Abstract
Hypofibrinogenaemia during cardiac surgery may increase blood loss and bleeding complications. Viscoelastic point-of-care tests provide more rapid diagnosis than laboratory measurement, allowing earlier treatment. However, their diagnostic test accuracy for hypofibrinogenaemia has never been reviewed systematically. We aimed to systematically review their diagnostic test accuracy for the identification of hypofibrinogenaemia during cardiac surgery. Two reviewers assessed relevant articles from seven electronic databases, extracted data from eligible articles and assessed quality. The primary outcomes were sensitivity, specificity and positive and negative predictive values. A total of 576 articles were screened and 81 full texts were assessed, most of which were clinical agreement or outcome studies. Only 10 diagnostic test accuracy studies were identified and only nine were eligible (ROTEMdelta 7; TEG5000 1; TEG6S 1, n = 1820 patients) (ROTEM, TEM International GmbH, Munich, Germany; TEG, Haemonetics, Braintree, MA, USA). None had a low risk of bias. Four ROTEM studies with a fibrinogen threshold less than 1.5–1.6 g/l and FIBTEM threshold A10 less than 7.5–8 mm had point estimates for sensitivity of 0.61–0.88; specificity 0.54–0.94; positive predictive value 0.42–0.70; and negative predictive value 0.74–0.98 (i.e. false positive rate 30%–58%; false negative rate 2%–26%). Two ROTEM studies with higher thresholds for both fibrinogen (<2 g/l) and FIBTEM A10 (<9.5 mm) had similar false positive rates (25%–46%), as did the two TEG studies (15%–48%). This review demonstrates that there have been few diagnostic test accuracy studies of viscoelastic point-of-care identification of hypofibrinogenaemia in cardiac surgical patients. The studies performed so far report false positive rates of up to 58%, but low false negative rates. Further diagnostic test accuracy studies of viscoelastic point-of-care identification of hypofibrinogenaemia are required to guide their better use during cardiac surgery.
Keywords
Introduction
Excessive blood loss during cardiac surgery is associated with an increased incidence of blood transfusion, re-operation, prolonged hospital stay and death.1–4 Excessive blood loss may be due to surgical factors alone, but may be compounded by impaired coagulation if plasma coagulation factors fall below critical levels. 4 This is more likely if cardiopulmonary bypass (CPB) is used, due to the associated haemodilution.4–7 Ongoing blood loss may exacerbate the haemodilution resulting in a vicious cycle of coagulopathy and further bleeding.
Over the past decade, the critical role of normal fibrinogen levels (e.g. ≥1.5 g/l) in promoting effective coagulation during cardiac surgery has been recognised.8–16 It is now considered a priority to correct fibrinogen levels before other plasmatic coagulation factor deficiencies. 17 This is endorsed by many anaesthetic and surgical organisations.17–21 Moreover, long shelf-life purified fibrinogen concentrates are now available for this purpose in many countries. 22
Unfortunately, obtaining laboratory confirmation of impaired coagulation, including low fibrinogen levels, typically takes 45 minutes or more. 23 , 24 During this time further blood loss and bleeding complications could occur. 25 , 26 In contrast, viscoelastic tests (e.g. thrombelastography, thromboelastometry) can provide rapid (<15 minutes) results at the point of care (POC). 23 , 24 Moreover, both thrombelastography (TEG; Haemonetics, Braintree, MA, USA) and thromboelastometry (ROTEM; TEM International GmbH, Munich, Germany) now include assays for estimation of fibrinogen levels. For the TEG this is known as ‘functional fibrinogen’ (FF), and for the ROTEM this is known as FIBTEM. 23 , 24 These assays have become part of the routine panel of viscoelastic tests available during cardiac surgery, 27 and have been incorporated into many perioperative coagulation management algorithms.12,28–30
There have been many reviews,12,28–31 including systematic reviews 32–35 and health technology assessments, 36 , 37 on the correlation or agreement of these viscoelastic POC tests with laboratory standards in cardiac surgical patients, or on their influence on outcomes. Most have supported the use of viscoelastic POC tests in patients who are acutely bleeding, with many professional organisations and consensus guidelines recommending their use to reduce blood loss perioperatively.17–21,38 Yet correlation and clinical agreement studies do not provide information on the ability of a new test to identify a target condition accurately.39–41 For example, high correlation can occur despite large differences in the absolute values of two tests. 39 Similarly, clinical agreement estimates such as bias and precision refer to agreement across the full range of possible values, without focusing on the threshold that would identify a target condition39–41 and prompt further diagnostic or therapeutic interventions. For this reason the US Food and Drug Administration (FDA) recommends the use of diagnostic test accuracy (DTA) studies (i.e. sensitivity, specificity and positive predictive value (PPV) and negative predictive value (NPV) at the threshold for clinical interventions) to assess new diagnostic tests when possible. 42
To our knowledge, there have been no previous systematic reviews of the diagnostic accuracy of viscoelastic POC estimates of hypofibrinogenaemia in cardiac surgical patients. Yet the value of viscoelastic POC estimates of hypofibrinogenaemia depends on their diagnostic accuracy, as false positives could result in unnecessary fibrinogen replacement, and false negatives could leave patients at continued risk of excessive bleeding.
We therefore undertook a systematic review of the DTA of viscoelastic POC tests (versus a laboratory standard) for the identification of hypofibrinogenaemia in cardiac surgical patients. The primary aim was to assess their sensitivity, specificity and PPV and NPV, in order to calculate false positive and false negative rates. The secondary aims were to perform a meta-analysis if possible and to identify potential sources of heterogeneity.
Methods
This review is reported as per the recommendations of the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement. 43 The protocol was registered in the International Prospective Register of Systematic Reviews (PROSPERO, https://www.crd.york.ac.uk/PROSPERO/) on 7 January 2019 as ‘The diagnostic accuracy of viscoelastic point of care identification of hypofibrinogenaemia in cardiac surgical patients’ (CRD42019119223).
Eligibility criteria
All DTA studies reporting the sensitivity and specificity of one or more viscoelastic POC assessments (index test) of fibrinogen (target condition) versus a laboratory standard (reference test) in cardiac surgical patients of any age (participants) were potentially eligible. Studies on participants with known pre-existing coagulation defects were ineligible. The index tests must have been performed at the same time as the reference test, or within an interval during which no further blood loss, dilution, or fibrinogen administration occurred. Studies in which all tests were completed before surgery commenced were ineligible. The reference test must have been a laboratory standard, such as the Clauss method or equivalent. 44 Hypofibrinogenaemia was defined as a plasma fibrinogen level less than 1.5 g/l or less than 2 g/l, based on the most common thresholds recommended by professional organisations for supplementation of fibrinogen in cardiac surgical patients who are bleeding.17–21,38
Search strategy
The following electronic databases were searched for studies in English published between 1 January 2000 and 31 December 2018. No DTA filters were used.
Cochrane Library PubMed Embase DARE Health Technology Assessment Database Web of Science Science Citation Index Conference Citation Index Clinical Trials.gov
The search terms were (viscoelast* OR thromboelast* OR TEG OR thrombelast* OR ROTEM OR FIBTEM OR Sonoclot OR SCT) AND (fibrinogen OR hypofibrinogen*). If more than 500 studies were identified in a database, terms for participants were added, i.e. AND (cardio* OR ‘cardiac surgery’).
Non-electronic literature searches included correspondence with manufacturers of commercially available viscoelastic POC tests and authors of related DTA studies. Reference lists of relevant primary and secondary source articles were hand searched.
The search results were exported into a custom Endnote reference library (Endnote X8, Clarivate Analytics, Philadelphia, PA, USA) using Endnote reference management software to remove duplicates, matching on title, and if necessary, authors, and year of publication.
Selection of studies
One author (NMG) screened all titles and abstracts and obtained full-text versions of all studies that included diagnostic accuracy terms (i.e. sensitivity, specificity, positive or negative predictive value, or receiver operator characteristic) in their title, abstract, or key words. Studies on correlation, clinical agreement, or broader impact (i.e. outcome) were excluded, unless DTA data were included and could be reliably extracted. Both authors then independently scrutinised all selected full-text versions for eligibility. All studies for which one or more eligibility criteria were scored as ‘unclear’ by either author and all studies in which assessments of eligibility were scored differently between authors were discussed, and if consensus could not be reached a third reviewer was available to make the final decision on inclusion or exclusion.
Data extraction
Both authors independently extracted details of each study’s participants, recruitment strategy, surgical complexity and flow and timing of blood sampling. For each index and reference threshold comparison, the sensitivity and specificity of the index test for the identification of hypofibrinogenaemia was recorded, along with the prevalence of hypofibrinogenaemia at the reference threshold specified; 2 × 2 diagnostic tables were constructed for calculation of predictive values.
Assessment of methodological quality and applicability
Risk of bias and applicability of each eligible study was assessed by both authors independently using the quality assessment of diagnostic accuracy studies-2 (QUADAS-2) tool untailored. 45 All studies for which one or more assessment items were scored as ‘unclear’ by either reviewer and all studies in which any items were scored differently between reviewers were discussed by the two reviewers, and if consensus could not be reached a third reviewer was available to make the final decision on the risk of bias or applicability in relation to the item.
Data analysis
All analyses and plots were performed using Stata (Stata/IC v15.1; College Station, TX, USA).
For studies in which data were provided for more than one index test threshold, only data for the prespecified index test threshold or the threshold identified by the authors as providing the best balance of sensitivity and specificity were analysed.
If studies involved pooling of results of samples taken at repeated timepoints in the same patients, for purposes of analysis, display and calculation of 95% confidence intervals (CIs), the number in each cell of the diagnostic 2 × 2 table was divided by the number of repeated samples. In this way, the 95% CI reflected the sample size based on the number of participants, not the number of samples.
Forest plots were constructed to display the sensitivity and specificity, and PPV and NPV (with their 95% CI) of the index tests for their reference comparisons across studies. Due to the anticipated heterogeneity, no weighted summary estimates were calculated. Heterogeneity across studies using similar index tests and thresholds, and between studies using different index tests and thresholds, was assessed when possible by visual inspection.
If there was sufficient homogeneity across studies in relation to index tests and reference thresholds, a meta-analysis was planned using the hierarchical summary receiver operator characteristic approach. 46 , 47 Sensitivity analyses were planned for studies in which index or reference thresholds were relative outliers to other studies in the meta-analysis. Further subgroup analyses were planned only if there were sufficient studies identified.
Reporting bias
If there was homogeneity across studies, a Deeks test was planned to assess reporting bias.48–50
Results
Search results
The search of electronic databases identified 1312 potentially relevant studies. Hand searching identified a further seven studies. After removal of duplicates, there were 576 studies. After screening of titles and abstracts (if published electronically), 495 were excluded. Full-text versions of the remaining 81 studies were assessed. There was a total of ten studies with diagnostic accuracy data and nine met the eligibility criteria (n = 1820 patients).51–59 The search results are summarised in the PRISMA flow diagram (Figure 1). 43
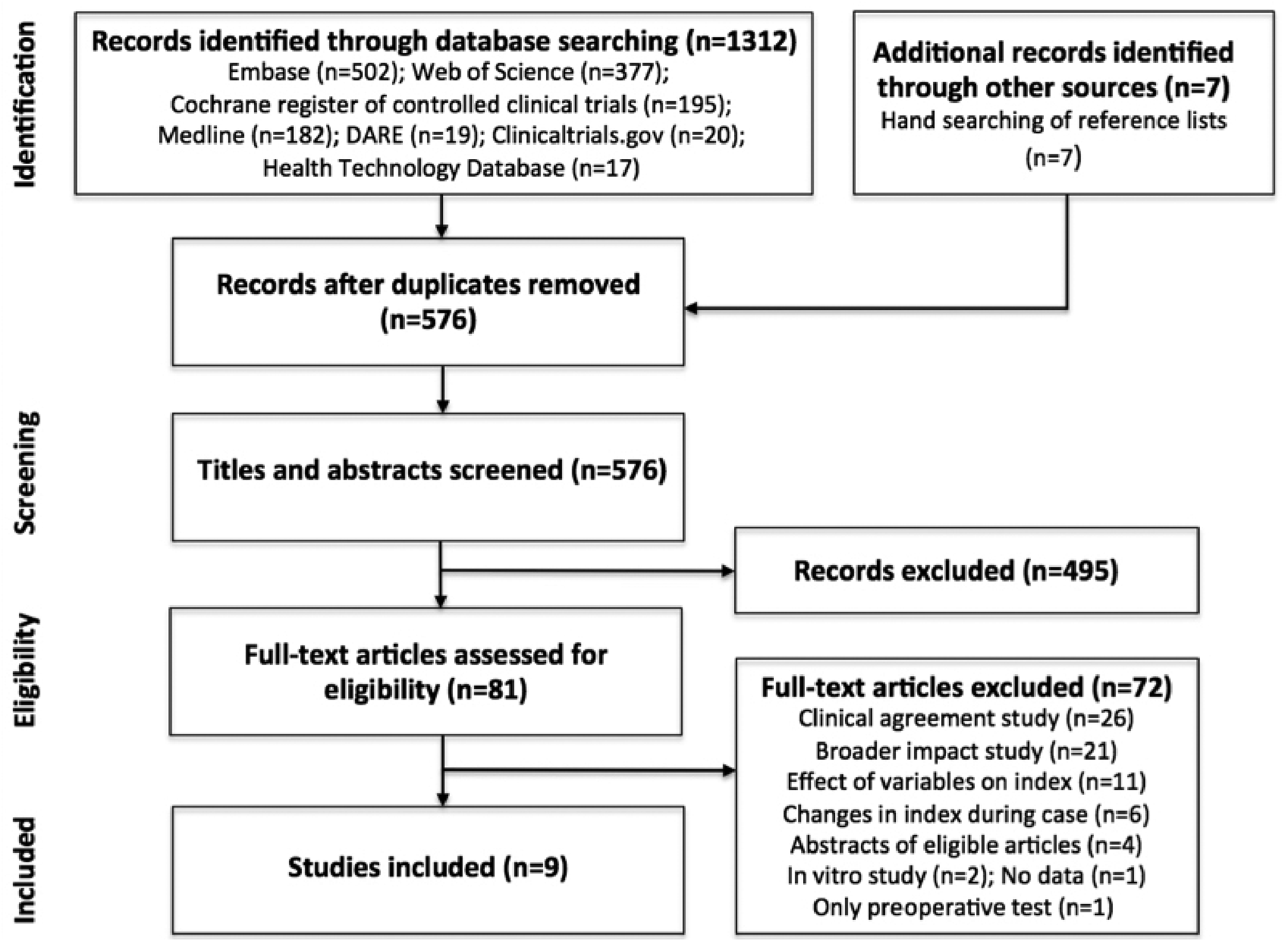
Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) diagram. 43
While this systematic review involved a formal search for studies published only up to 31 December 2018, a subsequent post hoc search by the authors using the same search criteria in the same databases for studies published during 2019 revealed no further diagnostic accuracy studies of viscoelastic POC identification of hypofibrinogenaemia in cardiac surgical patients
Details of included studies
There were five prospective observational studies, two retrospective observational studies, one retrospective audit and one prospective audit (Table 1). All were journal articles published between 2012 and 2018. Seven studies were on the ROTEMdelta device,51–57 with one study was on the TEG5000 58 and one was on the TEG6S. 59 The reference standard was Clauss fibrinogen in all studies. The setting for all studies was a cardiac surgical centre in a major metropolitan or university hospital. None of the studies acknowledged funding support from a manufacturer of a viscoelastic device.
Details of included articles.
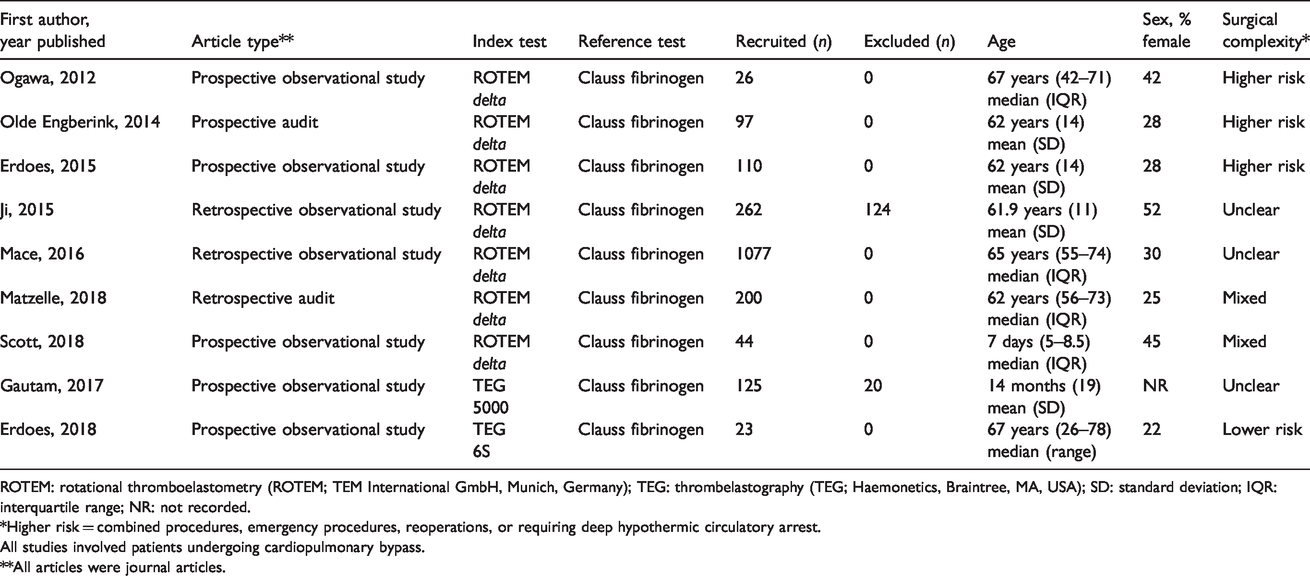
ROTEM: rotational thromboelastometry (ROTEM; TEM International GmbH, Munich, Germany); TEG: thrombelastography (TEG; Haemonetics, Braintree, MA, USA); SD: standard deviation; IQR: interquartile range; NR: not recorded.
*Higher risk = combined procedures, emergency procedures, reoperations, or requiring deep hypothermic circulatory arrest.
All studies involved patients undergoing cardiopulmonary bypass.
**All articles were journal articles.
Characteristics of participants and procedures
The number of patients in each included study ranged from 23 to 1077 (total 1820 patients) (Table 1). Seven studies were in adults and two were in children or neonates. 57 , 58 The proportion of female participants ranged from 0.22 to 0.52, although one study did not report this proportion. 58 Three studies were on higher risk surgical patients (i.e. combined procedures, emergency procedures, reoperations, or requiring deep hypothermic circulatory arrest); one study specifically excluded higher risk patients; two included a combination of higher risk and lower risk patients, and three did not specify higher risk or lower risk (Table 1). All nine studies involved patients undergoing CPB.
Two studies included data for two fibrinogen thresholds (i.e. <1.5 g/l and <2 g/l). 52 , 58 The remainder included data for only one fibrinogen threshold (<1.5 g/l or <2 g/l). Three studies involved analysis of data obtained from repeated samples within the same patient. 51 , 57 , 59 The remainder involved analysis of data obtained from only one sample point per patient.
Details of studies excluded after assessment of their full texts
Seventy-two studies were excluded after assessment of their full-text versions. Twenty-six of these were on clinical agreement (reporting correlation, summary differences, or Bland–Altman plots). Twenty-one were on outcomes (e.g. on blood loss, blood transfusion, or bleeding complications), or on predicting adverse outcomes. Four studies were abstracts of other articles identified in the search. Six studies reported changes in index tests during cardiac surgical procedures with no diagnostic accuracy component. Eleven studies focused on the effect of other variables on fibrinogen measurements. Two studies were in vitro. One study was a DTA study but reported only preoperative results. One letter to the editor commented only on methods.
Details of studies excluded after assessment of their full texts, including the reasons for exclusion, are provided in Supplementary Appendix A (available online).
Risk of bias and applicability concerns
None of the studies had a low risk of bias in all domains of the QUADAS-2 tool (Figure 2). In relation to patient selection, only two studies specified consecutive recruitment, 53 , 59 and none specifically commented on whether the tests were blinded. In two studies, several patients were excluded after study completion, rendering these studies at high risk of patient selection bias.54–58 In only four studies did the methods confirm that the index and reference tests were performed on blood samples collected at the same time. 52 , 53 , 56 , 59 In one study, there was a 45 minute interval between the index and reference test. 55 In the remaining five studies the flow and timing were not specified.
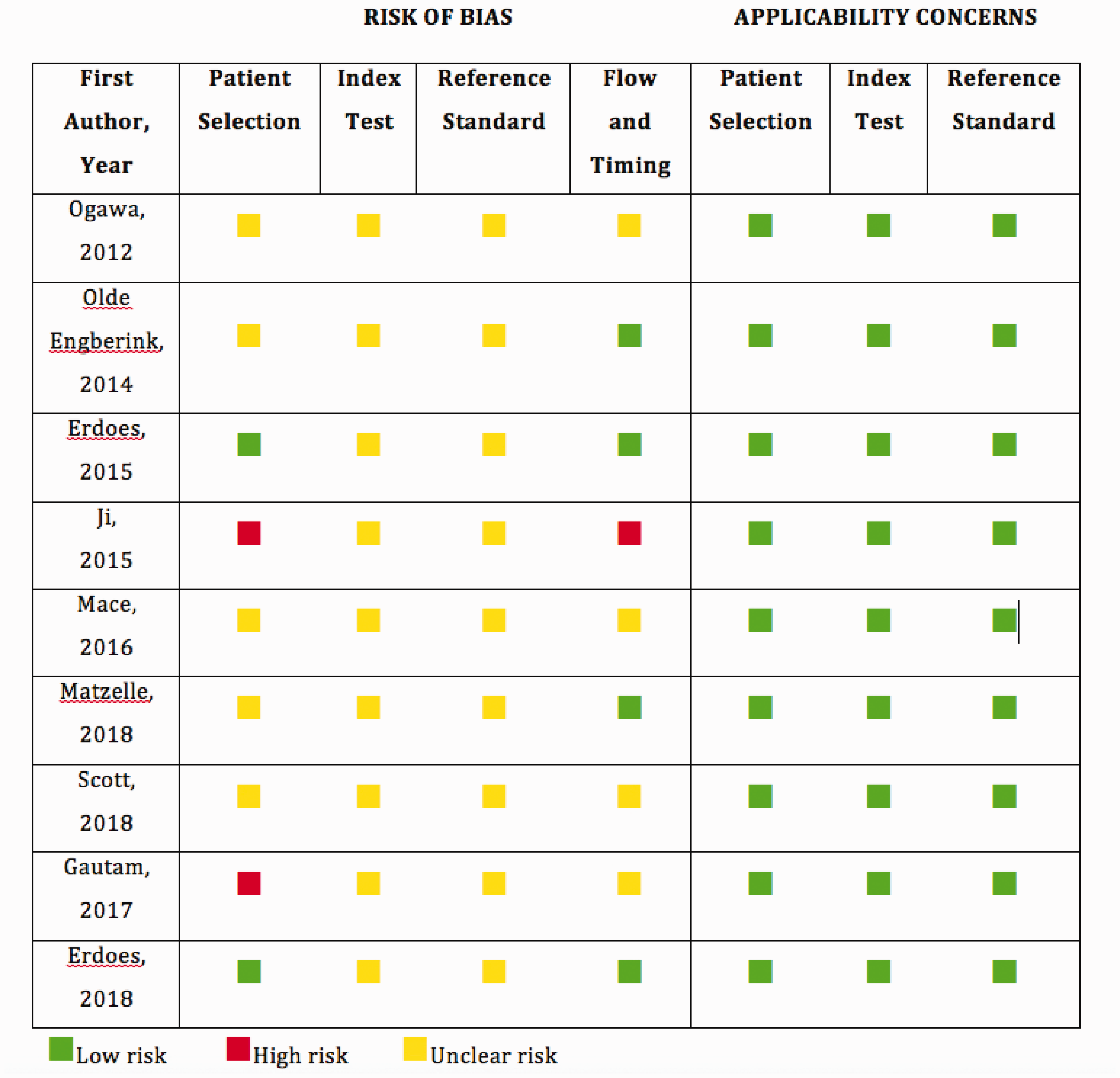
Quality assessment of diagnostic accuracy studies-2 (QUADAS-2) risk of bias and applicability concerns of included studies. 45
In contrast to the risk of bias, there were no applicability concerns in relation to patient selection, index tests, or reference standards, in any of the studies. 45
Diagnostic accuracy of ROTEM studies (n = 1692 patients)
There were four ROTEM studies reporting the DTA for a reference fibrinogen threshold less than 1.5–1.6 g/l and a FIBTEM A10 threshold of less than 7.5–8 mm (n = 1525 patients, Figure 3(a)).54–57 The point estimates of their sensitivities ranged from 0.61 to 0.88 (see Figures for 95% CIs), and specificities from 0.88 to 0.94. The PPVs ranged from 0.42 to 0.81 and the NPVs ranged from 0.74 to 0.98 (Figure 3(b)). These data represent false positive rates of 19%–58% and false negative rates of 2%–26%. A fifth study (n = 97 patients) with a higher and earlier FIBTEM threshold (A5 <9.5 mm) had a sensitivity of 1.0, but a specificity of only 0.54 and a PPV of only 0.44. 52 There were two studies with a reference fibrinogen threshold less than 2 g/l and a FIBTEM threshold A10 less than 9.5 mm (n = 70 patients, Figure 4(a)). 51 , 58 These demonstrated similar false positive (25%–46%) and false negative (7%–10%) rates to the four ROTEM A10 less than 7.5–8 mm studies for the lower fibrinogen threshold (<1.5–1.6 g/l) (Figure 4(b)).
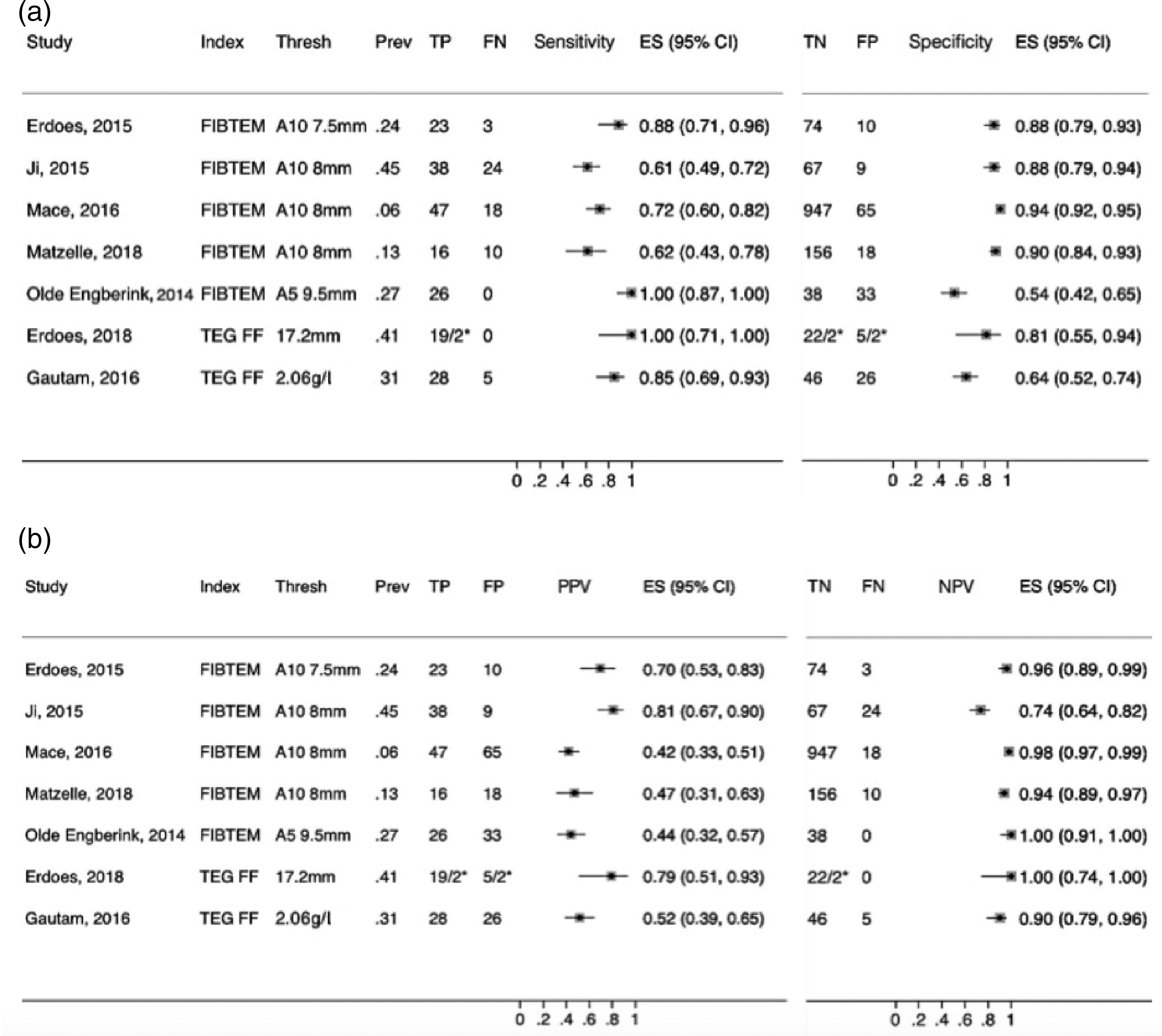
Sensitivity and specificity (a) and positive predictive value (PPV) and negative predictive value (NPV) (b) of index tests for the identification of reference fibrinogen less than 1.5 g/l (except Erdoes, 2015 <1.6 g/l) by index test (Index) and threshold (Thresh).
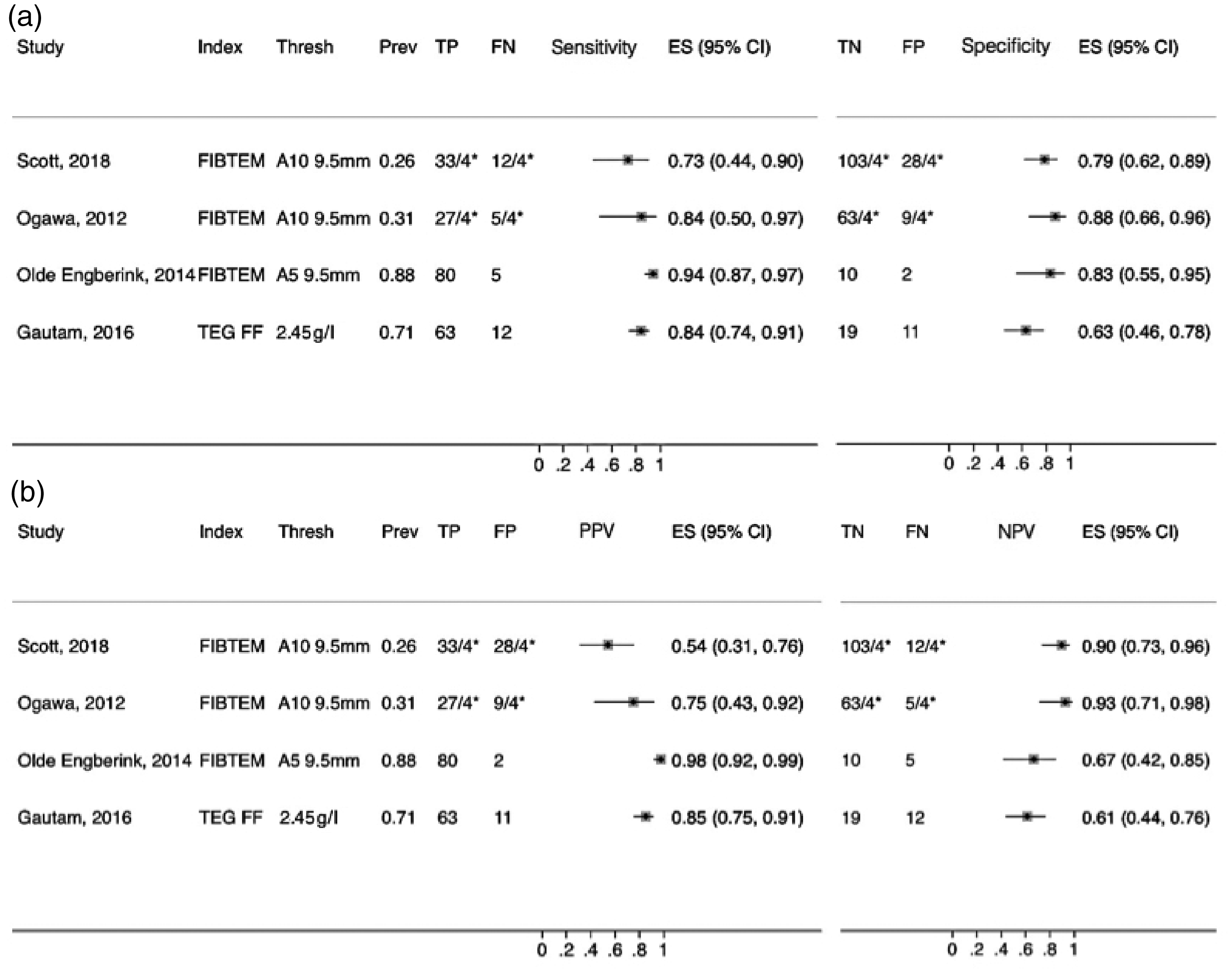
Sensitivity and specificity (a) and positive predictive value (PPV) and negative predictive value (NPV) (b) of index tests for the identification of reference fibrinogen less than 2 g/l by index test (Index) and threshold (Thresh).
Diagnostic accuracy of TEG studies (n=128 patients)
For the single TEG5000 study (n = 105 patients), the sensitivity point estimate for identifying fibrinogen less than 1.5 g/l was 0.85, specificity was 0.64, with a PPV of 0.52 and NPV of 0.90 (Figure 3). 58 For the single TEG6S study (n = 23 patients), the sensitivity was 1.0 and the specificity was 0.81, with a PPV of 0.79 and a NPV of 1.0. 59 The TEG5000 study also included data for a higher fibrinogen threshold (<2 g/l) with similar sensitivities, specificities and PPVs, but with lower NPVs (Figure 4). Overall, the TEG studies demonstrated similar false positive (21%–48%) and false negative (0–10%) rates to the ROTEM studies.
Meta-analysis
A meta-analysis was performed on the five ROTEM studies with similar fibrinogen thresholds (<1.5–1.6 g/l).52–56 The summary sensitivity for the five studies using a hierarchical summary receiver operator characteristic (HSROC) model 46 , 47 was 0.84 with a summary specificity of 0.87 (Figure 5(a)). The 95% confidence and prediction regions were both wide. (Figure 5(a)).
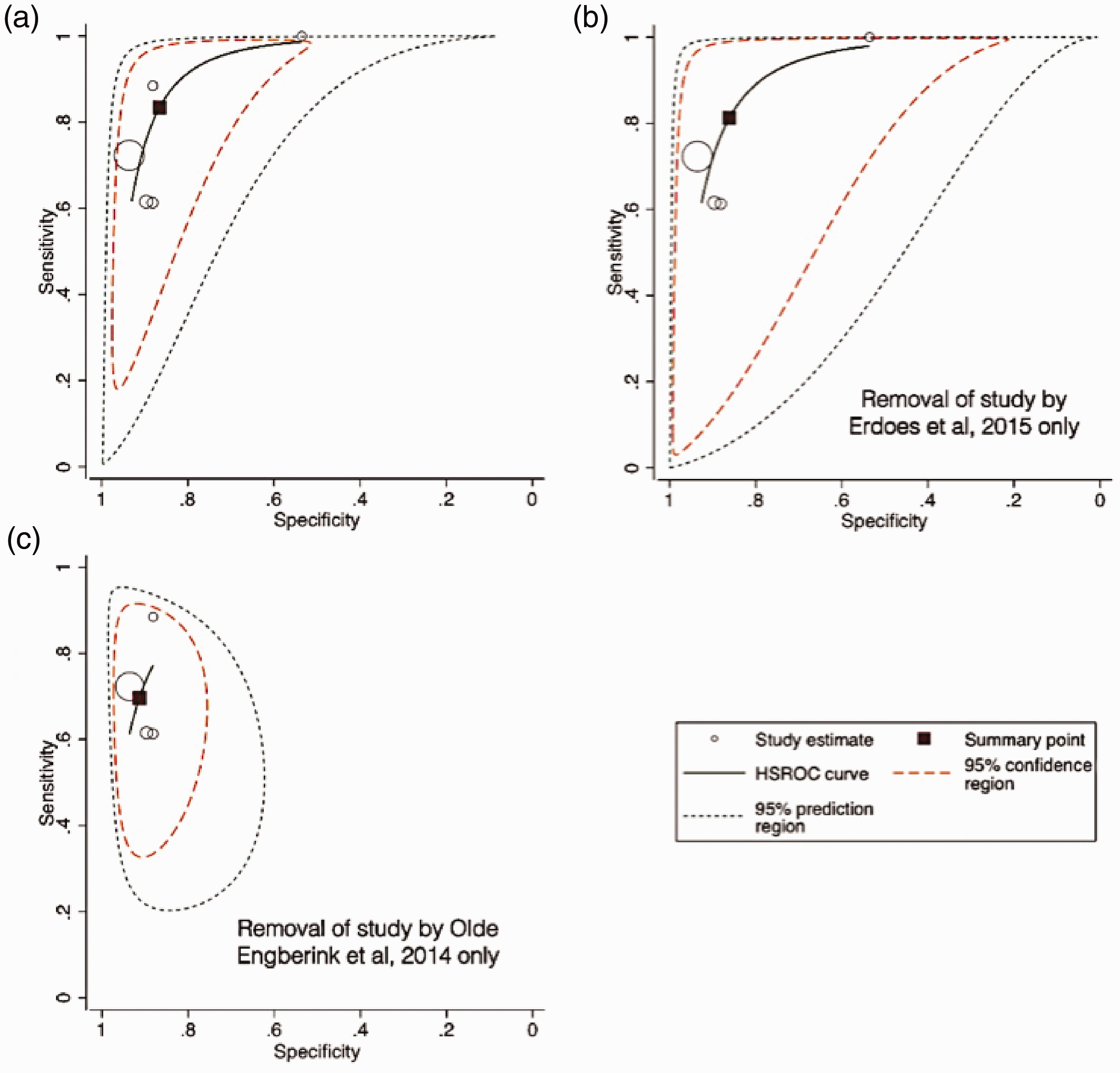
(a) Hierarchical summary receiver operator characteristic (HSROC) curves of five included ROTEM studies (ROTEM; TEM International GmbH, Munich, Germany) with similar index thresholds (FIBTEM A10 <7.5 mm, n = 1; FIBTEM A10 <8 mm, n = 3; FIBTEM A5 <9.5 mm, n = 1) and reference thresholds (<1.5 g/l, n = 4; <1.6 g/l, n = 1). The plot includes the summary point with its 95% confidence region and 95% prediction region, and a HSROC curve. (b) The same plot as (a) after removal of the study by Erdoes et al., 2015, which had a slightly lower index threshold (<7.5 mm) and a slightly higher reference threshold (<1.6 g/l) than the other four studies. (c) The same plot as (a) after removal of the study by Olde Engberink et al., 2014, which had an earlier and higher amplitude index threshold (A5 <9.5 mm) than the other four studies.
As the study by Erdoes et al. 53 had a slightly higher reference threshold to the other four studies (<1.6 g/l versus <1.5 g/l), a sensitivity analysis was performed, with the results presented in Figure 5(b). Removing this study had little effect on the summary points: sensitivity 0.81 versus 0.84; specificity 0.86 versus 0.87. Similarly, as the study by Olde Engberink et al. 52 had an earlier and higher amplitude FIBTEM threshold (A5 9.5 mm) than the other four studies (A10 7.5–8 mm), another sensitivity analysis was performed, with the results presented in Figure 5(c). In contrast to removing the study by Erdoes et al., removing the study by Olde Engberink et al. had a larger effect on the summary point: sensitivity 0.70 versus 0.84; specificity 0.91 versus 0.87.
Meta-analysis of studies with a reference fibrinogen less than 2 g/l or using the TEG were not possible given the small number of studies identified.
Heterogeneity, subgroup analysis, and reporting bias
Due to the small number of studies, assessments of heterogeneity and subgroup analysis were not possible. Given the small number of eligible studies and their heterogeneity, a Deeks test would not have been informative and was not performed.48–50
Discussion
The findings of this systematic review indicate that there are very few studies and no high quality studies of the DTA of viscoelastic POC tests for the identification of hypofibrinogenaemia in cardiac surgical patients (ROTEM 7, n = 1692 patients; TEG 2, n = 128 patients). Using a comprehensive and sensitive search strategy, only 10 DTA studies were identified and only nine were eligible. All had a risk of bias in relation to patient selection, flow and timing of the tests, or blinding of test results. These findings indicate that current assessments of the diagnostic accuracy of viscoelastic POC tests for the identification of hypofibrinogenamia during cardiac surgery are based on little or low quality evidence.
From the few DTA studies performed so far, the results indicate that viscoelastic POC identification of hypofibrinogenaemia, when compared to laboratory fibrinogen measurement, is associated with high false positive rates. For example, the false positive rate for ROTEM FIBTEM identification of a laboratory fibrinogen less than 1.5–1.6 g/l was 30%–58%. This means that if the clinical intention is to administer fibrinogen only if the laboratory fibrinogen is less than 1.5–1.6 g/l, then using the ROTEM FIBTEM at typical thresholds (e.g A10 <7.5–8 mm) would result in unnecessary fibrinogen administration in about 30%–58% of patients. This should prompt a reappraisal of the use of viscoelastic POC tests alone to guide fibrinogen replacement in stable cardiac surgical patients when there is an option to obtain a laboratory fibrinogen measurement within a safe time period.
These findings are supported by the meta-analysis of four ROTEM studies (total 1525 patients) with similar index and reference thresholds (Figure 5(c)). The summary sensitivity point in the meta-analysis was 0.70 (95% CI 0.58 to 0.79) with a summary specificity point of 0.91 (95% CI 0.88 to 0.94). These data indicate that for a hypothetical prevalence of laboratory fibrinogen less than 1.5–1.6 g/l of 12%, the false positive rate would be about 50%. 61 For a prevalence of 24%, the false positive rate would still be about 28%. 61
In contrast to the high false positive rates, the false negative rates for excluding hypofibrinogenaemia were low. For a laboratory fibrinogen less than 1.5–1.6 g/l, the four ROTEM studies using a FIBTEM A10 threshold less than 7.5–8 mm had a false negative rate of 2%–26%. However, for three of the four studies this rate was only 2%–4%. These findings suggest that the ROTEM FIBTEM A10 less than 7.5–8 mm is more accurate in excluding a laboratory fibrinogen less than 1.5–1.6 g/l than confirming it.
For two studies with a higher fibrinogen threshold (<2 g/l) and a higher FIBTEM threshold (A10 <9.5 mm), the findings were similar (Figure 4). 51 , 57 However, for a third study using an earlier FIBTEM threshold (A5 <9.5 mm) with a hypofibrinogenaemia prevalence of 81%, the findings were inverse (i.e. higher PPV, lower NPV, Figure 4). 54 This is to be expected in a study with a high prevalence of a target condition. 40 , 61
The two TEG FF studies had PPVs and NPVs and false positive and false negative rates in a similar range to the four ROTEM studies, with a FIBTEM A10 threshold less than 7.5–8 mm (Figures 3 and 4). However, these findings should be interpreted with caution, given that they involved a total of only 128 patients.
It has been argued correctly that laboratory fibrinogen measurement, even when performed to the highest standards, may not always represent the true fibrinogen level, and may vary between laboratories and with different clinical scenarios. 62 Nevertheless, this source of variation could also apply to any alternative form of fibrinogen measurement, including viscoelastic POC techniques. In any event, no studies have shown that viscoelastic POC techniques are less variable across sites or across scenarios than the Clauss fibrinogen.
It could also be argued that viscoelastic tests provide a more relevant index of hypofibrinogenaemia than laboratory measurements, because they assess the functional contribution of fibrinogen to clot formation, not only a plasma level. However, this has never been shown unequivocally in any study. Therefore, until a more accurate and reliable method of fibrinogen measurement has been proved and taken up by haematology laboratories broadly, the Clauss method remains the reference standard of fibrinogen measurement, acknowledging the variations in results that may occur. 44 , 62
Another possibility is that viscoelastic fibrinogen estimates measure a different aspect of coagulation to laboratory fibrinogen and that the tests are not directly comparable. However, like the laboratory fibrinogen, both the ROTEM FIBTEM and the TEG FF provide information to assist clinicians in their decision-making on ‘whether or not to administer fibrinogen’. 23 This is supported by several reviews,28–37 including a British Society of Haematology guideline: ‘VHA [viscoelastic haemostatic assays] can be used as a valid assessment of fibrinogen concentration to guide fibrinogen replacement. Grade 1B’. 24 Whether the methodology is based on measurement of ‘plasma fibrinogen’ or ‘fibrinogen contribution to clot’ the implications and immediate therapeutic options for a low fibrinogen are the same. This makes all three tests comparable for diagnostic purposes. Of course, irrespective of the source of information, the decision to administer fibrinogen will depend on the clinical scenario and the rate of bleeding.
Yet another argument is that the diagnostic accuracy of viscoelastic tests should be based on a clinical reference condition such as their identification of ‘bleeding responsiveness to fibrinogen replacement’. However, ‘bleeding responsiveness to fibrinogen replacement’ may prove difficult to measure clinically, because in practice low fibrinogen is only one of many potential causes of abnormal bleeding in cardiac surgical patients. Others include low platelets, residual heparin, deficiency of other clotting factors, fibrinolysis, or a combination of these causes.5–7 The cause may also be entirely surgical and unrelated to a coagulation deficiency. Therefore, correction of low fibrinogen may not necessarily reduce bleeding in all cases. Yet, it is still incumbent on clinicians to correct low fibrinogen in bleeding patients to remove hypofibrinogenaemia as a possible cause.8–11
The laboratory fibrinogen thresholds in this review were based on consensus guidelines published by professional organisations.17–21, 38 Including both recommended thresholds (<1.5 g/l and <2 g/l) increases the ‘generalisability’ of the results. The range of viscoelastic test thresholds also allows clinicians to compare their practice to published studies. It is notable that one study (Mace et al.), 55 provided about 60% of all patients included in the meta-analysis, thereby having a large influence on the meta-analysis component of the review (Figure 5). However, the results of all nine studies are also presented individually with no summary component (Figures 3 and 4). Clinicians can assess for themselves whether the coagulation practices in the included studies are relevant to their own practice and experience.
There have been no previous systematic reviews on the DTA of viscoelastic POC tests for the identification of hypofibrinogenaemia in cardiac surgical patients. However, there have been two recent narrative reviews of their role in the estimation of fibrinogen concentration across patient groups. 62 , 63 Both reviews were based on a search of only one database. The review by Peng et al. concluded that TEG FF and ROTEM FIBTEM discrimination of fibrinogen deficiencies varied ‘depending on patient population and fibrinogen concentrations’. 63 The review by Ranucci et al. 62 included DTA data but identified only three DTA studies in cardiac surgical patients; 53 , 55 , 60 other DTA data included in their review was derived post hoc from ‘rough data in graphic form’ from other studies.64–66 In any event, with the exception of one study that reported only preoperative data, 60 the range of PPVs (42–80) and NPVs (64–99) was similar to those obtained in our systematic review. The authors concluded that viscoelastic POC tests ‘should probably be used more as a trend monitor than as tools for identifying specific cut-off levels’ of fibrinogen. 62
The findings pertain mostly to the ROTEMdelta, as seven of the nine studies involved this device. This is an important caveat. Recent studies comparing ROTEMdelta and ROTEMsigma values have reported different results. 67 , 68 Shenk et al. found high correlation (r>0.8) for all variables measured by the devices in 30 healthy volunteers and 30 intensive care unit patients. 67 In contrast, Gillissen et al. reported poor correlation between the devices for FIBTEM assays in 23 women with postpartum haemorrhage. 68 As such, it is not clear that the findings for ROTEMdelta will apply to the ROTEMsigma. Similarly, it is not clear that results for the TEG5000 and TEG6S are interchangeable: while Lloyd-Donald et al. found close agreement between the two devices in 25 intensive care unit patients, 69 Robson et al. reported that results from the two devices were not quantitatively interchangeable in ten liver transplant patients. 70
A recent study reported the sensitivity and specificity of ROTEMsigma FIBTEM A5 less than 12 mm values for identifying laboratory fibrinogen thresholds of less than 1.5 g/l and less than 2 g/l in 43 patients, ten of whom were undergoing cardiac surgery. 71 The sensitivity was 100% for both thresholds, with specificities of 63.6% and 71.4%, respectively. However, the authors did not report data separately for the ten cardiac surgical patients, and also it was not possible to calculate PPVs or NPVs from their data.
While there were very few DTA studies identified in this review, there were a large number of studies on correlation or clinical agreement or on the influence of viscoelastic POC tests on outcomes (Figure 1). However, correlation and clinical agreement studies do not provide clinicians with the ability to assess the likelihood of false positives and false negatives when making clinical decisions. 39 , 40 , 61 ,72–75 Moreover, most of the outcome studies have reported only a modest benefit of using viscoelastic POC devices, typically limited to a small reduction in blood loss or blood transfusion.28–37,62,63,76–80 Most reviews of viscoelastic POC tests have also commented on the low quality of evidence currently available, and have called for further studies of their accuracy and role.
The strengths of this systematic review include the high sensitivity of the search strategy, and the low threshold for scrutinising full-text versions of potentially eligible articles. However, there were limitations due to the small number of eligible studies, reducing the scope for analysis of subgroups that may have differing coagulation parameters (e.g. neonates and children) or investigation of sources of heterogeneity. The findings also apply only to cardiac surgical patients as other groups of patients may have other causes for coagulation abnormalities. Due to the low quality of the studies, it was also not possible to exclude observer bias, which would produce an overestimate of the true DTA. 49 , 50 Another important source of bias that could not be excluded is publication bias. 48
The findings apply only to the identification of hypofibrinogenaemia, and not to other aspects of impaired coagulation. Viscoelastic POC tests measure other variables including the influence of other coagulation factors and platelet function. 25 , 26 However, often these can be interpreted only once hypofibrinogenaemia has been excluded as a cause. 17 Moreover, the clinical priority if coagulopathy is suspected in bleeding patients is to correct hypofibrinogenaemia in the first instance, so the requirement for accurate fibrinogen estimation underpins the value of the tests.8–16
A potential confounder when comparing coagulation measurements during CPB is the presence of heparin. While, heparin does not affect FIBTEM or TEG FF measurements (due to the presence of polybrene and heparinase, respectively), 23 , 24 it may reduce the Clauss fibrinogen. 81 This typically occurs at heparin levels greater than 2 IU/ml, but may occur at lower concentrations, depending on the assay used. 81 Therefore, laboratory fibrinogen levels during CPB may have been artificially low in this review, confounding the assessment of diagnostic accuracy. However, in three of the studies (Olde Engberink et al., 52 Gautam et al. 58 and Erdoes et al., 2018), 59 all measurements were made before or after CPB (when little or no heparin was present), and in a fourth study (Scott et al.) 57 samples were obtained at four timepoints, only one of which was during CPB. Moreover, in the studies by Ji et al., 54 Ogawa et al. 51 and Erdoes et al., 2015, 53 laboratory fibrinogen levels were similar before separation from CPB and after administration of protamine (post-CPB), indicating that the reversal of heparin had little or no effect on the laboratory fibrinogen measurements. In the remaining two studies by Mace et al. 55 and Matzelle et al., 56 the prevalence of hypofibrinogenaemia during CPB was very low (6% and 13%, respectively) indicating that artificially low fibrinogen levels would be unlikely. In any event, artificially low reference laboratory fibrinogen would increase false positive rates of the viscoelastic tests (by reducing the number of true positives), thereby reducing, not improving their diagnostic accuracy.
Several other viscoelastic devices have been developed for the assessment of coagulation and the identification of hypofibrinogenaemia. These include SEER technology 82 (HaemoSonics LLC, Charlottesville, VA, USA), FIBTEM Plus 83 (ROTEM; TEM International, GmbH, Munich, Germany), ReRox 84 (Medirox AB, Nykoping, Sweden), the oscillating viscometer, 85 steel ball viscometry 86 and the Quantra device 87 (HaemoSonics LLC, Charlottesville, VA, USA). However, we found no studies of their DTA for the identification of hypofibrinogenaemia (versus a laboratory standard fibrinogen) in cardiac surgical patients. Further studies are required to confirm the DTA of all viscoelastic POC tests in cardiac surgical patients, but particularly for the newer ROTEMsigma, the TEG and other recently developed devices. Another source of variability could be improvements in technology or reagents used by the various devices over time.
Despite the low PPVs obtained in this systematic review, and those reported in a previous narrative review, 62 the use of the ROTEM FIBTEM and TEG FF may still have an important role in cardiac surgical patients. Apart from their use as a trend monitor as suggested by Ranucci et al., 62 their faster turnaround time may improve clinical decision-making when clinical conditions are such that waiting up to 45 minutes for a laboratory result is not possible. For example, in unstable patients or those with particularly brisk bleeding, a decision on fibrinogen replacement may be required within a much shorter time frame. In these situations it is not only a pragmatic reality to make decisions about fibrinogen replacement without laboratory confirmation of hypofibrinogenaemia, it is also recommended by consensus guidelines. 17 The results of this review support the use of viscoelastic POC tests in these scenarios. With the low false negative rates (NPV 96%–98% in most studies, Figure 3), negative findings (e.g. ROTEM FIBTEM A10 >8 mm) would reassure clinicians that the likelihood of hypofibrinogenaemia was low, giving them the option of withholding fibrinogen pending laboratory confirmation, clinical review and consideration of other causes of bleeding. At the same time, positive findings would support early fibrinogen supplementation, accepting that while this could be unnecessary in a proportion of patients, the alternative strategy would be the administration of fibrinogen on clinical grounds alone. In this way, the results may provide valuable information when a rapid clinical decision on fibrinogen supplementation is required. On the other hand, the results should prompt a reappraisal of the use of viscoelastic POC tests alone to guide fibrinogen replacement in stable cardiac surgical patients when there is an option to obtain a laboratory fibrinogen measurement within a safe time period.
Supplemental Material
sj-pdf-1-aic-10.1177_0310057X20948868 - Supplemental material for Diagnostic accuracy of viscoelastic point-of-care identification of hypofibrinogenaemia in cardiac surgical patients: A systematic review
Supplemental material, sj-pdf-1-aic-10.1177_0310057X20948868 for Diagnostic accuracy of viscoelastic point-of-care identification of hypofibrinogenaemia in cardiac surgical patients: A systematic review by Neville M Gibbs and William M Weightman in Anaesthesia and Intensive Care
Footnotes
Acknowledgement
This work was the basis for a dissertation submitted to the University of Oxford by NMG in part fulfilment for the degree of MSc in Evidence-Based Health Care in September 2019.
Author contribution(s)
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
