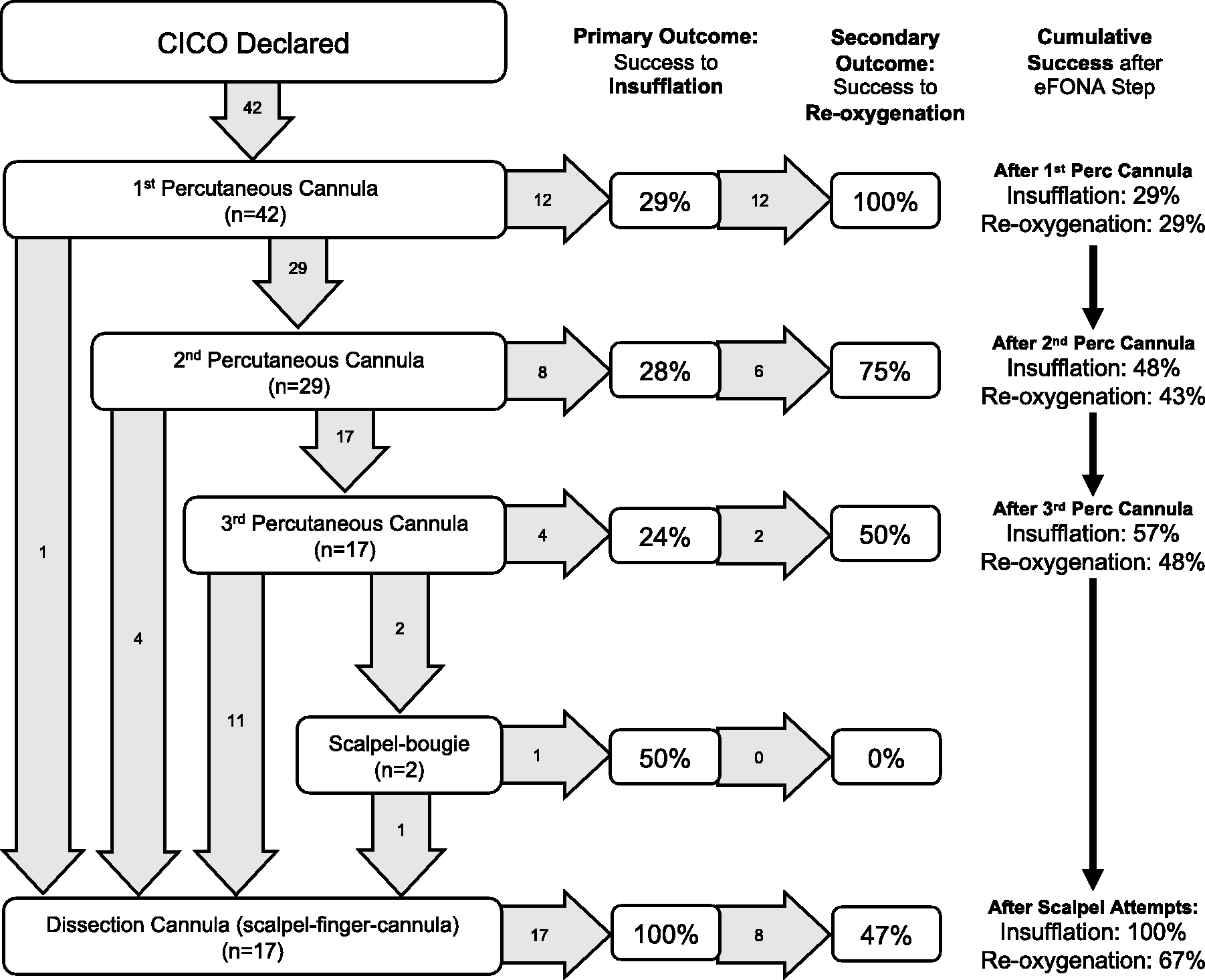
Flow chart of emergency front-of-neck access (eFONA) techniques performed by 42 anaesthesia teams following the Royal Perth Hospital ‘cannot intubate, cannot oxygenate’ algorithm. Success rates to insufflation (primary outcome) and re-oxygenation (secondary outcome) for each step of the algorithm are expressed as the proportion of attempts made by all teams (% attempts). Cumulative success rates are listed as the proportion of teams achieving insufflation and re-oxygenation by each step of the algorithm (% teams). Numbers in arrows indicate the number of teams progressing to the next step (n is the number of teams undertaking eFONA step).
In the event of failed or abandoned percutaneous cannulation attempts, 18 teams undertook scalpel-based techniques. Dissection cannulation was performed by 16 teams as the only scalpel technique and by one team after a failed scalpel–bougie technique. Dissection cannulation led to successful insufflation in all cases (100% success (17/17)) and successful re-oxygenation in 47% (8/17) of attempts. One team undertook a scalpel–bougie technique as the only scalpel technique, which was successful to ventilate oxygen but not re-oxygenate due to critical hypoxia necessitating the administration of euthanasia. Overall success across all teams following the RPH CICO algorithm was 100% to insufflation (42/42) and 67% (28/42) (95% CI 0.52 to 0.79) to re-oxygenation.
The median times to attempts, insufflation and re-oxygenation are listed in Table 1. From the start of simulations, teams took a median (IQR (range)) time of 42 (35–50 (24–93)) s to reach the point of initial needle-to-skin, representing the time taken to prepare cannula equipment and start the first percutaneous cannulation attempt. Median (IQR (range)) SpO2 at the point of initial needle-to-skin was 71% (62%–77% (40%–85%)). When successful, first percutaneous cannulation attempts achieved insufflation at median (IQR (range)) 74 (64–91 (46–110)) s and re-oxygenation at 107 (92–119 (72–157)) s. Second and third percutaneous cannulation attempts were made at median (IQR (range)) 76 (68–88 (49–152)) s and 116 (105–168 (78–202)) s, respectively, and achieved insufflation at 111 (95–136 (79–150)) s and 141 (127–159 (122–179)) s, respectively. When scalpel techniques were undertaken, the median (IQR (range)) knife-to-skin time was 142 (133–218 (97–293)) s with insufflation occurring at 232 (205–303 (152–344)) s.
Median times to emergency front-of-neck access attempts and outcomes of 42 anaesthesia teams following the Royal Perth Hospital ‘cannot intubate, cannot oxygenate’ algorithm.

Times from the start of simulations to the start of eFONA attempts, insufflation (primary outcome) and re-oxygenation (secondary outcome) when successful for each step of the algorithm.
Times in seconds (s) are median (interquartile range (range)). eFONA: emergency front-of-neck access.
When euthanasia was administered prior to re-oxygenation, this occurred at a mean time of 199 (95% CI 165 to 233) s. One complication occurred (1/42 teams (2%)), a single incident of surgical emphysema during one team’s first percutaneous cannulation attempt, most likely due to cannula tip migration. This occurred despite positive air aspiration checks from both the needle and cannula, with insufflation resulting in subcutaneous oxygen delivery. This was immediately recognised by the team who abandoned further percutaneous cannulation attempts and performed dissection cannulation, which was successful to insufflate but not re-oxygenate. Mean (SD) pig weight was 15.6 (3.5) kg and mean (SD) pig tracheal internal diameter was 11 (1.4) mm.
Discussion
By following the stepwise approach of the RPH CICO algorithm, all anaesthesia teams (100%) were able to insufflate tracheal oxygen in the live anaesthetised pig model. Across all teams, 67% were able to achieve re-oxygenation to SpO2 of 90% or greater, with the need to administer euthanasia due to critical hypoxia preventing the remainder from achieving re-oxygenation. First-pass insufflation success of percutaneous cannulation was low (29%). By repeating percutaneous cannulation, insufflation success improved in a cumulative fashion (57% allowing for three attempts across all teams), but second and third attempts were individually less successful to insufflate oxygen than the first pass. Successful insufflation led to re-oxygenation in all first percutaneous cannulation attempts, but second and third percutaneous attempts were increasingly less able to re-oxygenate due to the increasing risk of hypoxia necessitating euthanasia with time. Teams that failed with percutaneous cannulation predominantly performed dissection cannulation which was universally successful to insufflate oxygen.
A 2019 study by Rees and colleagues reported high percutaneous cannulation success (97%) in a live anaesthetised sheep model similar to that described by the RPH group.10,18 Although Rees and colleagues do not define their model as ‘palpable’, they do describe the trachea of their sheep model as ‘longer, more mobile and superficial’ compared to human airways. 18 During their original work, the RPH group developed an impalpable anatomy model using subcutaneous fluid infiltration over the sheep airway. 10 In this impalpable model, success to insufflate tracheal oxygen was 40% with percutaneous cannulation and 100% with dissection cannulation. 10 More recently in the same impalpable sheep model, 31 Heard and colleagues demonstrated superiority of the dissection cannulation technique over scalpel–bougie (after scalpel dissection). They cited improved tactile feedback, the air aspiration endpoint and ergonomic equipment manoeuvrability as advantages of dissection cannulation in deep soft tissue structures. A palpable airway is arguably easier to cannulate, therefore an initial scalpel incision and blunt dissection in the impalpable neck would allow direct location of the airway for cannulation. Some authors have recommended a scalpel incision prior to any attempt at cannula cricothyroidotomy, to aid location and cannulation of the airway.32,33
Animal models do not perfectly imitate human anatomy, 16 which limits the direct translation of our results to the human CICO scenario. More broadly, any standardised airway model cannot imitate the anatomical variation that exists between patients or simulate the many reasons why an airway may be difficult. Although NAP4 did not specifically comment on the presence of palpable or impalpable front-of-neck anatomy, it did find obese patients to be disproportionately represented in airway crises. 1 Obesity is associated with both increased depth of 34 and inability to locate 35 the airway. We therefore consider the impalpable and deep airway anatomy of the porcine model to be appropriate as a front-of-neck anatomy model to simulate CICO crises. Beyond anaesthesia, live porcine models have been validated by ear, nose and throat specialists as suitable for tracheostomy and open-airway surgery training. 36
Aside from anatomical comparisons, the fidelity gained from a live, bleeding and desaturating animal model with resultant effect on team performance is difficult to achieve by alternative simulation means, offering a unique advantage over human cadaver models. 16 An important component of our simulations was the lack of faculty intervention, with scenarios allowed to run to the natural endpoint of survival (re-oxygenation) or death (euthanasia). Participants’ knowledge that survival was dependent on their timely actions significantly contributed to the level of stress experienced and the fidelity of the simulation. We are unaware of any other airway simulation study using a similar methodology. Teamwork is considered essential for optimal CICO management, 37 therefore trained anaesthetic assistants were included to simulate team-based crisis management. Although the decision to undertake CICO had already been made, we would consider the resultant team behaviours and interactions to reflect closely those of a real-life crisis. Given the fidelity of simulations, the recency of training and the readiness of teams to perform eFONA, a median time of 42 s to prepare cannula equipment from original packaging probably represents a best case scenario for the time taken to prepare equipment in real life.
The high percutaneous cannulation failure rate also highlighted the difficulty in performing multiple percutaneous attempts within the recommended 60 s time frame. The challenging anatomy of the impalpable pig model exposed many of the inherent flaws of percutaneous techniques. Issues such as inability to locate the airway, cannula malposition, cannula kinking and lumen blockage have previously been described.1,24 Additional issues observed in our simulations included obstruction when attempting to feed the cannula off the needle, premature cannula–needle dislodgement and inconsistent air aspiration. The resultant troubleshooting, indecision and need to prepare a new cannula further delayed repeat attempts, which ultimately led to delay in transitioning to a scalpel technique. Comparing median times, only successful first percutaneous cannulation attempts were performed to insufflation within 60 s of initial needle-to-skin time (32 s after initial needle-to-skin time). Successful second percutaneous cannulation attempts achieved insufflation at a median time after this 60 s time frame (69 s after initial needle-to-skin time), and third percutaneous cannulation attempts were started at a median time after this 60 s time frame (74 s after initial-needle-to-skin time). Scalpel attempts were therefore delayed, starting at a median time of 100 s after initial needle-to-skin rather than immediately after 60 s. Given the recency of training, this behaviour likely reflects a lack of temporal awareness, with teams prioritising the ‘three attempts’ over the need to move to a scalpel technique at 60 s. Such delays are likely to be longer in real life, as coronial enquiries have highlighted that doctors often fail to progress to the next airway step under the cognitive load of a crisis. 38 The importance of transitioning has therefore become a key element of modern airway training. 39
Our results would suggest that dissection cannulation may be the preferred technique in the CICO crisis with impalpable anatomy. A limitation of our study though, is that these were not performed in isolation, as all dissection cannulation attempts were preceded by a (failed) percutaneous cannulation attempt. It is difficult to interpret how these failed attempts may have influenced subsequent success. Another limitation of our study was that the decision to transition to the neck had already been made, with ‘declaration of CICO’ signifying the start of simulations and teams primed to perform a percutaneous cannulation attempt. Transitioning to the neck is an important aspect of CICO management, 38 and the ‘cannula-first’ approach is often cited as offering an advantage with the view that anaesthetists are more familiar with cannulae than scalpels, 40 and that choosing a cannula technique may promote earlier intervention. 41
What we can say with reasonable confidence from our results is that multiple percutaneous cannulation attempts are inadvisable. Regarding transition, only one cannula attempt is required to aid the anaesthetist to move to the neck. Therefore, we would strongly recommend that those following a ‘cannula-first’ approach to CICO management in the impalpable neck should adopt a ‘single best effort’ strategy, with failure prompting immediate transition to a scalpel technique, namely the dissection cannulation technique. Our results provide some additional justification for this approach. Although first-pass percutaneous cannulation success was low, successful insufflation occurred 32 s after needle-to-skin, compared to dissection cannulation in which successful insufflation occurred 90 s after knife-to-skin. When insufflation was achieved in these initial percutaneous attempts, all were able to re-oxygenate the pig model, that is they were performed rapidly enough to avoid critical hypoxia and the need to administer euthanasia. Our study observed a single complication from high pressure oxygen delivery, which did not impede a subsequently successful dissection cannulation. This complication rate (2%) was much lower that the 51% rate reported in a 2015 systematic review of ‘transcutaneous tracheal jet ventilation’, 24 presumably due to standardised equipment, the safe RPH techniques and team training immediately prior to simulations.
In our opinion, the ‘single best effort’ strategy may also be advantageous from a human factors perspective. By emphasising rapid transition after the percutaneous attempt, failure itself would act as the trigger to transition to a scalpel technique, offering a more explicit prompt than relying on a specific time frame (shown to be unreliable in our study). Considering the RPH CICO algorithm as a whole, a ‘single best effort’ strategy for impalpable anatomy maintains the cannula-first approach for all patients (a core principle of the algorithm) and ensures standardised immediate management prior to the ‘palpable neck airway anatomy?’ decision step which occurs after percutaneous attempts. Whether the same ‘single best effort’ strategy would be suitable for patients with palpable front-of-neck anatomy would require more study. Following this study though, our own group continues to base CICO teaching on the RPH CICO algorithm, but we now recommend a ‘single best effort’ at percutaneous cannulation for all patients, with rapid transition to a scalpel technique in the event of failure.
There were other limitations to the current study. A randomised trial would have allowed for direct comparison of the success and timing of different eFONA techniques, but this would have significantly impacted the teaching commitments of the airway course and would not have allowed for appraisal of these techniques as part of a stepwise algorithm. Also, despite the wide adoption of the scalpel–bougie technique, too few attempts were observed to draw specific conclusions. Scalpel–bougie equipment was provided to mirror the standard ‘CICO pack’ provided at our institution, but the two attempts observed represent deviations away from the impalpable side of the RPH CICO algorithm. Our results have demonstrated that even when a crisis algorithm is taught and teams are instructed to follow it, participants may still perform alternative techniques in a high-pressure simulation. While these deviations could reflect human error or preference for an alternative technique (i.e. a scalpel–bougie over dissection cannulation), our study did not collect data on why deviations were made. Another limitation was that the physiological condition of the pigs resulted in rapid desaturation after apnoea, which alongside the need to euthanase before cardiac arrest, may have contributed to a higher failure rate to re-oxygenate. The ability to insufflate was therefore used as the primary outcome, as this was deemed independent of porcine physiology and veterinary influence. Finally, a CICO algorithm designed for adults was studied using pigs comparable in size to small children. 42 While anatomical comparisons have been discussed, the demonstrated success of the dissection cannulation technique in narrow porcine airways warrants further consideration of its potential role in the rare but challenging scenario of paediatric CICO with impalpable anatomy.
This prospective, observational study of the RPH CICO algorithm for impalpable anatomy has found differing success rates between the percutaneous cannulation and dissection cannulation techniques when used by anaesthesia teams in a live anaesthetised pig model. While percutaneous cannulation could result in rapid re-oxygenation, success rates were low and repeated attempts delayed progression to the more successful dissection cannulation technique. Percutaneous cannulation as the initial eFONA technique for all patients is only justifiable if it does not delay transition to a scalpel technique, and a ‘single best effort’ strategy may offer a more reliable trigger for this in a crisis. The RPH CICO algorithm remains the gold standard for stepwise, ‘cannula-first’ CICO management, but more study is needed continually to develop CICO techniques, and live anaesthetised animal models, particularly with impalpable anatomy, provide an invaluable resource for this.
Footnotes
Data availability statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Acknowledgements
The author(s) would like to acknowledge the contributions made by past and present faculty of the airway rescue course, especially veterinary and administrative staff at Murdoch University.
Author contributions
Declaration of conflicting interests
The author(s) have no conflicts of interest to declare.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Appendix 1: Live pig anaesthesia
Live pig anaesthesia was maintained by dedicated veterinary anaesthetists throughout veterinary student surgical training and live airway simulations. This included:
sedation with intramuscular zoletil (2 mg/kg) and xylazine (4 mg/kg); propofol induction via an auricular intravenous cannula; tracheal intubation; anaesthesia maintenance using isoflurane in oxygen; supine positioning for surgical training; intra-abdominal surgery via midline laparotomy incisions; total surgical time—up to two hours; post-surgery—pigs either euthanased and used for cadaveric training or anaesthesia maintained and transferred to the airway simulation area; for pigs used in simulations, anaesthesia converted to total intravenous anaesthesia (TIVA); TIVA continuous infusion—propofol (0.2–0.4 mg/kg/min) and fentanyl (20–30 μ/kg/hour); muscle relaxation—combination of propofol and fentanyl sufficient to prevent movement or spontaneous ventilation; avoidance of paralytic agents—standard veterinary practice is to avoid muscle-relaxant drugs, allowing clinical signs to be used to assess depth of anaesthesia; mechanical ventilation with 100% oxygen (GE Aestiva 5 anaesthetic machines, GE Medical, Wauwatosa, WI, USA) volume-controlled ventilation adjusted to achieve an end-tidal carbon dioxide (EtCO2) of 40–45 mmHg; arterial cannulas were placed in the medial saphenous artery; vital signs monitored using a SurgiVet multiparameter monitor (Sound Veterinary Equipment Pty Ltd, Rowville, Victoria) including peripheral oxygen saturation (SpO2), electrocardiography (ECG), invasive blood pressure and EtCO2; pigs were rendered apnoeic by cessation of mechanical ventilation and continuation of TIVA; the tracheal tubes were withdrawn over a bougie through which EtCO2 was measured; bougie was placed just through the vocal cords with the tip at the level of the glottis, leaving tracheal lumen free for interventions; euthanasia was administered in the event of critical hypoxia during simulations and after the completion of all simulations; critical hypoxia was characterised by clinical signs such as piloerection, skin plethora and ECG changes including tachycardia, T-wave changes and arrhythmias; euthanasia was administered by intravenous phenobarbitone (100 mg/kg), with appropriate dose prepared prior to simulations.
Appendix 2: ‘can’t intubate,can’t oxygenate’ (CICO) pack
CICO pack on airway trolley included:
four 14 gauge 45 mm intravenous cannulae (BD Insyte™ Autoguard™ BC; Becton Dickinson, Franklin Lakes, NJ, USA); Rapid-O2 cricothyroidotomy insufflation device (Meditech Systems Ltd., Shaftesbury, UK); 5 ml syringe; 20 ml 0.9% sodium chloride; size 10 scalpel; Frova intubating bougie (Frova intubating introducer; Cook Medical, Bloomington, IN, USA); Rapi-fit connector (bougie to 15 mm circuit) (Cook Critical Care, Bloomington, IN, USA); tracheal tube (internal diameter 6.0 mm); self-inflating ventilation bag; size 5.0 Melker cuffed emergency cricothyroidotomy catheter set (Seldinger) (Cook Critical Care, Bloomington, IN, USA).
