Abstract
The Australian and New Zealand College of Anaesthetists (ANZCA) recently reviewed and updated the guideline on equipment to manage a difficult airway. An ANZCA-established document development group, which included representatives from the Australasian College for Emergency Medicine and the College of Intensive Care Medicine of Australia and New Zealand, performed the review, which is based on expert consensus, an extensive literature review, and bi-nationwide consultation. The guideline (PG56(A) 2021, https://www.anzca.edu.au/getattachment/02fe1a4c-14f0-4ad1-8337-c281d26bfa17/PS56-Guideline-on-equipment-to-manage-difficult-airways) is accompanied by a detailed background paper (PG56(A)BP 2021, https://www.anzca.edu.au/getattachment/9ef4cd97-2f02-47fe-a63a-9f74fa7c68ac/PG56(A)BP-Guideline-on-equipment-to-manage-difficult-airways-Background-Paper), from which the current recommendations are reproduced on behalf of, and with the permission of, ANZCA. The updated 2021 guideline replaces the 2012 version and aims to provide an updated, objective, informed, transparent, and evidence-based review of equipment to manage difficult airways.
Keywords
Summary of PG56(A) recommendations
Equipment required to manage difficult airways should be stored in difficult airway trolleys (DATs), which should be easily recognisable, readily accessed and logically laid out with clear labelling to facilitate efficient use.
Equipment for DATs should be fit for purpose. A minimum standard of evidence should support new equipment to justify its purchase. New equipment should be scrutinised by the Hospital Airway Committee or nominated airway expert and trialled by medical staff before purchase and introduction into clinical service.
Essential equipment for DATs should be standardised throughout all clinical areas where airway management is conducted to facilitate familiarity and recognition, particularly in a crisis.
Essential equipment for DATs should allow for redundancy. Redundancy provides backup when first-line ventilation or intubation equipment fails.
Guidelines, cognitive aids and emergency contact numbers should be attached to the DATs.
A culture of safety should apply to DATs and grab bags (see 10 below). The contents of DATs and grab bags should be audited regularly and checked for absence, malfunction, damage, contamination, expiry and misplacement of equipment and resources.
Equipment within the DAT should meet recognised infection control standards and comply with national standards and industry recommendations for handling.
Effective and timely training before patient contact is recommended for all equipment in any DAT. Regular in-service education should be conducted to teach practitioners about the proper use of airway management equipment.
Capnography should be immediately accessible wherever emergency airway management occurs.
A grab bag is a portable emergency container with recommended difficult airway management equipment for rapid deployment to clinical areas not readily serviced by the DAT. Grab bags are not a replacement for DATs in areas where routine airway management is regularly conducted.
What other resources are available on this topic?
The Australian and New Zealand College of Anaesthetists (ANZCA) PS55(A) position statement on minimum facilities for safe administration of anaesthesia in operating suites and other anaesthetising locations 1 describes the immediately required essential equipment for the institution, restoration and maintenance of oxygenation wherever airway management is or may be required.
Why were these guidelines updated?
All published guidelines risk becoming outdated. Since 2012, several devices for airway management have been withdrawn and new technology has been introduced. Guidelines for managing difficult airways have been updated and the increasing importance of human factors in airway management has been recognised. The 4th National Audit Project of the Royal College of Anaesthetists and the Difficult Airway Society (NAP4) highlighted the need for significant changes to airway management. 2 To this end, the revision of PS56 2012 examined all aspects related to DATs and simplified and standardised contents when possible.
How and why does this statement differ from existing guidelines?
The fundamental principles of PS56 2012 still apply. However, the current review has been extended to encompass adult, paediatric, obstetric, intensive care, and emergency medicine airway management, in both public and private hospital environments, including small stand-alone single procedural rooms. In addition, the diverse range of requirements encountered from tertiary centres through to very remote areas has been considered.
Summary of PG56(A) background paper
Introduction
In 2012 ANZCA released the PS56 Guideline on equipment to manage a difficult airway during anaesthesia based on recommendations from an expert panel established in 2008. This was in response to concerns raised in an audit of airway management equipment in a metropolitan region published in 2007. 3 The guideline was accompanied by a background paper published in Anaesthesia and Intensive Care in 2011. 4 While the fundamental principles of PS56, namely standardisation, redundancy, and a culture of safety still apply, significant changes have occurred during the intervening years. For example, several devices mentioned have now been withdrawn and new technology has been introduced. 5 A multidisciplinary expert team encompassing anaesthetists, intensive care medicine specialists, and emergency medicine specialists performed the review, updating adult, paediatric and obstetric difficult airway management practice requirements. Both public and private hospital environments were considered, including small stand-alone single procedural rooms, and the diverse range of requirements encountered from tertiary centres through to very remote areas. This article is an abbreviated version of the updated PG56(A) background paper (PG56(A)BP 2021), which is available on the ANZCA website. 6
Aim
The aim of updating PS56BP was to provide a current, objective, informed, transparent, and evidence-based review of equipment to manage difficult airways, including standardisation of the equipment throughout operating suites and areas beyond, including emergency departments (EDs) and intensive care units (ICUs).
Methods
A document development group (DDG) was formed from members of the airway management special interest group (SIG) as well as representatives from the College of Intensive Care Medicine of Australia and New Zealand and the Australasian College of Emergency Medicine.
An initial literature search was conducted from 1 January 2009 to 30 June 2019 using databases (Medline Ovid, Cochrane Database of Systematic Reviews) and a search engine (Google Scholar). The websites of the Difficult Airway Society (UK: https://das.uk.com/), Society of Airway Management (https://samhq.com/), and the European Airway Management Society (http://eamshq.net/index.php) were searched for equipment used in difficult airway management. In addition, the following websites were searched for additional information: American Society of Anesthesiologists (http://www.asahq.org), Australian and New Zealand College of Anaesthetists (http://www.anzca.edu.au), the former European Society of Anaesthesiology (http://www.esahq.org/euroanaesthesia), Canadian Anesthesiologists’ Society (http://www.cas.ca), and the Scandinavian Society of Anaesthesiology and Intensive Care Medicine (http://ssai.info/guidelines/).
English-only language articles and abstract publications were identified using keywords (Appendix 1) and filters. Devices and techniques that are not available in Australia and New Zealand were excluded. The initial search results were presented to the entire group for discussion to ensure all aspects of equipment to manage a difficult airway were incorporated in the repeat searches. Each of the subheadings from the draft paper were allocated to two independent committee members for an exhaustive keyword search, assessment of research and collation of results specific to their subheadings. These assessments were then presented to the entire committee for final evaluation, avoidance of duplication, identification of possible bias, collation and integration into the review. The results of the literature searches are summarised in Figure 1.
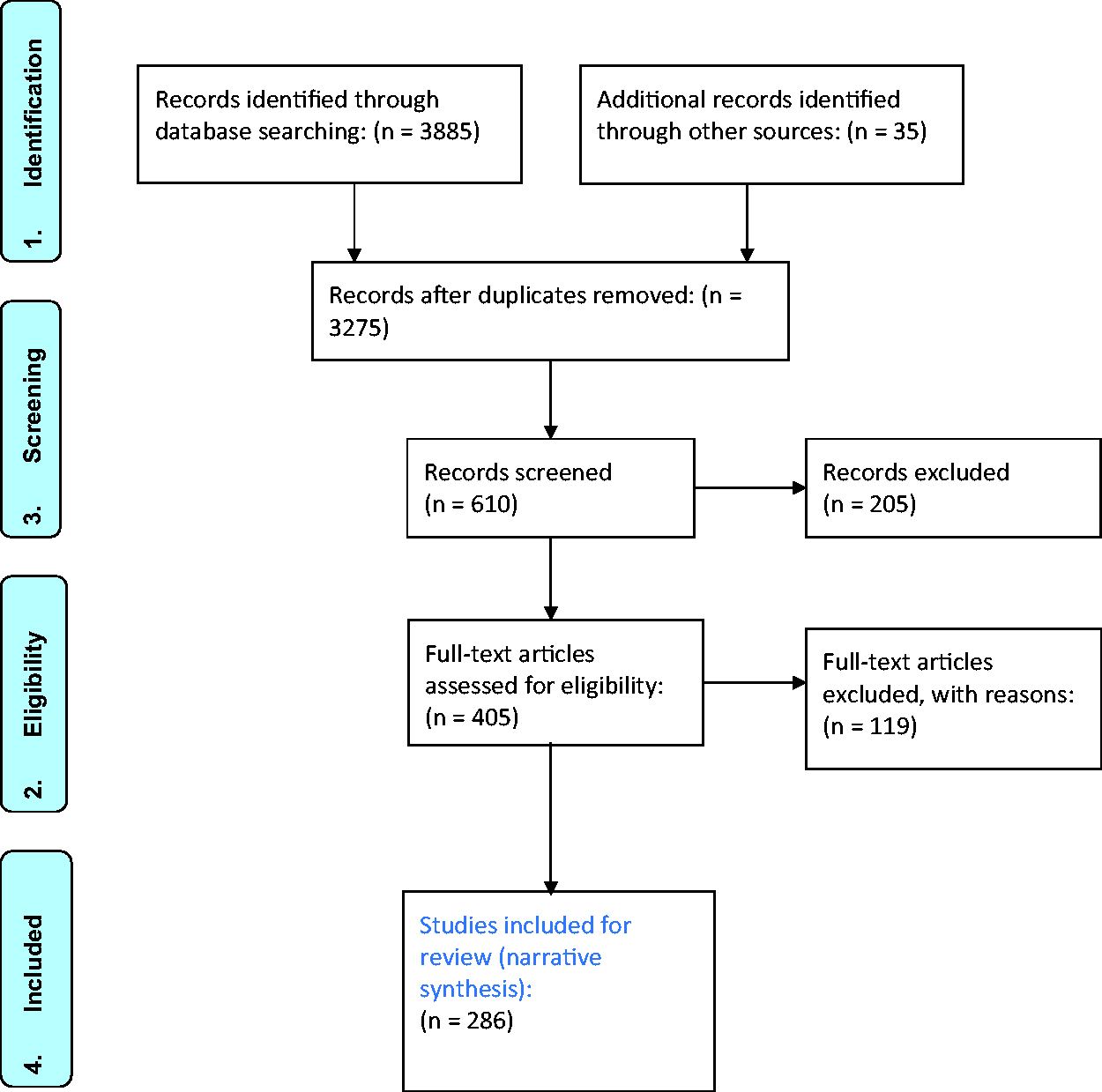
Results of literature search for PG56(A)BP 2021, 6 Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) diagram. As the current article is an abbreviated version of the PG56(A)BP, only selected references are included.

Recommended labels for difficult airway trolleys (DATs).
Subgroups of the DDG were asked to review publications relevant to their assigned subjects both from articles resulting from their searches, and from the references within those articles. Reviewed articles were given a rating according to the Grading of Recommendations, Assessment, Development and Evaluations (GRADE) system.7,8 Subgroups reported back to the DDG by teleconference and group discussion was recorded. This led to the development of informed expert consensus. 9
Results
The literature search results were screened for relevance by the DDG before allocation to subgroups (Figure 1). The ANZCA PG61(A) Guideline for the management of evolving airway obstruction: transition to the can’t intubate can’t oxygenate (CICO) airway emergency, 10 continuing professional development resources and previous work developing PS56BP 2012 formed the basis of the review. National and international guidelines and recommendations were considered during this process. Recommendations were based on the level of evidence from the review of the literature across all clinical applications from rural to tertiary level health institutions. Special areas of focus included obstetric, paediatric, intensive care and emergency medicine. The DDG assessed all previous recommendations as well as new proposals to ensure recommendations were up to date.
The DDG then made recommendations based on the evidence available, classifying their recommendations as follows.
The strength of recommendations and their descriptions are as follows:
Strong recommendation for – majority of practitioners would require the device. Weak recommendation for – many practitioners would require the device, but some would not; the choice may depend on local expertise. Strong recommendation against – majority of practitioners would not require the device. Weak recommendation against – many practitioners would not require the device, but some would require the device.
The quality of evidence was scored according to the GRADE system7,8 with the following definitions:
High quality – further research is very unlikely to change our confidence in the estimate of effect. Moderate quality – further research is likely to have an important impact on our confidence in the estimate of effect and may change the estimate. Low quality – further research is very likely to have an important impact on our confidence in the estimate of effect and is likely to change the estimate. Very low quality – any estimate of effect is very uncertain. In these cases, recommendations reflect the consensus opinion of the working group.
Discussion
DATs have evolved from heavy steel constructed sets of drawers confined to operating theatres to the modern equivalent, lightweight, highly manoeuvrable containers present in all clinical areas where airway management is performed. The ‘footprint’ of modern DATs has increased, however, as more bulky equipment is required. Large items such as flexible bronchoscopes, videolaryngoscopy screens, ultrasound, and various airway exchange catheters have made rapid transport of DATs problematic
It is recommended that the contents of each drawer be labelled to facilitate rapid and accurate access. This labelling system should be both printed and pictorial. Standards Australia and National Aeronautics and Space Administration (NASA) flight-deck typography recommend that printed labels are concise and easy to read at a distance.11,12 Lettering should use a clean and simple font and avoid using all capitals, underline or italics. The size of the font and spacing of the words should be maximal to fit the drawer front. Pictographs are preferred because they resemble pictures of what they signify, and are, therefore, more likely to be recognised by staff members. These pictographs should be simple and easily recognisable as stand-alone features
Equipment for difficult airway trolleys
Equipment for DATs should be fit for purpose and its purchase supported by a minimum standard of evidence, as is detailed in the Difficult Airway Society’s ‘airway device evaluation project team’ (ADEPT) article.
13
In addition, chosen equipment should comply with known international standards and recommendations.
14
Before purchase and introduction into clinical service, new equipment should be scrutinised by the hospital airway leads and trialled by medical staff according to committee standards
15
(
Essential equipment for DATs should be standardised throughout all clinical areas where airway management is conducted. Familiarity and confidence with any chosen equipment are key factors contributing to successful outcomes. Changes to their contents should be evidence based, or at least guided by expert advice
16
(
It is necessary to recognise that every device and technique is associated with a failure rate, and therefore structured plans and backup equipment, when first-line ventilation or intubation fails, are essential. Safety culture is a crucial component of the PG56(A) Guideline on equipment to manage a difficult airway during anaesthesia. Patient safety should always override considerations of convenience or economy.
Previous adult and paediatric surveys have shown a lack of confidence with DATs because of missing or faulty equipment or lack of confidence using the equipment.3,17 Morbidity and mortality have been reported due to practitioners using equipment without adequate training.
18
Examples include videolaryngoscopes, airway exchange catheters, and jet ventilators.19–21 Formal training and recent use of the equipment are directly correlated with confidence in using that equipment.
16
It is recommended that regular in-service education is conducted to teach practitioners about the proper use of airway management equipment (
Unfortunately, the removal of airway equipment from airway containers is widespread. For example, in a study of 22 DATs, 14 had content lists, but only 50% had contents corresponding with the checklists.
3
The authors recommend that DATs’ contents be checked for absence, malfunction, damage, contamination and expiry of equipment and resources at least weekly. This check should follow a checklist, which should be permanently attached to the trolleys, and an additional check should occur whenever any DAT has been used. Particular areas of concern are laryngoscopes, in which light emission should comply with international standards
22
(
Capnography should be immediately accessible wherever airway management occurs.4,23 It is recommended that waveform capnography should be used during emergency airway management (including emergency tracheal intubation and emergency front-of-neck access (eFONA) both inside and outside the operating theatre, including in the post-anaesthesia care unit (PACU) and off-site healthcare institutions such as stand-alone gastroenterology units (
Access to equipment
Wherever airway management is required, essential equipment for the institution, restoration and maintenance of oxygenation must be available and ready to use. This ‘point-of-care’ equipment includes an oxygen supply, suction, and essential airway management devices.
1
In a crisis, time is critical and includes the time taken to make a decision that a DAT is required, time to access and deliver the DAT to the point of care, and finally, time to restore oxygenation effectively and secure the patient’s airway.
26
The time taken for this process should be as short as possible to optimise the chance of a good outcome. Therefore, it is recommended that essential equipment should be portable and rapidly available within 60 seconds (
Recognised guidelines,10,27–32 cognitive aids,10,33–35 (Figure 3), emergency phone numbers, difficult airway alert letters and audit forms are recommended to be attached to the DATs. In addition, orientation, education, and regular interdisciplinary training based on chosen guidelines and cognitive aids should be performed (
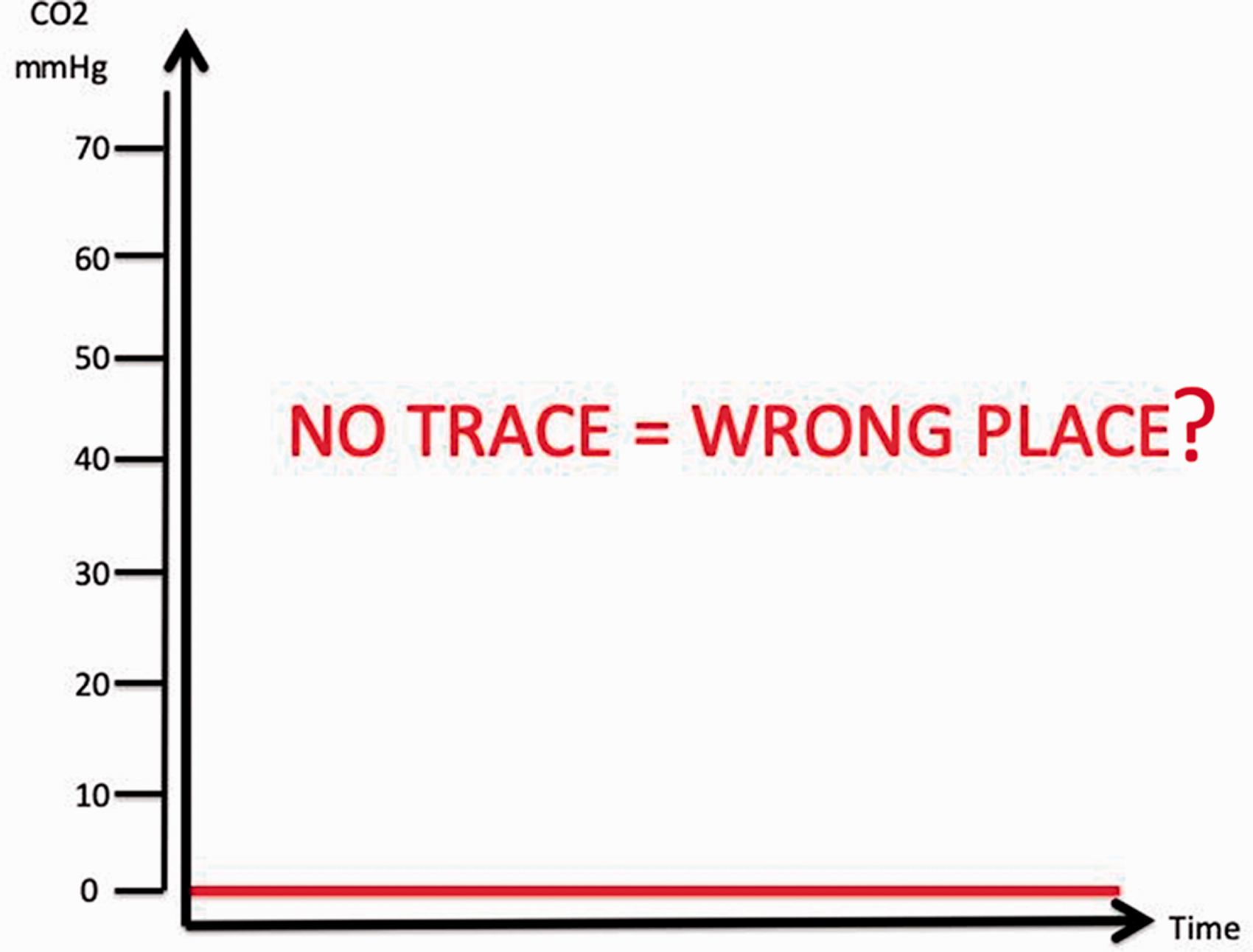
‘No trace = wrong place?’ cognitive aid reminding the operator that no capnograph tracing requires immediate removal of airway device and ongoing management.
Contents of difficult airway trolleys
Facemasks and self-inflating resuscitation bags
Difficult facemask ventilation incidence has been described as one in 100.36–39 The basic design of facemasks has not changed in 100 years, 40 and a wide range of sizes and designs are available. Ideally, a good seal should be achievable with a minimum leak and minimal dead space, while being transparent, lightweight and low cost. Designs include facemasks with inflatable air cushions, with or without inflation valves and those that mould to the face. Paediatric facemasks and laryngeal masks may provide an excellent seal over adult tracheostomy stomas. Consideration may be given to storing this equipment in adult DATs.
Single-use airway management equipment has increased significantly over the years, 41 and anaesthesia facemasks are the most commonly used single-use airway devices. Future aims should include reducing plastic waste for environmental reasons, and health practitioners should ensure that the impact on global climatic conditions and water scarcity becomes a significant consideration in the choice of equipment.42,43
Self-inflating resuscitation bags do not need a gas source to operate. However, the operator needs to be aware of their main disadvantage: the lack of operator ‘feel’ when there are changes in respiratory compliance or resistance. Several studies have indicated a high tendency for hyperventilation with currently used adult self-inflating bags, and a recommendation was made to use paediatric-sized bags instead.44,45 Appropriately sized self-inflating bags should be available where airway management occurs (including general anaesthesia, sedation, and intensive care and EDs) (
Laryngoscope blades
When initial tracheal intubation by direct laryngoscopy has been declared unsuccessful, ANZCA PG61(A) 10 recommends varying the type and size of the laryngoscope or converting to videolaryngoscopy.
The two common Macintosh blade designs are the English and the standard (or American) blade.
46
Compared to the standard blade, the English blade is longer, and its curve is more continuous across the entire blade length. In addition, the height of the blade flange is shorter, and the flange continues to the tip. These design features are advantageous in difficult airway scenarios.
47
Therefore, ANZCA recommends that DATs for adult use contain size 3 and 4 English style Macintosh blades with handles (
Advanced equipment, such as videolaryngoscopy and the availability of flexible bronchoscopes and intubation guides/bougies, may prove to be better alternatives to difficult airway management than straight and McCoy laryngoscope blades. Therefore, the latter are not required in DATs unless operators have been trained in their use and have an ongoing volume of practice (
Currently, there are no videolaryngoscopes with short handles commercially available. However, short-handled laryngoscopes may be useful for managing patients with large anteroposterior chest diameter and obstetric patients. Therefore, it is recommended that a short-handle laryngoscope is included in DATs (
Given that the size of paediatric patients in the adolescent age group can approach that of adults, it is recommended that Macintosh blades sizes 1, 2, 3, and 4 should be included in paediatric DATs in addition to straight blades, such as Miller blades, sizes 0, 1, and 2 (
Tracheal tube introducers, guides and catheters
Uses for tracheal tube introducers include difficult intubation, difficult extubation, placement of second‐generation laryngeal mask airways (LMAs) and cricothyroidotomy. 48 In addition, they come in different styles, including angled distal tips, malleable and newer devices, including a steerable tip. Unfortunately, success rates vary,49,50 and there is a lack of comparative studies to examine their performance 13 and peak force. 49 There is concern that some introducers may be responsible for airway trauma, mainly when ‘hold-ups’ and ‘clicks’ are elicited.49,51,52 Other studies describe shearing of the introducer tip, especially when railroading double-lumen tubes.53–55
Airway exchange catheters can be used as a safety adjunct when exchanging tracheal tubes or as part of an extubation technique.56,57 However, it is recommended that hollow exchange catheters are not used for oxygenation using insufflation by way of oxygen tubing connected via Luer lock due to the potentially high pressure created, causing a significant risk of barotrauma.18,20,58–61 The most widely available intubation catheter is the Aintree intubating catheter (Cook Medical, Bloomington, IN, USA), a hollow catheter 56 cm long, with a 6.5 mm outer diameter and 4.7 mm inner diameter. It is used over flexible bronchoscopes (preferably 4 mm or less) and in association with supraglottic devices for difficult tracheal intubation.
62
The smallest tracheal tube that can be easily railroaded over Aintree intubating catheters has an internal diameter of 7 mm. It is recommended that tracheal tube introducers with a 35° Coudé tip, intubation catheters and 11 Fr and 14 Fr catheters be stored in the DAT. They should be stored straight and not folded (
Videolaryngoscopes
Research comparing videolaryngoscopy with direct laryngoscopy is heterogeneous with variable outcomes. Videolaryngoscopy has been shown to improve the glottic view and to reduce the number of failed intubations and the incidence and degree of airway trauma.
63
There is, however, limited evidence that videolaryngoscopy reduces the number of intubation attempts, the incidence of hypoxia, respiratory complications,64,65 or the time required for intubation.66,67 This may also equally apply to difficult airways68–70 in the paediatric cohort, but further research is required.
71
Nevertheless, the use of videolaryngoscopy has been suggested as a first-line laryngoscopy device during elective anaesthesia71,72 and in specific clinical areas such as obstetric anaesthesia,32,73,74 bariatric anaesthesia,
75
intensive care,30,65,76 and emergency medicine.76–80 This argument is supported by the challenging task for all specialties to reliably predict airway difficulties, and the need to be prepared for unexpected difficulties.
68
Not a lot of evidence exists to support any particular type of videolaryngoscope over another in adult practice. Therefore, the choice of any particular device depends on individual case scenarios, local resources, operator familiarity, and dexterity with the device.
81
Both Macintosh style and hyperangulated blades, including tracheal introducers, are recommended to be available on all DATs (
Supraglottic airway devices
Supraglottic airway devices (SADs) are an essential component of any DAT. Although there is a large amount of research on SADs, many studies have limitations, including small numbers, the use of manikins only, consideration of minor outcomes, or being performed on patients with normal airways. 82 For DATs, the particular scenario of airway difficulty rather than routine clinical use should be the consideration. In the difficult airway setting, ventilation is paramount, and classic design LMAs with their low profile and lack of preformed curve have several advantages. Placement may be easier in common difficult airway scenarios such as reduced mouth opening, limited neck extension and reduced space in the anterior column. 83 Flexible intubation using an intubating catheter remains a robust solution to converting this device to a tracheal tube. 62
Second-generation SADs generally include a gastric access channel and an inbuilt bite block, and offer higher airway seal pressures than first-generation devices,29,84–86 providing better protection from aspiration than the classic LMA. When considering which second-generation device to include, expertise with use will significantly influence choice. 29
The classic design offers some advantages in managing difficult airways and should continue to be included on DATs. The inclusion of a second-generation SAD that can serve as a conduit for easy, flexible bronchoscope-guided intubation is recommended. The range of sizes to be stocked on DATs will depend on the population of the individual hospitals. Inclusion of the intubating LMA (iLMA or LMA-Fastrach; Teleflex Medical, Co., Westmeath, Ireland) on DATs is not recommended (
Flexible, fibreoptic and video bronchoscopes
Flexible, fibreoptic and video bronchoscopes provide a pivotal role in tracheal intubation and extubation, evaluation of upper and lower airways, examination of supraglottic airways, as well as tracheal tubes and tracheostomy tubes.87–91 In addition, they are valuable tools for lower airway suctioning, foreign body retrieval and obtaining specimens.
91
Flexible bronchoscopes should be readily available for all areas performing advanced airway management. The actual types and sizes chosen for any location will depend ultimately on the case mix, the volume of patients, and their physical status. Low-cost single-use flexible bronchoscopes may provide a realistic alternative for clinical environments with low usage but adequate staff training and experience. There is a wide range of ancillary equipment that is required to supplement flexible bronchoscopes. Airway experts should be involved in deciding what equipment and drugs should be stored in DATs to facilitate the performance of awake and asleep flexible bronchoscopy (
A range of tracheal tubes such as the Parker Flex- Tip92-94, the reinforced tracheal tube accompanying the intubating LMA 95 (iLMA or LMA-Fastrach; Teleflex Medical, Co., Westmeath, Ireland), reinforced tracheal tubes and nasal Ring-Adair-Elwyn (RAE) tracheal tubes may be better tubes to use than conventional PVC tracheal tubes for bronchoscope-assisted intubations. They position better on the scope and are less likely to cause epistaxis or hanging up on the vocal cords or arytenoids.
Front-of-neck access
Front-of-neck access procedures in anaesthesia include transtracheal or cricothyroid injection of local anaesthetic in preparation for awake intubation, pre-emptive cannula cricothyroidotomy or tracheotomy before induction of anaesthesia, retrograde intubation, awake surgical cricothyroidotomy and awake tracheostomy.96–98 eFONA is more commonly associated with percutaneous and surgical techniques associated with the CICO scenario.99,100 Clinical scenarios progressing from airway obstruction with low oxygen delivery to hypoxic cardiac arrest have escalating patient morbidity and mortality.
101
Based on these studies, it can be postulated that in the presence of hypoxia and ischaemia, eFONA should be performed within approximately 3–4 minutes.
102
It is recommended that a method of timekeeping is incorporated into future DAT designs to ensure this critical part of eFONA and difficult airway management is managed optimally (
Both cannula and scalpel techniques have been described. The cannula should be simple to use for eFONA and needs to allow passage of the Melker kit (Cook Medical, Bloomington, IN, USA) guidewire (16G minimum). A slip tip (non-Luer lock tip) 5 ml syringe is preferred for its length and volume. Using 2 ml 0.9% saline in the 5 ml syringe provides a positive endpoint of bubbles when aspirating air from within the airway. Oxygenation devices are flow-regulated T-piece variants, and should allow for expiration and provide auditory and tactile feedback concerning oxygen delivery. When attached to an oxygen flowmeter set at 15 l/min, they deliver oxygen at flows of 250 ml/s. However, flow rates above 15 l/min are discouraged as excessive flows may be obtained if the oxygen valve is opened beyond this level. 103
Pressure-regulated jet ventilation devices require extreme caution if used in the presence of upper airway obstruction.20,104 There is no tactile or visual feedback in terms of oxygen delivery, and there is no route for expiratory flow or pressure relief by way of these devices during expiration when connected directly to a cannula
105
(
A size 10 scalpel has a maximum width at a shallow depth required for the stab incision and successful performance of the scalpel-bougie technique. A hollow intubating introducer enables rapid oxygenation by attaching a 15 mm adapter to a self-inflating bag or anaesthetic circuit. Barotrauma risk is significantly reduced when using a low-pressure circuit compared with jet oxygenation by a Luer lock connector. A size 6.0 mm cuffed tracheal tube passes easily over the tracheal tube introducer with a Coudé tip of 35°. The tube dimensions ensure that it will pass through both the stab incision and the cricothyroid membrane in patients aged 12 years and older. Due to the importance of immediate access to eFONA equipment, it is recommended that it should be available at the point of care and duplicated on DATs (
The incidence of CICO in otherwise healthy children is exceedingly rare.109,110 Nevertheless, consideration needs to be given to significant anatomical differences in children of different ages. In neonates and infants, the cricothyroid space is underdeveloped. The narrow gap between the cricoid and the thyroid cartilage does not enable the passage of a size 2.0 mm inner diameter tracheal tube without significantly damaging the laryngeal cartilages. 111 The Association of Paediatric Anaesthetists of Great Britain and Ireland (APAGBI) paediatric airway guidelines recommend needle, cannula or surgical tracheostomy below the level of the cricoid ring. 112 Cricothyroidotomy sets are too large and potentially traumatic to laryngeal structures in children under the age of five years and are not recommended. 113 In children over eight to ten years of age, their cricothyroid dimensions allow for many of the commercially available percutaneous cricothyroidotomy devices to be used. eFONA may be extremely challenging even for the experienced ENT surgeon. Nevertheless, if equipment and the expertise of an experienced ENT surgeon are immediately available, then surgical tracheostomy may still be the better option.114,115
High flow nasal oxygen
There is increasing evidence that high flow nasal oxygen (HFNO) can provide a safe, convenient, well-tolerated and effective adjunct to difficult airway management. This technique provides oxygen flow rates that match or exceed a patient’s inspiratory flow rate and allows titration of warmed humidified inspired oxygen concentration up to 95%–100%. 116 Alongside the ability to provide a titratable high fractional inspired oxygen, other physiological benefits of HFNO have been described, 117 including reduced anatomical dead space, decreased airway resistance, increased functional residual capacity, improved respiratory mechanics, reduced alveolar de-recruitment, and flow rate–related positive end-expiratory pressure while closed mouth breathing. In addition, a variety of clinical applications have been described, including alternative methods of facemask pre-oxygenation,118–120 increased apnoeic oxygenation time, 121 as an adjunct providing oxygenation during awake tracheal intubation, 122 and as a means of providing ongoing oxygenation during ‘shared airway’ procedures.123,124
HFNO has recently transformed modern day anaesthetic practice and is an evolving area of practice in anaesthesia. There are several cautions concerning the use of HFNO, including the risk of airway fires.125–127 A patent upper airway is a prerequisite, and the degree of CO2 washout in apnoeic patients has not been fully validated.
128
It is advised to take precautions in high-risk patient populations susceptible to respiratory acidosis.
128
Other contraindications may include complete nasal obstruction, the presence of a base of skull fracture, cerebrospinal fluid leak, midfacial fractures and untreated pneumothorax. A tight-fitting mask attached to a closed breathing circuit should not be applied to a patient’s face when HFNO is used to avoid high airway pressures, barotrauma and possible gastric insufflation. ANZCA recommends that HFNO may be considered for difficult airway management and should be kept in a secure but easily accessible (unlocked) location close to, but not necessarily attached to, DATs (
Difficult airway equipment for paediatric anaesthesia
The same principles concerning quality and safety that apply to adult airway management equipment also apply to paediatric equipment. The specific details of equipment will depend on the scope of services provided, ranging from tertiary paediatric referral centres to general rural clinics. Ideally, equipment to manage difficult paediatric airways should be stored in a dedicated paediatric difficult airway trolley (PaedDAT) and should be suitable for the range of ages and sizes of patients undergoing care in the facility. For simplicity and easy access, it may be reasonable to design two trolleys–one PaedDAT for children up to eight years and an adult DAT for larger older children. SADs are recognised as essential tools for managing difficult paediatric airways functioning as primary ventilation airways, conduits for tracheal intubation and rescue ventilation devices. 129 The incidence of hypoxaemia is lower when continuous ventilation occurs through supraglottic airways during flexible bronchoscope–guided intubation, and higher first-attempt success than videolaryngoscopy in infants with difficult airways has been described. 130 Results from the Pediatric Difficult Intubation (PeDI) registry suggest a 2% difficult intubation rate. 131 Complications were associated with more than two intubation attempts, weight less than 10 kg, short thyromental distance and more than three direct laryngoscopy attempts before resorting to any indirect technique. 131
eFONA equipment should be provided to suit the full spectrum of ages from premature neonates to morbidly obese adolescents. Paediatric practice guidelines do not consider infants or neonates. 132 A recent systematic review of the emergency paediatric surgical airway included only five papers, all only involving animal studies. 133 The analysis of mean time for placement of a definitive airway showed ‘catheter over needle’ was the most rapid technique but with a high failure rate; the ‘wire-guided’ technique had a high success rate but a high complication rate; the ‘cannula’ technique had fewer complications but a high failure rate; the ‘scalpel-bougie’ technique had a high success rate but longer procedural time. The other important issue after eFONA relates to oxygenation techniques. Very little clinical or laboratory evidence exists to confirm the efficacy or safety of the most recommended techniques. 108
Difficult airway equipment for obstetric anaesthesia
Equipment to manage difficult airways in obstetric patients does not need to be different from that recommended in the remainder of the accompanying guideline.134,135 The guiding principles are simplicity and familiarity. The use of videolaryngoscopes should be first-line, given the higher baseline risk of difficult airways and the emergent nature of many obstetric intubations.32,74,134,136,137 There are no large-scale studies in obstetrics regarding the use of videolaryngoscopy and no evidence-based guidance as to the best device.134,138 However, there are multiple reports of successful use, and one unit reported no failed intubations following the introduction of videolaryngoscopy to their DAT. 139 A standard laryngoscope with a stubby or short handle should be available. At present, no videolaryngoscopes fulfil this requirement. Second-generation SADs are beneficial in obstetrics providing a higher seal pressure facilitating ventilation and a gastric drainage port. 137 As recommended for adults, a first-generation device needs to be available in DATs if problems with intubation, facemask ventilation, or seating of any second-generation supraglottic device occur. All SADs should allow an easy transition to intubation without the need to remove the airway, which may be required during general anaesthesia for caesarean section. 140
Aside from a ready supply of pillows for routine ramping, no new or alternative equipment is required for DATs in the obstetric difficult airways setting
141
(
Difficult airway equipment in intensive care medicine
The NAP4 2 highlighted deficiencies in airway management and higher rates of adverse patient outcomes in ICUs in the UK. How these results reflect intensive care medicine in Australia and New Zealand is debatable. Nevertheless, the ICU environment poses unique challenges that may encompass various combinations of anatomical, physiological142,143 and psychological144,145 difficult airway factors.
Airway management in the ICU often involves time-critical airway management in deteriorating patients with multiple comorbidities. While no specific equipment exists for ICU difficult airways, the rationale for choice or how it is presented often reveals significant differences from operating theatre environments.30,51 For example, patients with a limited physiological reserve are unlikely to tolerate slow airway management. Waking up patients when difficulty is encountered is rarely an option. In general, patients are often not fully conscious and cooperative and, therefore, rapid sequence or modified rapid sequence is commonly selected. This, in turn, has direct implications on the type of rescue device and techniques used.
Equipment stored on DATs in the ICU provides an important ‘infrastructure’ for any airway management team. These devices should have high success rates, be ‘user friendly’, and be intuitive to use. While various airway management devices are commercially available, stocking a large variety of different devices should be avoided. 146 Recommended devices include a range of second-generation SADs, Macintosh-style and hyperangulated videolaryngoscope blades, and equipment for a surgical airway. In addition, the need for specialised tracheal extubation equipment should be considered. 147
Currently, there is a wide range of commercially available videolaryngoscopes, and therefore their selection should reflect essential nuances of the intensive care environment. Videolaryngoscope monitors should be large enough for the entire team to view while being easily manoeuvrable to avoid reflected lighting from nearby windows on the video image. In addition, they should provide a high-resolution image viewable at angles allowing assistants a good view of the image.
The videolaryngoscope blades should be low profile, allowing sufficient space for manipulating tracheal tubes and/or adjuncts, especially in narrow upper airways.
The cannula for eFONA may be inadequate to provide oxygenation in patients with profound abnormalities of pulmonary compliance or gas exchange. Therefore, the choice of equipment available for eFONA should include a cuffed tube positioned by either a scalpel-bougie technique or a wire-guided percutaneous dilational technique (the Seldinger technique).
The lack of experienced assistance makes the labelling of intensive care DATs a critical issue. Clear labelling with both printed labels and pictographs is essential.
Flexible bronchoscopes should be available. Operators need to be aware of the different diameters of these scopes. ‘Quality improvement bundles’ 148 and high-fidelity simulation programmes 149 have been shown to improve patient care. DATs may be considered an integral part of these programmes. Therefore, familiarisation and regular training using DAT equipment should be encouraged.
Difficult airway equipment in emergency medicine
Airway management in EDs, as with other areas outside of the operating theatre environment, carries significantly increased risks and can be technically challenging.2,29,150 Patients requiring intubation in EDs often have severe physiological derangement, have had limited pre-intubation airway assessment, and may require expedient intubation. It is well recognised that there is a higher incidence of difficult airways in EDs, many of which will be unpredicted.151,152 It is, therefore, recommended that for all intubations in EDs, DATs are available within the time frames specified in this document.
Given the variations in patient volume and acuity, staffing and skill mix in EDs throughout Australia and New Zealand, the specific configuration of DATs in individual EDs will largely be determined by local factors. It should, however, be consistent with the underlying principles and recommendations contained in this document and should be configured in a standardised manner consistent with other DATs in the institution.
A recent multicentre study of intubations in 43 EDs in Australia and New Zealand highlighted that videolaryngoscopes were used in the first attempt in over half of the intubations. 153 With this and other studies demonstrating higher first-pass success rates with videolaryngoscopy over direct laryngoscopy, even in expert hands, this proportion is likely to increase in coming years significantly. 63 Therefore, for EDs that use videolaryngoscopes for first intubation attempt, a second videolaryngoscope is recommended to be available on the DAT.
Cricothyroidotomy in Australasian EDs is a rare event, with rates around 0.3%.153,154 However, all clinicians should be familiar with the eFONA equipment on their DATs, and regular simulated practice is strongly encouraged.
No data are currently available regarding flexible bronchoscope use in EDs in Australia and New Zealand. A large US registry study reported low ED utilisation, 155 likely reflective of Australian and New Zealand practice. It may, therefore, be neither feasible nor desirable for every ED to procure a flexible bronchoscope. Alternative solutions, such as rapid access to flexible bronchoscopy and skilled personnel from the operating theatres or ICUs, may be reasonable alternatives in low demand EDs.
Limitations of review
This review focused on English language publications and therefore may have omitted important articles in other languages. In addition, the level of airway management research is often moderate or low quality. Therefore, the strength of the recommendations will generally reflect these levels.
Conclusion
Standardisation of difficult airway equipment commonly used in anaesthesia, intensive care and emergency medicine in DATs is seen as a vital and fundamental step towards improving patient safety and a collaborative multidisciplinary approach to airway management. While each clinical environment may require variants of these recommendations, the provision of core airway management equipment in all DATs throughout Australia and New Zealand is a fundamental focus of this review. All aspects of airway management for general anaesthesia and sedation were considered as well as the various clinical scenarios ranging from stand-alone gastroenterology units providing sedation to tertiary level hospitals providing care to high acuity patients with airway problems. It is expected that close adherence to these guidelines will provide an infrastructure of suitable airway equipment to operators managing difficult airway scenarios. The review has been extensive and comprehensive, as evidenced by the length of this background paper, to ensure that it is contemporary and reflects the range of opinions when making any recommendations. It is strongly recommended that airway leads are consulted in implementing the recommendations in the accompanying guideline.
Supplemental Material
sj-pdf-1-aic-10.1177_0310057X221082664 - Supplemental material for Updated guideline on equipment to manage difficult airways: Australian and New Zealand College of Anaesthetists
Supplemental material, sj-pdf-1-aic-10.1177_0310057X221082664 for Updated guideline on equipment to manage difficult airways: Australian and New Zealand College of Anaesthetists by Endlich Yasmin Conceptualization Methodology Writing – original draft Writing – review & editing Hore Phillipa J Conceptualization Methodology Writing – review & editing Baker Paul A Conceptualization Writing – review & editing Beckmann Linda A Conceptualization Writing – review & editing Bradley William P Conceptualization Writing – review & editing Chan Kah L E Conceptualization Writing – review & editing Chapman Gordon A Conceptualization Writing – review & editing Jephcott Christopher G A Conceptualization Writing – review & editing Kruger Peter S Conceptualization Writing – review & editing Newton Alastair Conceptualization Writing – review & editing Roessler Peter Conceptualization Project administration Writing – review & editing in Anaesthesia and Intensive Care
Footnotes
Author Contribution(s)
Acknowledgements
The author(s) wish to thank Laura Foley (Manager, Knowledge Resources at ANZCA library) for her tireless efforts in coordinating the literature search. The author(s) also wish to thank Vanessa Hille for her support in coordinating the review and the editing process of the document. Finally, the author(s) acknowledge the work and support of current and past committee members and would also like to thank the large number of anaesthetists providing feedback to the PS 56BP.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: YE, PAB, LAB, WPB, KLEC and GAC have received free airway equipment for research and teaching from several manufacturers listed in this article. PSK provides consultancy work for Smiths Medical Pty. Ltd. PJH has conceptualised and collaborated with the development of an intubating bougie with a flexible tip. The remaining author(s) have no conflicts of interest to declare.
Funding
The author(s) received no financial support for this article's research, authorship, and/or publication.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
