Abstract
Māori are the indigenous people of New Zealand, and suffer disparate health outcomes compared to non-Māori. Waikato District Health Board provides level III intensive care unit services to New Zealand’s Midland region. In 2016, our institution formalised a corporate strategy to eliminate health inequities for Māori. Our study aimed to describe Māori health outcomes in our intensive care unit and identify inequities. We performed a retrospective audit of prospectively entered data in the Australian and New Zealand Intensive Care Society database for all general intensive care unit admissions over 15 years of age to Waikato Hospital from 2014 to 2018 (
Keywords
Introduction
Māori are the indigenous people of Aotearoa New Zealand (NZ), constituting 16.5% of the population in 2018. 1 Māori experience inequitable health outcomes in NZ, including an increased burden of chronic disease.2,3 This is attributable to multiple upstream determinants in the post-colonial setting, including intergenerational social disenfranchisement, socioeconomic disparity and increased barriers to access at all levels of healthcare.4–7
Inequities are reversible, unjust, and perpetuate other inequalities in society. 8 Furthermore, inequities can have consequences for the health of a population across all socioeconomic strata, with more inequitable societies experiencing worse outcomes as a whole compared to less inequitable societies.8,9 Previous literature has identified inequity in critical illness admissions for Māori, including trauma,10,11 stroke 12 and severe infections.13,14 There is, however, no literature describing Māori health outcomes in a general intensive care unit (ICU) population not specified to a type of presentation.
Waikato Hospital is the tertiary referral centre for five district health boards (DHBs) across the Midland region of the North Island (Lakes, Bay of Plenty, Tairāwhiti, Taranaki and Waikato DHBs), providing cardiothoracic, neurosurgical and trauma services supported by a 16-bedded closed model level III ICU and a 12-bedded open model high dependency unit (HDU). Waikato Hospital serves a total population of 951,965 people across the Midland, of whom 25.75% identified as Māori in 2018. 15
The NZ Public Health and Disability Act 2000 provides a mandate for the NZ health sector to tackle ethnic health inequities actively, according to the principles of partnership, protection and participation of the nation’s founding document, te Tiriti o Waitangi.16
We aimed to audit the care of critically ill patients in our institution’s ICU, focusing on risk-adjusted mortality as a measure of equity.
Methods
The study was conducted as a systems performance audit at Waikato DHB (registration number 3988DIC), following the principles of the audit cycle. 17 Ethical consideration was provided by our institution’s Clinical Audit Support Unit, and Māori consultation by our institution’s Māori Research Review Committee. In 2016, Waikato DHB formalised a six-goal corporate strategy policy for the provision of healthcare, one of which was to ‘eliminat[e] health inequities for Māori’. 18 This was chosen as the standard for our audit, with the target being no difference in risk-adjusted mortality between Māori and non-Māori.
Adult patients aged 16 years or older admitted to Waikato ICU between 2014 and 2018 inclusive were identified through the prospectively entered adult patient database (APD) of the Australian and New Zealand Intensive Care Society (ANZICS), and admission characteristics were extracted. Elective cardiothoracic patients were excluded due to their unique characteristics and morbidity, and are examined in a separate study. HDU patients did not form part of the inclusion criteria due to the open model nature at our institution; only ICU admissions contribute to the ANZICS APD. Admission diagnoses, as defined by the ANZICS APD data dictionary, 19 were further aggregated by major diagnostic groups after Paul et al. 20 and Secombe et al., 21 with the exception of cardiovascular diagnoses, in which the distinction between operative and non-operative was retained. Due to some infection-related diagnoses being classified under organ system rather than ‘sepsis’ in the APD – for example, bacterial pneumonia under ‘respiratory’ – admissions were further stratified in a binary manner for infection across all diagnostic groups. Only the first admission for each patient during a single hospital episode was included in analysis.
Acute illness severity is defined by established ICU scoring systems extracted from the APD, including Acute Physiology and Chronic Health Evaluation (APACHE) II, 22 APACHE III 23 and the Australian and New Zealand Risk of Death (ANZROD) score. 20 Chronic morbidities are identified in the APACHE III score. Our unit additionally collected data for insulin-dependent diabetes mellitus. Chronic illness severity is described by multimorbidity, defined as two or more morbidities, in which all immunosuppressive states and all liver-related morbidities are each considered one morbidity, after Secombe et al. 21
Ethnicity is self-identified by patients according to the NZ National Health Index system. When a patient identifies with more than one ethnicity, Māori ethnicity is prioritised for the purposes of analysis. 24 Other demographic data were extracted from the hospital internal database. This included rurality, as matched to domicile code through previous work by Edwards et al., 25 and the NZ Index of Deprivation 2018 (NZDep2018), a nationalised centile of incremental socioeconomic deprivation, as matched to Statistics NZ geographical meshblocks. 26 Renal replacement therapy (RRT) requirement is identified by International Classification of Disease (ICD) version 10 coding.
The primary outcomes were adjusted in-ICU and in-hospital mortality. The secondary outcome was one-year mortality, as a proxy measure for post-ICU quality of health and life. We additionally looked at the utilisation of RRT, invasive ventilatory requirement within the first 24 hours and length of stay as other measures of equity.
Statistical analysis was performed using R Statistical Software (version 3.6.2; The R Foundation, Vienna, Austria). Continuous data were assessed for normality using the Shapiro–Wilk test. Non-normal data are presented as median with interquartile ranges, and analysed using the Wilcoxon rank sum test. Normal data are presented as mean with standard deviation, and analysed using appropriate
Binary logistical regression for in-ICU and in-hospital mortality were separately performed, and controlled for clinically selected variables, including gender, ethnicity, ANZROD, NZDep2018, rurality, admission source, infection-related diagnosis, admitting specialty and organ support requirement (RRT and invasive ventilation within the first 24 hours, separately). Model goodness-of-fit was assessed by the chi-squared likelihood ratio test and discrimination indices, including C-index and Brier score. Results are presented as adjusted odds ratio (AOR) with 95% CI. Separate sensitivity analysis was performed for relative outcome of ethnicity by diagnostic group, after Secombe et al. 21
Patients who transferred to another facility on discharge were excluded from the analysis for all mortality outcomes, including adjusted. One-year mortality uses the same denominator as in-ICU mortality. Missing data were not imputed, due to the audit nature of this study.
A
Results
Between 2014 and 2018 inclusive, there were 3009 admissions to Waikato ICU that met the study criteria, of which 982 (32.6%) were for Māori. Patient characteristics are summarised in Table 1.
Patient characteristics.
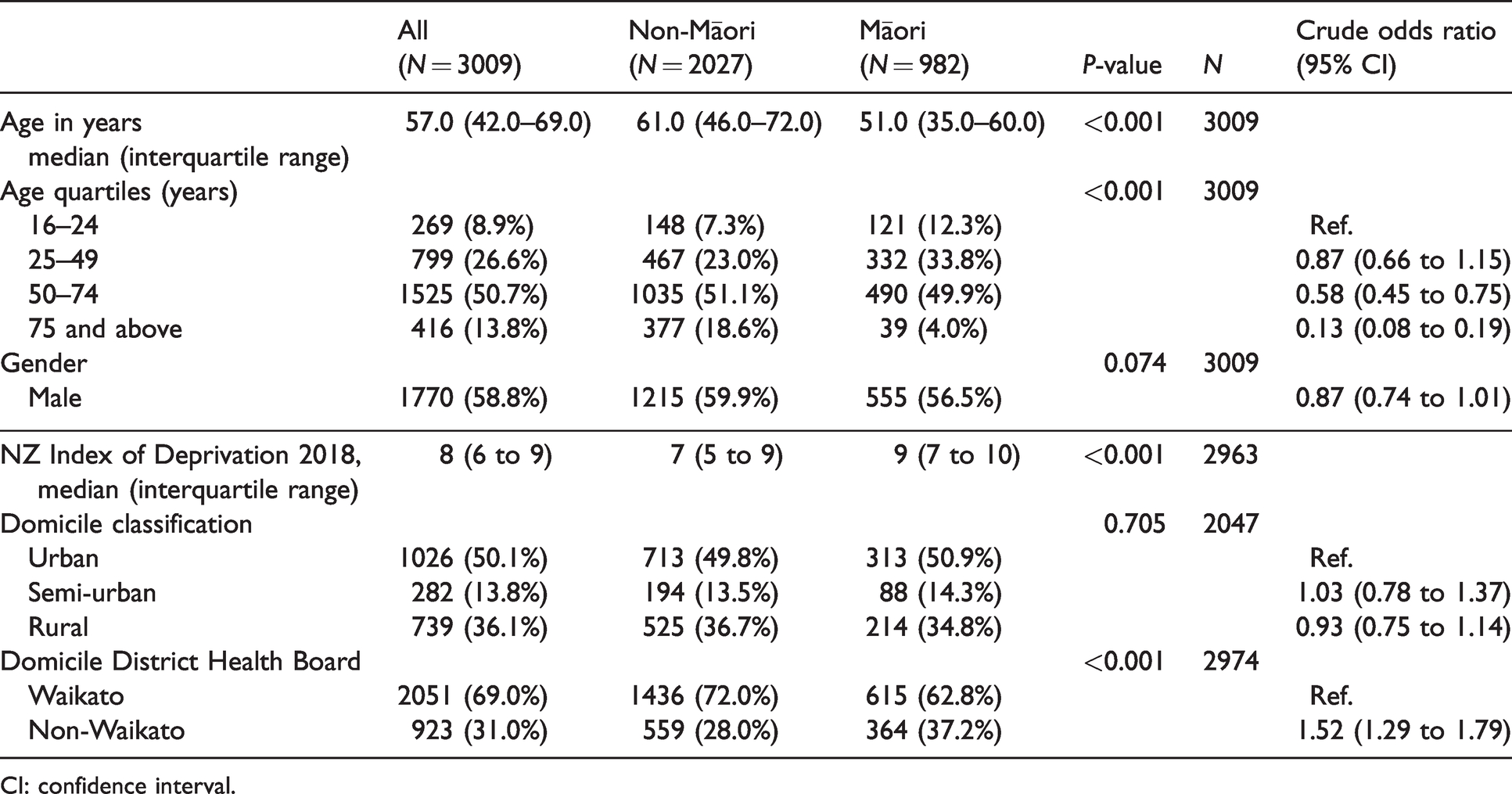
CI: confidence interval.
Māori patients had a younger median age (51 versus 61 years,
Māori were more likely to be domiciled in a more deprived area (NZDep2018 of 9 versus 7,
Admission and baseline characteristics are summarised in Tables 2 and 3. Baseline severity scores were similar between Māori and non-Māori, including ANZROD (8.72 versus 9.41;
Admission characteristics.
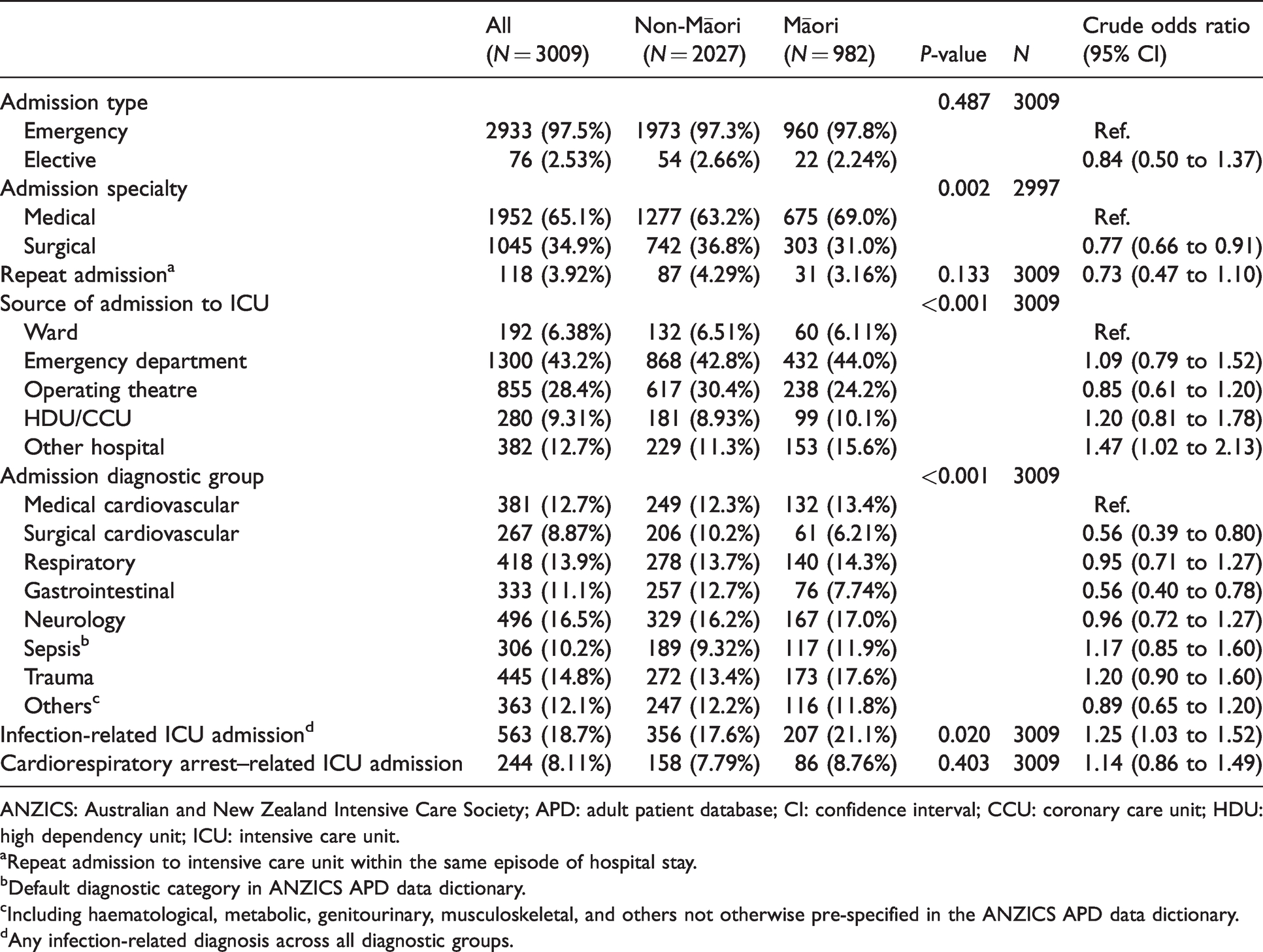
ANZICS: Australian and New Zealand Intensive Care Society; APD: adult patient database; CI: confidence interval; CCU: coronary care unit; HDU: high dependency unit; ICU: intensive care unit.
aRepeat admission to intensive care unit within the same episode of hospital stay.
bDefault diagnostic category in ANZICS APD data dictionary.
cIncluding haematological, metabolic, genitourinary, musculoskeletal, and others not otherwise pre-specified in the ANZICS APD data dictionary.
dAny infection-related diagnosis across all diagnostic groups.
Baseline characteristics.
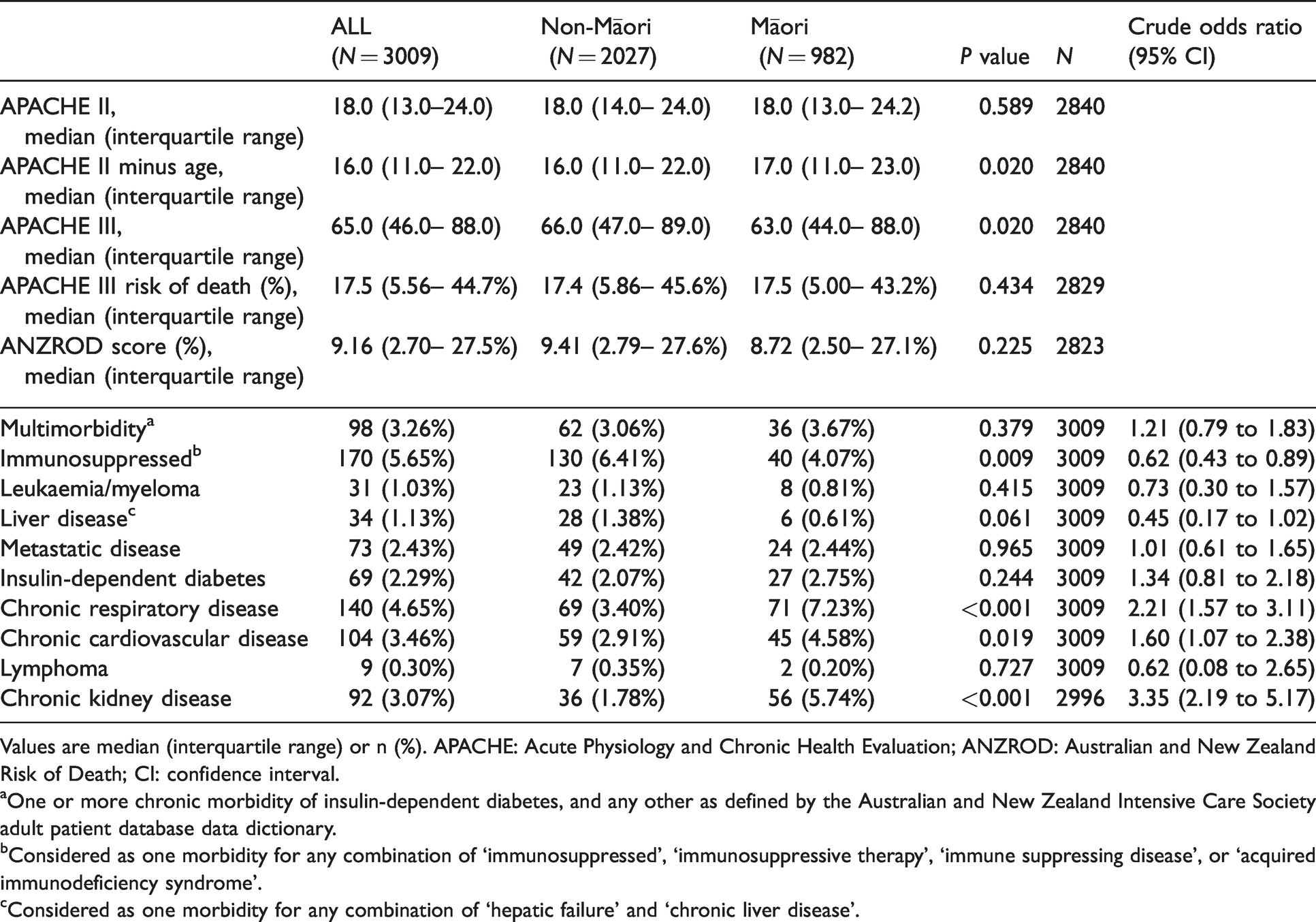
Values are median (interquartile range) or n (%). APACHE: Acute Physiology and Chronic Health Evaluation; ANZROD: Australian and New Zealand Risk of Death; CI: confidence interval.
aOne or more chronic morbidity of insulin-dependent diabetes, and any other as defined by the Australian and New Zealand Intensive Care Society adult patient database data dictionary.
bConsidered as one morbidity for any combination of ‘immunosuppressed’, ‘immunosuppressive therapy’, ‘immune suppressing disease’, or ‘acquired immunodeficiency syndrome’.
cConsidered as one morbidity for any combination of ‘hepatic failure’ and ‘chronic liver disease’.
Admission diagnosis by ethnicity is summarised in Figure 1. Māori had higher rates of admission for APD-defined sepsis and trauma compared to non-Māori (
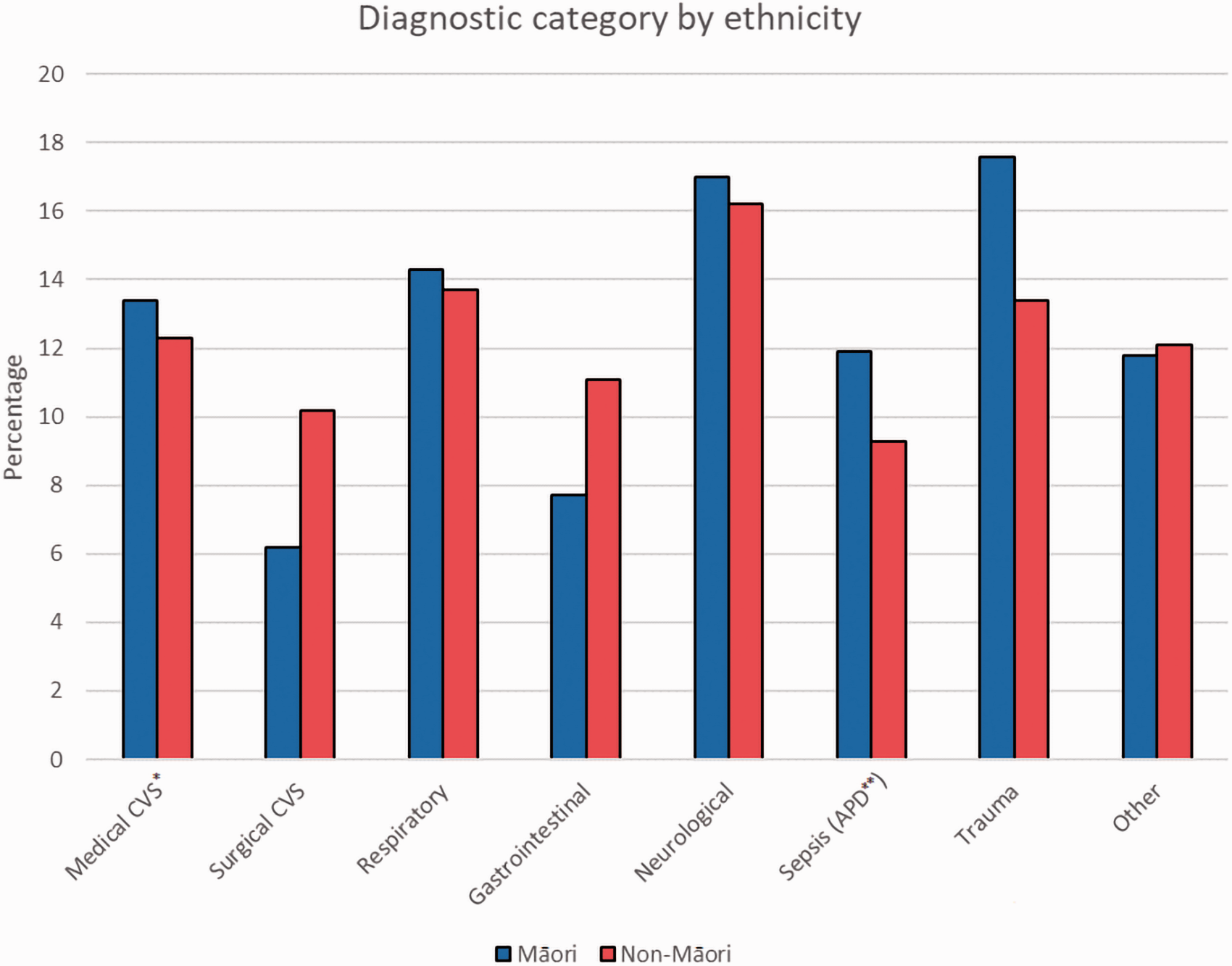
Diagnostic category by ethnicity.
Study outcomes, including ICU and hospital lengths of stay, are summarised in Table 4. Māori were more likely to require RRT at any time in ICU (15.9% versus 11.3%,
Study outcomes.
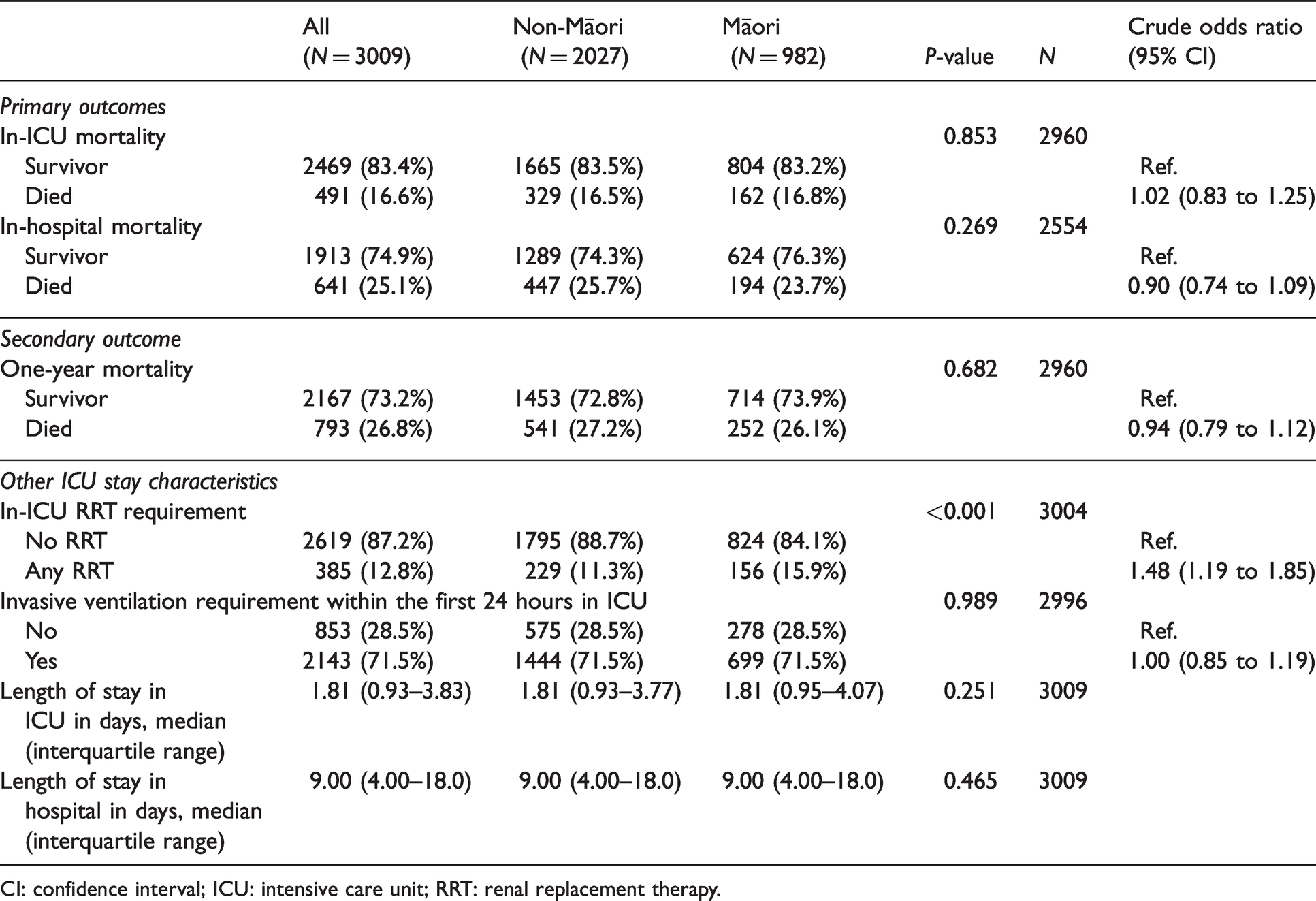
CI: confidence interval; ICU: intensive care unit; RRT: renal replacement therapy.
After adjusting for clinically relevant factors in logistic regression analysis, ethnicity was not predictive of in-ICU or in-hospital mortality (
AOR for all variables for in-ICU and in-hospital mortality are summarised in Table 5 and Supplementary Table 1, respectively. Further adjustment by diagnostic category and ethnicity did not reveal any significant difference in in-ICU mortality (see Supplementary Figure 1).
Adjusted odds ratio for in-ICU mortality.
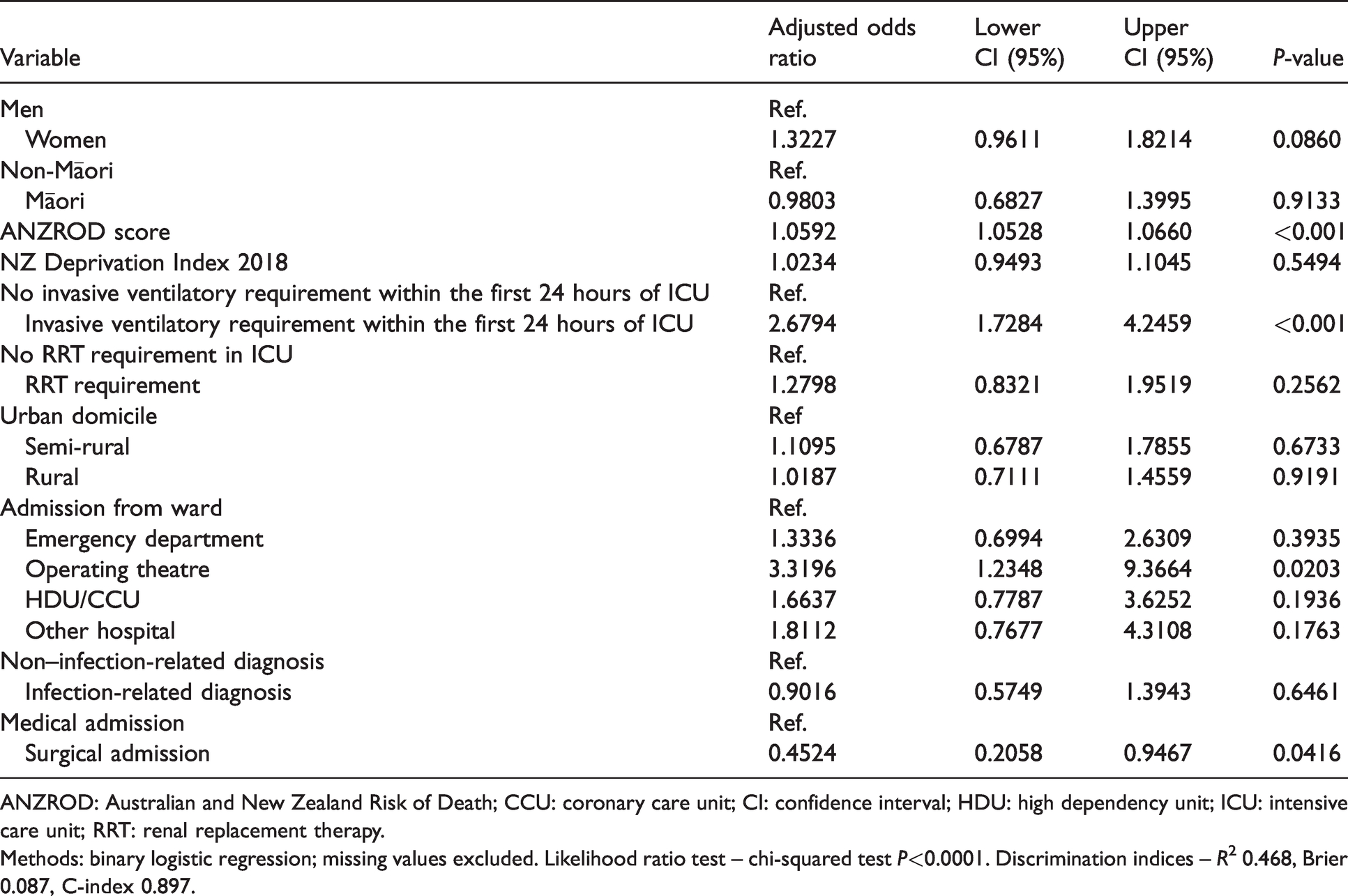
ANZROD: Australian and New Zealand Risk of Death; CCU: coronary care unit; CI: confidence interval; HDU: high dependency unit; ICU: intensive care unit; RRT: renal replacement therapy.
Methods: binary logistic regression; missing values excluded. Likelihood ratio test – chi-squared test
Discussion
The Midland region served by our unit has one of the highest proportions of Maori in NZ at 25.75% of the population, or 21.48% amongst adults aged 16 years and over.15 Māori are over-represented at 33.1% of admissions to our unit, after excluding patients not domiciled in the region.
Critically ill Māori admitted to our unit are younger and come from areas of worse socioeconomic deprivation. They more frequently reside in regions outside of Waikato DHB primarily served by secondary health services with level I or level II ICUs. Our findings suggest that Māori are more likely to require direct transfer to our ICU from other hospitals, and therefore experience social displacement due to an admission for critical illness. This may have important consequences to the ICU experience for Māori patients, such as reduced access to social support due to the distance and financial costs of travel for whānau (family). 27 The overall male predominance in our cohort is consistent with international observations.28
Although the higher rates of chronic respiratory, cardiac and renal disease among Māori in our ICU reflect known community inequities,29–31 there was no significant ethnic difference in the rate of diabetes nor overall multimorbidity in our cohort, which are counter to epidemiological findings.3,32 This discrepancy may be due to the strict qualification of a significant morbidity in the ANZICS APD, as previously described by Secombe and colleagues.19,21 In addition, our unit’s collection of data only for insulin-dependent diabetes probably excluded many patients with non–insulin-dependent diabetes, hence under-reporting.
More admissions among Māori were attributable to infection, a criterion in our audit that spans multiple APD diagnostic categories. Despite not reaching statistical significance, this is consistent with a previous analysis of sepsis in the Waikato region, 13 and in combination with the higher rates of admission with APD-defined sepsis for Māori in our study, probably reflects a real finding. Māori are also over-represented in trauma admissions, consolidating previous findings at other NZ centres.10,11,33 An increased risk of both trauma and sepsis admissions has been described internationally as being associated with socioeconomic disadvantage,34–39 further highlighting the possible downstream consequences of income inequality on the health of Māori in NZ.
Reassuringly, however, crude and adjusted mortality outcomes did not differ between Māori and non-Māori in our audit, with similar in-ICU and in-hospital lengths of stay. Following admission to the ICU, Māori had similar rates of invasive ventilatory requirements, but higher rates of RRT, mirroring known inequities in renal health. 29 Comparable one-year crude mortality rates infer no major inequity in ICU survivorship in our cohort. ANZROD underestimated in-ICU mortality in our cohort, which is 1.8 times predicted. This may reflect factors at systemic, institutional and population levels unique to our unit’s cohort. One disadvantage of our outcome measures is that they do not capture quality of life and return of function, both of which are important dimensions in defining ICU survivorship.40,41 However, these are beyond the scope of our systems performance audit and require further prospective research.
Previous smaller studies have identified inequities for Māori in NZ requiring critical care for specific presentations, including necrotising fasciitis,
42
acute pancreatitis,
14
trauma
11
and influenza
43
among adults, and
Regionally, Secombe et al. 21 recently published a nationwide analysis of ethnic inequity in Australian ICU admissions for Indigenous Australians. They did not find differential mortality for Indigenous Australians. Their overall in-ICU mortality, however, was lower (5.0%) compared to our findings. This is most likely explained by lower baseline illness severity compared to ours (median ANZROD of 1.7% and AP3ROD of 4.6% in their study), as well as their larger case mix, including elective cardiothoracic patients with lower expected and observed mortality.
Critical illness exists on a continuum, influenced by individual and population health in the community.52 Research in critical care should be concerned as much about the outcomes of patients whilst in the ICU, as it is about the prevention of critical illness,52 and the quality of survivorship on discharge from the ICU.41 Disparate health outcomes for NZ Māori and the complex role of social determinants of health are well described in the literature. For example, Māori have lower life expectancy, 53 and more Māori than non-Māori experience socioeconomic deprivation and barriers to accessing healthcare, including culturally appropriate care.5–7,54 Our findings add to the literature by demonstrating how the extent of health inequities for Māori also include the experience of critical illness.
Our audit importantly demonstrated no mortality difference for Māori in the ICU. However, we identified significant inequities for Māori requiring critical care, including over-representation, increased admission rates for trauma and sepsis and increased RRT requirement. The differences in baseline morbidity, regional domicile and socioeconomic deprivation index in our study speak to the pervasive influence that social inequities may have on the risk of critical illness for Māori. Our findings add to the compelling body of evidence for a concerted, multi-pronged approach in addressing inequities in health outcomes for Māori in NZ, and reinforce the need for cooperation and coordination of multiple stakeholders across the continuum of healthcare, from policy-makers to community health personnel to healthcare providers by the ICU bedside.
Supplemental Material
sj-pdf-1-aic-10.1177_0310057X21989715 - Supplemental material for Māori health outcomes in an intensive care unit in Aotearoa New Zealand
Supplemental material, sj-pdf-1-aic-10.1177_0310057X21989715 for Māori health outcomes in an intensive care unit in Aotearoa New Zealand by M Atif Mohd Slim in Anaesthesia and Intensive Care
Supplemental Material
sj-pdf-2-aic-10.1177_0310057X21989715 - Supplemental material for Māori health outcomes in an intensive care unit in Aotearoa New Zealand
Supplemental material, sj-pdf-2-aic-10.1177_0310057X21989715 for Māori health outcomes in an intensive care unit in Aotearoa New Zealand by M Atif Mohd Slim in Anaesthesia and Intensive Care
Footnotes
Acknowledgements
We thank Dianne Takiari, administrator (Intensive Care Unit, Waikato District Health Board), for her assistance with data extraction from the ANZICS Adult Patient Database, and Dr Veronique Gibbons, Clinical Effectiveness Manager (Quality and Patient Safety, Waikato District Health Board), for her advice and comments regarding audit methodology and processes.
Declaration of conflicting interests
The author(s) declare that they have no conflicts of interest.
Funding
The author(s) received no specific funding for this work.
Supplemental Material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
