Abstract
Opioids are a commonly administered analgesic medication in the intensive care unit, primarily to facilitate invasive mechanical ventilation. Consensus guidelines advocate for an opioid-first strategy for the management of acute pain in ventilated patients. As a result, these patients are potentially exposed to high opioid doses for prolonged periods, increasing the risk of adverse effects. Adverse effects relevant to these critically ill patients include delirium, intensive care unit–acquired infections, acute opioid tolerance, iatrogenic withdrawal syndrome, opioid-induced hyperalgesia, persistent opioid use, and chronic post–intensive care unit pain. Consequently, there is a challenge of optimising analgesia while minimising these adverse effects. This narrative review will discuss the characteristics of opioid use in the intensive care unit, outline the potential short-term and long-term adverse effects of opioid therapy in critically ill patients, and outline a multifaceted strategy for opioid minimisation.
Keywords
Introduction
Opioids are one of the most common medications administered to critically ill patients. In the intensive care unit (ICU), nearly 80% of mechanically ventilated patients receive opioids.1,2 The predominant indication for their use is the treatment of acute pain or discomfort. The mechanisms underlying the causes of acute pain (Table 1) are complex and synergistic and include the admission diagnosis to the ICU (e.g. sepsis, trauma, burns, surgery), procedures to facilitate supportive therapy (e.g. the presence of an endotracheal tube, urinary catheter, prolonged immobility), excess medications (e.g. opioids, sedatives) and procedures such as turning for pressure area care, suctioning and vascular access.3,4 Potential psychosocial causes of acute pain include delirium, sleep deprivation, fear, social isolation, depression, anxiety and post-traumatic stress disorder.4,5
Factors inducing or exacerbating acute pain in the intensive care unit.
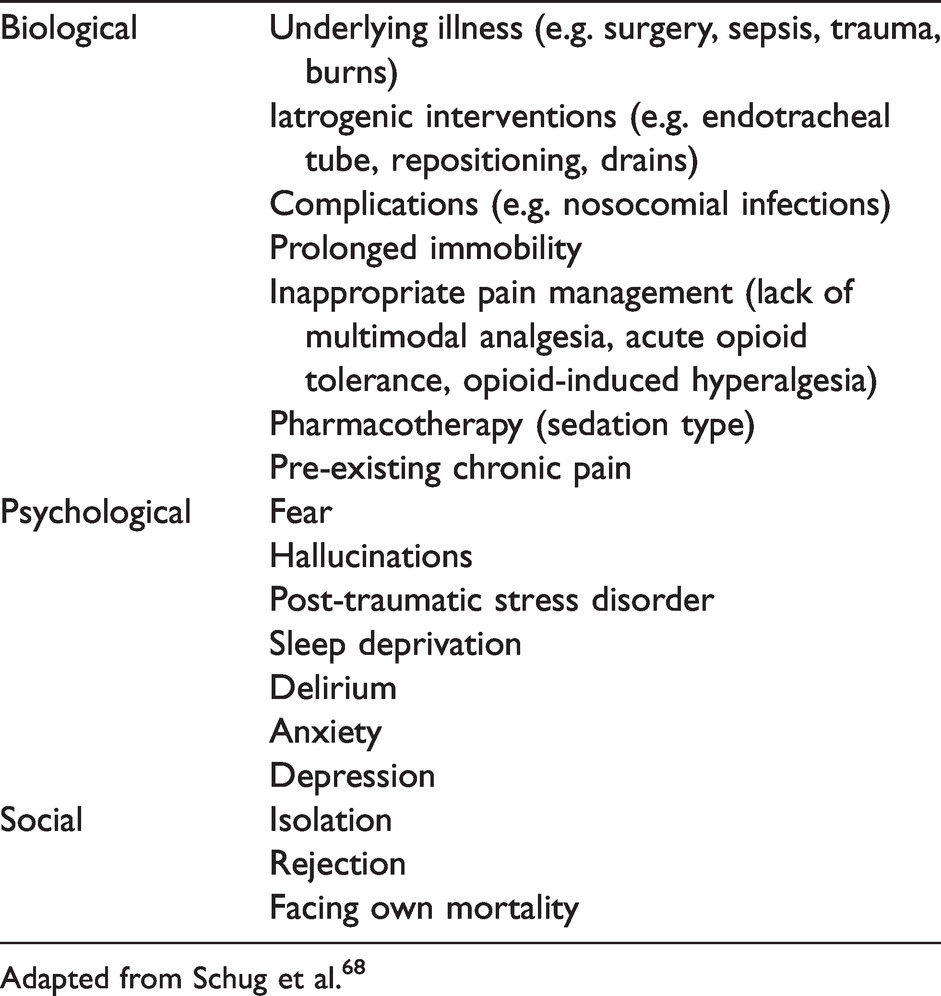
Adapted from Schug et al. 68
Acute pain is common in critically ill patients, 6 with 80% of critically ill patients reporting moderate to severe pain at some point in their ICU admission, and attributing it to a source of long-term psychological morbidity. 7 Approximately half of both medical and surgical ICU patients report moderate to severe pain at rest. 8 Treatment of acute pain within the ICU can lead to improved outcomes and decreased length of ICU stay, while inadequate analgesia can lead to chronic physical and psychological morbidity. 9 The importance of pain management in critically ill patients is highlighted by consensus guidelines, 10 which recommend an analgesia-first analgesic regimen. Opioids are recommended as the first-line analgesic for non-neuropathic pain in ventilated patients.
Despite the clear benefit of opioids in facilitating invasive therapies, prolonged infusions of opioids, as is common in patients receiving mechanical ventilation, can be harmful. Prolonged duration of opioid therapy may lead to short-term adverse effects such as delirium, acute opioid tolerance, opioid-induced hyperalgesia, immunosuppression with an increased risk of ICU-acquired infection, iatrogenic withdrawal syndrome on abrupt cessation, and long-term effects such as persistent opioid use and their potential role in the development of chronic pain in survivors of the ICU.
The objectives of this narrative review are to discuss the characteristics of opioid use in the ICU and outline the potential short-term and long-term adverse effects of opioid therapy in the general, adult critically ill population. In addition, we provide an opioid minimisation framework to assist intensive care clinicians in the management of opioids and pain in critically ill patients. Discussions relating to the pain management of the patient receiving chronic opioid therapy and opioid use in ICU subspecialties (e.g. burns, spinal cord injury) are outside the scope of this review and are reviewed elsewhere.11 –13
Characteristics of opioid use in the intensive care unit
Type of opioid
Over the past two decades, there has been a shift in the predominant opioid used for analgesia in ventilated ICU patients. Initially, morphine was favoured over synthetic opioids such as fentanyl, sufentanil, alfentanil and remifentanil.14 –16 Over time, however, fentanyl has become more popular than the other opioids.17 –22 The reasons for this have not been extensively investigated, but perceived favourable pharmacokinetics, lack of accumulation of active metabolites in end-organ dysfunction, and reduced incidence of adverse events such as nausea and vomiting, urinary retention and constipation 19 are likely to play a role in prescription decision-making.
There also exists regional variation in the type of opioid prescribed. The predominant opioids used in ICUs in Australia and New Zealand for analgesia are morphine and fentanyl, 23 with ventilated patients receiving fentanyl (80%), morphine (30%) and alfentanil (7.6%). 21 In France, 40% of patients received sufentanil, 25% received fentanyl and 20% received morphine. 17 In a survey of European ICUs, morphine was preferred over fentanyl and sufentanil in Norway, the UK, Ireland, Sweden, Switzerland, the Netherlands, Spain and Portugal. In contrast, fentanyl was preferred in France, Germany and Italy.14,24 Although this survey was 20 years ago and preferences may have changed, it highlights the regional variation in practice.
There is a lack of comparative effectiveness trials comparing opioids used for analgesia in the ICU. Casamento et al. 25 compared infusions of fentanyl to morphine in ventilated patients and found that patients receiving fentanyl had more ventilator-free days and a shorter ICU length of stay. A major limitation of this study is that opioid infusions were titrated using local protocols targeting light sedation. The targeting of sedation, rather than pain, make extrapolation of opioid type for analgesia difficult as higher doses may be required to achieve sedation.
The remaining studies compared remifentanil to fentanyl,26,27 morphine27,28 and sufentanil. 29 Compared with morphine and sufentanil, remifentanil resulted in a decreased duration of mechanical ventilation and more time in range of a target sedation score. Compared to morphine and fentanyl in patients admitted to the ICU with acute neurological injuries, remifentanil use resulted in a shorter time to neurological assessment. In addition, patients receiving remifentanil had a decreased time to extubation compared to those receiving morphine, but not fentanyl. 27 In a study comparing remifentanil to fentanyl, there was no difference in the time to liberation from mechanical ventilation. For patients who experienced pain, those treated with remifentanil had a longer duration of pain during extubation, post-extubation and post-treatment of pain. 26 An explanation of this observed effect is the increased risk of opioid-induced hyperalgesia (OIH) and rapid opioid tolerance seen with remifentanil. 30 In an otherwise pharmacokinetically ideal opioid, the potential for tolerance and OIH has potentially limited its widespread use.
It is unsurprising that there is a lack of evidence informing opioid use in ventilated patients. There is a large variation in practice, determined by differing clinical objectives, patient characteristics such as admission diagnosis and end-organ dysfunction, and an increasing choice of available opioids. Added to this is the misconception of ‘analgosedation’, in which opioids are used primarily as sedatives. Given opioids are analgesics, sedation in an opioid should be viewed as an adverse effect, not a primary effect. Also, the required opioid dose to achieve sedation would be likely to be higher than that required as an analgesic. In addition, there may be a misunderstanding of the appropriate opioid conversion factors, resulting in relative underdosing which may make comparison of opioids difficult.
Dose of opioid
One of the potential outcomes of an opioid-first approach to analgesia in ventilated patients is an increased cumulative dose of opioids. It is evident that small hourly doses of continuous infusion opioid may result in large daily cumulative opioid doses. In studies performed in France by Payen and colleagues,17,22,31 the fentanyl doses titrated against validated pain scores were 40–50 μg/kg/day (560–700 mg/day oral morphine equivalent daily dose (oMEDD) in a 70 kg person). Cumulative doses of morphine, however, were not of this magnitude, with a mean dose of 0.4–0.5 mg/kg/day (84–105 mg oMEDD in a 70 kg person). This may be due to the lower analgesic efficacy of fentanyl compared to morphine, or the accumulation of active metabolites of morphine. This high cumulative fentanyl dose was observed in other studies.21,32,33 More recently, however, the daily dose of fentanyl in ventilated patients receiving an opioid was 148.5 mg oMEDD. 34 Further studies are needed to determine whether lower opioid doses are more prevalent since the release of recent guidelines 10 advocating the lowest possible dose of opioid to achieve adequate analgesia.
There are conflicting results in studies investigating opioid doses over time. In a study comparing morphine and fentanyl in ventilated patients, the mean daily dose of both fentanyl and morphine remained constant over a period of seven days. 25 It should be noted that the opioid was titrated against a target of light sedation, so extrapolating these results to analgesic endpoints should be undertaken cautiously. However, this may reflect what is occurring at the bedside. This constant dose of opioid administration was also seen in a study in which the cumulative daily fentanyl dose was 500 mg oMEDD on day two and 600 mg oMEDD on day ten. 32 However, Burry et al. 18 found an increase in daily opioid doses over time in ventilated patients, with 23 mg oMEDD (<48 hours), 63 mg oMEDD (two to seven days) and 106 mg oMEDD (>seven days). The predominant opioid in that study was morphine.
As previously mentioned, the comparison of opioids and potential misconceptions about opioid conversion factors in the setting of end-organ dysfunction make inference from these studies on opioid dosage difficult. Nevertheless, the above studies are concerning for the following reasons. First, these high opioid doses are administered for a prolonged period to a patient cohort, in which at least 80% of ventilated patients are opioid naive. 1 Second, there appears to be a disconnect between opioid dose and pain, particularly over time. Longitudinal analysis of acute pain in general ICU patients shows a decrease in pain at rest but a relatively constant proportion of pain while turning. 35 Hence, it is concerning that although pain is decreasing over time, opioid doses are remaining constant or increasing, essentially resulting in a relative opioid overdose. Given that opioids are frequently given with sedation, it is plausible that the titration of opioids does not occur for analgesia alone, but also for sedation, essentially increasing the dose. It was not noted in the above studies whether opioid infusions were titrated to pain scores. The combination of inappropriately high opioid dosing, for prolonged periods in opioid-naive patients renders the critically ill patient at an increased risk of both short- and long-term adverse effects of opioids.
Short-term adverse effects of opioids in the intensive care unit
The potential short-term adverse effects of opioid therapy in the ICU include delirium, acute opioid tolerance and withdrawal, opioid-induced hyperalgesia, immunosuppression, gastrointestinal disturbance, and psychological or cognitive impairment. While these side-effects are identical to opioid therapy outside of the ICU, the effects may be exaggerated due to exposure to a higher opioid dose for prolonged periods, end-organ dysfunction, and concomitant critical illness.
Delirium
Delirium, or acute brain dysfunction, is a common ICU complication that affects 50%–75% of ICU patients. Pain and opioid use are two potential causal factors for the development of delirium.36 –39 Given the pathophysiology of ICU delirium is complex, it is unsurprising that there is no clear role of opioids in its development. While numerous studies confer a positive association between opioid use in the ICU and ICU delirium,36,37,39 –41 several other studies have found no association.38,42,43 However, it is plausible that both opioid dose and type may play a role in the development of delirium in the ICU.
Early studies looking at potential causal factors for developing ICU delirium identified a non-linear relationship between opioid dose and delirium. Dubois et al. 41 found an inverse U-shaped relationship between daily opioid dose and the risk of developing delirium. The dose range of daily opioid exposure that was associated with the highest risk of developing delirium (odds ratio (OR) 9.2) was 21.6–55.8 mg oMEDD, with a decreasing risk of delirium on either side of that range. Pain is a potential causal factor for developing delirium, and appropriate analgesia in the lower daily dose range is possibly an explanation for a decreased incidence of delirium in those patients. In addition, a higher opioid dose is likely to be used in patients who have a high acuity of illness, which increases the likelihood of developing delirium. Also, a higher dose of opioid is likely to be used in patients who are more deeply sedated, which may mask the diagnosis of delirium.42 This reflects a non-causative role of opioid use and delirium, in which sedatives, and the co-administration of opioids, are being used to manage delirium. It should be noted that that study did not differentiate between opioid and benzodiazepine use, which is an additional potential causal factor for developing ICU delirium.40,41 Another possibility is the differential effect of sedatives on pain perception. In a study by Frölich et al., 44 the use of midazolam for procedural sedation resulted in higher pain perception compared with propofol and dexmedetomidine. It is plausible that this may translate to the ICU, where an increase in pain perception may induce agitation and delirium.
It is feasible that different opioids result in a different risk of developing delirium. In surgical and trauma patients, morphine had a lower risk of delirium in both groups. 37 Multivariable analysis adjusting for sedative medications, but not pain scores, showed that in ventilated surgical ICU patients, the use of fentanyl was associated with a four times increased risk of developing delirium. This was in contrast to morphine, which had a 60% decreased risk. This morphine effect was also seen in trauma patients, with a nearly 80% reduced risk of developing ICU delirium. In that study, patients receiving morphine had a lower proportion of time with delirium. The observed effect may be due to the analgesic efficacy of morphine or its metabolites compared to fentanyl, particularly in a group that may experience increased neuropathic or somatic pain compared to general ICU patients.
One other potential factor in explaining the inconsistent reported effects of opioids on delirium is the adjustment of pain as a potential confounder. Ventilated patients are usually receiving an opioid, which is a potential risk factor for delirium. The presence of the endotracheal tube is known to cause moderate to severe pain for the ICU patient,7,45,46 and pain is a potential causal factor of delirium. 42 Hence, acute pain is a confounder in the causal pathway from opioid exposure to delirium.
When acute pain, defined as an average pain score or worst pain score, was adjusted for, opioid exposure was associated with 2.5 times the risk of developing delirium in an elderly ICU cohort. 39 Interestingly, neither average pain score nor worst pain score was associated with the development of delirium. Duprey et al. 47 determined the transitional probability of developing next-day delirium, adjusting for severe pain, in ventilated ICU patients. The administration of opioids was associated with an increased risk of developing delirium the next day (OR 1.45, 95% confidence interval (CI) 1.24 to 1.69), with an increase in the risk of 2.4% for each daily 30 mg oMEDD administered. In addition, the risk of developing delirium was greater for synthetic opioids such as fentanyl and remifentanil (OR 1.77) compared to morphine (OR 1.09). Interestingly, in that study severe pain was inversely associated with transitioning to delirium (OR 0.72, 95% CI 0.53 to 0.97), when adjusting for opioid use. This may be explained by the definition of severe acute pain in which pain only needed to have occurred once in the previous 24 hours, when the administration of appropriate opioid analgesia preventing delirium gave the fallacy of pain being protective.
One major limitation of studies investigating the role of opioids in the development of delirium is the concept of time-varying exposure and confounding. As can be seen in Figure 1, the causal pathway from opioid exposure at time (t0) to delirium is complex. Pain at t0 is a confounder for opioid exposure and delirium. There is potential for the opioid exposure to exacerbate acute pain at a point in the future (pain t1) through opioid-induced hyperalgesia or latent sensitisation. Pain at t1 is a confounder for opioid exposure at t1 and delirium, and is also potentially causal for future pain at tn. Hence, acute pain is acting as both a confounder and a mediator in the causal pathway of opioid exposure to delirium. It is evident that the use of a single cumulative variable for ICU admission (e.g. one episode of severe pain or total opioid dose) can result in biased estimates of effect. In addition, opioid exposure (i.e. opioid dose) may also change over time and this needs to be accounted for in the analysis.
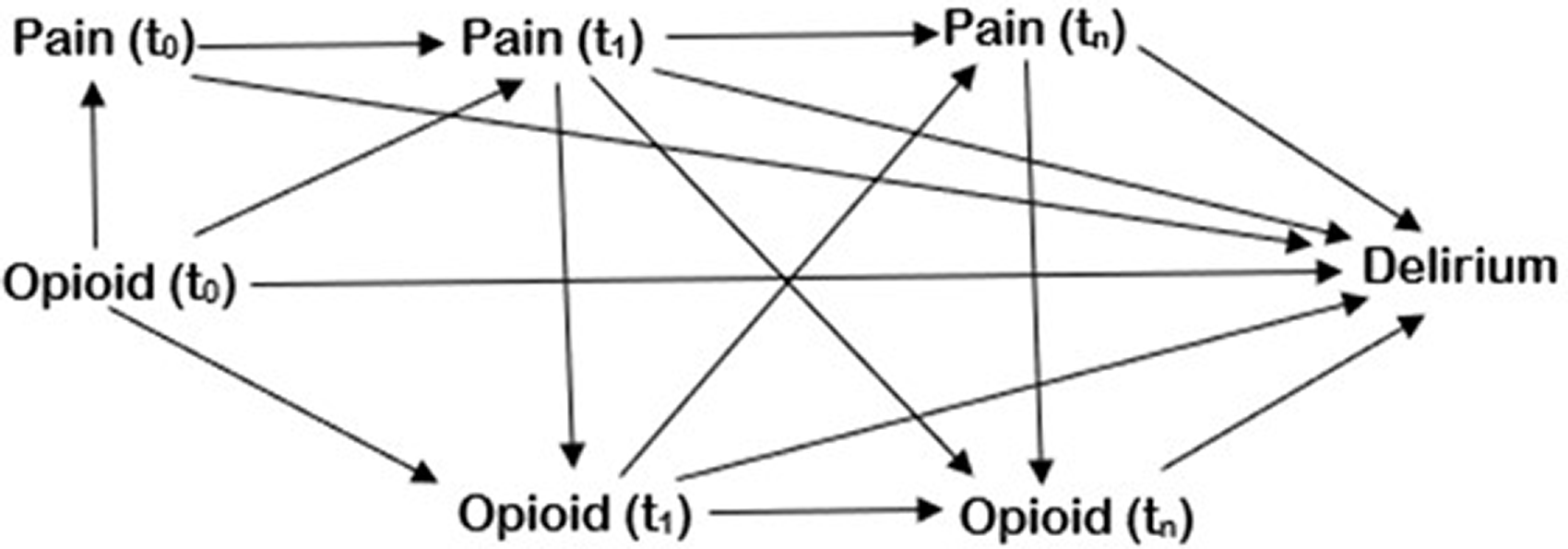
Causal diagram of opioid dose and the development of delirium. The directed acyclic graph (DAG) details the causal pathway from the time-varying exposure (opioid) to the outcome (delirium), with pain as a time-varying confounder. t0: time at baseline; t1: time at timepoint 1; tn: time at timepoint n.
The causal pathways from opioid exposure to the development of delirium are complex, reflecting the multitude of causes of ICU delirium and the time-varying nature of both exposures and confounders. Opioids most likely play a role in the development of delirium, but the specifics of opioid type, opioid dose, confounding by pain, sedative medications and admission diagnosis need to be evaluated further. Further studies should focus on time-varying exposure and confounding, utilising accepted causal inference methodology to ensure unbiased effect estimates and appropriate inferences can be made about the role of opioids in the development of delirium.
Intensive care unit-acquired infection
Infection is a common occurrence in patients in the ICU. The European Prevalence of Infection in Intensive Care (EPIC) III point prevalence study reported 54% of patients had a suspected or confirmed infection, including 22% with ICU-acquired infection. 48 The most common site of infection was the respiratory tract (61%), followed by abdominal (16%) and bloodstream (15%). 48 Patients who develop a nosocomial infection in the ICU are associated with the development of multi-organ dysfunction, have a longer ICU and hospital length of stay, and an increased risk of in-hospital mortality.48 –51
There are several mechanisms by which opioids may increase the risk of developing an ICU-acquired infection. These include immunosuppression, increased sedation resulting in increased duration of mechanical ventilation and microaspiration, and gastrointestinal motility dysfunction.
Immunosuppression
There is a substantial body of literature detailing the effects of opioids on both the innate and adaptive immune system.49,50,52 –55 Opioids may affect immune function with acute exposure, chronic exposure or during acute opioid withdrawal.
Acute exposure to morphine suppresses mitogen-stimulated proliferation of T- and B-cell lymphocytes, natural killer (NK) cell cytotoxic activity, primary antibody production, macrocyte phagocytosis, and interleukin-2, interferon-γ (IFN-γ), tumour necrosis factor α and nitric oxide production. 50 Many of these effects are inhibited by naloxone, suggesting that the effects are mediated primarily through mu-opioid receptors. Mu-opioid receptors are present on leukocytes, which is suggestive of a direct effect on immune function.50,53 Indirect effects may occur centrally, mediated through central opioid receptors, which activate the sympathetic nervous system and the hypothalamic–pituitary–adrenal axis. This activation increases the production of catecholamines and neuropeptides from sympathetic nerves and glucocorticoids from the adrenal glands, resulting in immunomodulation.50,53
The cellular effects of chronic exposure to opioids on immune function are conflicting in animal studies, and are prone to potential confounding in human studies. It is currently unknown whether chronic opioid use results in a return to baseline in immune function, or whether the acute effects continue. Animal models of chronic morphine administration demonstrated altered lipopolysaccharide-induced inflammation, with rapid progression to septic shock and increased HIV viral replication.50,53 However, there is a possibility of immune tolerance in the presence of chronic morphine use, in which lymphocyte proliferation, suppression of NK cell activity and hypothalamic–pituitary–adrenal axis activation return to baseline after as early as 96 hours after morphine exposure, 50 although this is not a consistent finding.50,53 Human studies investigating the effects on infections in patients with opioid addiction are confounded by the route of administration (e.g. intravenous drug use, which can predispose to soft tissue, vascular and cardiac infections), and effects of the opioid (e.g. due to sedation, increasing the risk of respiratory tract and musculoskeletal infections). 53 Hence, in an uncontrolled clinical environment, it is difficult to ascertain the cellular mechanisms of immunosuppression in humans who are chronically exposed to opioids. It is also unclear if acute tolerance of opioids in ventilated patients results in the same cellular mechanisms seen in chronic opioid exposure.
Acute opioid withdrawal has been shown in animal models to result in immunosuppression, through increased levels of corticosterone, suppression of lymphocytes and T-cell proliferation, and decreased IFN-γ production and NK cell activation. These changes were seen up to 72 hours after the abrupt withdrawal of chronically administered morphine, before returning to baseline levels. 50 Given that clonidine ameliorated some of these effects, it is hypothesised that a potential pathway for withdrawal-mediated immunosuppression includes activation of the sympathetic nervous system and the hypothalamic–pituitary–adrenal axis. Attempts to study opioid withdrawal on ICU-acquired infections are plagued by diagnostic uncertainty of opioid withdrawal syndrome and methodological limitations. In a retrospective cohort study in mechanically ventilated patients, remifentanil discontinuation was associated with a 2.5 times increased risk of ICU-acquired infection, after adjustment for mechanical ventilation and Simplified Acute Physiology Score. 54 Although the inference of causality is tenuous at best, it is worthy of consideration as opioid withdrawal may be a more common potential causal factor for the development of ICU-acquired infection than previously realised.
Given that the ventilated ICU patient may receive opioids acutely or chronically and opioid withdrawal may occur, the effects of opioids on immunosuppression may predispose the patient to an increased risk of nosocomial infection.
Prolonged duration of ventilation
The addition of opioids to sedative medications can result in an increase in the level of sedation and a prolonged duration of ventilation. 55 The level of sedation is a potential causal factor for the development of ventilator-associated pneumonia (VAP). One potential explanation for this relationship is microaspiration.49,50,55 Oral pooling of secretions may be exaggerated by a reduction in oesophageal propulsion in the presence of sedation.50,55 Contaminated oropharyngeal secretions may enter the lower respiratory tract through leakage around the endotracheal tube cuff, allowing bacterial seeding into the lower respiratory tract. The increased duration of mechanical ventilation prolongs the exposure to these potential causal factors for developing nosocomial infections (particularly VAP). However, the precise role of opioid dose or type resulting in prolonging ventilation and the effect on nosocomial infection requires further evaluation.
Gastrointestinal dysfunction
Gastrointestinal (GI) dysfunction is common in critically ill patients and the effects of opioids on the GI system are well documented. Mechanisms by which opioids can cause GI dysfunction, resulting in an increased risk of ICU-acquired infection, include alteration of the structural barrier and functional gut microbiome (dysbiosis) and GI motility dysfunction. 56 The gut microbiome is an integral component of the GI barrier to infection. Administration of opioids can reduce microbial diversity, resulting in an increase in the proportion of potential pathogenic bacteria, such as Pseudomonas aeruginosa, Enterococcus faecalis, Listeria monocytogenes, Citrobacter rodentium, Clostridium difficile, Streptococcaceae and Staphylococcaceae.56 –58 In animal models, the effects of opioid-induced GI dysbiosis occur in a receptor-dependent manner, which can be inhibited by opioid antagonism; 56 however, this is yet to be investigated in humans. In addition, opioids impair gut epithelial integrity through disruption of the intestinal barrier function and tight junction damage in mice, increasing the risk of bacterial translocation and systemic infection. 58
Opioids affect GI motility by the inhibition of intestinal neural excitability. The resultant faecal stasis can lead to microbiological imbalance and GI dysbiosis, resulting in a reduced diversity and dominance of harmful bacteria, a reduction in endogenous anaerobic and Gram-positive flora and an increase in endotoxin load. Translocation of bacteria and endotoxins can lead to infections and enhanced systemic inflammation.50,55 Again, the precise role of opioids in this is yet to be delineated.
Overall, there is potential for opioids to increase the risk of ICU-acquired infections, through the mechanisms of structural and functional dysbiosis, immunosuppression, prolonged ventilation, poor clearance of secretions and GI motility dysfunction. Despite this, the potential causal effect of opioid exposure on the development of ICU-acquired infection warrants further evaluation.
Acute opioid tolerance and opioid-induced iatrogenic withdrawal syndrome
Given the large opioid dose for prolonged periods, ventilated patients are at an increased risk of acute opioid tolerance and iatrogenic withdrawal. Acute opioid tolerance results from the desensitisation of antinociceptive pathways to opioids. This results in an increased requirement of opioid to achieve equianalgesia, with escalating doses of opioids increasing the likelihood of adverse effects.
Once acute tolerance occurs, abrupt cessation of opioid infusions can result in a withdrawal syndrome. The incidence of iatrogenic withdrawal syndrome in ventilated trauma and surgical patients was 32% 59 and 44%, respectively,, 60 although these studies were confounded by concomitant benzodiazepine use. This issue was addressed by Hyun et al., 61 who found that in ventilated patients with low or no benzodiazepine sedation, the incidence of opioid-induced withdrawal syndrome was 29.4%. The incidence of withdrawal was higher in patients receiving remifentanil (31%) and fentanyl (36%) compared to morphine (9.7%). Patients receiving remifentanil and fentanyl had a higher opioid dose compared to morphine, with a shorter duration of opioid therapy, indicating more rapid tolerance. In a prospective study by Wang et al., withdrawal symptoms were recorded in 16.7% of patients, suggesting opioid weaning prior to discontinuation may reduce the incidence of opioid-induced withdrawal syndrome.62 The authors reported a possible association between higher median opioid dose and longer duration of opioid therapy with the development of a withdrawal syndrome; however, they noted that their conclusions were likely limited by their small sample size.
Difficulties exist in the ICU population in determining the incidence of opioid-induced iatrogenic withdrawal syndrome. First, there is no validated withdrawal assessment tool for non-communicative patients. The withdrawal assessment tool 1 (WAT-1), an assessment tool that is validated in the paediatric ICU population, was evaluated in adult ICU patients. There was poor sensitivity (50%) and specificity (65.9%), with poor agreement between WAT-1 and the Diagnostic and Statistical Manual of Mental Disorders (DSM-V) (
Opioid-induced hyperalgesia
OIH is defined as an increase in sensitivity to noxious stimuli resulting in increased pain due to opioid use.
64
Although it is possible to acquire OIH with the use of any opioid, it is more prevalent after the use of phenylpiperidine derivatives, such as remifentanil, and to a lesser extent, fentanyl.26,30,64
–67 OIH is clinically similar to acute opioid tolerance resulting in the decreased analgesic efficacy of opioids, although the mechanisms differ. Although not completely understood, OIH occurs with sensitisation of pronociceptive pathways, leading to hyperalgesia. This differs from the mechanism for acute opioid tolerance. Both pathways are thought to involve central N-methyl-
Given the unique situation in ICU patients, the diagnosis of opioid-induced hyperalgesia may be problematic. In non-ICU patients, one diagnostic tool is the quantitative sensory test, which determines pain thresholds to differing opioids. This may help with differentiation between acute opioid tolerance and OIH. It is self-evident that it would be difficult to conduct such tests in sedated of mechanically ventilated patients. Other issues that may confound OIH diagnosis include opioid-resistant pain, multiple potential sources of acute pain (see Table 1) and psychological factors such as delirium and pain catastrophising. 68 On a practical note, if the patient has OIH, a reduction in opioid may reduce pain severity. If the patient has acute opioid tolerance, an increase in opioid may help. In these settings, alternative reasons for increased analgesic requirement should be sought. These include neuropathic pain, multi-site pain, psychological distress, and patients who receive chronic opioid therapy.
It may be difficult clinically to distinguish poor analgesia due to acute opioid tolerance from OIH. Increasing or decreasing the dose of opioid in a ventilated patient may not elicit any meaningful insight into the problem. Clinical management of these patients with non-opioid analgesia may attenuate these phenomena. One of the more useful interventions is NMDA antagonism with ketamine, although the risk of psychomimetic side-effects such as delirium needs to weighed up against the analgesic benefits. 68 In addition, the gabapentinoids and alpha-2 agonists may attenuate wound hyperalgesia in the postoperative setting 64 and may play a role in the ICU cohort.
Long-term adverse effects of opioids in the intensive care unit
The potential long-term adverse effects of opioid use during ICU admission include persistent opioid use and chronic post–intensive care unit pain (CPIP).
Persistent opioid use after the intensive care unit
Several studies of varying ICU cohorts have analysed the incidence and potential causative factors associated with persistent opioid use after the ICU.1,34,69 –79 Overall, the incidence of persistent opioid use after the ICU ranges from 2.6% to 70%. This observed variability is due to the inclusion of patients with pre-existing opioid use, variable time to follow-up, differing definitions of chronic opioid therapy and the patient cohort being studied.
The incidence of chronic post-ICU opioid use ranges from 2.6% to 16.3% in patients who were opioid-naive prior to ICU admission.1,34,70 –72,74 –79 Trials with a larger incidence of persistent opioid use used a definition of single prescriptions of opioids. Using a time-based prescription of 120 days of opioid supply within the first year after ICU hospitalisation, Wunsch et al. reported the incidence of chronic opioid therapy in ICU patients receiving mechanical ventilation to be 2.6%, 1 with prescriptions occurring more in surgical patients than medical patients (4.1% versus 1.6% at 12 months). This was similar to a study of postoperative ICU admission, in which the incidence was 4.1%. 71
In patients identified as chronic opioid users, admission to the ICU has a varied effect on opioid prescriptions after discharge. Yaffe et al. 80 describe a 44% relative reduction in opioid prescriptions in chronic opioid users in the first 12 months after ICU discharge, with a 71% relative reduction over four years. Wang et al. 69 found that 63% of elderly chronic opioid users had at least one opioid prescription six months after ICU discharge, with approximately one-quarter having a higher opioid dose. Dunn et al. 70 also found a high incidence of opioid prescription, with 61% and 44% of traumatic brain-injured patients still receiving an opioid prescription at six and 12 months, respectively. Wampole et al. 78 found that 70% of chronic opioid users had one opioid prescription filled at three months post-discharge, with similar proportions at six and 12 months. Again, these results should be interpreted with caution as the outcomes are based on single prescriptions of opioids.
These data suggest that there is a decrease in opioid use over time after ICU discharge for both patients who are opioid naive and those receiving chronic opioid therapy.70,74,80 This reduction may be a result of improved follow-up services, in which chronic opioid therapy may be weaned or discontinued. However, follow-up studies are notorious for loss to follow-up. Given the mortality rate of ICU survivors is approximately 15%–20% at 12 months,81 –83 this may contain a large proportion of patients who are on chronic opioid therapy, who are at an increased risk of 12-month mortality. 84 Von Oelreich et al. 79 found that approximately 21% of patients who died within six to 18 months after ICU discharge were receiving chronic opioid therapy. In addition, the use of non-prescription, illicit opioids in survivors of the ICU is currently unknown.
The relationship between ICU opioid exposure and persistent opioid use is not well studied. The few studies that have investigated this all used a definition of a single prescription, rather than a time-based definition of chronic opioid therapy.34,70,78 As there is no consensus definition of chronic opioid therapy, it is a difficult endpoint to study. In addition, the exposure of opioid dose was a cumulative dose for the entire ICU admission, which ignores the varying nature of opioid exposure and confounders.
Using a stringent definition of chronic opioid therapy, it is remarkable that less than 5% of ICU patients are receiving chronic opioid therapy at 12 months, especially given the exposure to high-dose, prolonged opioid by infusion. Standardised outcome definitions and methodological considerations will enable a more robust estimation of the causal effect of such opioid exposure and chronic opioid use in the ICU cohort.
Chronic post–intensive care unit pain
Improvements in the management of critically ill patients have resulted in improved survival, albeit with an increased risk of physical, cognitive and psychiatric disability, collectively known as post–intensive care unit syndrome (PICS). 85 One component of PICS is the development of CPIP. Currently, there is no consensus definition of CPIP, but the revised definition of chronic primary pain is ‘Pain in one or more anatomical regions that persists or recurs for longer than 3 months and is associated with significant emotional distress or functional disability and that cannot be better accounted for by another chronic pain condition’. 86
The precise prevalence of CPIP is difficult to establish, ranging from 22% to 94.1%.87 –91 Such diverse estimates are explained by variation in study cohorts, study locations, definitions of CPIP, time frames of evaluation, study designs, and analytical methods. The majority of long-term outcome studies use the EuroQOL-5D (EQ-5D), with the incidence of body pain present in 40%–50% of ICU survivors.92 –108 While the pain domain of the EQ-5D does not meet the definition of chronic pain, it is reasonable to suspect that the incidence of CPIP is high. It is also interesting to note that the incidence of chronic opioid use after ICU discharge is considerably lower than the incidence of CPIP. It is suggested that one possible reason for this is a reduction in the use of chronic opioid therapy in the treatment of chronic non-cancer pain. 80 In comparison, it should be noted that patients with chronic post-surgical pain have a prevalence of chronic opioid use of 4%–11%. 80 It is also interesting to note that the incidence of chronic opioid use after the ICU is considerably lower than the incidence of CPIP. The authors suggested that one possible reason for this is a reduction in the use of opioid therapy in the treatment of chronic non-cancer pain, which they suggest is supported by opioid prescriptions for chronic opioid use for pain accounting for approximately 30% of all opioid prescriptions in the community.80 Hence, it is not unexpected that the use of chronic opioid therapy in ICU survivors is lower than those with CPIP.
It remains difficult to determine whether opioid use within the ICU causes CPIP. Although there is a biological plausibility that high-dose, continuous opioid infusions could potentially sensitise to acute pain, resulting in a transition to chronic pain, it would be difficult to establish the contribution of opioids and acute pain to this. Animal studies 109 have shown that opioid administration can result in an enhanced hyperalgesic state, in which the balance of pronociceptive and antinociceptive systems favours pronociception and results in a state of latent pain sensitisation. A human volunteer study showed varied development of latent pain sensitisation from acute opioid exposure; however, the pathophysiology of opioid-induced chronic pain is worthy of further investigation.
The possibility of opioid exposure in the ICU resulting in CPIP was addressed in a study by Hayhurst et al. 89 In survivors of the ICU, the cumulative total fentanyl dose was associated with a 2.5 times risk (OR 2.58, 95% CI 1.26 to 5.29) of chronic pain at 12 months post–ICU discharge, although, debatably, the authors considered this to be non-significant after applying a multiplicity adjustment. It is unknown if this response is contingent on fentanyl, or if other opioids have a similar response. Pain was not measured to enable adjustment in the regression analysis; hence it is unknown whether the cumulative opioid dose was sub- or supratherapeutic in order to hypothesise the pathophysiological pathway in the development of CPIP.
Given the biological plausibility of opioid-induced chronic pain and the exposure of critically ill patients to high doses of opioids for prolonged periods, it is important to consider the potential role of opioids in the development of CPIP. Similar to the effect of opioids on ICU delirium, a study that adjusts for acute pain will be required to show a potential causal or contributory effect of opioids on CPIP.
Strategies to minimise opioid use in the intensive care unit
The need to manage acute pain is often offset with the potential adverse effects of opioid therapy, with both situations leading to increased morbidity. With the increasing use of opioids as first-line analgesic management it can prove difficult to achieve opioid minimisation, while ensuring adequate analgesia. Unfortunately, there is limited evidence to guide such a practice. Potential strategies to aid in opioid minimisation in the ICU should be targeted at the biological, psychological and social determinants of pain (see Table 2). Some of these strategies are discussed.
Opioid minimisation strategies in the intensive care unit.
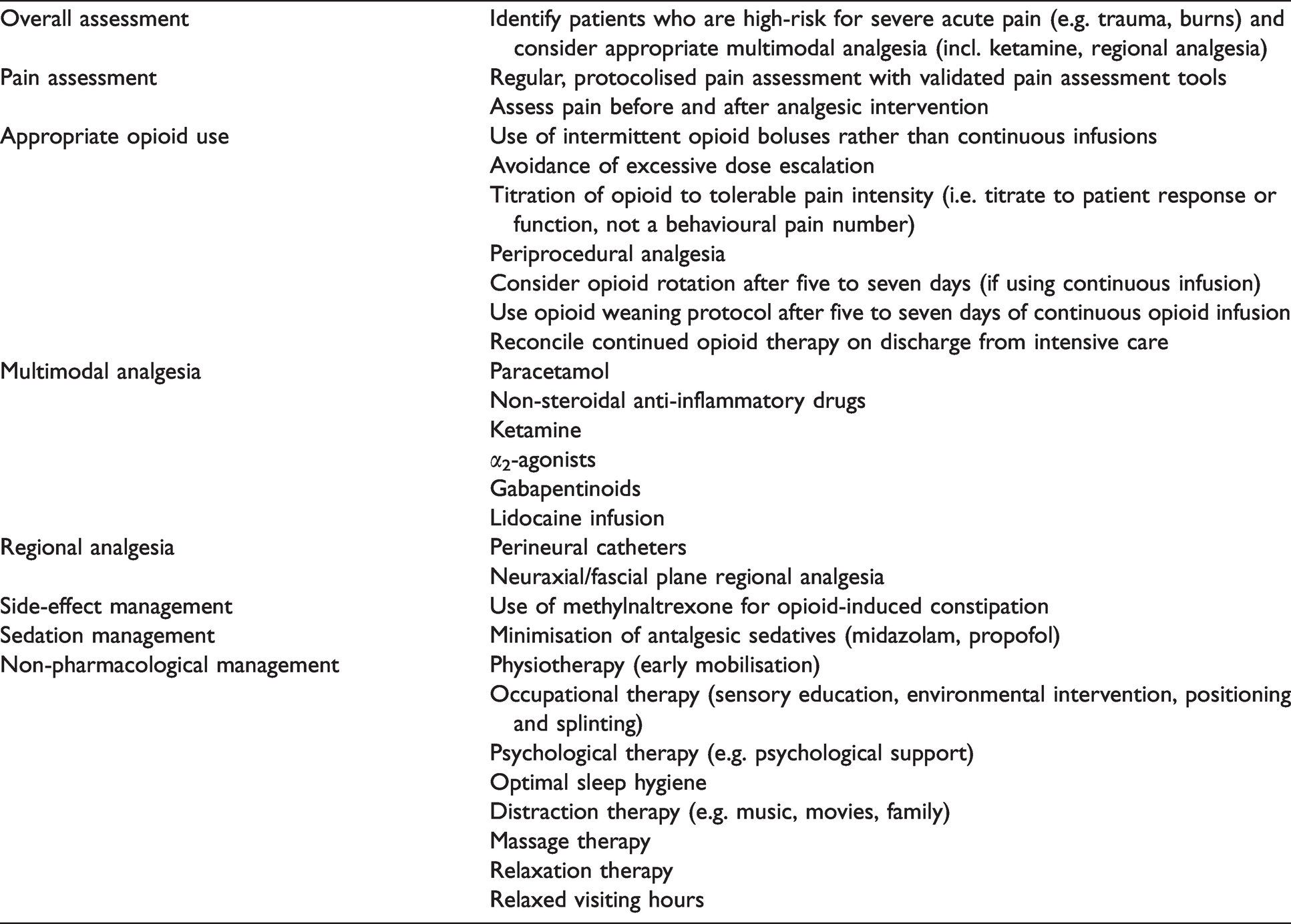
Approaches to pain assessment and management
It is important that clinicians have a clear sense of the presence of acute pain in patients in the ICU. Patients who have a superficially disproportionate response to pain should prompt a concerted search for additional contributory sociocultural and psychological factors. These include, but are not limited to, delirium, sleep deprivation, existential threat, post-traumatic stress disorder, anxiety and depression, previous trauma, and being from a cultural and linguistically diverse community. Addressing these factors may result in improved analgesia.
Approaches to analgesic pharmacotherapy
Intermittent versus continuous opioid infusions
Given that opioid infusions can produce acute opioid tolerance, OIH and opioid withdrawal syndrome, intermittent opioid boluses may reduce the incidence of opioid-related adverse effects. Intermittent bolusing of opioids also reduces the accumulation of active drug and metabolites, which can potentially result in a decreased duration of mechanical ventilation. In a retrospective analysis, patients initiated on a continuous infusion of fentanyl experienced more delirium than those receiving intermittent boluses of fentanyl. There was no difference in the duration of mechanical ventilation and ICU length of stay between the groups. 110
Periprocedural analgesia
Critically ill patients experience pain during standard care procedures, including turning, arterial line insertion, chest tube removal, endotracheal tube suctioning, and drain removal.3,46 Pre-emptive analgesia may result in less post-procedural postprocedural pain and opioid requirement, although this has not been studied. A strategy of preprocedural pain assessment and appropriately timed pre-emptive analgesia should be used for procedures known to cause pain. 10
Opioid weaning protocols
In patients tolerant to opioids, abrupt cessation usually results in a withdrawal syndrome. As mentioned previously, acute opioid tolerance may occur in ICU patients after five to seven days of continuous opioid infusion. In the setting of acute opioid tolerance, it would be prudent to institute an opioid weaning plan. Such plans implement a stepwise reduction of opioid tapering that may last as long as one week. The use of methadone replacement in the ICU to facilitate weaning from fentanyl infusions has been studied. In two studies, mechanically ventilated patients receiving a continuous fentanyl infusion, replacement of fentanyl with enteral methadone resulted in a higher probability of successful weaning with a decreased time to extubation.111,112 In addition, an opioid taper regimen has been suggested, 113 decreasing the infusion rate by 20%–40%, followed by 10% every 12–24 hours in patients at low risk of opioid withdrawal or 5%–10% reduction in dose per day in patients who have received high-dose opioids for greater than five days.
Opioid rotation
Opioid rotation to a different opioid occurs when opioid tolerance results in diminishing analgesia and there are concerns about the harms of opioid dose escalation. Opioid rotation takes advantage of incomplete cross-tolerance, whereby different receptor subtypes and specific opioid desensitisation result in improved analgesia and the use of a relatively lower dose of the new opioid. 114 Another potential effect of opioid rotation is the alleviation of the antalgesic effect of 3-glucuronide metabolites, seen with morphine and hydromorphone. Rotation to alternative opioids without active metabolites will decrease 3-glucuronide levels, potentially improving analgesic efficacy. 114 A feasibility study of ventilated ICU patients showed that opioid rotation was possible. 33 Patients were rotated from fentanyl infusions to hydromorphone infusions. Although there was improved analgesia, the cumulative opioid dose was similar after rotation, suggesting a disconnect between the opioid dose administered and pain. Nevertheless, opioid rotation should be considered for patients who are on continuous opioid infusions for longer than five to seven days, but this requires further investigation.
Opioid prescriptions on discharge from the intensive care unit
Administration of an opioid in the hospital setting is associated with an increase in opioid prescription at hospital discharge and in the community. 115 In a study examining the continuation of opioid therapy at the transitions of care of opioid-naive, ventilated ICU patients, it was found that 56.1% of patients continued opioid therapy from the ICU to the ward. 116 It was also found that 31.9% of all patients continued opioid therapy to hospital discharge. Given the increased risk of inappropriate continuation of opioid therapy and its associated morbidity, it is important to liaise with and refer to appropriate services (e.g. admitting team, acute pain team, drug and alcohol services) to implement appropriate opioid stewardship.
Non-opioid adjuvant analgesics
Given the variable aetiology of acute pain, the addition of non-opioid analgesics that target various nociceptive pathways may produce synergistic analgesic effects that result in a reduction in the required opioid dose.
Paracetamol is a commonly administered analgesic with a mechanism of action that is unclear but may include cyclo-oxygenase inhibition in addition to endocannabinoid effects. Investigation into its use is limited to small studies in specific patient cohorts. In postoperative ICU patients, the addition of paracetamol resulted in a reduction in pain scores, opioid consumption and time to extubation. 117 In one study of intravenous paracetamol in ICU patients post-craniotomy, there was no difference in either pain scores or opioid consumption. 118 Despite the lack of evidence in general ICU patients, the risk of harm is thought to be low. Administration intravenously did not result in an increase in the incidence of hepatic dysfunction, 119 but did increase the incidence of hypotension that required intervention. 120 Given the potential opioid reduction and improved analgesia, and a probable low risk of harm, the addition of paracetamol may be beneficial in opioid reduction strategies.
Non-steroidal anti-inflammatory drugs (NSAIDs) are effective analgesics; however, the perceived risk of adverse effects limits widespread use. When 24 hours of ketoprofen was added to an opioid infusion in postoperative major abdominal surgery patients, the result was a reduction in opioid consumption (by approximately 20%), pain scores, and nausea and vomiting. There were no reported bleeding events or episodes of acute kidney injury. 121 In addition, the use of ketorolac in patients with rib fractures was associated with a decrease in the incidence of pneumonia and shorter ventilation times, with no apparent increase in the risk of bleeding or renal failure. 122 The safety of ibuprofen in patients with sepsis was demonstrated with no increase in the incidence of acute kidney injury, GI bleeding or occult bleeding. 123 Coxib NSAIDs may be a useful adjunct due to the decreased risk of gastric ulceration and the lack of platelet inhibition, with the benefits of decreased opioid consumption and improved analgesia.
Continuous infusions of low-dose ketamine, an NMDA antagonist, is a commonly used analgesic strategy to decrease opioid consumption and hyperalgesia in the postoperative setting. Its use in the ICU is less well studied, with varying results. A retrospective before–after observational study of 390 mechanically ventilated patients who were administered a continuous infusion of ketamine showed an increase in achieved target scores for pain and sedation, with no difference in the duration of delirium. 124 This reduction in opioid dose was also observed in awake, postoperative ICU patients 125 as well as ventilated postoperative ICU patients. 126 In contrast, another retrospective, uncontrolled before–after study found an increase in daily opioid dose after the initiation of ketamine, 127 while a randomised trial found no difference in opioid consumption although there was a reduction in the incidence of delirium. 128 Methodological approaches may account for the observed differences in addition to the dose of ketamine used. Analgesic doses of ketamine (0.1–0.2 mg/kg/h) typically have a lower side-effect profile compared with sedative doses of ketamine (1–2 mg/kg/h). Nevertheless, patients at risk of moderate to severe acute pain may benefit from a continuous, low-dose ketamine infusion.
The alpha-2 agonists dexmedetomidine and clonidine are weak analgesics but may aid in analgesia, through the management of psychological causes of acute pain (e.g. delirium and agitation) and by the avoidance of potentially antalgesic sedatives. 44 A systematic review found that the addition of clonidine to standard sedation resulted in decreased opioid dose, with no difference in the duration of mechanical ventilation, ICU mortality or ICU length of stay. 129 In post–coronary artery bypass graft (CABG) patients, sedation with dexmedetomidine was associated with significantly lower morphine requirements than propofol-based sedation. 130 While the benefits of using alpha-2 agonists lie predominantly with sedation strategies, there may be some benefit with regard to opioid reduction.
The gabapentinoids, gabapentin and pregabalin, may be effective in improving analgesia and decreasing opioid consumption in the patient with neuropathic or unyielding nociceptive pain. In ventilated patients with Guillain–Barré syndrome, the administration of gabapentin resulted in a decrease in pain scores and opioid consumption.131,132 However, in a pilot study of critically ill patients with rib fractures, the addition of gabapentin compared with placebo to a multimodal analgesic regimen did not result in a difference in pain scores, opioid consumption, oxygen requirement or incentive spirometry. 133 In cardiac surgical patients, the use of pregabalin resulted in a decrease in opioid consumption, improved acute and chronic pain, and a decreased incidence of delirium in the elderly.134,135 As the gabapentinoids are eliminated unchanged in the urine, accumulation and increased sedation may occur in the setting of renal impairment, in which dose adjustment may be warranted. In addition, patients on long-term gabapentinoid therapy should be weaned prior to discontinuation, as abrupt cessation may result in a withdrawal syndrome. Overall, the potential benefits of gabapentinoids in the setting of neuropathic pain need to outweigh the risks of increased sedation in the setting of renal impairment. Clearly, further evaluation is needed in the general ICU cohort.
The use of lidocaine is well established as a useful analgesic adjunct in the treatment of acute pain. Intravenous lidocaine infusions have had varied success in the treatment of acute pain, neuropathic pain and chronic pain.136 –138 Studies examining the use of lidocaine infusions in the general ICU are limited to retrospective, case-controlled studies.136,139 In these studies, the addition of lidocaine infusion resulted in a reduction in opioid consumption and pain scores. However, acute neurological adverse effects (decreased conscious state, confusion) were noted in patients who developed acute kidney injury with resultant elevated serum lidocaine levels. The adverse effects resolved on discontinuation of lidocaine. Given that a large proportion of ICU patients develop end-organ dysfunction, the risk of lidocaine toxicity is increased. In this setting, regular serum lidocaine levels should be monitored, which may preclude widespread use. Needless to say, further randomised controlled trials are warranted to determine the role of lidocaine infusions for the treatment of acute pain.
Regional analgesia
The use of neuraxial analgesia (e.g. epidural, paravertebral blocks), fascial-plane catheters (e.g. erector spinae blocks) and peripheral nerve blocks may be of analgesic benefit for critically ill trauma and postoperative patients. Regional analgesia (RA) techniques can result in improved analgesia, and reduced opioid requirements and opioid-related adverse effects. The predominant indications for RA in the ICU are thoracoabdominal surgery and trunk trauma. 140
Thoracic epidural analgesia has shown benefit in abdominal aortic aneurysm surgery, 141 traumatic rib fractures,142,143 and thoracoabdominal procedures. 144 In geriatric trauma patients admitted to the ICU with rib fractures, the use of RA resulted in a decrease in the incidence of delirium, with a reduced cumulative daily opioid dose. 143 There was no effect on the incidence of attributable pneumonia.
Regional analgesic techniques for rib fractures include thoracic epidural, paravertebral, intercostal, erector spinae, and serratus anterior catheters. Transversus abdominis plane, rectus sheath or preperitoneal catheters may be used for abdominal procedures, reducing the potential morbidity of a thoracic epidural (e.g. hypotension, poor mobilisation). Specific nerve distributions may be targeted in trauma patients, such as brachial plexus blocks for upper limb pathology or sciatic nerve blocks for foot and ankle pathology. Close liaison with the acute pain team is essential to facilitate RA in the ICU.
Choice of sedatives
The choice of sedative in mechanically ventilated patients may affect pain perception. In a healthy volunteer study, patients were randomly assigned to receive moderate sedation with midazolam, propofol or dexmedetomidine, and the responses to experimental pain tasks (cold, heat, ischaemic or electrical) were recorded. 44 Midazolam increased cold, heat and electrical pain perception significantly. Dexmedetomidine reduced both cold and ischaemic pain perception significantly, and propofol had non-significant varied effects on all modalities. While it is not known if this translates to the ICU cohort, it is possible that midazolam and potentially propofol are antalgesic sedatives, and that alternatives such as dexmedetomidine should be used.
Future directions
As evidenced by the paucity of data investigating the effects of opioids during and after ICU admission, further research is warranted. This would further elucidate the short-term adverse effects of opioids such as delirium and ICU-acquired infections, and the long-term adverse effects such as the role of opioids in developing chronic opioid therapy and chronic post-ICU pain. The ethical limitations of conducting randomised controlled trials into opioids versus no opioids precludes the robust estimation of causal effects of opioids on outcomes. Hence, the use of observational data under a causal inference target trial framework is appropriate.
Short-term effects of opioids
Given the biological plausibility of the effects of opioids on delirium, acute tolerance and withdrawal syndrome, and ICU-acquired infection, these effects should be established. As previously mentioned, there exist many time-varying confounders in the causal pathway from opioid exposure to outcome. In critically ill patients, these may include acute pain, sedation, the presence of mechanical ventilation, adjuvant analgesics, sepsis, and delirium. In addition, factors such as admission diagnosis, socioeconomic factors, prior substance use and gender may also play a role. For robust estimation of the potential causation of opioids on short-term outcomes using observational data, specific methodology should be employed (discussed below).
Long-term effects of opioids
The role of opioids in the development of chronic opioid therapy and chronic post-ICU pain is currently unknown. As with the studies examining the potential short-term adverse effects of opioids, studies investigating the long-term outcomes suffer the same complexities of robust estimation of potential causal effects. In addition to the issues mentioned above, studies of long-term outcomes are susceptible to loss to follow-up, and this selection bias needs to be accounted for in any estimation of effect.
Methodological and statistical approaches
Approaches to the design and analysis of studies to determine unbiased causal effects of opioid exposure within the ICU to specified outcomes remain complex. Complexities include the presence of correlated data (e.g. daily opioid dose or daily pain scores), the presence of variables that are both mediators and confounders for the outcome (e.g. opioid dose, pain and delirium), time-varying confounding, and multiple time-varying exposures for the outcome. For this reason, standard multivariable regression techniques, using adjustment for time-invariant confounding or a single variable composite of complex exposures (e.g. total opioid dose for an ICU admission), are inappropriate and may lead to erroneous estimates. Likewise, the addition of all potential variables into a regression model and using P-values to determine ‘risk factors’ may result in effect estimation of mediators, confounders, proxy confounders and competing exposures (the Table 2 fallacy), 145 rather than an exposure–outcome causal effect.
These issues require specific analysis. Adjustment of time-varying confounders using a marginal structural model may give a more accurate estimate of potential causation. 146 The use of a potential outcomes framework with a target trial emulation of a randomised controlled trial enables transparency and reproducibility in observational data when a randomised trial cannot be performed.147,148 Target trials, utilising marginal structural models or g-computation may enable the determination of a less biased exposure–outcome causal effect.
Conclusion
Opioids remain an integral component of the management of acute pain in the ICU. However, opioid use may be associated with both short-term and long-term morbidity, including delirium, ICU-acquired infection, acute opioid tolerance, opioid-induced hyperalgesia, opioid-induced iatrogenic withdrawal syndrome, chronic opioid therapy, and chronic post-ICU pain. Both the large cumulative dose and prolonged duration of opioid therapy in mechanically ventilated patients in the ICU may increase the risk of opioid-related morbidity. Currently, the potential causal pathways between opioid use and the aforementioned adverse effects have yet to be fully elucidated, and precise measurement tools are lacking. In addition, a more nuanced, patient-specific approach to opioid management within the ICU is warranted, with the dual aim of opioid minimisation and optimisation of analgesia. Finally, it remains to be seen if the application of opioid minimisation strategies resulting in a reduction in opioid use in the ICU will lead to a reduction in the short- and long-term adverse effects of ICU opioid therapy.
Footnotes
Author contribution(s)
Acknowledgements
The author(s) would like to thank Meredith Craigie for assistance in the review of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
