Abstract
The ROTEM®delta (TEM Innovations GmbH, Munich, Germany) has been used extensively worldwide for the assessment of coagulation in cardiac surgical patients. Recently, a new cartridge-based ROTEM device (ROTEM®sigma, TEM Innovations GmbH, Munich, Germany) has become available. In this paper we report an audit of the diagnostic accuracy of the ROTEM sigma for the identification of hypofibrinogenaemia in cardiac surgical patients. We hypothesised that the diagnostic accuracy of the ROTEMsigma for the identification of hypofibrinogenaemia would be in a similar range to that previously reported for the ROTEMdelta. Simultaneous blood samples for Clauss laboratory fibrinogen and ROTEMsigma FIBTEM measurements were obtained immediately after heparin reversal post-cardiopulmonary bypass in 200 adult cardiac surgical patients. The sensitivity, specificity, and positive and negative predictive values for FIBTEM A5 and A10 for the identification of hypofibrinogenaemia (Clauss fibrinogen <1.5 g/l) were calculated. The prevalence of hypofibrinogenaemia across the 200 patients was 8%. The mean sensitivity and specificity of FIBTEM A10 ≤8 mm for the identification of hypofibrinogenaemia were 0.75 and 0.90 respectively, which are in a similar range to that reported in several previous studies using the ROTEMdelta. For FIBTEM A5 ≤6 mm the values were 0.63 and 0.98 respectively. The predictive values were also in a similar range to those previously reported for the ROTEMdelta, with low false negative rates (2% for A10 ≤8 mm; 3% for A5 ≤6 mm). These findings support the use of the ROTEMsigma as an alternative to the ROTEMdelta for the identification of hypofibrinogenaemia post-cardiopulmonary bypass in cardiac surgical patients. However, further studies are required in other settings.
Introduction
Thromboelastometry using the ROTEM®delta (TEM Innovations GmbH, Munich, Germany) has been used extensively worldwide for many years for the assessment of coagulation in cardiac surgical patients.1–3 Thromboelastometry provides both global indices of coagulation, such as the clotting time, INTEM, and EXTEM, and more specific indices of individual factor levels, such as the FIBTEM, which rapidly estimates the fibrinogen contribution to clot formation. 4 Thromboelastometry and other viscoelastic tests have also been used to predict blood loss following cardiac surgery.3,4 Recently, there has been increased interest in the FIBTEM, due to the recognition of the need to correct hypofibrinogenaemia before considering other factor deficiencies as a potential cause of abnormal bleeding.5–9 Also more recently, a user-friendly, cartridge-based ROTEM device has become available (ROTEM®sigma, TEM Innovations GmbH, Munich, Germany).10–12 However, while the diagnostic accuracy of the ROTEMdelta for the identification of hypofibrinogenaemia in cardiac surgical patients has been investigated in many previous studies, 13 there have been few studies using the ROTEMsigma. Moreover, initial studies in non-cardiac surgery patients suggest that the ROTEMdelta and ROTEMsigma do not provide equivalent results.11,14 Therefore, as we had moved to using the ROTEMsigma routinely, we audited its diagnostic accuracy for the identification of hypofibrinogenaemia in our cardiac surgical patients. Our hypothesis was that its diagnostic accuracy would be in a similar range to that previously reported for the ROTEMdelta.
Methods
The audit was performed at the Sir Charles Gairdner Hospital (SCGH), Nedlands, Western Australia, and the St John of God Hospital, (SJOGH), Subiaco, Western Australia. The audit was approved as a quality assurance activity at both sites (SCGH quality activity 37906; SJOGH quality activity 1727) with a waiver of the need for patient consent. The approval process required submission of the audit protocol. The audit was undertaken in compliance with the Standards for Reporting Diagnostic Accuracy (STARD) guidelines, 15 although as a quality assurance activity it was not registered in a clinical trials registry. Data from all patients who had simultaneous ROTEMsigma and laboratory fibrinogen measurements performed following the administration of protamine after separation from cardiopulmonary bypass (CPB) at both sites during the audit period were included. The measurement of both ROTEMsigma and laboratory fibrinogen post CPB was based on routine clinical practice of the anaesthetists participating in the audit, with no patients receiving additional testing or a change in management as a result of being included in the audit. We did not exclude patients who had received preoperative blood products or who had an abnormal fibrinogen preoperatively, because this would not have influenced the relationship between ROTEMsigma and fibrinogen measurements.
We planned to prospectively include 200 patients. This sample size was chosen to limit the 95% confidence intervals (CI) for specificities to <±7% (absolute) of the mean values (assuming a hypofibrinogenaemia prevalence of about 10%). All patients undergoing a cardiac surgical procedure requiring CPB were eligible. This included both elective and emergency coronary artery bypass (CABG) procedures, valve procedures (repair or replacement), atrial septal defect or atrial tumour excisions, combined procedures, redo procedures, or ascending aorta or aortic arch repairs, but excluded procedures in paediatric patients (<16 years) and transplant procedures (neither of which are performed in our institutions).
As per our previous audit, anaesthetic management was at the discretion of the anaesthetist. The heparin dose prior to CPB was 300 IU/kg with further doses of 5,000–10,000 IU given incrementally as necessary to maintain the kaolin activated clotting time >480 s. The CPB circuit was an Affinity Fusion™ Oxygenator with a Carmeda® bioactive surface (Medtronic, Minneapolis, MN, USA) primed with 2 l Plasma-Lyte (Baxter Healthcare Corporation, Deerfield, IL, USA) crystalloid and 10,000 IU heparin. The management of CPB included mild hypothermia (32°–35°C) with a target mean arterial pressure of 50–80 mmHg and a pump flow of 2.4 l/min/m2. Myocardial protection was achieved with a combination of antegrade and retrograde intermittent blood cardioplegia or antegrade crystalloid cardioplegia (Custodiol®, Bensheim, Germany) depending on surgical procedure and surgeon preference. The use of tranexamic acid (up to 15 mg/kg bolus followed by up to 4.5 mg/kg/h) was also dependent upon surgical procedure and surgeon preference. No gelatin- or starch-containing solutions were used.
Blood samples were taken from a radial arterial cannula using a two syringe technique after separation from CPB and the administration of protamine. The first syringe was used to remove 10 ml of dead space fluid. A second syringe was used to collect 5.4 ml of blood, which was transferred into two 2.7 ml citrated blood collection tubes (Vacutainer, Becton Dickinson and Company, Franklin Lakes, NJ, USA). One tube was transported immediately (<5 min) to a pathology laboratory where on receipt, fibrinogen concentration measurement commenced using the Clauss method 16 (STAR Max3, Diagnostica Stago, France). The other tube was used for ROTEMsigma assays, which were performed immediately in the operating theatre suite. We used the disposable ROTEMsigma ‘complete + hep cartridge’ (TEM Innovations GmbH, Munich, Germany). This cartridge runs four channels, although in this audit we were interested particularly in the FIBTEM channel. The FIBTEM channel contains recombinant tissue factor, phospholipids, polybrene (heparin inhibitor), calcium chloride, and cytochalasin D (which inhibits platelets and thereby provides an index of fibrinogen contribution to clot strength independent of platelets). 4 The other channels include the EXTEM, INTEM and HEPTEM. The ROTEMsigma has an automated sequence that initiates the test. The FIBTEM trace is displayed as a clot develops with the amplitude automatically measured at 5 min (A5) and 10 min (A10) after commencement of clot formation. The ROTEMsigma data in the operating theatre were not available to laboratory staff performing the fibrinogen measurements, or vice versa.
We calculated the sensitivity, specificity, and positive (PPV) and negative predictive values (NPV) of ROTEMsigma FIBTEM A5 ≤6 mm and A10 ≤8 mm for the identification of hypofibrinogenaemia, defined as a laboratory fibrinogen <1.5 g/l. This fibrinogen value is a common recommended trigger for fibrinogen administration in the presence of abnormal bleeding published in the consensus guidelines of many professional societies,17–21 and the A5 ≤6 mm and A10 ≤8 mm thresholds are typical thresholds used for the estimation of hypofibrinogenaemia.1–3,13 As both the ROTEMsigma and the laboratory results are reported numerically, no indeterminate values were expected. All data were de-identified.
Results
Simultaneous ROTEMsigma FIBTEM and laboratory fibrinogen measurements were performed in 200 consecutive eligible patients across the two sites over a ten-month period (July 2020 to May 2021). This represented 66% of the total of 304 patients who required CPB for their cardiac surgical procedure across both sites over this period. The remaining patients either had no simultaneous ROTEMsigma and laboratory measurements, or had these performed at other stages of the procedure. Summary demographic and procedural data on the included patients are presented in Table 1. Complete data were collected in all 200 patients, with no missing values.
Patient and procedural characteristics (n = 200).
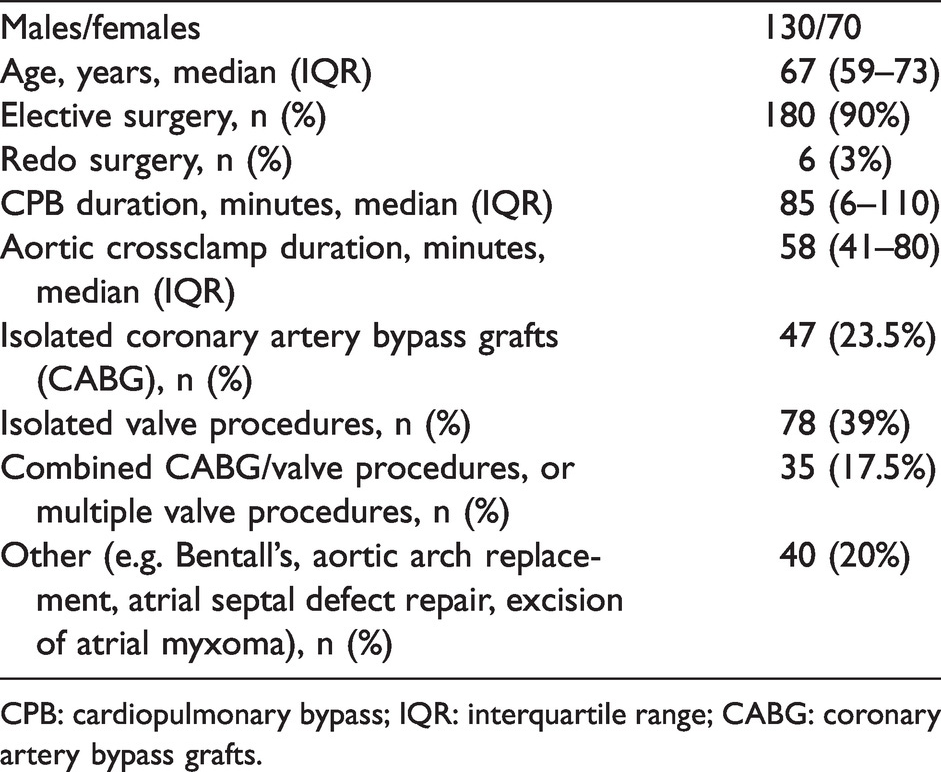
CPB: cardiopulmonary bypass; IQR: interquartile range; CABG: coronary artery bypass grafts.
The laboratory fibrinogen values for the 200 patients ranged from 1.0 to 5.8 g/l (median (interquartile range, IQR) 2.0 (1.7–2.4 g/l)). Sixteen patients had a laboratory fibrinogen <1.5 g/l (hypofibrinogenaemia prevalence 8%). This subset was broadly typical of the 200 patient cohort in terms of age (median (IQR) 60 (38–73 years)), sex distribution (50% female), CPB duration (median (IQR) 95 (69–125 min)), and procedure type (isolated CABG 12.5%, single valve 31.25%, redo procedure 12.5%, combined procedure 12.5%, other 31.25%). Fifteen of the 16 patients were undergoing elective procedures (94%).
The diagnostic accuracy of FIBTEM A5 and A10 for the identification of laboratory fibrinogen <1.5 g/l are shown in Table 2, with actual numbers provided in Tables 3 and 4. All diagnostic accuracy data were rounded up to two decimal places. The mean sensitivity and specificity of FIBTEM A10 ≤8 mm for the identification of hypofibrinogenaemia were 0.75 and 0.90 respectively. For FIBTEM A5 ≤6 mm the values were 0.63 and 0.98 respectively. Figures 1 and 2 present the STARD patient flow diagrams 15 for the A5 and A10 thresholds respectively.
Diagnostic accuracy of ROTEM®sigma (TEM Innovations GmbH, Munich, Germany) FIBTEM A5 ≤6 mm and FIBTEM A10 ≤8 mm for the detection of hypofibrinogenaemia (laboratory fibrinogen <1.5 g/l).
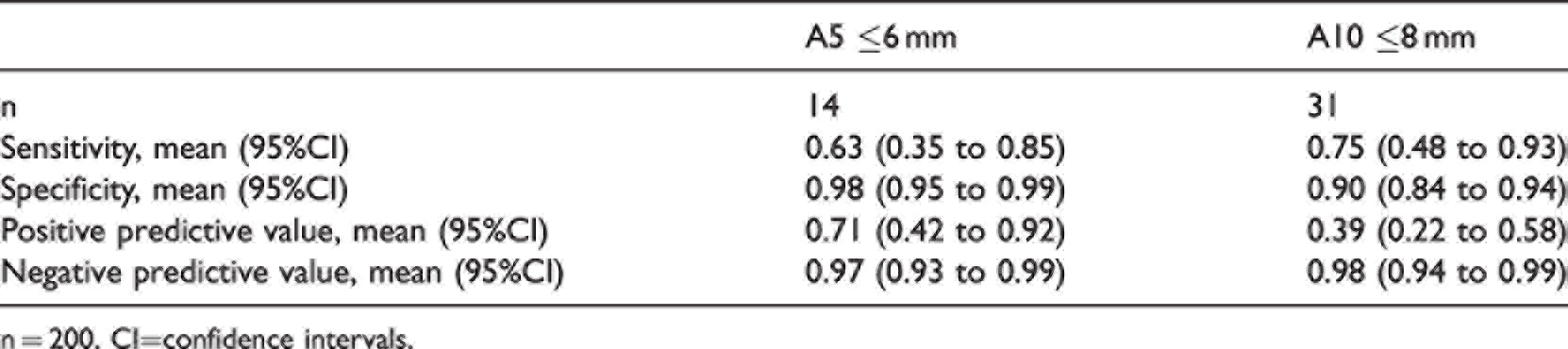
n = 200. CI=confidence intervals.
ROTEM®sigma (TEM Innovations GmbH Munich, Germany) FIBTEM A5 ≤6 mm versus Fibrinogen <1.5 g/l.
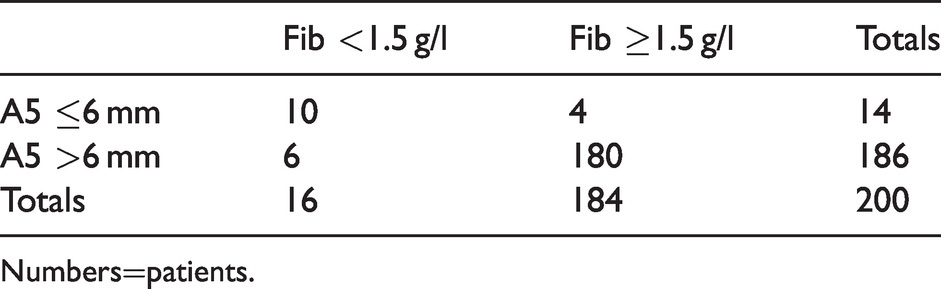
Numbers=patients.
ROTEM®sigma (TEM Innovations GmbH, Munich, Germany) FIBTEM A10 ≤8 mm versus Fibrinogen <1.5 g/l.

Numbers=patients.
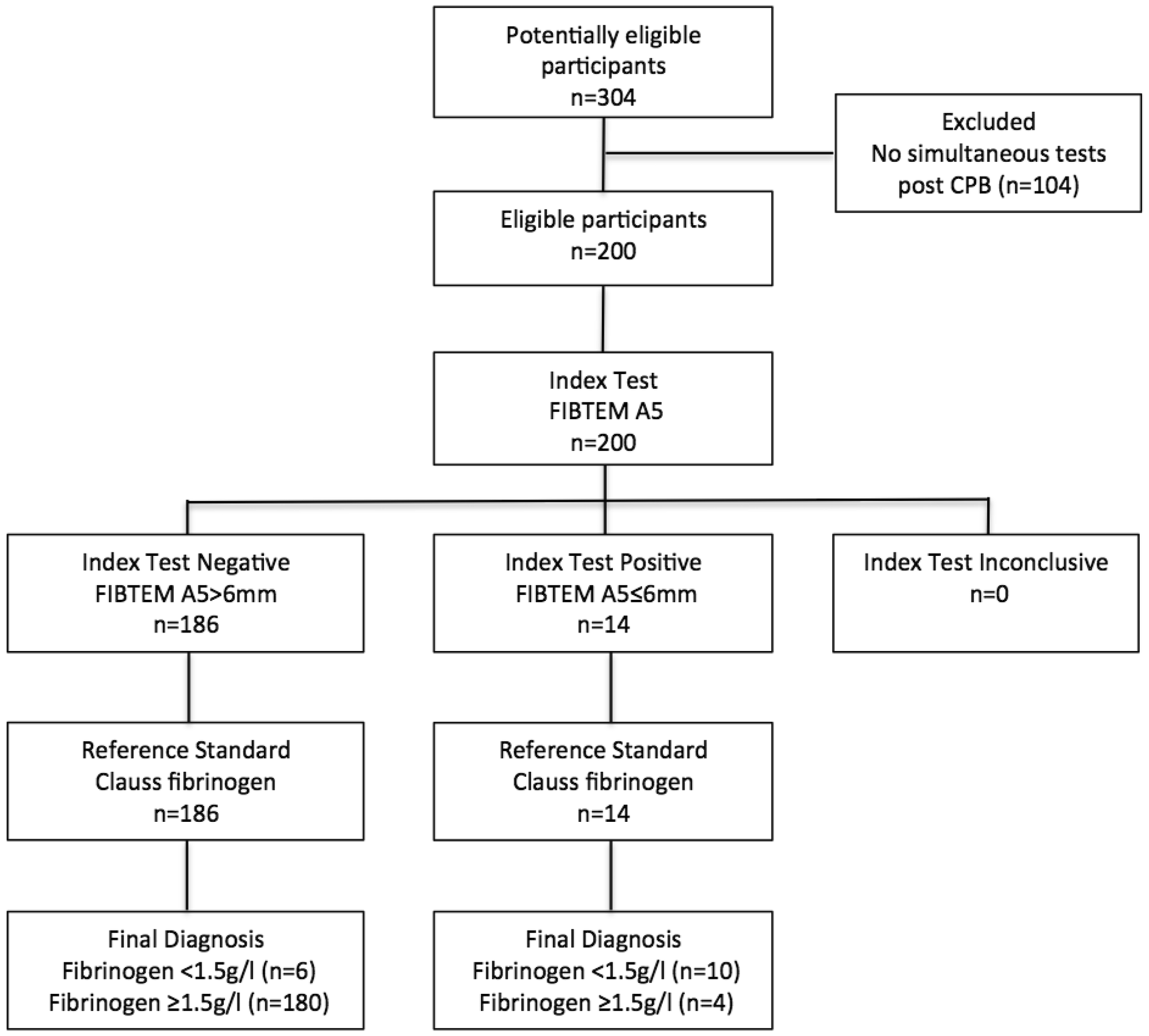
Standards for Reporting Diagnostic Accuracy (STARD) flow diagram 15 for ROTEM®sigma (TEM Innovations GmbH, Munich, Germany) FIBTEM A5 ≤ 6 mm (index test) versus Clauss laboratory fibrinogen (reference standard) for the identification of fibrinogen <1.5 g/l. CPB: cardiopulmonary bypass.
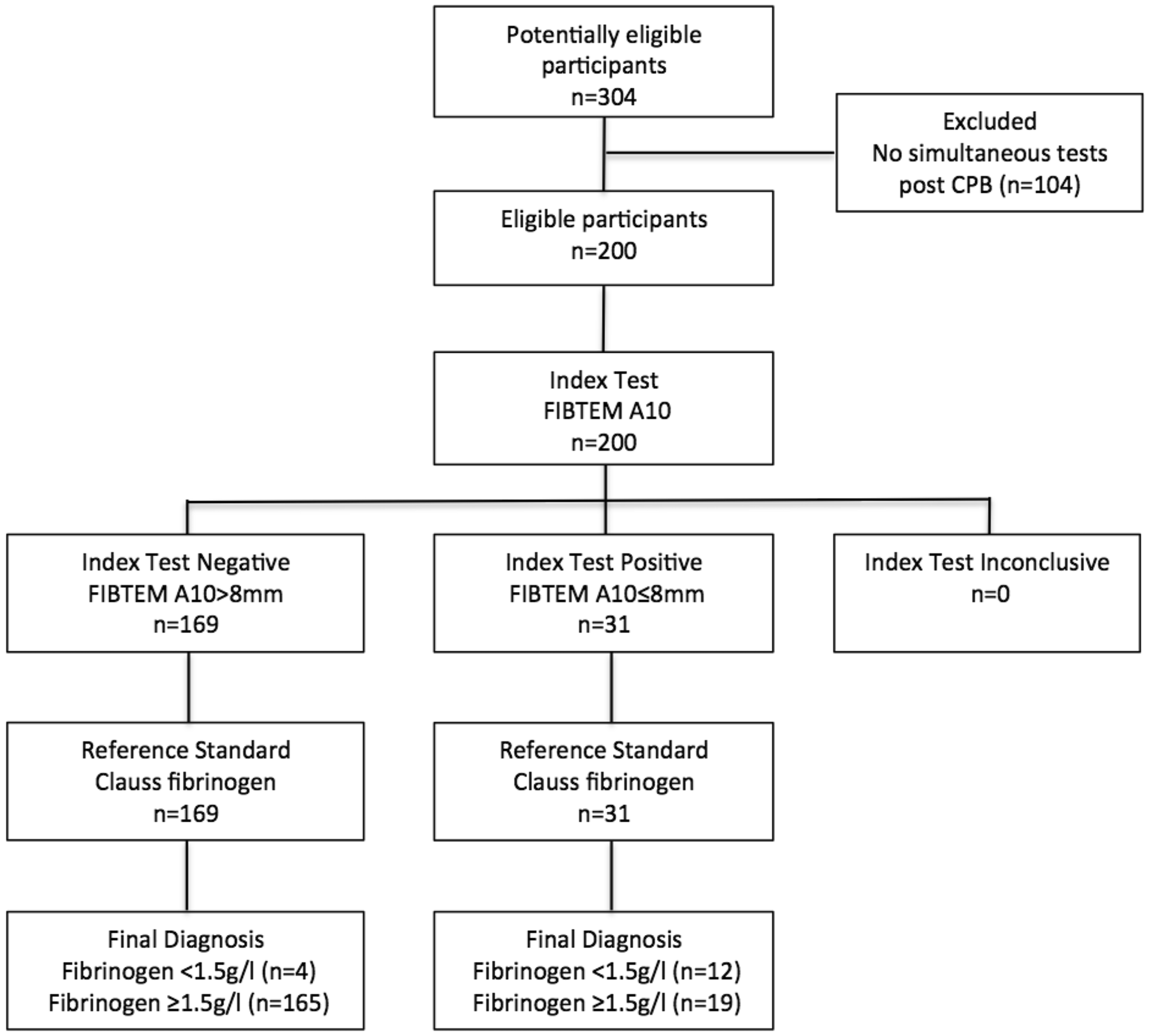
Standards for Reporting Diagnostic Accuracy (STARD) flow diagram 15 for ROTEM®sigma (TEM Innovations GmbH, Munich, Germany) FIBTEM A10 ≤ 8 mm (index test) versus Clauss laboratory fibrinogen (reference standard) for the identification of fibrinogen <1.5 g/l. CPB: cardiopulmonary bypass.
Discussion
We found that the diagnostic accuracy of the ROTEMsigma for the identification of hypofibrinogenaemia (Clauss fibrinogen <1.5 g/l) in this audit was in a similar range to that reported in several previous studies using the ROTEMdelta. 13 For example, the mean sensitivity for a FIBTEM A10 ≤8 mm was 0.75, which was within the range 0.61–0.88 reported in a recent meta-analysis of four ROTEMdelta studies involving 1525 patients. 13 Similarly, the mean specificity was 0.90, which was within the range 0.88–0.94 reported in the same meta-analysis. 13 The current results were also similar to a previous audit we conducted using the ROTEMdelta. 22 For example, the mean sensitivity and specificity in our previous audit for A10 ≤8 mm were 0.62 and 0.90 respectively (versus 0.75 and 0.90 in the current audit); for A5 ≤6 mm the mean sensitivity and specificity in the previous audit were 0.58 and 0.95 respectively (versus 0.63 and 0.98 in the current audit). 22 These findings suggested that the ROTEMsigma has similar diagnostic accuracy to the ROTEMdelta for the identification of hypofibrinogenaemia in cardiac surgical patients.
As in previous studies, the PPVs of the FIBTEM results were lower than the NPVs (Table 2). For example, the mean PPV of the ROTEMsigma A5 ≤6 mm was 0.71, which translates to a false positive rate of 29%. In contrast, the mean NPV was 0.97, which translates to a false negative rate of only 3%. For A10 ≤ 8 mm the mean PPV and NPV were 0.39 and 0.98 respectively, for a false positive rate of 61% and a false negative rate of 2%. This means that a negative result all but excluded the presence of hypofibrinogen-aemia as a cause for abnormal bleeding. A positive result was less definitive, but nevertheless increased the probability of hypofibrinogenaemia substantially (i.e. up to 71% for an A5 ≤6 mm). The A5 ≤6 mm had a similar or higher PPV to an A10 ≤8 mm (0.71 versus 0.39), with similar NPVs (0.97 versus 0.98).
This current prospective audit was similar to but not directly comparable to our previous retrospective audit. 22 In the current audit we obtained blood samples post CPB, after the administration of protamine, rather than prior to separation from CPB. This was to avoid potential artefactual reductions of laboratory fibrinogen by the high concentration of heparin during CPB.23–25 The current study also had a lower prevalence of hypofibrinogenaemia (8% versus 13% in our previous audit). 22 The hypofibrinogenaemia prevalence was closer to the 6% rate reported in a study of 1077 similar cardiac surgical patients by Mace et al. 26 The lower prevalence may explain the higher NPVs we observed.
To our knowledge there have been no previous studies of the diagnostic accuracy of the ROTEMsigma specifically in cardiac surgical patients, so we cannot compare our results to any previous studies. Scala et al. compared ROTEMsigma to standard laboratory coagulation tests, including fibrinogen, in 14 volunteers and 35 patients, ten of whom were undergoing cardiac surgery. 12 They found that with their pooled results the FIBTEM A5 ≤12 mm had 100% sensitivity and 63.6% specificity for the identification of laboratory fibrinogen <1.5 g/l. This finding would not be meaningful for most cardiac surgical patients, because an A5 ≤6 mm would be more typical as the threshold for identifying hypofibrinogenaemia. 25 Bouzat et al. reported the diagnostic performance of thromboelastometry in trauma-induced coagulopathy in two separate trauma centres, one of which used the ROTEMdelta, and the other the ROTEMsigma. 27 The centre using the ROTEMdelta found that an A5 ≤6 mm had 85% sensitivity and 90% specificity for the identification of fibrinogen <1.5 g/l, which was similar to the other centre using a ROTEMsigma (sensitivity 83%, specificity 82%). They concluded that the same ROTEM-based algorithms for trauma-induced coagulopathy could be used for either device. In contrast, Gillissen et al. found only moderate correlation between the ROTEMdelta and ROTEMsigma devices for FIBTEM A5 or A10 in women with postpartum haemorrhage (r ≤ 0.74). 11 Another study, by Schenk et al., found high correlations between ROTEMsigma and ROTEMdelta measurements in 30 healthy volunteers and 30 patients with abnormal coagulation, but did not include FIBTEM measurements. 10 Field et al. cautioned that the high correlations observed in the Shenk et al. study were to be expected using their methodology, but that this did not indicate that the results of the two devices were equivalent. 14 Given the queries raised in previous studies, our findings provide reassuring data on the diagnostic accuracy of the ROTEMsigma FIBTEM measurements for the identification of hypofibrinogen-aemia. in cardiac surgical patients. Nevertheless, further studies would be required to compare ROTEMsigma results to ROTEMdelta results in the same patients.
While the ROTEMsigma displays several coagulation indices, in this audit we were interested specifically in the accuracy of the FIBTEM for the identification of hypofibrinogenaemia. This is because most other ROTEM coagulation indices, including the EXTEM, are influenced by the fibrinogen level,9,28 so other abnormalities are difficult to interpret unless hypofibrinogenaemia has first been excluded as a cause. Moreover, the current clinical priority in the event of abnormal bleeding (after ensuring reversal of heparin and adequate functional platelets) is to correct hypofibrinogenaemia before investigating other potential coagulation factor deficiencies.6,9,17-21, 25
There are several limitations in this study. Importantly, we assessed only diagnostic accuracy aspects of the ROTEMsigma, not its clinical utility in predicting or reducing blood loss in cardiac surgical patients. Far larger studies would be required to assess these outcome measures. There are also other limitations that might influence the ‘generalisability’ of our results. Our findings apply mostly to the early post CPB period after the reversal of heparin, and may not apply to other periods or after blood products have been administered. Fibrinogen levels are also likely to be lowest during this period due to the haemodilution occurring during CPB, and are likely to increase over the following 24 h unless further bleeding occurs. 29 Therefore, serial measurements of ROTEM and other coagulation parameters are recommended. Clauss fibrinogen values may also differ between laboratories depending on the reagents used, so clinicians should consider the normal ranges and target values for their own institution. 25
There was also the potential for patient selection bias, given that not all patients undergoing CPB were eligible for inclusion. 30 However, recruitment was consecutive for all patients who had simultaneous ROTEMsigma and laboratory fibrinogen measurements post CPB. Moreover, the included patients covered a broad range of demographics and procedures (Table 1) and the subset of patients with hypofibrinogenaemia was similar to the full cohort in terms of age, sex, CPB duration and procedure mix, making significant selection bias unlikely. Our findings also apply only to a hypofibrinogenaemia threshold of <1.5 g/l, our observed hypofibrinogenaemia prevalence of 8%, the demographic and procedural mix of our patients, and the spectrum (spread) of fibrinogen levels we observed. 30 While we anticipate that our patients and practice would be typical of many cardiac surgery centres, further studies would be required to assess the diagnostic accuracy of the ROTEMsigma using other thresholds or other patient groups or scenarios with a differing prevalence or definition of hypofibrinogenaemia (e.g. postoperatively) or for massive transfusion. Nevertheless, our sensitivities and specificities could be used to calculate positive (sensitivity/(1-specificity) and negative (1-sensitivity/specificity)) likelihood ratios, which could be used to estimate post-test probabilities of hypofibrinogenaemia (at the thresholds we used) for differences in prevalence (pre-test probability) using a Fagan’s nomogram. 30 We could also provide area under the receiver operator characteristic curve data, 30 on request, for investigators undertaking pre-registered meta-analyses or systematic reviews.
The strengths of our audit relate to the close compliance to the STARD guidelines, 15 including the blinding of those performing the ROTEMsigma tests to the results of the laboratory fibrinogen tests (and vice versa), thereby excluding observer bias. 30 Another strength was taking a diagnostic accuracy approach rather than a clinical agreement approach. The United States Food and Drug Administration recommends taking a diagnostic accuracy approach to diagnostic tests, because this provides information that will influence clinical decision-making in relation to clinically relevant thresholds. 31 In contrast, correlation and clinical agreement studies apply to the whole range of possible results, including those that would not prompt a clinical intervention (e.g. very high values).30,32
While the Clauss laboratory method remains the current reference standard for fibrinogen measurement at an institutional level, its clinical utility is limited by the minimum 45–60 min delay before obtaining a result. 25 This delay may be of little concern in stable patients. However, in patients with excessive or abnormal bleeding a decision has to be made sooner about whether to correct hypofibrinogenaemia as a first line of coagulation therapy. The benefit of ROTEM technology is that it can provide information on the likelihood of hypofibrinogenaemia within seven to 12 min of commencement of the test. The additional benefit of the ROTEMsigma is its ease of use at the point of care, facilitating even earlier commencement of the test, and thereby even more rapid results. Still, these more rapid results would have little merit if they were less accurate than ROTEMdelta results. Our findings indicated that the diagnostic accuracy of the ROTEMsigma for the identification of hypofibrino-genaemia in cardiac surgical patients was in a similar range to previous reports for the ROTEMdelta. This supports the use of the ROTEMsigma as an alternative to the ROTEMdelta in cardiac surgical patients. We would encourage further audits of the diagnostic accuracy of the ROTEMsigma in other settings and in other patient groups (e.g. obstetrics, trauma, liver transplantation).
Footnotes
Author Contribution(s)
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
