Abstract
Misuse of prescription opioids forced an inevitable response from authorities to intervene with consequences felt by all.
In the Australian community one person will die for approximately every 3600 adults prescribed opioids, while in the hospital setting a postoperative patient managed primarily with opioids, as opposed to epidural analgesia, has an additional risk of death as high as between one in 56 to 477.
Opioids maintain a valid role in acute pain management when use is reasoned and with full awareness of the harms and how they are to be avoided, such as in those at risk of ongoing use, the opioid naïve, and when opioid-induced ventilatory impairment may occur.
Clinicians managing acute pain can focus on assessing pain versus nociception, strategically apply antinociceptive medications and neural blockade when indicated, assess pain with an emphasis on the degree of bothersomeness and functional impairment and, finally, optimise the use of framing and placebo-enhancing communication to minimise reliance on medications.
Introduction
Much has been published on the genesis of the over-reliance on opioids for the patient with pain and the ensuing opioid crisis.1,2 By the start of this century there was rising awareness of severe unrelieved pain causing serious morbidity and mortality in patients with acute, chronic, and cancer pain, with valid arguments put forward on ethical and medical grounds to view chronic pain as a disease entity, and pain management as a universal human right. 3 This presented acute care clinicians with a difficult challenge because the management of pain was yet to be fully developed, and they often grappled to comprehend fully or best utilise all the tools at their disposal. Neural blockade when correctly utilised was known to be capable of providing effective analgesia. 4 Unfortunately, awareness arose that neural blockade could also harm patients, sometimes to a devastating extent, even when performed proficiently. 5 The readily identifiable link between an intervention and subsequent harm in an increasingly litigious and risk-averse world saw clinicians searching for seemingly ‘safer’ analgesic methods. Opioids when used for acute pain were reported as rarely addictive; 6 although the scant evidence for this assertion never underwent the necessary degree of scrutiny. It seemed when faced with a lack of suitable alternatives and no apparent downside, the response of clinicians increasingly to look towards opioids became inevitable. The harm from opioids was largely out of sight as well as readily explained away, so also out of mind.
An additional influence contributing to the use of opioids was the promotion of ‘pain as the fifth vital sign’. Pain was now to be assessed and documented as a score on the observation chart alongside the four vital signs of heart rate, blood pressure, respiratory rate and temperature. The intention was to bring awareness to clinicians of the patient in pain, with the aim of preventing pain in the first place, and to improve the use of analgesia. The resultant outcomes were contrary to that intended. Opioid prescribing merely escalated as ward staff sought to rescue patients and more harm was caused from opioid side-effects. 7 In response the push for pain to be documented as the fifth vital sign has been largely abandoned. 8
The response now required of acute care clinicians to the opioid crisis is not simply to restrict or eliminate opioid prescribing; rather it is better to equip themselves and their patients with knowledge of safe and effective prescribing of opioids.
Opioid prescribing in the acute phase: how much of a problem is it?
The greatest burden of deaths from prescription opioid misuse has been borne by the chronic pain patient population. However, we are increasingly becoming concerned about the contribution of opioid prescribing initiated in the acute phase of pain. 9
Notably, follow-up studies reveal patients remain on opioids for periods of time well beyond that intended by the initial prescriber. For example, in one USA study, 6% of patients on average were still filling scripts for strong analgesics 90 days after surgery—including after even minor surgeries such as carpal tunnel release. 10 Medications included pure opioid agonists, tramadol, and codeine (personal communication with author). This study was limited to 13 soft-tissue surgery types and, importantly, excluded orthopaedic surgery.
Australian data covering a broader range of surgery types including orthopaedics identified ongoing analgesic use at a higher rate of 11% at 90 days post surgery. 11 In that study nearly all patients were taking a medication requiring a doctor’s prescription, as either a pure opioid agonist (41%), tramadol (20%), or high-dose codeine (30%). If allowance is made for the key difference in study populations of inclusion or exclusion of orthopaedic surgery—a group of patients with greater rates of long-term analgesic use—then the Australian data for ongoing opioid prescribing becomes not too dissimilar to the USA. This is despite opioid prescribing in the USA, measured as milligrams per capita, being three times greater than that of Australia, 12 and suggests known patient risk factors determine the ongoing use of opioids, rather than simply opioid overprescribing. These risk factors are summarised in Table 1.11,13–15
Patient risk factors for ongoing opioid use.
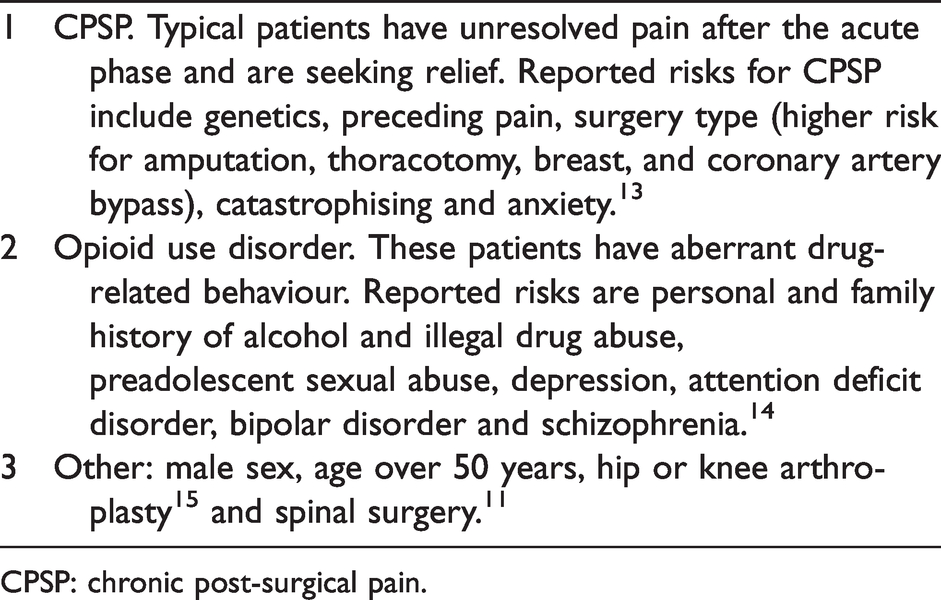
CPSP: chronic post-surgical pain.
Patients at increased risk require close supervision of their opioid use and consideration of follow-up with a specialist pain medicine physician post discharge. Rational opioid prescribing as stewardship programmes can form part of the in-hospital strategy for all patients, and many approaches have been described including providing procedure-specific guidelines on opioid prescribing 16 and, when necessary, intervention with targeted clinician education. 17
Opioid use and the associated risk of death
Patients maintained on opioids outside of hospital are known to be at increased risk of death. Determining individualised risk for an outpatient when prescribing an opioid is difficult because although data are collected on prescription opioid deaths, whether the implicated opioid was diverted, or intentionally prescribed to the deceased is not always known. Through combining data on opioid prescribing with death rates we can make a general population estimate; that for approximately every 3600 Australian adults prescribed an opioid there will be one associated death in the population per year (based on calculations using data from 2017 to 2018).18,19
Knowing the risk of death from opioid prescribing in inpatients is similarly challenging due to gaps in data capture. A patient who, for example, dies of aspiration pneumonia caused by opioid-induced ileus and sedation may not have this death recorded as opioid related. However, an estimate of opioid risk is a vital component of our response to the opioid crisis to better weigh up the risks of opioids versus the alternatives. One approach is to compare mortality outcomes for an opioid-sparing analgesic technique, such as an epidural, with opioid-based controls to provide the clinician with a sense of to what degree opioid-based therapies can have harmful consequences. One large study of epidural use showed a small reduction in mortality, deriving a number needed to treat (NNT) to save one life with an epidural of 477, 20 while a meta-analysis of published studies and including a greater proportion of thoracic epidurals derived a NNT of 56. 21 Alternatively, we can choose to view these NNTs differently and say, when choosing to forgo an epidural in preference for opioid-based therapy in a distinct population of patients, there may be one extra death for every 56 to 477 patients so treated.
Understanding these data in this way, bearing in mind the deficiencies of generalisability, provides the clinician with a familiar method of determining the risk:benefit of choosing opioid-based therapy for individuals in the inpatient setting.
The pain experience
Misunderstanding of the human experience of pain underpins much of the misdirection in the management of pain and the reliance on opioids.
The definition of pain of the International Association for the Study of Pain aims to focus our thinking with the first few words: ‘(pain is) an unpleasant sensory and emotional experience’, and by stating further that ‘pain and nociception are different phenomena’. 22 In simplistic terms, the human body’s afferent pathways convey the sensation of a noxious stimulus (nociception) to the thalamus, and along the way modifications including from the limbic system add an emotional, affective component. This interaction then influences the experience of pain at the cerebral cortex. 23
Conceptualisation of this difference between pain and nociception may be aided by reference to the lowest order animals, which when injured will respond (perhaps by moving away from the stimulus), but they are unlikely to be in pain—rather a response to nociception is being witnessed. The higher-order brain of mammals such as humans possesses complex emotion-controlling structures allowing us to perceive nociception as something unpleasant—to be pain.
Differentiating pain from nociception in our patients can be aided by careful questioning. The McGill pain questionnaire (MPQ) is a widely adopted pain assessment tool with three major classes of word descriptors to aid differentiation: sensory, affective and evaluative. 24 Sensory descriptors best relate to nociception and include sharp, cutting, lacerating, and crushing. The affective and evaluative descriptors, which focus on the degree of unpleasantness, include troublesome, annoying, distressing, and unbearable. The word ‘bothersomeness’ was not included in the original description of the MPQ and has since been promoted to evaluate the affective component. 25 Research indicates assessment of bothersomeness aids in detecting patients with the greatest pain and disability. 25
Differentiating between the sensory and the affective components aids our selection of analgesic options, especially the indication for opioids. Nociception can be most effectively managed with a focus on antinociceptive strategies utilising non-steroidal anti-inflammatory drugs (NSAIDs) and neural blockade as these act potently and predominantly on prespinal structures (discussed in the next section). In contrast, systemic opioids gain efficacy as pure antinociceptive agents as dose is increased, while at lower doses they hold their greatest efficacy as analgesics in a broad sense removing the affective unpleasant experience of pain. 26
Clinical management of nociception and pain
A noxious stimulus, most commonly acutely due to an injury such as surgery or trauma, will result in nociception. This nociception will always have the potential to be reported as pain by the patient, so the primary strategic approach of pain management is first to limit nociception as much as is feasible.
The two key biological approaches available to limit nociception are described below.
Neural blockade
Neural blockade, especially major regional or neuraxial techniques, is in theory the most effective means to prevent transmission of nociception beyond the second-order neuron. An analgesic benefit with neural blockade is reported across a wide range of techniques.
4
Neuraxial blockade has the added benefit of limiting the neuro-endocrine stress response to injury, so providing for morbidity and mortality benefits beyond purely analgesia.
21
In order to deliver the known benefits of neural blockade to their patients a clinician needs to ensure the block utilised is appropriate, evidence based, and is a technical success. It is critical therefore for a clinician to be familiar with methods to evaluate the success and effectiveness of neural blockade. The likely clinical efficacy of a chosen neural block can be judged by seeking confirmation of at least four key endpoints:
27
Dynamic (movement) analgesia: pain should not increase if the blocked region is moved. Analgesia: pain should be reduced after performance of the block. This assessment is not always possible if, for example, the block is placed prior to surgery or during a general anaesthetic. In these circumstances, the absence of pain after surgery may be misconstrued by the clinician as proof of neural block success when instead the patient may merely have no pain resulting from the surgery. Sensory anaesthesia: if the nerves being blocked innervate the source of nociception as well as having a cutaneous sensory distribution (nearly always the case), then sensory anaesthesia should be present. Opioid sparing: if the site of nociception is meaningfully blocked then opioid requirements should be zero or substantially reduced.
Non-steroidal anti-inflammatory drugs
Of systemic medications, the NSAIDs including the COX-2 inhibitors have potent antinociceptive properties. They act predominantly peripherally at the level prior to the primary order neuron, limiting the generation of the inflammatory response to injury, thus minimising peripheral sensitisation and preventing the amplification of nociceptive activity. This mechanism of action results in NSAIDs dominating the upper ranks of the Oxford league table of analgesic efficacy for acute pain, with NNTs mostly in the range of 1.5–2.1 (for comparison, oxycodone immediate release 15 mg has a NNT of 2.3). 28 The potency of NSAIDs as antinociceptive agents is further demonstrated by their capacity to effectively minimise exacerbations of pain on movement (dynamic analgesia)29–31—a time of intense nociceptive stimulus. Unfortunately, the analgesic benefit of the NSAID class is often not realised for patients due to excessive caution in the presence of relative contraindications.
Patients who report predominantly pain on movement will be most effectively managed with the above antinociceptive approaches. In most circumstances management is first to ensure the NSAIDs are optimised, and if this fails or alternatively if it is a suitable option, utilise neural blockade. Second-line agents can be introduced when indicated for antinociception and include atypical opioids, lidocaine, gabapentinoids, ketamine and opioids.
Non-biological approaches to managing acute pain: enhancing endogenous analgesia
In 2015 Loeser wrote ‘chronic pain is not a sign of either morphine or lidocaine deficiency’. 32 A similar way of thinking is warranted for acute pain. The ‘biopsychosocial’ catchphrase used to describe a clinician’s intended pain management plan is frequently heavily weighted towards the ‘bio’ component. The psychosocial component can, however, be readily addressed with communication approaches which require only limited training and are intuitive. There are overt verbal and non-verbal cues which form part of communication with our patients and impact their pain reports. Two simple, effective methods can be utilised by the clinician:
Placebo-enhancing communication
Placebo can be simply defined from its Latin origins of ‘I shall please’. In pain management, placebo can result in substantial analgesia. Placebo-mediated analgesia is underpinned by biological processes, as evidenced by placebo being negated in research settings by administering naloxone, 33 indicating an actual mind–body interaction. 34 Placebo does not need to be hidden or done by trickery to be effective. Study patients openly administered inert substances which they know are placebo (either saline infusions or sugar pills) report improvement in their pain.35,36
When an analgesic medication or intervention is trialled and shown to be beneficial, there are three components to that benefit. 37 First, there is a placebo response due to regression to the mean or the natural history of pain, which acutely is generally to improve on its own. Second, there is the placebo effect due to patient expectations and conditioning that they will improve from the medication or intervention. 38 Third, there is the direct biological response from the intervention or medication. Therefore, clinicians will only achieve the same clinical effect of an analgesic intervention or medication if they recreate the circumstances of the positive clinical trial through attention to placebo effects.
The environment and the ritual of the doctor–patient interaction leads to conditioning. A positive conditioned response to the therapeutic ritual surrounding caregiving can be enhanced by the clinician presenting themselves as caring, trustworthy and knowledgeable, which are perceived by patients through such things as their attire and manner. 39
Expectations are positively enhanced by clinicians by their choice of wording. Analgesics therefore should be prescribed accompanied by the use of positive language 40 such as, ‘patients report these medications as being very effective at helping them be more comfortable’. Opioid-sparing approaches may be realised with wording such as, ‘most patients find because the other medications they are on are so effective, they only rarely need to use the opioid pain medications to cope with what they are feeling’.
Better use of such placebo-enhancing communication strategies could form an important basis for reducing the over-use of opioids in acute pain. 41
Awareness of the power of placebo goes hand in hand with knowledge of nocebo. Nocebo can be interpreted from its origins as ‘I shall harm’. Nocebo is detrimental and is to be avoided. 42 One very common nocebo term is the word ‘pain-killer’ when prescribing analgesics. It is very rare for any analgesic to truly ‘kill’ pain, rather in most instances a varying reduction in pain is seen. When medication is administered to a patient with the expectation it will ‘kill’ their pain and it does not achieve this, an expectation mismatch occurs for the patient and they have in essence been harmed. Harm can be avoided by the use of wording which provides a realistic expectation; for example, the clinician can instead choose to say, ‘these medications will help you feel more comfortable’. If shorter wording is desired, a simple description of ‘pain medications’ can be chosen. Another common but frustratingly powerful nocebo is forewarning about itch. 43 Informing a patient they might get itch after intrathecal opioids such as morphine ensures this clinical outcome is a certainty. Itch is a highly suggestible symptom; once itch enters the mind it becomes very recalcitrant to therapy, mirrored by the paucity of effective remedies. Reports of itch are perceived to be so problematic by some clinicians they change their practices entirely. As a result, opioid-sparing techniques such as neuraxial morphine for caesarean section are abandoned in favour of systemic opioids, with the potential for far greater doses being required and in the absence of close supervision, leading to the ongoing use of opioids at discharge. 44
Message framing
Message framing can be utilised beneficially for the patient in pain, with the careful use of wording to provide positive instructions and suggestions. 45 Early attempts at framing may be viewed as somewhat blunt but were effective. In the seminal paper by Egbert et al. in 1964, patients who were informed prior to surgery that it is normal to have pain after surgery had lower opioid dose requirements than those who were given no instructions. 46
Since then, message framing techniques in acute pain have become more finessed to allow the clinician subtly to convey to the patient the concept of expected nociception, or ‘sensations’, rather than only expecting ‘pain’. Examples of this approach include clinicians enquiring, ‘how are your healing sensations after surgery?’ or referring to comfort scores rather than pain scores with ‘how comfortable are you?’. 47 Similarly, with the above example of itch, framing can be employed by the clinician to allay further complaints and emphasise the benefits. For example, ‘some patients report a short period of itch which soon goes away, and most patients prefer to have only had the small dose of opioid pain reliever we gave you in the spinal rather than the alternative of needing many times larger doses of opioids through your drip or as a tablet’.
The temptation to frame the expected post-surgical nociception to a patient by describing it as ‘good pain’ is arguably best avoided because it presents a mixed message, an oxymoron even. The word ‘pain’ carrying as it does such strong negative implications and the use of the preceding ‘good’ presents too confronting a juxtaposition. The preferable approach for the clinician is use of the word ‘sensations’ to describe nociception. If these sensations have become painful and unpleasant then they warrant appropriate management as previously discussed.
Clinical challenges of pain scoring systems
Pain scores were developed with the aim of objectively quantifying what is a subjective qualitative experience. 24 The use of ‘pain as the fifth vital sign’ came under heavy criticism because it led to over-relying on the numbers as steadfast measures of the underlying state. Pain management protocols emerged, which although well intended were misguided, for example linearly increasing ‘sliding’ scales of opioid dose were stipulated as the pain score rose. Such protocols worked on the assumption that both the patient and the number collector had a comprehensive understanding of pain, and they failed to allow for the fact that pain scores will not necessarily reduce in a linear fashion as the opioid dose is increased.
Despite the known flaws of pain scores being well publicised, 8 clinicians continue to find themselves being presented with scores deemed ‘out of range’. Finding a way forward requires continual education on pain and a knowledge of alternative measures which can be easily applied.
The defense and veterans pain rating scale (DVPRS) is one example of the development of such a measure which combines nociception with function and pain and is simple to understand and easy to apply clinically (Table 2). 48 The DVPRS was a response to the over-reliance on opioid analgesia which had arisen when pain management algorithms utilised only the unidimensional classic verbal numerical rating scale (NRS). The DVPRS utilises the familiar 0–10 scoring range but critically anchors each number with function or the degree of unpleasantness—thereby more truly representing our clinical concerns.
Defense and veterans pain rating scale.

Although the DVPRS is yet to be more widely adopted, the key aspects of such an approach with the focus on function and evaluative wording are being more widely adopted and warrant consideration by clinicians for integration into their routine practice. Patients report a preference for the use of the DVPRS compared with other pain scales to report their pain. 49
Pain endpoints in research
The aforementioned complexity surrounding pain scores means clinicians relying solely on an NRS endpoint when interpreting research can be misguided. For example, when measured solely as pain at rest, we may see a distress-altering opioid faring much better than neural blockade. A different conclusion could arise if measurements of function (movement) and of side-effects (e.g. nausea, constipation) were reported, so when the analgesia methods being compared act through different mechanisms, more endpoints are required for true differentiation.
Important considerations for clinicians when evaluating research in acute pain include:
NRS using 11 points from 0 to 10 are preferred over the categorical verbal rating scale of: no, mild, moderate, or severe pain.50 Baseline pain (if present) should reach a minimum of a numerical rating score of 5 to allow for a treatment effect to become evident.50 Pain measurements should include dynamic movement or activity-evoked scores.51 Measurements are made at specific times which are relevant to demonstrate efficacy, with particular emphasis on the early phase when a difference is most likely to be detectable.50 Time to rescue medication and consumption of rescue medication.51 Decrease in opioid consumption.51 Functional performance.51 Use of quality of recovery (QoR) scores after anaesthesia and surgery. The QoR-15 score, a 15-point questionnaire which can be undertaken in less than six minutes, is widely utilised and supported.52 Questions relate to patient experiences in the prior 24 hours including the ability to breathe easily, enjoyment of food, feeling rested, sleep, moderate or severe pain, anxiety and depression. Responses are recorded as a numerical rating score from 0 to 10, and after summation a maximal score of 150 would be seen in a patient with perfect health. In their original study across a wide range of surgery types the authors reported mean QoR-15 scores preoperatively of 123, falling to 101 postoperatively. Patients who rated their recovery overall as ‘poor’ had a mean QoR-15 score of only 85.53 The minimum clinically important difference in QoR-15 score is reported as 8.5.2
Clinical considerations for improving the safe use of opioids
While much of the focus in dealing with the opioid crisis has been on reducing the misprescribing of predominantly oral opioids to outpatients, there are aspects of in-hospital practice in which applying current knowledge may improve safety.
Opioid-induced ventilatory impairment
Opioid-induced ventilatory impairment (OIVI) is the mechanism by which most opioid-related deaths occur. As the opioid effect increases, the minute ventilation (MV) decreases and arterial carbon dioxide partial pressure (PCO2) rises while the oxygen partial pressure (PO2) falls. 54 Death culminates when the opioid-induced sedation (narcosis) reaches a point at which the upper airway becomes obstructed and/or ventilatory effort ceases entirely (apnoea) due to the opioid. Death by either mechanism is due to a failure of ventilation. Death from opioids will hence be stopped by a definitive strategy to restore ventilation such as the administration of naloxone or opening the airway with or without positive pressure ventilation.
Detection of the pre-terminal signs of opioid-induced sedation (narcosis) by regular sedation score checks has long been advocated. 55 To date, more advanced early detection systems such as chest plethysmography, oximetry and capnography have not been shown to be infallible. 56
The increased risk of OIVI has been most closely associated with age over 60 years, male sex, opioid naïvety, sleep-disordered breathing, and chronic heart failure. 57
Historically, after the introduction of patient-controlled intravenous opioid analgesia (PCIA), the mandatory use of supplemental oxygen (SO) therapy via nasal prongs or Hudson mask at 2–6 l/min was advocated as a measure to reduce harm from OIVI. Applying SO to patients using PCIA, the predominant form of opioid delivery postoperatively, can seem logical, especially in high-risk patients. However, applying SO was based on the erroneous assumption that hypoxaemia is the primary cause of harm in OIVI rather than sedation and failure of ventilation. All routes of opioid delivery can cause OIVI. 58 Deaths in the community witnessed as part of the opioid epidemic were mostly from oral opioids. Therefore, any strategy adopted to manage OIVI should entail a consistent approach across all forms of opioid delivery.
Hypoxaemia following surgery is seemingly very common and occurs not only in those on intravenous opioids. There have been several studies on the frequency of postoperative hypoxaemia. 59 In one such study, utilising continuous pulse oximetry on 833 postoperative patients, a remarkable 42% of patients experienced an episode of hypoxaemia (peripheral oxygen saturation (SpO2) <90%) lasting at least 30 minutes. 60 With hypoxaemia occurring so commonly, and despite SO administration to 66% of patients, the authors appropriately questioned whether preventing hypoxaemia improves patient outcome and that hypoxaemia may instead be an indicator of underlying disease rather than a treatable mechanism. 60
Intermittent oximetry when taken on average five and six times per day by ward staff has been shown to detect only 5% of the hypoxaemic episodes detected through continuous oximetry. 60 This knowledge has perhaps seen an eagerness to provide SO routinely as a proactive measure in the hope of improved safety when continuous oximetry is not available. However, a Cochrane review of outcomes for continuous oximetry versus intermittent oximetry strongly suggests flaws in such an approach. 61 While the use of continuous oximetry decreased the incidence of hypoxaemia—as expected, because perturbations in oximetry were corrected by staff applying SO—this did not translate to a reduction in complications of cognitive dysfunction, mortality, length of stay, or transfer to the intensive care unit. This finding indicates a focus solely on hypoxaemia prevention through the application of SO would be insufficient with outcome. The guideline of the British Thoracic Society on oxygen use emphasises the need for a considered approach with postoperative opioids, stating ‘oxygen should be administered to correct hypoxaemia rather than prevent it’. 59
Of further relevance is the association of all common modalities of postoperative opioid delivery with hypoxaemia. Clinicians at times seem overly focused on the intravenous route even though hypoxaemia (defined as SpO2 <85% or <90%) has been reported in 37% of patients with intramuscular opioids, compared with only 12% with PCIA and 15% with epidural. 62
To optimise safety, the management of hypoxaemia postoperatively requires a consistent approach across all routes of opioid delivery, with a focus on detecting and managing the cause of hypoxaemia rather than relying on the approach of preventing hypoxaemia in selected patients with SO.
An opioid-induced reduction in alveolar ventilation to approximately two-thirds of normal resting values can be predicted to result in a corresponding decline in the alveolar PO2, and an approximate SpO2 of 90% when breathing room air.
63
The fall in alveolar PO2 is matched by the increase in arterial PCO2 and can be readily corrected by only a small increase in the fraction of inspired oxygen (FiO2) to 0.3 using supplementation so normalising the SpO2. This relationship is described mathematically by the alveolar gas equation, in which alveolar PO2
Administering SO in the presence of OIVI, through impairing hypoxic ventilatory drive, depresses ventilation even further. This was demonstrated in the first and only study of its type conducted on 20 volunteers administered a short remifentanil infusion. 66 On room air 10% of the volunteers became apnoeic. After receiving oxygen supplementation at FiO2 0.5 apnoea occurred in 50% with the remifentanil infusion. Immediate desaturations did not occur due to oxygen supplementation and were averted by the researchers stimulating the volunteers to breathe. This study demonstrated that in the earlier stages of OIVI, ventilation is maintained by hypoxic ventilatory drive. This is then over-ridden by hyperoxia, leading to cessation of ventilation. Based on this research, there is concern about oxygen supplementation pushing the patient with OIVI towards an even more precarious state. 56
An analysis of adverse events (including death) ascribed to OIVI identified the pre-event use of SO in 15% of patients. 58 The patient with obstructive sleep apnoea (OSA) also requires considered use of SO. In an analysis of postoperative OSA-related adverse events including death, the use of SO was reported in 52% of patients. 67 These findings point to the unreliability of SO as a ‘failsafe’ in patients with OIVI or OSA.
In keeping with current knowledge of OIVI, Prielipp et al. 56 recently published key recommendations for postoperative patients receiving opioids by any route as listed in Table 3.
Recommendations for postoperative patients receiving opioids.

Ideally, once hypoxaemia on room air is detected medical staff are alerted, a diagnosis of cause is made, specific therapy instituted, and SO applied if indicated as supportive therapy. 65 Comprehensive guidance on oxygen use from expert groups has been promulgated including from the Thoracic Society of Australia and New Zealand, 68 with key messages as summarised in Table 4. 69
Adult acute oxygen use guidelines.

SpO2: peripheral oxygen saturation.
Opioid naïvety
The response of clinicians to the opioid epidemic has seemingly included an increased hesitancy when prescribing to opioid-naïve patients, often leading to undertreatment of their pain. Strictly speaking, naïvety to opioids means a state of ‘lack of experience’ and therefore applies only to someone who has never had opioids before. Naïvety denies the clinician the ability to be forewarned of the patient’s response to opioids.
Confusion has been generated by researchers often applying the term ‘naïve’ to those devoid of recent opioid use, to identify them as being in a predicted state of lack of opioid tolerance. Clinicians would view these patients as not strictly naïve, because they may well have had previous experience with opioids on which we can gauge their response, but instead, as patients who are unlikely to have developed analgesic tolerance.
Greater clarity to avoid confusion and misdirected clinical management may arise from using terminology with three options: (a) Opioid-tolerant: patients who have been regularly and recently administered opioids and are therefore expected to have tolerance to some extent; (b) Non-tolerant: patients who have experienced opioids in the past but have not received them so recently or regularly for tolerance to be predicted; and (c) Opioid naïve: reserved for patients never exposed to an opioid. An alternative term is ‘no opioid experience’.
Opioid dose requirements show a marked variation. Several factors including age and genetically determined expression of the opioid receptor are thought to contribute. 70 This variation in dose requirements adds complexity to clinical management when using opioids. Some opioids show far greater variability than others, with fentanyl being most pronounced showing up to a ten-fold variation in analgesic dose requirements.71,72 This marked variation in dose requirements for fentanyl leads to a perception that it has fewer side-effects when it is under-dosed or is responsible for frequent analgesia failures when clinicians fail to appreciate the equianalgesic fentanyl dose, and become uncomfortable extending doses to the outliers.
This wide opioid dose-range required to achieve analgesia occurs irrespective of being tolerant or non-tolerant (including naïve). Tolerant patients will, as a group, have a greater mean analgesic dose requirement than those without tolerance, but there is a great deal of overlap between the two groups.
The development of tolerance to the analgesic actions of opioids is well described and can require significant dose escalation. In contrast, the development of tolerance to the ventilation-impairing action of opioids is believed to be slower and less marked. 73 This has important implications for the clinician who can become complacent towards the tolerant and chooses only to be vigilant towards the naïve. Interestingly, tolerance to the ventilation-impairing action of opioids is largely a clinical perception, with evidence for tolerance in human volunteers and animal studies lacking. 73 Rather, what is being witnessed is a compensatory increase in the HVR to maintain ventilation in the presence of sustained depression of the hypercapnic ventilatory response.74,75 Central sleep apnoea, defined as the cessation of airflow without respiratory effort for more than ten seconds, has been diagnosed in 14%–60% of chronic opioid users, 75 and is caused by a sustained increase in HVR. The opioid-tolerant patient will require greater doses of opioid to achieve analgesia, but this analgesic tolerance is not matched by a similar degree of tolerance to ventilatory impairment when an equianalgesic dose of opioid is administered. 76 Consequently, the opioid-tolerant patient can paradoxically be at greater danger of OIVI perioperatively due to this differential tolerance. 73
The risk of prescribing to the opioid tolerant was highlighted in a Canadian study of over 600,000 patients, in which those prescribed opioid doses of greater than 200 mg oral morphine equivalents per day, and with a mean duration of opioid exposure of five years (therefore expected to be tolerant), were at a three times greater risk of opioid-related death than those maintained on less than 20 mg morphine equivalents per day. 77 The use of benzodiazepines, antidepressants and other central nervous system depressant medications was more frequent in those with opioid-related deaths.
The commonly recommended initiating doses of opioid are not those which have no chance of causing OIVI, rather they are doses which provide analgesia to a significant number of patients. Patients who are opioid naïve may best be, in effect, ‘tested’ with a dose of an immediate-acting opioid and then closely observed for their response. PCIA with the use of small incremental dosing and frequent observation exemplifies this approach. Longer acting and slow-release (SR) opioids are hence best avoided in the naïve patient due to the peak effect being delayed to a timepoint when observation is often inadequate to detect OIVI. The concern for the dangers posed by the use of SR opioids (including fentanyl patches) as the first opioid exposure in naïve patients contributed to the position statement of the Australian and New Zealand College of Anaesthetists cautioning against the use of SR opioid preparations in the treatment of acute pain. 78
Use of slow-release opioids in acute pain
The use of SR opioids in acute pain management is complex and controversial. 79 Over the past decade, the enthusiastic uptake of enhanced recovery after surgery (ERAS) programmes saw a move towards removal of mobility-encumbering PCIA machinery to deliver the opioid component of analgesia, and replacement with the use of regular SR opioids for a range of surgery types. ERAS-focused analgesia protocols, for example in total knee arthroplasty, commonly utilise SR opioids. 80 Typically, such protocols emphasise a multimodal pharmacological analgesic approach and the inclusion of an SR opioid is but one of many agents. The use of SR opioids in such a circumstance was argued as being acceptable because the pain following knee arthroplasty is characteristically constant and responsive to opioids. Concerns for safety were countered by clinicians considering the patient’s previous response to opioids before prescribing an SR preparation, and when encountering the naïve or sensitive, reverting to the use of PCIA with regular observations. Rational prescribing practices included ensuring use was for a short duration (commonly 3–5 days) limited to the in-hospital period, 8 ensuring use is endorsed by a group of peer clinicians, and provision of education.
In 2020 the Australian Therapeutic Goods Administration revised the indications for SR opioids to include the statement, ‘only be used where the pain is opioid-responsive and the patient requires daily, continuous, long-term treatment’. 81 Simultaneously the Pharmaceutical Benefits Scheme restricted the funding of SR opioids only to pain which is ‘chronic’. 82 Definitions of ‘long-term treatment’ and ‘chronic’ were not provided. These regulatory changes coinciding with greater improvements in the provision of multimodal analgesia may possibly reduce the use of SR opioids in such ERAS-focused guidelines.
Clinical judgement can be applied to assess the risks and benefits of SR opioids for patients. Examples include the prolonged inpatient stays of major burns and complex trauma patients who have constant opioid-responsive pain, and may transition to SR opioids for periods of their recovery under the supervision of experienced clinicians. 79
Opioid switching
Opioid-related harms can manifest when clinicians managing patients whose pain is poorly responsive to the prescribed opioid seek a resolution by simply changing the opioid or adding another opioid.
Opioid rotation is undertaken when side-effects or tolerance to an opioid occurs and is most common in the chronic cancer pain population in the setting of prolonged exposure to high doses. 83 Changing the opioid in acute pain (when dosing has often been lower and for only a short duration) is in contrast better referred to as an opioid switch. A switch can be a worthwhile endeavour when the experienced clinician judges that the lack of response or intolerability is related to the pharmacodynamics of the opioid. Clinicians have observed subtle differences in the neurocognitive pharmacodynamics of the opioids, but to date these have evaded clear documentation in the literature. 84 However, switching can be more problematic in some circumstances. When the lack of response is due to factors beyond the capability of opioids always to moderate, such as excessive patient anxiety, then a switch may merely add unnecessary complexity and risk.
Opioid switching is often unrewarding, only adding confusion and therapy gaps when prescribing opioids in the same structural grouping with similar actions, such as a switch from morphine to hydromorphone. 85 At other times, an apparent improvement in analgesia with an opioid switch may be merely due to a placebo response (including regression to the mean) or patient preference driven by the likeability of a particular opioid.
The limited data supporting analgesic benefit with the simultaneous administration of two differing opioids86,87 is best weighed by the clinician against the potential for harm. Taking a pragmatic approach of using only one agent at a time allows the clinician the best opportunity to assess opioid responsiveness clearly. When more than one opioid is used concurrently a greater potential for patient harm exists. 58 The sequence of events is typically confusion in the clinical assessment leading to frustration in both the clinician and patient, and rapid escalation of all doses until an unforeseen collision of side-effects finally occurs.
Conclusion
Our approach to the use of opioids in acute pain management does not need to be one of curtailment driven by fear, but instead one driven by greater knowledge and better assessment of pain from a sociopsychobiomedical perspective. Opioids can be used very effectively in certain circumstances and with least risk when we understand those risks, assess them on a case-by-case basis, and understand how best to avoid them for the individual patient.
