Abstract
Most research in this field has focused on finding oxytocin doses for initiating uterine contractions. Only limited data are available regarding the optimal rate of oxytocin infusion to maintain adequate uterine tone. This randomised, double blind study included 120 healthy term pregnant patients with uncomplicated, singleton pregnancy undergoing elective caesarean section under spinal anaesthesia. Following an initial 1 IU bolus, the patients received oxytocin infusion at 1.25 IU/hour (group 1.25), 2.5 IU/hour (group 2.5) or 5.0 IU/hour (group 5) for four hours. Uterine tone was assessed as adequate or inadequate at various intervals. If found inadequate, additional uterotonics were administered. Estimated blood loss was mean (standard deviation) 499 (172) ml, 454 (117) ml and 402 (151) ml in groups 1.25, 2.5 and 5, respectively (P value groups 1.25 versus 5 = 0.012). Oxytocin infusion at 5 IU/hour resulted in a significantly lower incidence of minor postpartum haemorrhage, defined as blood loss greater than 500 ml, than 1.25 IU/hour (P = 0.009). No patient had major/severe haemorrhage (>1000 ml blood loss). No significant difference was seen in haemoglobin levels (P = 0.677) and uterine tone. Fifteen, six and nine patients, respectively, required additional oxytocin (P = 0.151). The incidence of tachycardia (P = 0.726), hypotension (P = 0.321) and nausea/vomiting (P = 0.161) was comparable. To conclude, 5 IU/hour was more effective than 1.25 IU/hour in reducing total blood loss and the incidence of minor postpartum haemorrhage. Thus 5 IU/hour appears to be an optimal oxytocin infusion rate following 1 IU slow intravenous oxytocin injection for the maintenance of adequate uterine contraction in patients undergoing elective caesarean section under spinal anaesthesia.
Introduction
Oxytocin is considered the drug of choice for the prevention and treatment of uterine atony. Earlier it was used in large bolus doses ranging from 5 IU to 10 IU during caesarean section. 1 The recent literature has established that doses much smaller than 5 IU are sufficient to initiate adequate uterine contraction, with relatively fewer adverse effects.2–6 However, it is advisable to administer a maintenance infusion after using low bolus doses of oxytocin, to maintain uterine contraction during caesarean section.7,8 Most of the research conducted until now has focused on finding the doses for initiating uterine contractions and only limited data are available regarding the optimal rate of oxytocin infusion to maintain adequate uterine tone. 9 An international consensus statement on the use of uterotonic agents has recently recommended oxytocin bolus 1 IU followed by an infusion at 2.5–7.5 IU/hour during elective caesarean section. 8 However, the recommendations for infusion rates are based on limited literature.9,10
Duffield et al. compared two infusion rates of oxytocin, namely 2.5 IU/hour and 15 IU/hour following an initial 1 IU bolus in women undergoing elective caesarean section. 9 They did not find any significant difference in the estimated blood loss, frequency of postpartum haemorrhage (PPH), adequacy of uterine tone, need for additional uterotonics or incidence of hypotension. Consequently, they advocated the need for further studies evaluating various doses of oxytocin infusion, with the aim of maintaining adequate uterine tone to minimise blood loss while also mitigating its associated adverse effects.
In another study, Kovacheva et al., while promulgating the rule of 3, found an oxytocin infusion of 3 IU/hour to be effective and safe following 3 IU oxytocin boluses. 10 The aim of that study, however, had been to evaluate the doses of oxytocin needed to initiate adequate uterine tone.
In view of the paucity of evidence in this field, the present study was conducted to evaluate the efficacy of three different intravenous infusion rates after an initial low-dose bolus of oxytocin for minimising blood loss by maintaining uterine tone during elective caesarean section under spinal anaesthesia.
Materials and methods
This randomised, double blind study, having a parallel design and 1:1 allocation ratio, was conducted after approval from institutional ethics committee and written informed consent from all patients. The trial was prospectively registered on 13 November 2017 at www.ctri.nic.in (CTRI/2017/11/010462). This paper is in accordance with the Consolidated Standards of Reporting Trials (CONSORT) guidelines. 11
A total of 120 healthy term pregnant patients with uncomplicated, singleton pregnancy undergoing elective caesarean section under spinal anaesthesia were studied. Patients with ruptured membranes; known risk factors for PPH; for example, multiple gestation, abnormal placentation, uterine fibroid, macrosomia, hydramnios, history of PPH or uterine atony; and those in active labour or having received oxytocin infusion were excluded. Patients with known drug allergy to oxytocin, any absolute or relative contraindications for spinal anaesthesia or maternal complications; for example, pre-eclampsia, diabetes mellitus, cardiovascular disease, cerebrovascular disease or bronchial asthma were also excluded.
The patients were randomly divided into three groups of 40 patients each, using a computer-generated random numbers table. Sealed envelopes were prepared according to the random numbers table to conceal allocation. After an initial oxytocin injection of 1 IU for the initiation of uterine contraction, the patients received oxytocin infusion at rates of 1.25 IU/hour (group 1.25), 2.5 IU/hour (group 2.5) or 5.0 IU/hour (group 5) for the maintenance of uterine contraction. The infusions were prepared in different dilutions in a 20 ml syringe so as to administer a constant rate of 5 ml/hour in all the groups. The anaesthesiologist preparing the infusion was not involved in the study, and the patient as well as the investigator were blinded to the group allocation.
The patients were kept fasting for eight hours for solids and two hours for clear liquids. Routine aspiration prophylaxis in the form of ranitidine 50 mg and metoclopramide 10 mg intravenously was provided. The preoperative haemoglobin level was measured on the day of surgery.
In the operating room, the baseline values for maternal heart rate and non-invasive blood pressure were recorded with a standard sized wedge under the right buttock. Coloading with Ringer’s lactate solution 10 ml/kg was started. Lumbar puncture was performed in the sitting position by midline approach at L2–L3 or L3–L4 vertebral interspace using a 25G spinal needle and 2.2 ml of hyperbaric 0.5% bupivacaine was injected. The volume of bupivacaine was reduced to 2 ml if the patient’s height was less than 150 cm. The patient was immediately turned supine and the wedge was placed under the right buttock. Oxygen was administered by clear facemask until delivery. Monitoring included continuous electrocardiography (ECG), heart rate, non-invasive blood pressure and pulse oximetry. Hypotension was defined as a 20% or greater fall in systolic arterial pressure from the baseline or a systolic arterial pressure of less than 100 mmHg, and was treated with bolus doses of intravenous 6 mg mephentermine. Tachycardia was defined as a 20% rise in heart rate from the baseline value.
At the time of clamping the umbilical cord, oxytocin 1 IU was administered intravenously over a period of one minute to all the patients. Immediately after this injection, oxytocin infusion was started at a rate depending on the group allocation, namely 1.25 IU/hour, 2.5 IU/hour and 5 IU/hour. This infusion was continued for four hours. Heart rate and non-invasive blood pressure were recorded every three minutes.
Uterine tone was assessed by a blinded obstetrician as adequate or inadequate two minutes after the end of the bolus injection (three minutes after cord clamping) and then every three minutes until 30 minutes after cord clamping. If uterine tone was found to be inadequate, a rescue dose of oxytocin 1 IU was given over one minute. Two such rescue doses of 1 IU oxytocin were given, if required. In case the uterus was not well contracted despite administering two rescue doses of oxytocin, 15-methyl PGF2α (carboprost) 250 µg was administered intramuscularly in consultation with the obstetrician. After an initial 30 minutes, the monitoring continued every 15 minutes until four hours.
The primary outcome measure was estimated blood loss during the intraoperative period. This was calculated by adding the volume of blood in the graduated suction canister after excluding the initial volume of amniotic fluid in it, the weight of blood in the blood-soaked sponges measured by subtracting the dry weight of standard size sponges from the weight of blood-soaked sponges, and estimating blood in the surgical field and drapes by visual estimation. Before starting the study, the extent of spread of 10 ml blood on a regularly used drape was noted, and this approximate measurement was used to estimate the volume of blood visually on the drapes. Weight to volume conversion for the blood loss in soaked sponges was taken as 1 ml for 1 g because the density of blood is generally estimated to be one gram per millilitre. 12
The secondary outcome measures included the incidence of PPH, adequacy of uterine tone, need for additional uterotonics, change in haemoglobin level at four and 24 hours, and any oxytocin-related side-effects; for example, tachycardia, hypotension, ECG changes, nausea, vomiting, flushing, headache, chest pain or dyspnoea after the administration of oxytocin. In keeping with a previous recommendation, minor PPH was defined as blood loss greater than 500 ml and major PPH as blood loss greater than 1000 ml. 13 For defining post-oxytocin tachycardia and hypotension, heart rate and systolic arterial pressure values at the time of cord clamping were taken as baseline values. Hypotension was defined as a 20% or greater fall in systolic arterial pressure and tachycardia as a 20% increase in heart rate from these baseline values.
Sample size calculation
Considering the standard deviation (SD) of blood loss in elective caesarean sections as 225 ml, 14 34 patients in each group were required to detect a difference of 200 ml blood loss with a two-sided significance of 5% and a power of 90%. To account for fallouts, 40 patients in each group were studied.
Statistical analysis
The statistical analysis was performed using SPSS statistical software version 20.0 (SPSS Inc., Chicago, IL, USA). The data were presented as mean (SD), median (interquartile range (IQR) (range)) or number (proportion). One-way analysis of variance (ANOVA), followed by Tukey’s test wherever applicable, was used to compare demographic profile and blood loss among the various groups. The general linear model of ANOVA, followed by Tukey’s test for post hoc analysis, was used to compare the repeated variables including haemodynamic parameters and haemoglobin values. The incidence of PPH, uterine atony and complications were compared using the chi-square test. A P value less than 0.05 was considered significant. However, in view of three comparisons, Bonferroni’s correction was applied for obtaining significance after post hoc tests when using chi-square and a P value less than 0.017 was considered significant.
Results
The patients were recruited from November 2017 to February 2019. The CONSORT flow diagram for the study is shown in Figure 1. Table 1 shows the demographic profile and other patient characteristics. All these were statistically comparable except for baseline systolic arterial pressure.
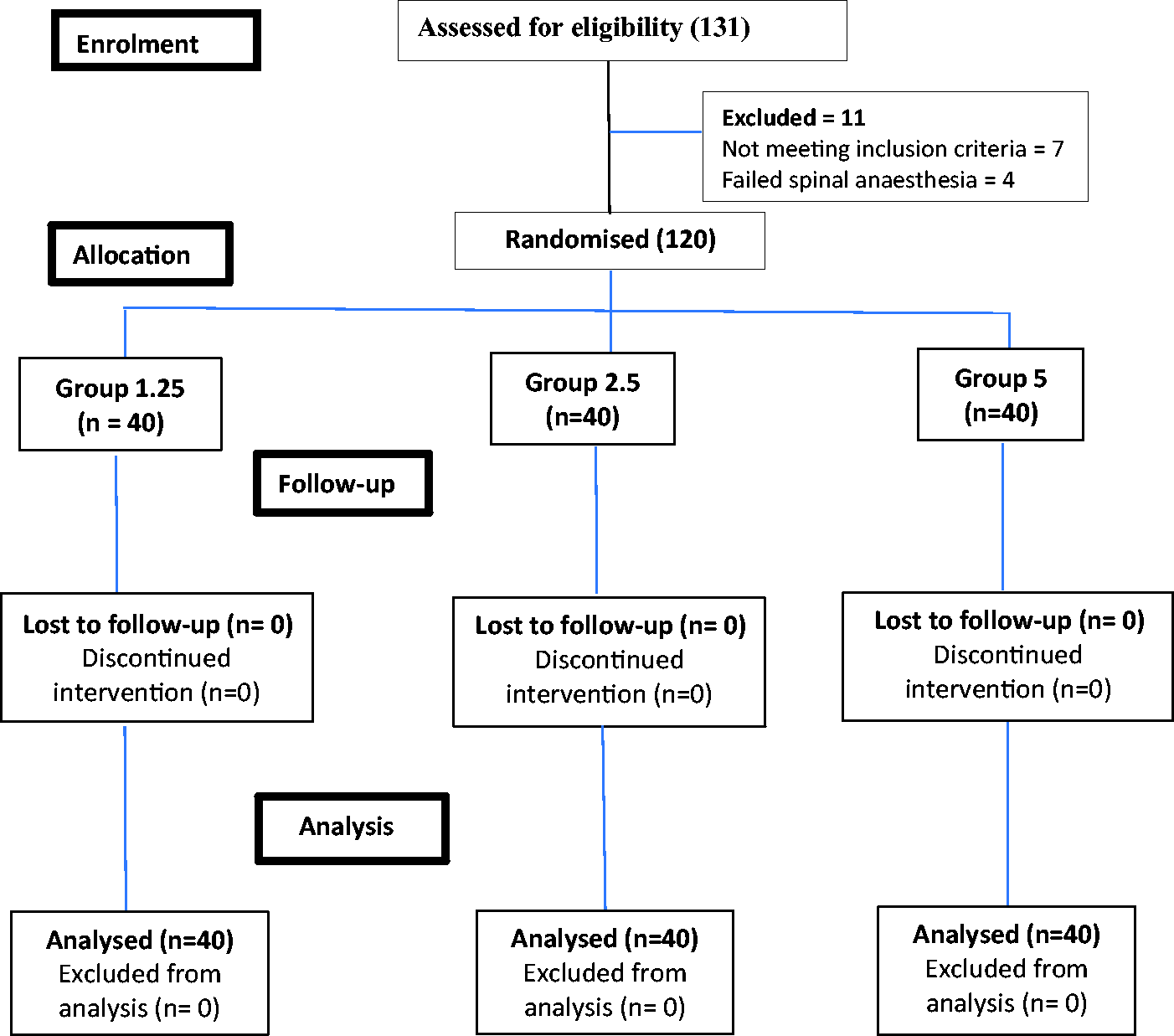
Consolidated Standards of Reporting Trials flow diagram. Groups received intravenous oxytocin 1.25, 2.5 or 5 IU/hour.
Demographic profile and other patient characteristics.
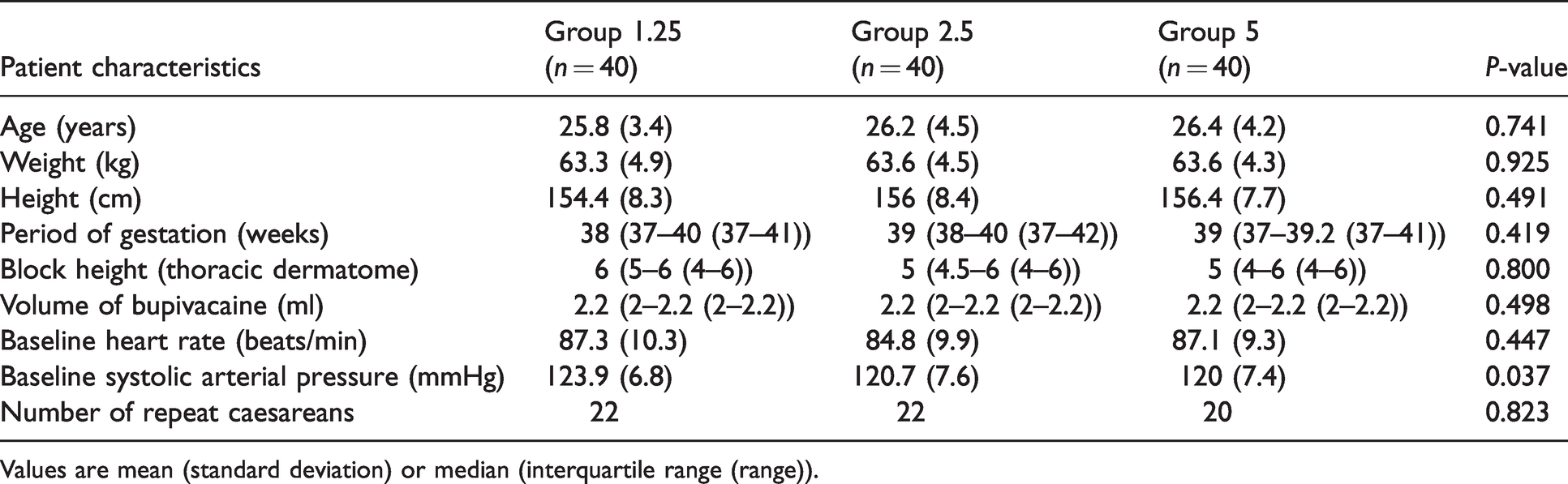
Values are mean (standard deviation) or median (interquartile range (range)).
Blood loss in the three groups is shown in Table 2. There was a decreasing trend in blood loss with increasing oxytocin infusion rates. Total blood loss was significantly lower for group 5 as compared to group 1.25; the estimated volume of blood lost on drapes was significantly lower in group 5 as compared to both group 2.5 as well as group 1.25. The number of patients with minor PPH, defined as blood loss greater than 500 ml, was also significantly lower in group 5 than in group 1.25. There was no statistically significant difference in total blood loss or the incidence of minor PPH between 1.25 IU/hour versus 2.5 IU/hour and 2.5 IU/hour versus 5 IU/hour (Table 2). No patient in any group had major/severe haemorrhage, defined as blood loss greater than 1000 ml. The maximum blood loss was 975 ml, 750 ml and 790 ml in groups 1.25, 2.5 and 5, respectively.
Blood loss and haemoglobin values.
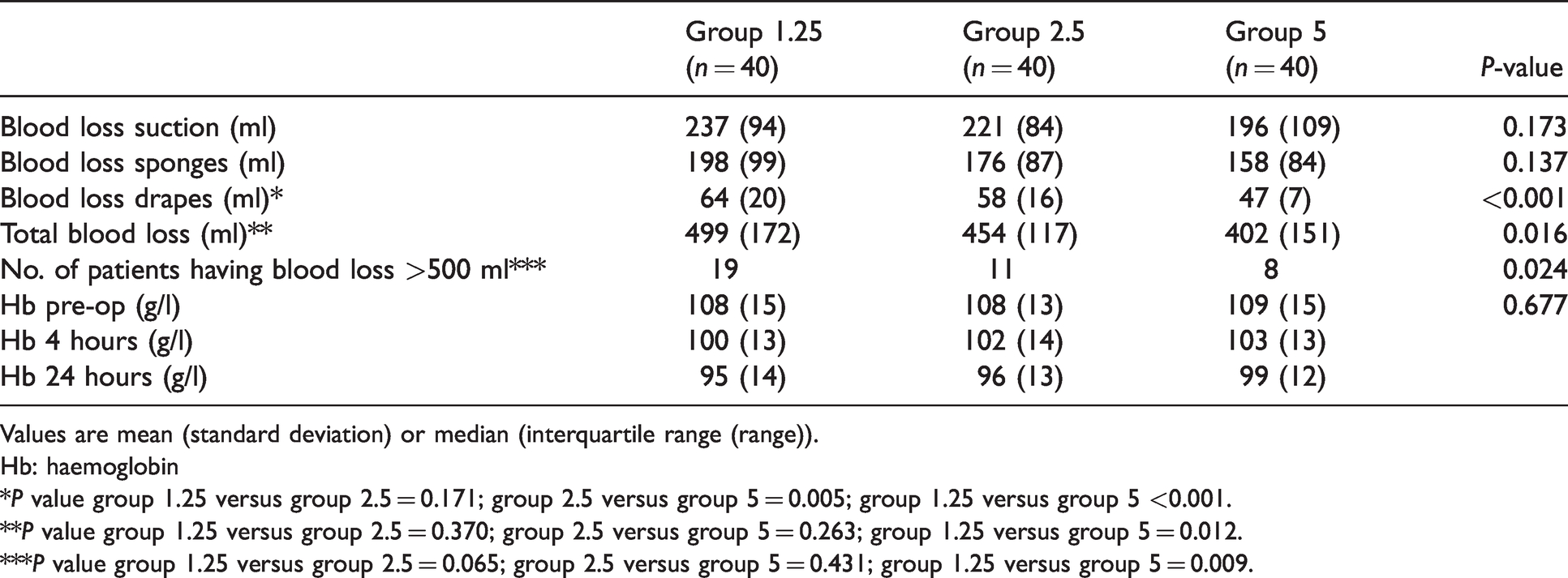
Values are mean (standard deviation) or median (interquartile range (range)).
Hb: haemoglobin
*P value group 1.25 versus group 2.5 = 0.171; group 2.5 versus group 5 = 0.005; group 1.25 versus group 5 <0.001.
**P value group 1.25 versus group 2.5 = 0.370; group 2.5 versus group 5 = 0.263; group 1.25 versus group 5 = 0.012.
***P value group 1.25 versus group 2.5 = 0.065; group 2.5 versus group 5 = 0.431; group 1.25 versus group 5 = 0.009.
Haemoglobin levels measured in the preoperative period, at four hours postoperatively and at 24 hours postoperatively, were not statistically significantly different among the groups (Table 2).
Uterine tone was comparable at all timepoints in all the groups except at nine minutes, when the tone was inadequate in seven patients in group 1.25, compared to one patient each in the other two groups (P = 0.019). However, using Bonferroni’s correction, this difference was not considered statistically significant. All the groups were statistically comparable with respect to the use of rescue oxytocin and carboprost (Table 3).
Requirement for additional uterotonics.

Values are numbers of patients.
There was no significant difference in mean heart rate (P = 0.186) and systolic arterial pressure (P = 0.057) at any timepoint after cord clamping in the three groups (Figures 2 and 3). Tachycardia after oxytocin administration was seen in 16, 16 and 13 patients receiving 1.25 IU/hour, 2.5 IU/hour and 5 IU/hour oxytocin, respectively (P = 0.726). The incidence of post-oxytocin hypotension was also similar, with eight, eight and 13 patients developing hypotension in groups 1.25, 2.5 and 5, respectively (P = 0.321).
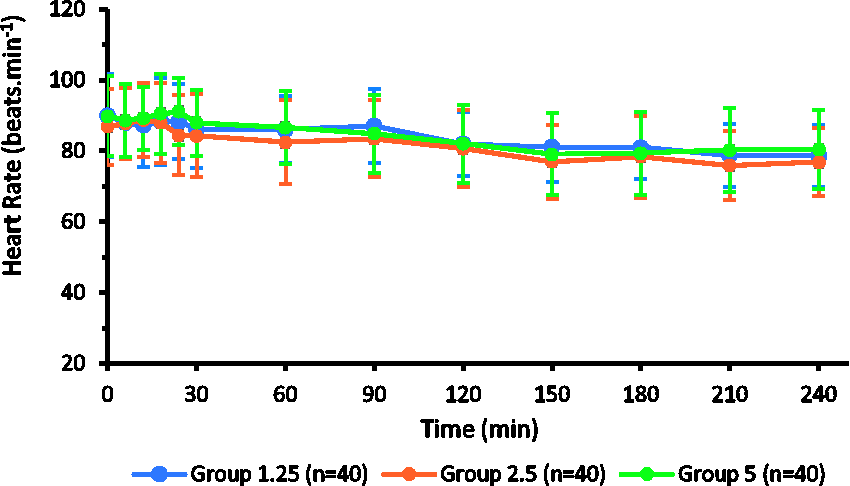
Heart rate trends (minute) after cord clamping. Groups received intravenous oxytocin 1.25, 2.5 or 5 IU/hour.
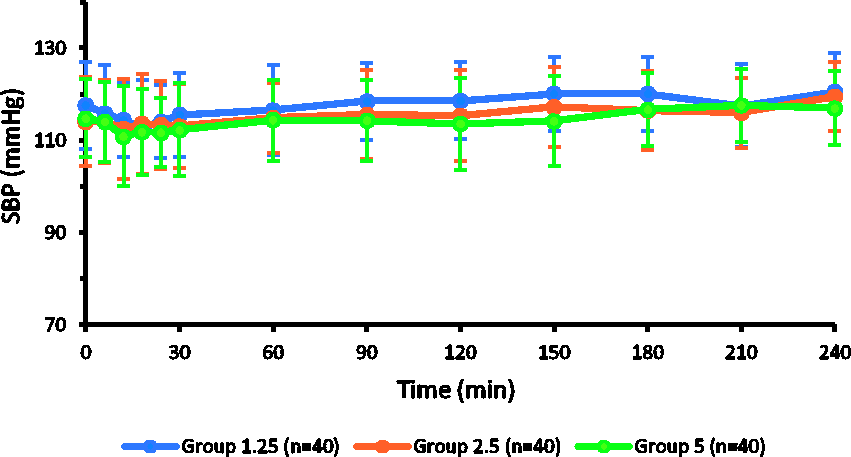
Systolic blood pressure trends (mmHg) after cord clamping. Groups received intravenous oxytocin 1.25, 2.5 or 5 IU/hour .
A total of seven, nine and 15 patients from groups 1.25, 2.5 and 5, respectively, complained of nausea/vomiting (P = 0.161). Of these, four, six and ten patients complained of nausea only and three, three and five patients developed vomiting in groups 1.25, 2.5 and 5, respectively. Two patients from group 5 had two episodes of vomiting each. No patients in any group developed ECG changes, flushing, headache, chest pain or dyspnoea.
Discussion
The results of this study demonstrated that the total intraoperative blood loss, the primary outcome, and the number of patients having minor PPH were significantly lower with oxytocin infusion at a rate of 5 IU/hour as compared to 1.25 IU/hour; whereas 2.5 IU/hour was statistically not different from 5 IU/hour or 1.25 IU/hour. Other secondary outcomes; that is, the adequacy of uterine tone, need for additional uterotonics and changes in haemoglobin levels at 4 and 24 hours after delivery were similar with all three doses.
Various researchers have used oxytocin maintenance infusions varying from 0.3 IU/hour to 15 IU/hour.2–4,15,16 However, most of them have focused mainly on studying the effect of bolus or infusion doses of oxytocin on the initiation of adequate uterine contractions. The research to determine an optimal rate of infusion for the maintenance of adequate uterine tone has been scarce. 9
In a study conducted by Duffield et al., to compare a low dose oxytocin infusion rate of 2.5 IU/hour with a high dose oxytocin infusion rate of 15 IU/hour after an initial 1 IU bolus, the estimated blood loss was found to be comparable in the two groups. 9 The incidence of PPH, defined as blood loss greater than 1000 ml in this study was similar in both groups; the incidence of blood loss greater than 500 ml was not reported. However, the study was terminated prematurely due to slow accrual of patients and change in the clinical practice. The calculated power of the study was only 68%, with approximately 23 patients in each group. For this reason, the authors stressed the need for further studies with larger sample sizes.
The lowest dose studied by Duffield et al. was 2.5 IU/hour, which was found to be as effective as the higher dose. 9 Their results support our observations that 2.5 IU/hour was not significantly different from the higher dose. However, the lowest dose used by us, that is, 1.25 IU/hour, was not studied by Duffield et al.
PPH is commonly defined as minor PPH with blood loss greater than 500 ml, and major/severe haemorrhage with blood loss greater than 1000 ml.8,13,17 According to Meher et al., the core outcome set for blood loss reporting should include the number of patients with 500 ml or greater, 1000 ml or greater loss and median or mean blood loss in each group. 17 Therefore, we calculated the number of patients having blood loss greater than 500 ml as well as 1000 ml. Although none of the patients had major haemorrhage, the incidence of minor PPH was higher in the patients receiving 1.25 IU/hour when compared to 5 IU/hour. Considering this finding along with the total blood loss, 5 IU/hour was more effective than 1.25 IU/hour. However, oxytocin infusion at a rate of 2.5 IU/hour was significantly not different from 5 IU/hour.
The blood loss in the suction canister was measured after the exclusion of the volume of amniotic fluid in it. Although an effort was made to note down the exact volume of amniotic fluid in the suction bottle, there may have been some mixing of blood with amniotic fluid. The blood loss on drapes was also assessed by visual estimation. However, these are the most practically feasible methods for the measurement of blood loss during caesarean section and have been used by earlier workers.9,18 Due to the problems involved in these methods, an accurate measurement of blood loss in the obstetric setting is considered challenging. 18
In the present study, although the 5 IU/hour oxytocin group was associated with a reduction in intraoperative blood loss and the incidence of minor PPH in comparison to the 1.25 IU/hour group, there was no significant inter-group difference in the adequacy of uterine tone or the use of rescue uterotonics. One explanation for this discrepancy could be that blood loss was measured by the same anaesthesiologist every time; whereas uterine tone was assessed in different patients by different obstetricians. This could have introduced some individual variations in the results. Although the number of patients requiring rescue oxytocin doses was more in group 1.25 (37.5%) compared to the other two groups (15% in group 2.5 and 22.5% in group 5), these differences did not achieve statistical significance. This could have been because the sample size was calculated on the basis of our primary outcome of blood loss estimation and our study was not adequately powered to detect differences in uterotonic requirements.
As there was no statistically or clinically significant difference in uterotonic requirements in patients receiving 2.5 IU/hour or 5 IU/hour oxytocin infusion, a dose of 2.5 IU/hour was as effective as the higher dose. Our results are supported by Duffield et al., who found no between-group difference in the use of additional uterotonics while comparing oxytocin infusions at rates of 2.5 IU/hour and 15 IU/hour. 9
We did not find any significant difference in the incidence of tachycardia or hypotension after delivery. Hypotension is reported to be one of the most common dose-related side-effects of oxytocin. 5 Our results indicate that a slow bolus of oxytocin 1 IU followed by infusion up to 5 IU/hour is quite safe and does not increase the incidence of hypotension when compared to lower infusion rates.
Oxytocin 1 IU was given slowly over one minute following cord clamping as this dose exceeds the minimum effective dose of oxytocin, has been found to be effective in initiating adequate uterine contractions,2,3,6 and has shown a 100% response rate. 2 The same dose for the initial bolus has also been recommended by the recent consensus statement on the use of uterotonics. 8
This study has certain limitations. First, this was a single-centre study. Second, the blood loss was measured only intraoperatively. Although the infusion was continued in the postoperative period, its effect on postoperative blood loss could not be assessed. Third, our results may not be applicable to those receiving large bolus doses of oxytocin initially by the “Rule of Three”.4,10 Finally, as our study included only non-labouring patients with uncomplicated pregnancies undergoing elective caesarean section, it may not be applicable for women who have been in labour or who are at high risk of uterine atony.
To conclude, oxytocin infusion at a rate of 5 IU/hour was more effective than 1.25 IU/hour in reducing intraoperative blood loss and the incidence of minor PPH. Thus 5 IU/hour appears to be an optimal infusion rate following a 1 IU slow intravenous oxytocin injection over one minute for the maintenance of adequate uterine contraction in patients undergoing elective caesarean section under spinal anaesthesia. Keeping in mind the continuous efforts being undertaken to use the minimum required dose of oxytocin, it may be suggested to use 2.5 IU/hour rather than 5 IU/hour as the two doses were statistically similar in terms of blood loss and minor PPH. However, based on the lack of a significant reduction in blood loss with 2.5 IU/hour as compared to 1.25 IU/hour, it would be appropriate to recommend 5 IU/hour as the optimal dose. Larger multicentre studies may be required to investigate this issue further.
Footnotes
Author contribution(s)
Data sharing
The data that support the findings of this study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
