Abstract
Carbetocin and oxytocin are commonly recommended agents for active management of the third stage of labour. Evidence is inconclusive whether either one more effectively reduces the occurrence of important postpartum haemorrhage outcomes at caesarean section. We examined whether carbetocin is associated with a lower risk of severe postpartum haemorrhage (blood loss ≥ 1000 ml) in comparison with oxytocin for the third stage of labour in women undergoing caesarean section. This was a retrospective cohort study among women undergoing scheduled or intrapartum caesarean section between 1 January 2010 and 2 July 2015 who received carbetocin or oxytocin for the third stage of labour. The primary outcome was severe postpartum haemorrhage. Secondary outcomes included blood transfusion, interventions, third stage complications and estimated blood loss. Outcomes were examined overall and by timing of birth, scheduled versus intrapartum, using propensity score-matched analysis. Among 21,027 eligible participants, 10,564 women who received carbetocin and 3836 women who received oxytocin at caesarean section were included in the analysis. Carbetocin was associated with a lower risk of severe postpartum haemorrhage overall (2.1% versus 3.3%; odds ratio, 0.62; 95% confidence interval 0.48 to 0.79; P < 0.001). This reduction was apparent irrespective of timing of birth. Secondary outcomes also favoured carbetocin over oxytocin. In this retrospective cohort study, the risk of severe postpartum haemorrhage associated with carbetocin was lower than that associated with oxytocin in women undergoing caesarean section. Randomised clinical trials are needed to further investigate these findings.
Introduction
Caesarean section is one of the most common major surgeries performed on women worldwide and, as such, health systems should provide safe standardised bundles of care for this procedure.1,2 Postpartum haemorrhage (PPH) rates, a major cause of maternal mortality, are on the rise internationally, with uterine atony consistently identified as the most common cause of PPH.3–6 Nonetheless, it is noteworthy that there is significant discord amongst published guidelines regarding the prophylactic administration of uterotonics to prevent postpartum haemorrhage at caesarean section. 7
A recent survey by our group found that the practice of administering uterotonics varied significantly in the anaesthesia community, coupled with different professional associations around the world positing differing counsel. 8 Royal College of Obstetricians and Gynaecologists guidelines recommend the administration of oxytocin. 7 In contrast, Society of Obstetricians and Gynaecologists of Canada guidelines recommend that carbetocin, a long-acting oxytocin analogue, is the preferred uterotonic agent to prevent PPH at scheduled caesarean section. 7 Conversely, a more recent consensus guideline on the use of uterotonic agents during caesarean section recommended the administration of either oxytocin or carbetocin immediately following the delivery of the fetus at caesarean section. 9 These guideline disparities may be explained by a lack of Level 1 evidence to assess the impact of first-line uterotonics at caesarean section on important maternal outcome measures such as serious maternal morbidity. Moreover, the evidence base is inconclusive whether carbetocin or oxytocin is more effective at caesarean section for preventing PPH.10,11
A Cochrane systematic review by Su et al. most recently updated in 2012 included 11 studies (2635 women). Four of the six studies comparing oxytocin with carbetocin were conducted in women undergoing caesarean section. This review provided uncertain evidence that favoured carbetocin at caesarean section, showing a possible reduction in PPH rates (risk ratio (RR) 0.66, 95% confidence interval (CI) 0.42 to 1.06) and a reduction in the need for additional uterotonics, with carbetocin administered as a 100 µg intravenous dosage across the trials (RR 0.62, 95% CI 0.44 to 0.88). 10 Likewise, a recent Cochrane network meta-analysis, which included 140 randomised trials with data from 88,947 women, and considered all amalgamations of prophylactic uterotonic drugs, after both vaginal and caesarean delivery, was inconclusive in terms of PPH rates (≥1000 ml), with prophylactic carbetocin in comparison with oxytocin. However, the point estimate was in favour of carbetocin (direct evidence RR 0.73, 95% CI 0.45 to 1.19). 11
The aim of this study was to examine the effectiveness of prophylactic carbetocin in comparison with oxytocin for preventing severe postpartum haemorrhage (defined as blood loss of 1000 ml or more). This endpoint is a precursor of significant maternal morbidity and mortality in women undergoing either elective or intrapartum caesarean section.
Materials and methods
Study design, setting and study population
This was a retrospective cohort analysis from one of Australia’s largest tertiary maternity centres, the Mater Mothers’ Hospital, Queensland. This work adhered to the reporting guidelines set by the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement and the Ethical Principles for Medical Research Involving Human Subjects outlined in the Declaration of Helsinki. 12 The study was conducted according to an a priori designed protocol and was approved with waiver of consent by the Mater Health Services Human Research Ethics Committee (HREC/13/MHS/115).
Inclusion criteria were adult participants who underwent scheduled, that is, before labour, or intrapartum, after a trial of labour or induction, caesarean section between 1 January 2010 and 2 July 2015, and who received either carbetocin or oxytocin immediate uterotonic therapy at the third stage of delivery. Exclusion criteria were: age less than 16 years or patients who received both carbetocin and oxytocin, as it was not possible to determine from clinical records which drug was administered first. The initial protocol did not include provisions for inclusion/exclusion of combination therapy, therefore the decision to exclude participants who were administered both carbetocin and oxytocin was made post hoc by the study team.
Participants were identified through the hospital clinical information system (MATRiX®) electronic database. This is a repository for antenatal information entered into the study institution’s electronic maternity record system (Meridian Health Informatics, Surry Hills, New South Wales, Australia) and included comprehensive individual level data on demographics, obstetric history, family history, medical and surgical history, allergies, standard observations, routine laboratory and ultrasound results, antenatal admissions and visit data.13,14 For the present study, deidentified data were electronically extracted by the MATRiX® database manager based on the study inclusion and exclusion criteria.
Exposure variable
The uterotonic was administered according to obstetrician preference, a decision that was not part of a research protocol, at the third stage of labour immediately after the birth of the infant and was recorded as oxytocin or carbetocin. At the time, there was no departmental guideline in place to inform the administration of uterotonics at caesarean section. Type and dosage of uteronic administered was at the discretion of the individual anaesthetist. Information was not available on the dose of carbetocin or oxytocin that was administered, nor whether the initial uterotonic was immediately followed by an oxytocin infusion.
Outcomes
The primary outcome was the incidence of severe postpartum haemorrhage defined by the Royal Australian and New Zealand College of Obstetricians and Gynaecologists as blood loss of 1000 ml or more during the postpartum period. 15 This outcome was clinically estimated by midwives after agreement between the anaesthetic, surgical and midwifery teams. Staff were very familiar with the visual estimation of blood loss technique and therefore they did not receive specific training. This method ensured high data entry rates and, to the best of our knowledge, no other methods of measurement were used to measure blood loss. Various secondary outcomes related to postpartum haemorrhage were also assessed: (1) total estimated blood loss using the method described above; (2) the composite endpoint that comprised all events recorded as ‘postpartum haemorrhage interventions’, which required one or more of: (i) bimanual compression; (ii) blood transfusion; (iii) insertion of a compression suture; (iv) hysterectomy; (v) internal iliac ligation; (vi) administration of additional uterotonic drugs, for example, ergometrine; (vii) administration of additional prostaglandin agents, for example, carboprost or; (viii) insertion of a tamponade balloon; (3) blood transfusion; (4) the composite endpoint of all events recorded as ‘third stage complications’, which required one or more of: (i) atonic uterus, defined as loss of tone in the uterine musculature not responsive to uterine massage; (ii) diffuse intravascular clotting; (iii) hypovolaemic shock; (iv) inverted uterus or; (v) cardiac arrest; (5) timing of caesarean section: scheduled (pre-labour) versus intrapartum (after a trial of labour or induction).
Statistical analysis
A sample size of 4634 participants with 2317 per group provided 90% power to detect a 1.5% decrease in the rate of severe postpartum haemorrhage in the carbetocin group, at a two-sided alpha level of 0.05, on the assumption of a 3.3% occurrence of blood loss of least 1000 ml in women who received oxytocin. 16 This occurrence was based on a meta-analysis of major PPH rates in randomised trials of prophylactic interventions. 16
A post hoc decision was made to exclude all those women who received both carbetocin and oxytocin from the statistical analysis as we were unable to determine which treatment women received first when given both drugs.
Descriptive statistics were calculated for all demographic and baseline levels of all clinical outcome variables or endpoints. 17 A multivariable logistic-regression model was used to estimate the propensity score, with third stage carbetocin used as the dependent variable and the following baseline characteristics included as covariates: (i) age; (ii) gestation; (iii) body mass index; (iv) private health insurance; (v) ethnicity; (vi) socioeconomic class (Index of Relative Socio-economic Advantage and Disadvantage); (vii) parity; (vii) timing of caesarean section: scheduled (pre-labour) versus intrapartum (after a trial of labour or induction); (viii) plurality; (ix) attendance at antenatal clinics; (x) method of anaesthesia for caesarean birth and; (xi) method of previous birth.
For the primary analyses, propensity score matching with replacement was conducted. This method includes all carbetocin (treated) patients and selects with replacement the oxytocin (control) patient whose propensity score is closest to the propensity score for each treated patient. Standardised difference was estimated before and after matching to assess the imbalances of the baseline characteristics. The standardised difference provides a method to compare the means between treatment groups in the propensity score matched sample. 17 This was done by calculating the difference in means between treatment groups as parts of the pooled standard deviation. 17 This permits different variables that are measured in different units to be compared. Importantly, this calculation remains independent of the sample size. 17 A standardised difference less than 10% for a covariate indicates a small imbalance in the mean or prevalence of a covariate between the treatment groups. 18
Comparisons of dichotomous outcomes were conducted using weighted logistic regression with cluster robust standard errors to account for controls being potentially included multiple times after matching with replacement. Similarly, for blood loss we conducted a weighted comparison between matched groups and estimated cluster robust standard errors.
A subgroup analysis was conducted by the timing for caesarean section, scheduled or intrapartum. 19 A subgroup analysis for the type of anaesthesia was planned; however, due to the low number of patients who underwent general anaesthesia this analysis was not performed.
The reported P-values are two-sided and P-values <0.05 were considered significant. All analysis was performed using SPSS, version 14.0 for Windows (SPSS Inc., Chicago, IL, USA) and SAS version 9.1 for Windows (SAS Institute, Cary, NC, USA).
Results
A total of 21,027 eligible participants were identified, of whom 10,696 received carbetocin and 10,331 received oxytocin for the active management of the third stage of labour (Figure 1). Numerous baseline characteristics showed significant differences before propensity score matching (Table 1). Propensity score matching with replacement resulted in 10,564 participants who received carbetocin that were matched with 3836 participants who received oxytocin (Table 1). The standardised difference was less than 10% after matching for all baseline characteristics (Table 1, note a). 18 Table 2 shows the maternal outcome data after propensity score matching.
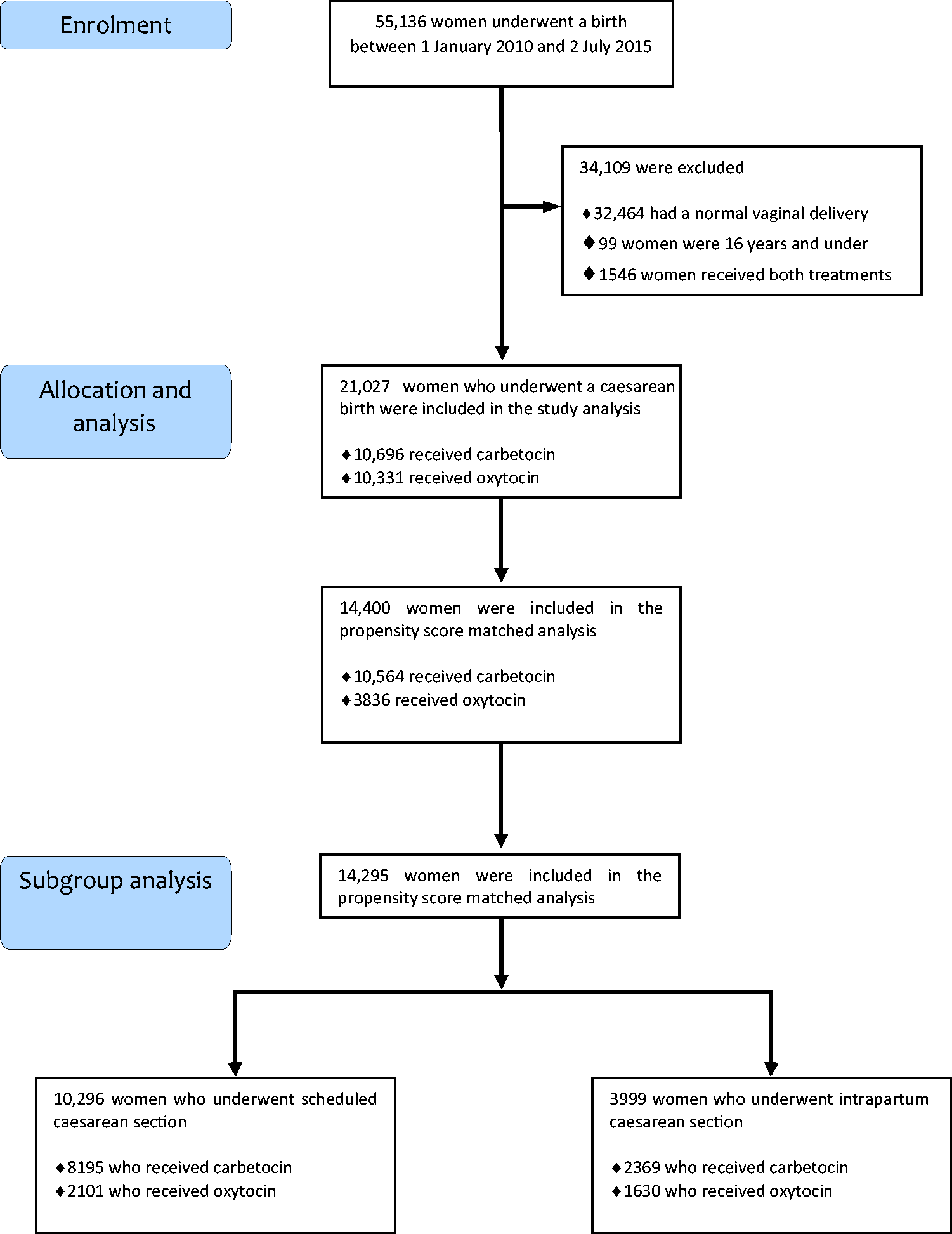
Flowchart of the women included in the retrospective cohort study.
Baseline maternal demographic and clinical characteristics of study participants in the unmatched and the matched samples.
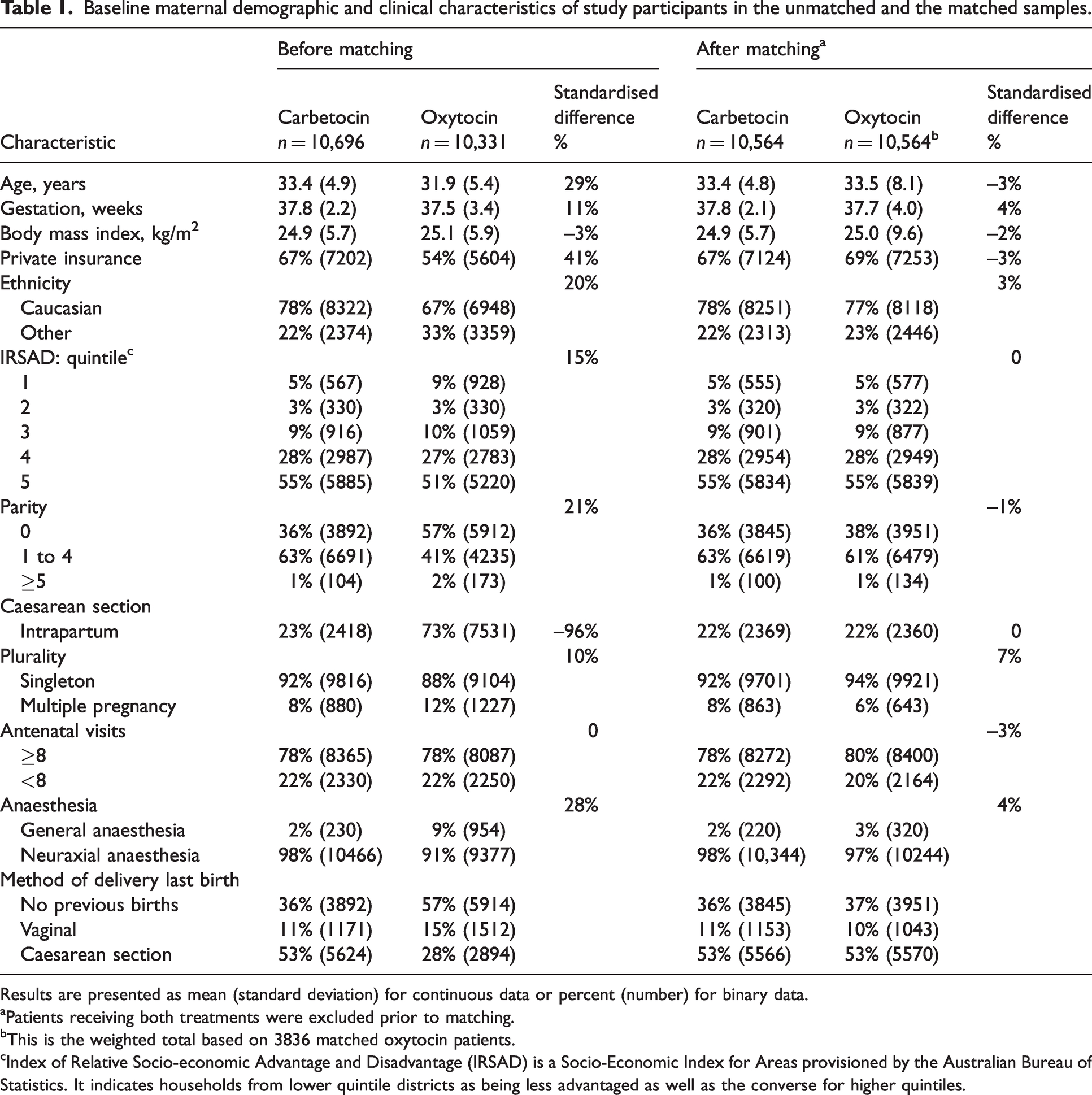
Results are presented as mean (standard deviation) for continuous data or percent (number) for binary data.
aPatients receiving both treatments were excluded prior to matching.
bThis is the weighted total based on 3836 matched oxytocin patients.
cIndex of Relative Socio-economic Advantage and Disadvantage (IRSAD) is a Socio-Economic Index for Areas provisioned by the Australian Bureau of Statistics. It indicates households from lower quintile districts as being less advantaged as well as the converse for higher quintiles.
Maternal outcome following caesarean section with carbetocin or oxytocin administration.
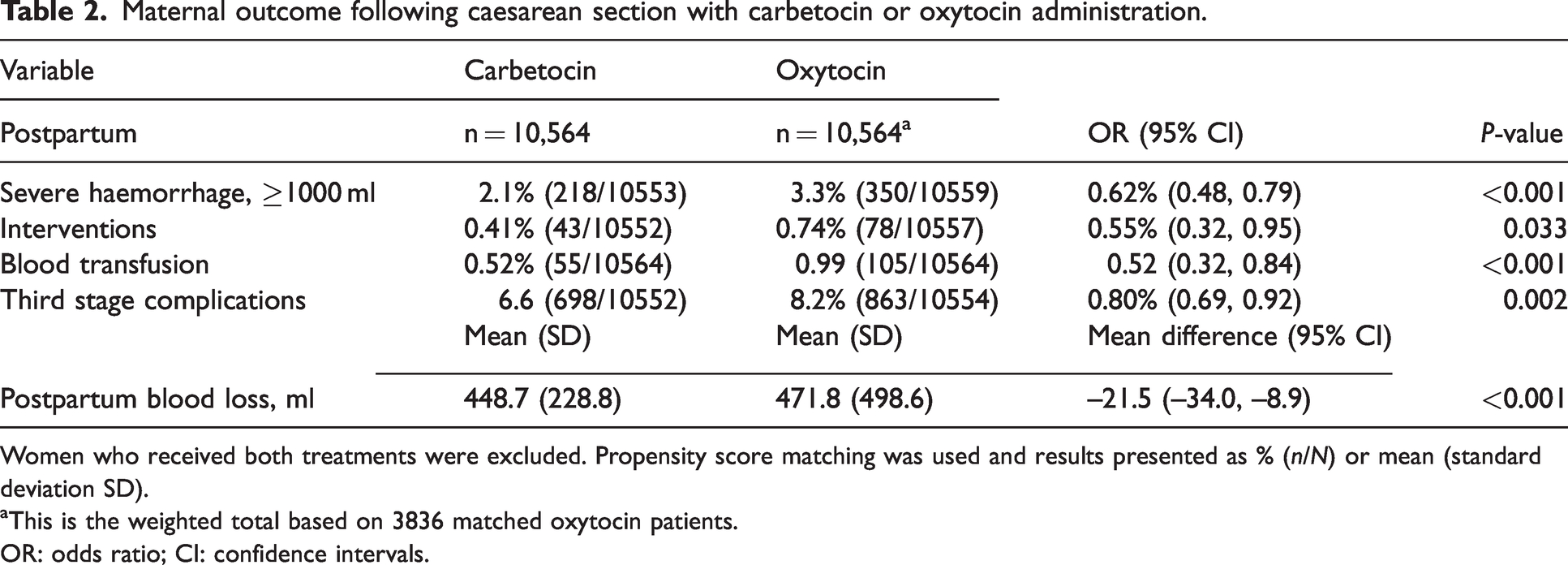
Women who received both treatments were excluded. Propensity score matching was used and results presented as % (n/N) or mean (standard deviation SD).
aThis is the weighted total based on 3836 matched oxytocin patients.
OR: odds ratio; CI: confidence intervals.
Primary outcome (matched cohort)
Carbetocin was associated with a lower risk of severe postpartum haemorrhage (2.1% versus 3.3% respectively; odds ratio, 0.62; 95% CI, 0.48 to 0.79; P < 0.001) compared with oxytocin for the active management of the third stage of labour.
Secondary outcomes (matched cohort)
Carbetocin was associated with a lower risk of postpartum interventions (0.41% versus 0.74% respectively; odds ratio, 0.55; 95% CI 0.32 to 0.95; P = 0.033) and a lower risk of blood transfusion (0.52% versus 0.99% respectively; odds ratio, 0.52; 95% CI 0.32 to 0.84; P < 0.001). Blood loss was decreased in the carbetocin cohort with mean (standard deviation, SD) blood loss of 449 (229) versus 472 (499) ml in the oxytocin cohort; P < 0.001. This corresponded to a mean difference in blood loss between the third stage oxytocic groups of −22 ml (95% CI –34 to –9; P < 0.001), favouring the administration of carbetocin. There was also a lower risk of ‘third stage complications’ after the administration of carbetocin compared with oxytocin (6.6% versus 8.2% respectively; odds ratio, 0.80; 95% CI 0.69 to 0.92; P = 0.002).
Subgroup analysis of primary outcome (Table 3)
For scheduled caesarean sections, carbetocin was associated with a lower risk of severe haemorrhage (1.5% versus 2.5% respectively; odds ratio, 0.59; 95% CI 0.41 to 0.84). Similarly, for intrapartum caesarean sections, carbetocin was associated with a lower risk of severe postpartum haemorrhage (4.2% versus 6.7% respectively; odds ratio, 0.60; 95% CI 0.43 to 0.84).
Severe haemorrhage in scheduled caesarean section or intrapartum caesarean following administration of carbetocin or oxytocin.
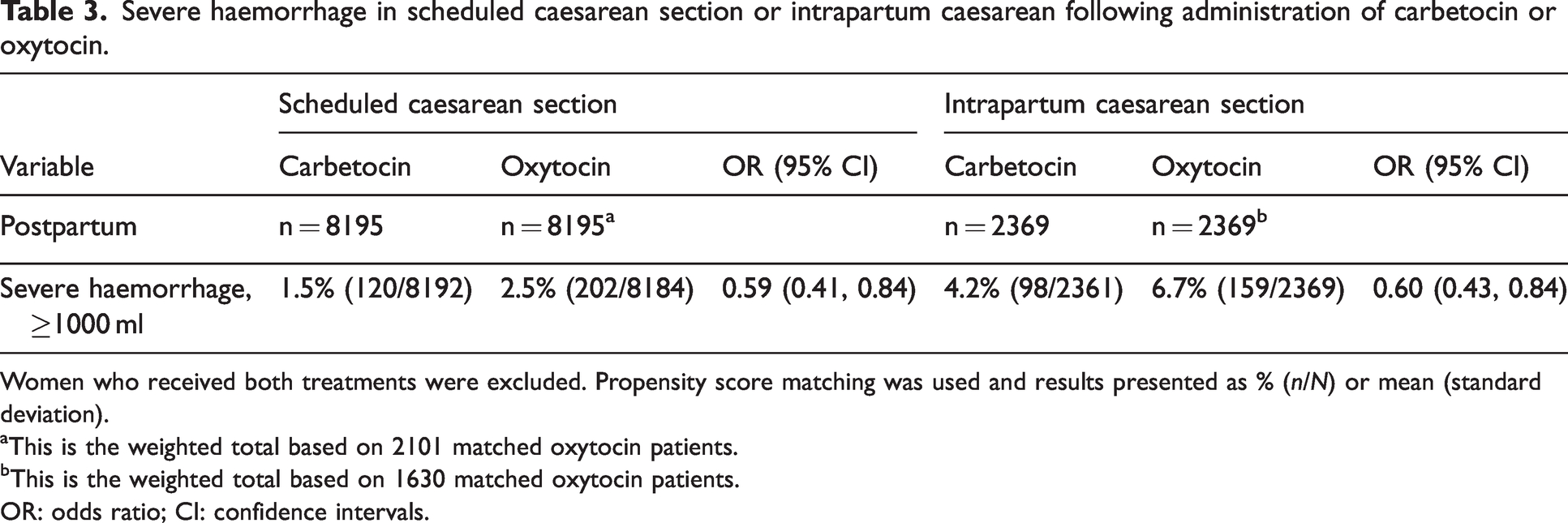
Women who received both treatments were excluded. Propensity score matching was used and results presented as % (n/N) or mean (standard deviation).
aThis is the weighted total based on 2101 matched oxytocin patients.
bThis is the weighted total based on 1630 matched oxytocin patients.
OR: odds ratio; CI: confidence intervals.
Discussion
The principal finding of this study was that the incidence of severe postpartum haemorrhage was lower with carbetocin administration compared with oxytocin for the active management of the third stage of labour in caesarean section. Importantly, the subgroup analysis for the population of women undergoing scheduled or intrapartum caesarean section did not differ materially and showed a consistent reduction in the risk of severe postpartum haemorrhage. Furthermore, secondary postpartum haemorrhage–related outcomes also favoured carbetocin. These findings favour the use of carbetocin at caesarean section, but require further investigation by randomised controlled study design. The results of this study are noteworthy considering that previous postpartum haemorrhage outcome data on the efficacy of carbetocin at caesarean section are inconclusive. The treatment effect estimated for severe postpartum haemorrhage in our study of odds ratio 0.62 (95% CI 0.48 to 0.79) is consistent with estimates reported in two Cochrane reviews based on point estimates and confidence intervals.10,11
For this present study, propensity score matching with replacement was conducted. This method means that oxytocin controls can be used as matches more than once. There was no calliper required as the closest control was matched. There were 10,564 cases and controls but some controls were used more than once. Therefore, there were 3836 unique controls in total that had been matched. The clustering effect of controls being used multiple times has been accounted for in the statistical analysis. Overall, matching with replacement can result in a reduction in bias by using similar controls to the treated individuals several times. 20
There was no difference in mean blood loss between the two groups. While average difference between treatment groups for blood loss in our study was statistically significant, it is pragmatically insignificant in the clinical setting, where measurement of blood loss relies upon consensus estimate.
The impact of excluding women who received both carbetocin and oxytocin should be considered. A possible explanation for why some women received both uterotonics is that the initial uterotonic inadequately contracted the uterus, resulting in further bleeding from uterine atony and therefore required an additional uterotonic. This may have resulted in a higher incidence of severe PPH in these patients. Excluding these patients could have had a significant effect on bleeding risk, with the direction of the systematic difference dependent on which uterotonic was administered first.
Another strategy that appears effective in the prevention of severe postpartum haemorrhage is the early administration of tranexamic acid. The WOMAN trial, a large international multicentre trial (N = 20,060), showed that tranexamic acid reduced the mortality from postpartum haemorrhage after vaginal delivery or caesarean section when compared with placebo (1.5% versus 1.9% respectively; risk ratio, 0.81; 95% CI 0.65 to 1.0; P = 0.045). 21 These promising interventions should hopefully prompt further investigations and funding to find preventive measures that will reduce the number of women who die from postpartum haemorrhage to a level below the 661,000 that were deceased worldwide after obstetric haemorrhage between 2003 and 2009. 22
Limitations
Due to the non-randomised, observational nature of the study, it is possible that selection bias by the attending obstetrician has influenced findings. The uterotonic was administered according to obstetrician preference rather than protocolised at the third stage of birth, and although this reflects real-life clinical practice, causality cannot be inferred from the results. The absence of data on the exact dose of carbetocin and oxytocin makes it difficult to compare the true bleeding risk between the groups. Furthermore, it is possible that the apparent association between carbetocin and severe PPH was confounded by the indication, for example, there is a possibility that women at lower risk of severe PPH were more likely to receive carbetocin. 19 Conceivably, the propensity score matching was unable to balance all unobserved confounders. A few risk factors for uterine atony for each participant could not be extracted, including polyhydramnios and prolonged labour. However, it is worth noting that significant PPH requiring a blood transfusion may occur in the absence of any risk factors in up to 60% of deliveries. 23 Generalisability of findings may have been compromised by including only around 38% of eligible controls in the matched comparison; however, matching with replacement matches the control with the closest propensity score to each ‘treated’ patient, hence the unmatched controls are not necessarily that different from the matched controls.
Measurement of the primary outcome, blood loss due to PPH, was performed via visual estimation and consensus from the treatment team. This method allowed for a high reporting rate in cases; however, it can be criticised for its poor reliability. This is routine for clinical determination of the degree of PPH at Mater Hospital, and due to the retrospective nature of this study, was unavoidable.
Conclusions
This study found that in a large cohort of women undergoing caesarean section, carbetocin was associated with a lower risk of severe postpartum haemorrhage compared with oxytocin. Our data also suggest that carbetocin is associated with a lower risk of postpartum haemorrhage interventions, blood transfusion and third stage complications, overall suggesting improved efficacy to oxytocin when administered for the active management of caesarean sections. Large pragmatic randomised controlled trials are needed to further investigate these results.
Footnotes
Acknowledgement
Assistance with the study: Mr Tyrone Humphries assisted with addressing revisions from the reviewers.
Author Contributions
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JES was supported by a National Health and Medical Research Council of Australia Career Development Award (reference 1045373). For the remaining authors none were declared.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
