Abstract
Medication error is a well-recognised cause of harm to patients undergoing anaesthesia. From the first 4000 reports in the webAIRS anaesthetic incident reporting system, we identified 462 reports of medication errors. These reports were reviewed iteratively by several reviewers paying particular attention to their narratives. The commonest error category was incorrect dose (29.4%), followed by substitution (28.1%), incorrect route (7.6%), omission (6.5%), inappropriate choice (5.8%), repetition (5.4%), insertion (4.1%), wrong timing (3.5%), wrong patient (1.5%), wrong side (1.5%) and others (6.5%). Most (58.9%) of the errors resulted in at least some harm (20.8% mild, 31.0% moderate and 7.1% severe). Contributing factors to the medication errors included the presence of look-alike medications, storage of medications in the incorrect compartment, inadequate labelling of medications, pressure of time, anaesthetist fatigue, unfamiliarity with the medication, distraction, involvement of multiple people and poor communication. These data add to current evidence suggesting a persistent and concerning failure effectively to address medication safety in anaesthesia. The wide variation in the nature of the errors and contributing factors underline the need for increased systematic and multifaceted efforts underpinned by a strengthening of the current focus on safety culture to improve medication safety in anaesthesia. This will require the concerted and committed engagement of all concerned, from practitioners at the clinical workface, to those who fund and manage healthcare.
Introduction
WebAIRS is an online voluntary incident reporting system for anaesthetists in Australia and New Zealand maintained by the Australian and New Zealand Tripartite Anaesthetic Data Committee (ANZTADC). It was established in September 20091 with the objective of improving the safety and quality of patient care.1,2 By July 2016, over 4000 anaesthesia incident reports had been received. A cross-sectional overview of these reports has been published, 1 followed by three reviews focused on specific topics.3–5
Medication errors have featured strongly in previous reports of critical incidents, 6 and were the second most common category of incidents in the first 4000 webAIRS reports. 1 They continue to be common in anaesthetic practice7–9 with occasional harm to patients 10 despite ongoing efforts to minimise their incidence.9,11,12 In the current study, we aimed to analyse the incidents related to medication error in the first 4000 cases reported to webAIRS and thereby to advance further webAIRs’ overall objective of improving patient safety by learning from incident reporting.
Methods
The methods used in this study, the categorisation of the first 4000 incidents and the relevant ethics committee approvals have been described in detail previously.1,3
The webAIRS software provides a drop-down list of seven primary categories, including ‘medication’. Reporters can also enter narrative text descriptions of the incident.
The first 4000 reports in the database were searched by MDC using structured query language (SQL), excluding those reported as not involving patients, to identify reports in which the reporter had selected ‘medication’ as the main category. Reports that had the word ‘medication’ or ‘drug’ in the text narrative were manually reviewed to identify those pertaining to medication errors. Several additional relevant reports were identified through reference to analyses for previous papers3–5 or serendipitously during the various scans of the database. The reports selected in this way were evaluated primarily by JAH, JYK and AFM, and in some instances also by MRM and MDC. These reviewers paid particular attention to the text narratives in finalising selection and categorising the selected reports, and also in identifying contributory factors, incorrect dosages, intended and actual administration routes and medications, and the level of patient harm and the potential preventability associated with each incident (see definitions in Table 1). Note that a case could be coded with multiple contributory factors. This process was iterative and any discrepancies were resolved by discussion and repeated cross-checking.
Operational definitions used in this analysis.
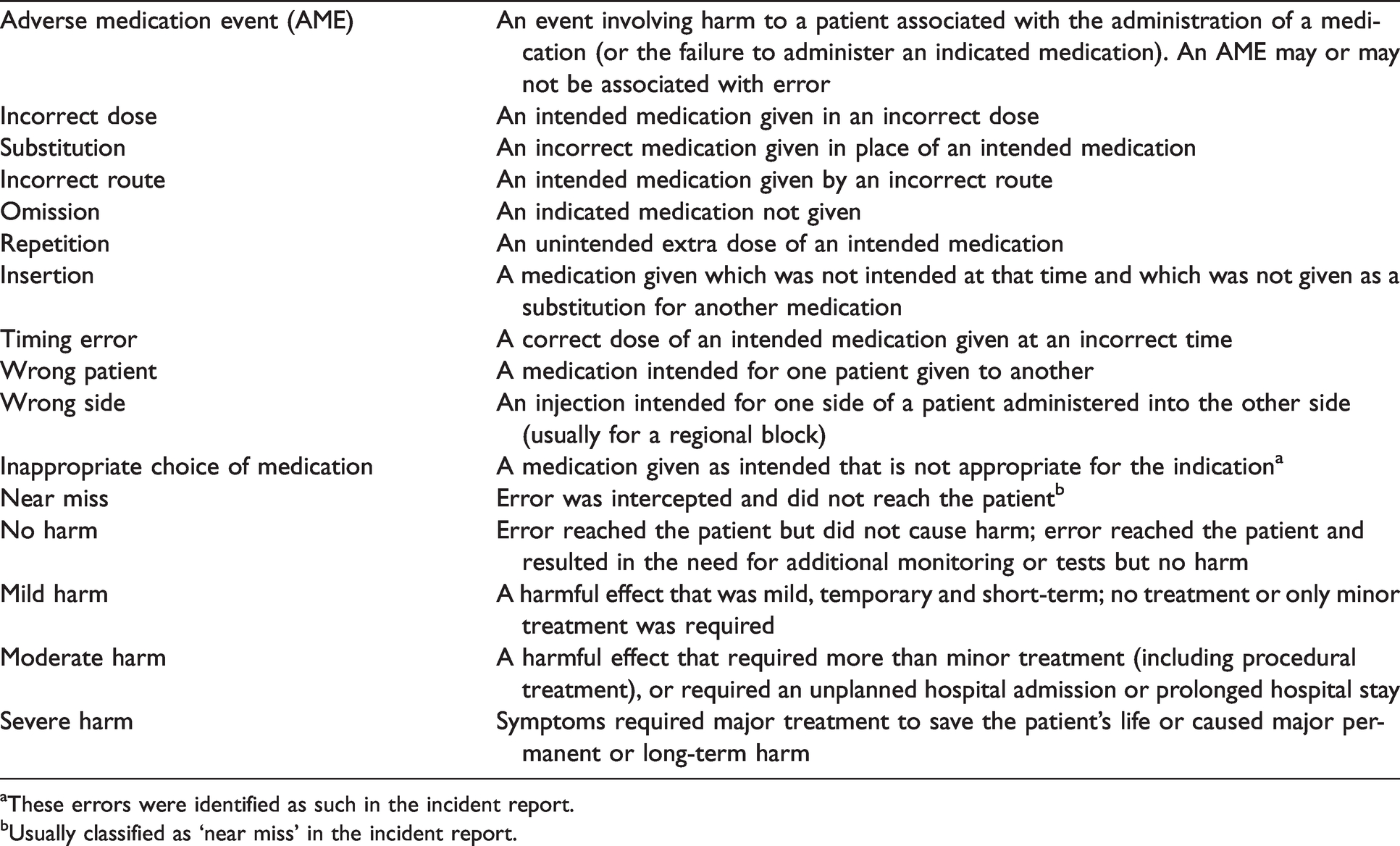
aThese errors were identified as such in the incident report.
bUsually classified as ‘near miss’ in the incident report.
As described previously,3,5 we calculated the proportion of incidents in the reports with a medication error and those with no medication error pertaining to selected characteristics potentially relevant to the risk of medication error (e.g. age band, American Society of Anesthesiologists physical status (ASA PS), time of day, etc.). When numbers were small, categories were amalgamated (e.g. ASA PS 4 and 5). In 18 cases of medication error in which patient gender was not reported it was derived from the narrative. We then calculated the risk ratio (RR) for each of these characteristics, by dividing the proportion of reports with that characteristic among the medication error incidents by the proportion of reports with that characteristic among the incidents with no medication error, with 95% confidence intervals (CIs) calculated using the Wald method of continuity correction. Note that this is the RR of a report being of a medication error, given that an incident has been reported, rather than the RR of making a medication error. Reports with no patient involved were not included in the analysis. As with previous studies evaluating webAIRS data, we deemed a factor to be potentially more or less likely to be associated with medication error reports if the 95% CIs did not overlap and the RR was greater than 1.5 or less than 0.67, respectively.3,5 All analyses were performed by MRM using R 4.0.3 13 in RStudio 1.3.1093. 14
Results
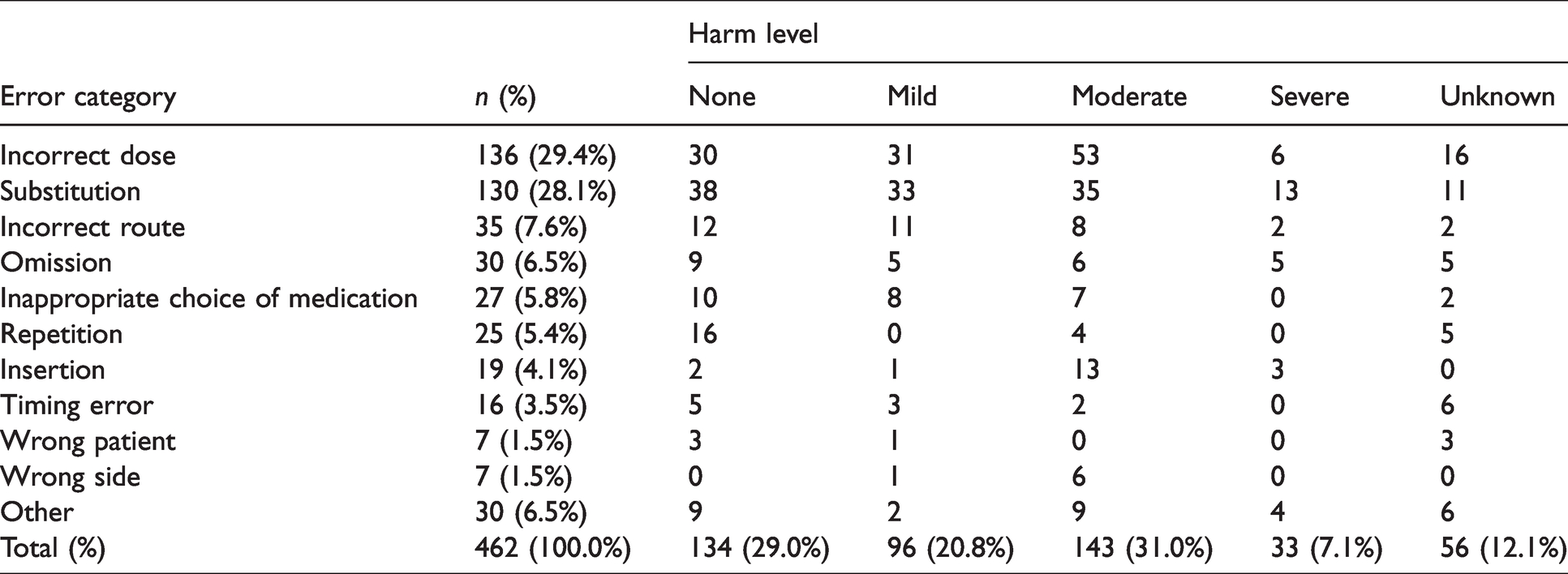
From the first 4000 reports, we excluded 71 reports that were coded as not involving a patient. In the remaining 3929, 687 reports had ‘medication’ as their main category. In addition, 336 reports included the words ‘drug’ or ‘medication’ in their narratives, and eight were found on manual review to contain a potential medication error. A further 17 reports were identified as involving medication errors through other processes (see Methods). Thus 712 records were included in a more detailed manual evaluation. This revealed seven more that did not involve a patient, eight reports of the use of sugammadex without error, 164 that involved adverse medication events (AMEs) with no error, 28 narratives of complex cases without an obvious medication error and 43 reports of near misses (the reports of near misses were excluded from the error reports, but analysed separately). Thus, 462 reports involved medication errors of various types (see Figure 1). Of these, 272 (58.9%) resulted in at least some harm, rated as mild in 96 (20.8%), moderate in 143 (31.0%) and severe in 33 (7.1%) (see Tables 2 and 3). The 462 reports involving medication errors with patients were compared with the 3460 reports that did involve a patient but not a medication error in relation to patient characteristics, procedural factors, outcomes and preventability (see Table 4). There was a wide range of contributory factors (see Figure 2), and those factors which featured prominently for a particular category of error are noted in the text below.
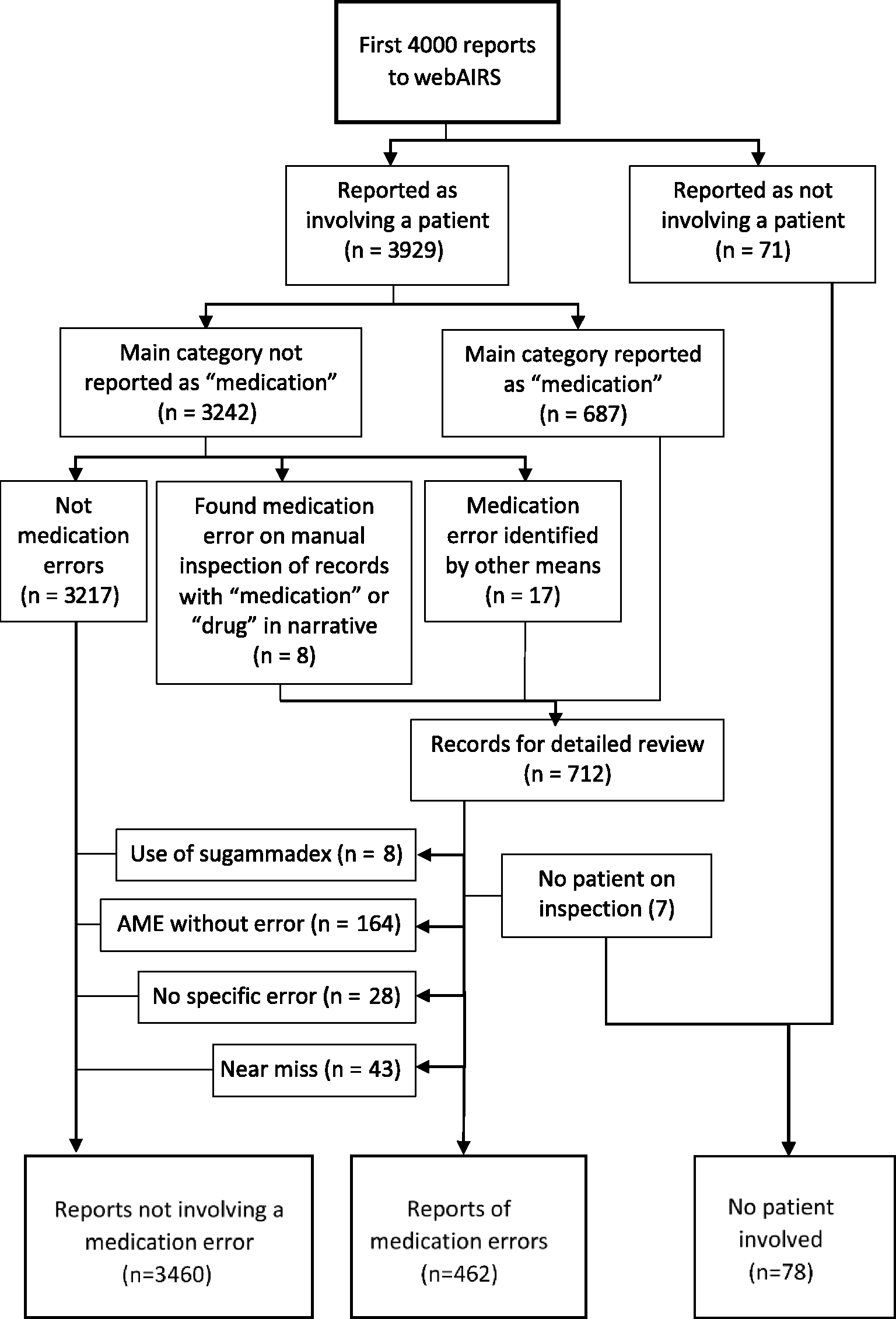
Study flowchart. AME: adverse medication event.
Medication errors included in the final analysis by category and level of harm as assessed by our raters.
Details of cases in which severe harm was reported by category of medication error.

n: number of cases.
aBaxter International Inc., Deerfield, IL, USA.
Patient characteristics, procedural factors, outcomes and preventability associated with a medication error and no medication error, and risk ratio for medication error versus no medication error (78 reports did not involve a patient and are excluded from this table).

Data presented as proportions and 95% confidence intervals (CIs).
RR: risk ratio; PAC: pre-admission consult; PACU: post-anaesthesia care unit.
aRR versus no medication error >1.5 or <0.67, and proportion outside the 95% CI for no medication error.
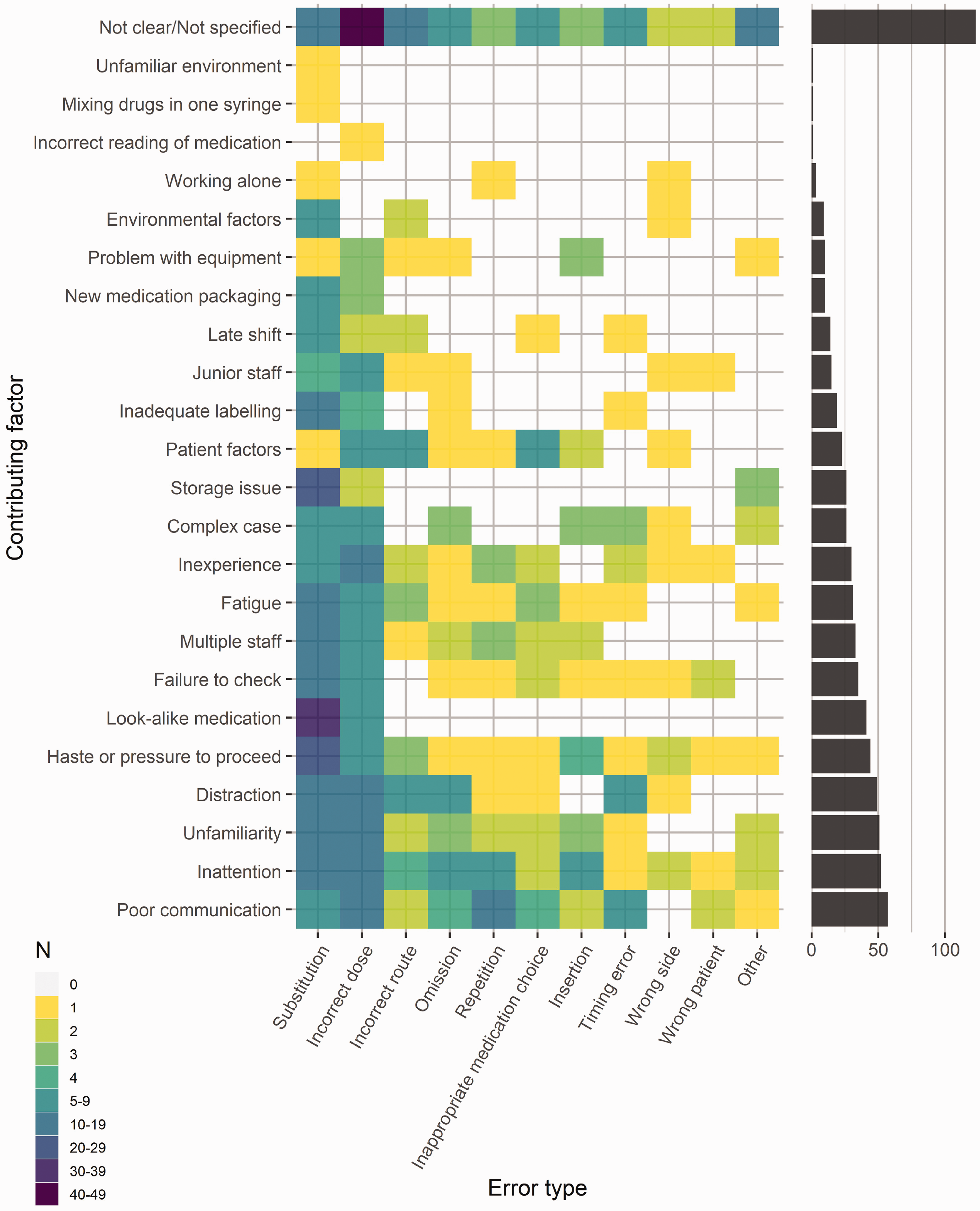
Heat diagram of contributory factors by category of error. The number of each combination of common error types (x-axis) and contributing factors (y-axis) is denoted by a coloured square, with darker shades being more common and lighter shades being less common. A white square indicates no cases. Total numbers for each error type and contributing factor are indicated by bar charts at the right and top, respectively. Note that each case was coded as having one error type, but may have had from zero to many contributing factors.
Incorrect doses were the commonest category of error (n=136, 29.4%), predominantly overdoses and most often involving opioids (n=46), sympathomimetics (n=13), local anaesthetics (LAs; n=12) and insulin (n=11) (see Table 5). Factors commonly found to contribute to incorrect dose errors included lack of experience, poor communication and inattention. Of the 46 opioid dose errors, 17 involved remifentanil infused in a wrong concentration or via an incorrectly programmed syringe pump. In most of these, harm was avoided or mitigated by early recognition of appropriate titration. Other opioid incorrect dose errors included incorrect charting or programming of patient-controlled analgesia pumps (n=4) and dose calculation errors in paediatric patients (n=4). In two reports involving intrathecal administration, substitution of a standard 10 mg/ml vial of morphine for one containing 50 µg or 100 µg in 0.1ml resulted in a ten-fold overdose, with consequent postoperative respiratory depression in one case. Substantial (i.e. ten-fold) dilution errors were also reported for insulin, reported in one case as being contributed to by the anaesthetist’s lack of familiarity with this medication.
Incorrect dose errors by class of medication and magnitude of the error.
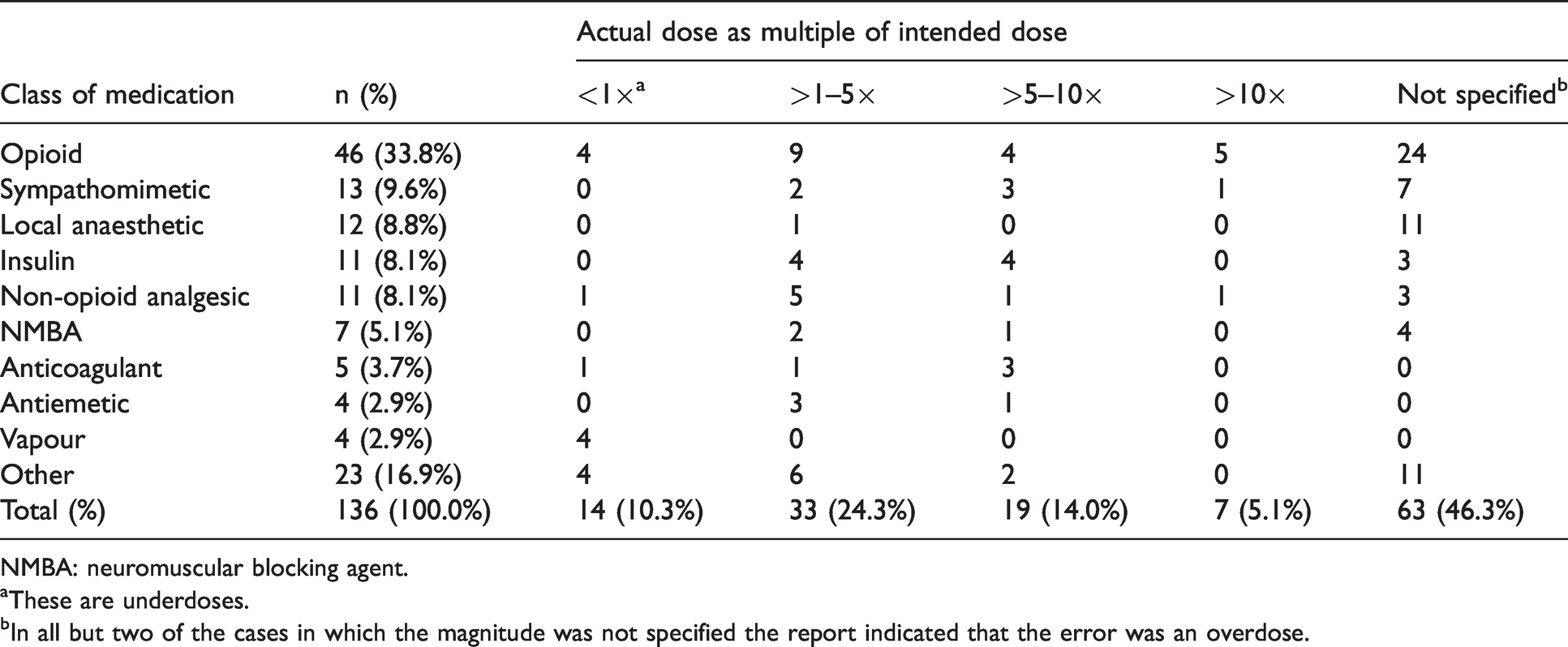
NMBA: neuromuscular blocking agent.
aThese are underdoses.
bIn all but two of the cases in which the magnitude was not specified the report indicated that the error was an overdose.
There were 130 (28.1%) substitution errors, with various pairings of intended and actually administered medications (see Table 6 and Figure 3). Most involved the intravenous (IV) route of administration (n=98), but substitutions also occurred with IV infusions (n=16), and with epidural (n=8), subcutaneous (n=3), inhalational (n=2), spinal (n=2) and ophthalmic (n=1) routes of administration. The commonest IV substitution errors involved administering a non-depolarising neuromuscular blocking agent (ND-NMBA) instead of a benzodiazepine (n=15), all involving midazolam pre-induction, and all but one rated as causing moderate or severe harm, and administering a different NMBA (either depolarising or non-depolarising) from the one intended. Sympathomimetics (n=4), opioids (n=2), an antibiotic and an analgesic were substituted for an intended LA in epidurals, and there were two substitutions of one LA for another by spinal injection. Common contributing factors included look-alike medications (n=35), the storage of medications in the incorrect compartment or next to look-alike medications (n=21), haste (n=21), having multiple anaesthetists involved in the same anaesthetic (n=17), omitting to double check the medication (n=17) and distraction (n=16).
Most common substitution errors presented in Figure 3.
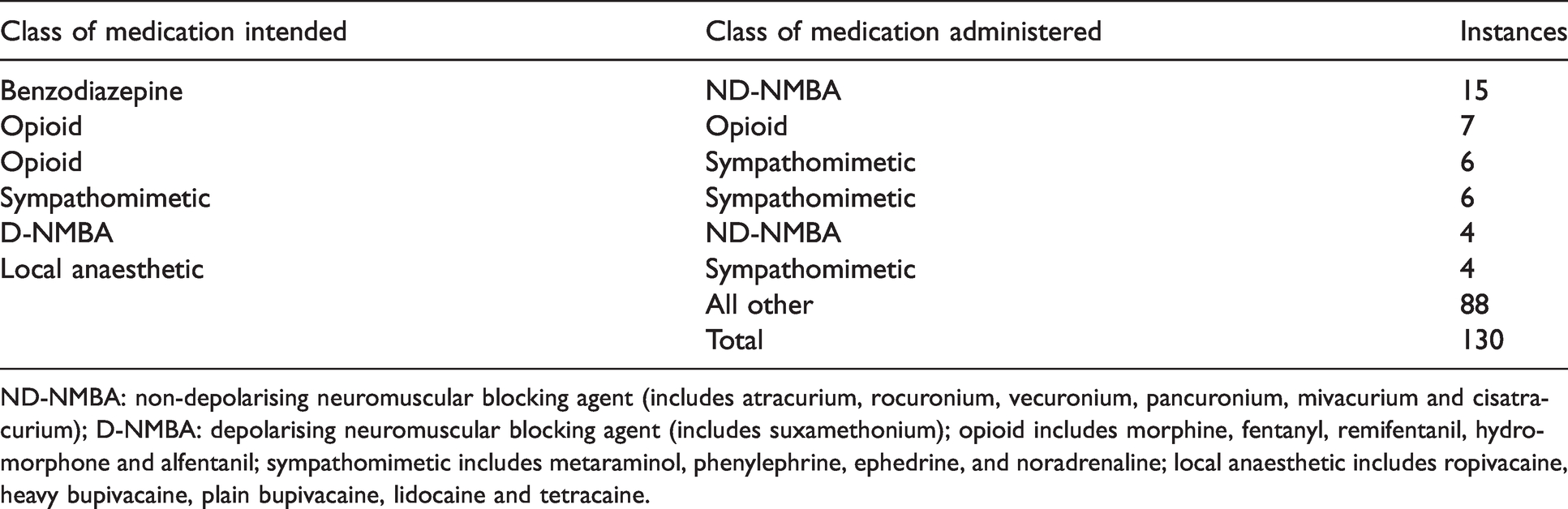
ND-NMBA: non-depolarising neuromuscular blocking agent (includes atracurium, rocuronium, vecuronium, pancuronium, mivacurium and cisatracurium); D-NMBA: depolarising neuromuscular blocking agent (includes suxamethonium); opioid includes morphine, fentanyl, remifentanil, hydromorphone and alfentanil; sympathomimetic includes metaraminol, phenylephrine, ephedrine, and noradrenaline; local anaesthetic includes ropivacaine, heavy bupivacaine, plain bupivacaine, lidocaine and tetracaine.
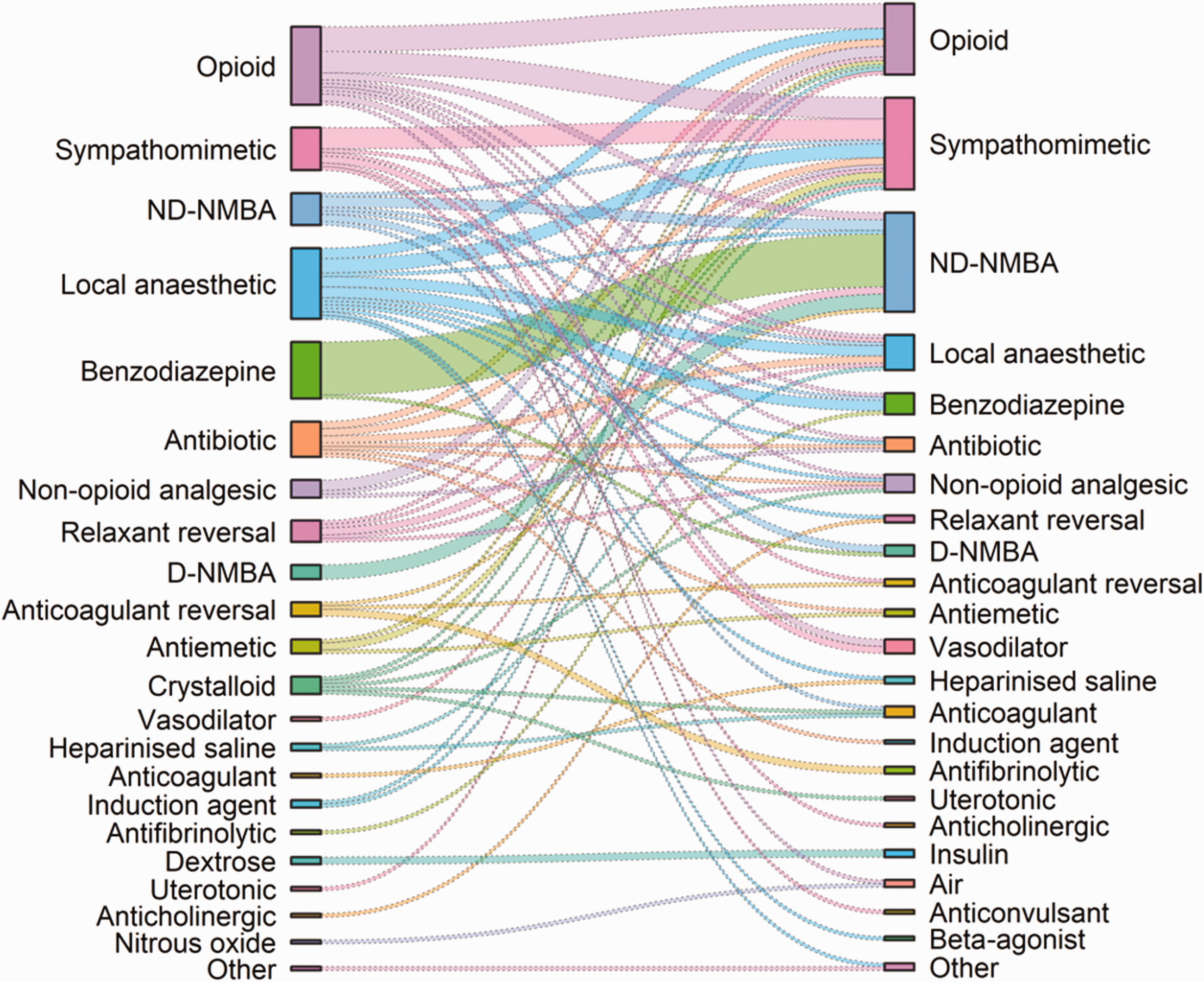
Sankey diagram showing the relationship between medications intended (on the left) and medications actually administered (on the right) in the substitution errors in this study. ND-NMBA: non-depolarising neuromuscular blocking agents; D-NMBA: depolarising neuromuscular blocking agents.
There were 35 (7.6%) reports of medications administered by an incorrect route, with several combinations of the route intended and the route actually employed (see Figure 4). Most frequently, medications intended for IV injection were administered by another route, but medications intended for the epidural route were administered by IV (n=6), intrathecal (n=1) and subdural (n=1) routes. In addition, one medication intended for subcutaneous administration was delivered via the IV route (n=1), a medication intended for a regional block was administered intra-arterially (n=1) and one medication intended for intranasal use was given IV. There were ten instances of extravasation of IV medications into a patient’s tissues, and seven of failed delivery of IV infusions due to failed connection of the line and cannula. In four cases, the extravasation of induction agents caused pain and failure of prompt induction, two in the context of rapid sequence induction (RSI). There were also reports of IV medications being administered via a leaking IV (n=1), epidural (n=1) or intra-arterial cannula (n=3), and one of a central line misplacement that resulted in intrathoracic infusion of noradrenaline. A notable contributing factor in many of these incorrect route reports was concealment of the IV cannula, often under the surgical drapes.
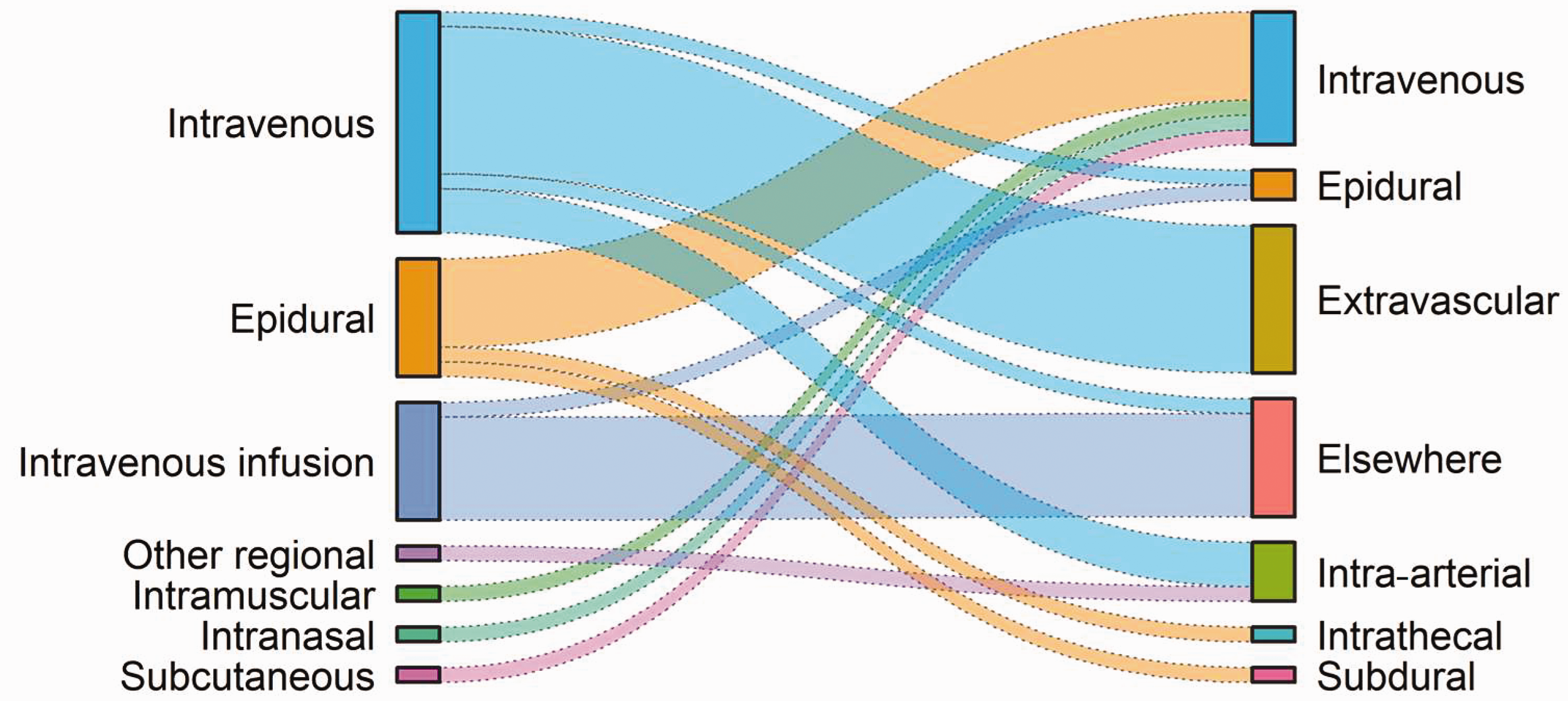
Sankey diagram showing the relationship between the route intended (on the left) and route by which medications were actually administered (on the right) in the wrong route errors in this study.
There were 30 (6.5%) omissions of an intended medication, most commonly an inhalational agent (n=10). The anaesthetist failed to start an intended volatile agent in four cases, and to resume after refilling the vaporiser in three, after a change in volatile agent in one, and after cessation during cardiopulmonary bypass in one. There was a failure to confirm the selection of a volatile agent on the electronic screen in one case. Opioids were omitted in seven patients. In five of these cases, remifentanil was not added to a saline solution for infusion; these errors were recognised when a lack of clinical response prompted a check of the medication vial, found to be full of powder. Other omitted medications included induction agents (n=2), and ND-NMBA reversal agents (n=2). There were single reports of omission of LA in a neuraxial block (consequently performed with opioids only), and of omission of oxytocin, an NMBA, prostacyclin, an antihypertensive agent, an anticoagulant and insulin. Common contributing factors for the errors with inhalational agents were distraction, inattention and poor communication. In another case, use of high-flow nasal oxygen for preoxygenation resulted in the omission of bag-mask ventilation, which was reported as a normal procedural cue for that particular anaesthetist to start a volatile agent.
There were 27 (5.8%) reports of inappropriate choice of medication. Many of these involved administration of medications for which there was a known history of allergy, including antibiotics (n=6), non-steroidal anti-inflammatory drugs (NSAIDs; n=3), opioids (n=2), iodine (n=1), chlorhexidine (n=1), paracetamol (n=1) and an antiemetic. Other reports included the administration of an NSAID to a patient with renal impairment, ephedrine to a patient on a monoamine oxidase inhibitor, and suxamethonium to a tetraplegic patient and to a patient with a family history of probable pseudocholinesterase deficiency. Patient factors (n=5), poor communication (n=4) and fatigue (n=3) were notable as contributing factors to these errors.
There were 25 (5.4%) repetition errors, notably of paracetamol (n=11) and LAs (n=5). A patient who had received epidural LA for a bilateral knee replacement was subsequently administered high volume LA in each knee by the surgeon, resulting in a toxic cumulative dose. Notable contributory factors included poor communication and inattention.
There were 19 (4.1%) insertion errors, notably involving the flushing of residual NMBA in the IV line resulting in residual neuromuscular blockade (n=8); five of these cases occurred in the post-anaesthesia care unit (PACU) and one in the ward (with two locations unspecified). In one awake patient, emergency tracheal intubation was required. In another patient, flushing residual ketamine in the IV line led to distressing hallucinations.
There were 16 (3.5%) reports of timing errors in administrations of medications, including administration of prophylactic antibiotics after surgical incision (n=3), an anticoagulant medication soon after neuraxial blockade (n=2) and neuraxial blockade soon after the administration of antiplatelet medication (n=2). A high dose oxytocin infusion prepared for post-delivery administration during emergency caesarean section was started prior to delivery of the baby, a volatile agent was administered during pre-oxygenation of a patient undergoing a rapid sequence induction (increasing the risk of aspiration), an induction agent was given in the preoperative bay prior to a planned neuraxial block, insulin was repeated too soon in a hyperglycaemic patient, and an opioid was given before delivery of a baby during general anaesthesia for caesarean section. The administration of an NMBA was delayed post-induction because it had not been drawn up. Poor communication and distraction were common contributing factors in these errors.
There were seven (1.5%) reports involving the administration of medications to the wrong patient. In five such cases, the medication was intended for the previous patient but not disposed of at the end of that case. These medications included metaraminol, fentanyl, midazolam and IV fluid. There was also a report of blood prepared against a different patient’s documentation and administered; the blood happened to be compatible with the patient and caused no harm. In another report naloxone was administered to the wrong patient on a ward.
Regional anaesthesia for lower limb orthopaedic operations was administered on the wrong side in seven (1.5%) patients. This resulted in abandoning the surgery in one case, and failing to receive regional anaesthesia on the correct side in another. In the others, surgery proceeded with a second block on the correct side after recognition of the error. Notable contributing factors included time pressure, failure to mark the surgical site, not doing time-out before the block and distraction.
There were 30 (6.5%) other reports of medication errors that did not fit into any of the above categories. Notable examples included errors in documentation and labelling (n=4), use of an expired medication (n=2), failure to have vasopressors on hand during a transfer of a critically ill patient (n=1), failure to have suxamethonium available in the operating room (OR) (n=3), opioids found on patients’ beds (n=3), a missing opioid (n=1), and a propofol syringe found still attached to the IV line of a patient in the PACU (n=1). There was also a case where air was entrained in an IV line when giving IV paracetamol in the PACU, causing air embolism in a patient.
In assessing the characteristics of medication error reports against reports containing a patient with no medication error (excluding cases with no patient, see Table 4), we found several factors that seemed to be consistently more or less frequently associated with medication error (defined as an RR >1.5 or <0.67, and proportion outside the 95% CI for no medication error). Trainee anaesthetists in years 1–2 were more likely to be involved in medication error reports (RR 1.52). Patients between one and 16 years or older than 80 years were less likely to be involved in medication error reports (RR 0.63 and 0.65, respectively), as were patients with a body mass index (BMI) higher than 40 (RR 0.43), or with ASA PS 4–5 (RR 0.60). Medication errors were less likely to be reported from the PACU (RR 0.66) and pre-admission consult (PAC) or reception (RR 0.66) than other locations. Medication errors were far more likely to be reported as preventable than other error types (89% versus 47%, RR 1.91).
In the 43 reports of near misses, 34 related to IV boluses, seven to IV infusions, one to an epidural and one to an oral medication. A wide range of medication classes (n=17) was involved. The potential errors included substitutions (n=22), incorrect doses (n=15), wrong choices of medication (n=2), an omission (n=1), an expired medication (n=1) and a documentation error (n=1). The potential error was detected by the same person second checking (n=21), by a second person checking (n=20) and by scanning of a barcode (n=1). The second person check was by a consultant anaesthetist (n=7), a nurse (n=5), an anaesthetic technician (n=2), a midwife (n=1), a staff member in the intensive care unit (ICU) after the transfer of a patient from the OR (n=1), and unspecified (n=4).
Discussion
Nearly 12% of the first 4000 reports to webAIRS involved a medication error; 89% of these were considered preventable and 38% caused moderate or severe harm to a patient. These reports add to an ever-increasing body of evidence testifying to a persistent failure to address the safe management of medications adequately during anaesthesia and the perioperative period despite numerous calls to arms in regard to this fundamental aspect of anaesthetists’ practice.8,15–21 At the same time, the reports of near misses provide an opportunity to learn from cases that have gone well, thereby adding a Safety-II approach to that of Safety-I.22,23 These near miss reports strongly reinforce the value of repeated checking, whether by the individual practitioner, with a second practitioner (be it another anaesthetist or a colleague from another discipline), or with a device such as a barcode reader. 24 The importance of checking is also reinforced by the contributory factors identified in the reports of actual medication errors: failure to check was commonly reported (see Figure 2), as were inattention, distraction, haste or pressure to proceed, the presence of multiple staff, and poor communication. Taken together, these reports provide a reminder of the undisputed professional and legal responsibility of every medical practitioner to check each medication administered, and its dose and route. However, they also provide testimony to the challenges faced in anaesthesia: anaesthetists are often required to undertake parallel processing in relation to current, previous and impending patients, while also teaching, continuously assessing dynamic clinical situations and at the same time interacting with surgeons and other staff. NetworkZ and the Effective Management of Anaesthetic Crises courses are examples of important initiatives to improve communication and the management of factors such as these during anaesthesia in general,25,26 but perhaps more thought is needed on how to do this effectively in the context of medication management in particular.
Many of the reported medication errors had no consequence. Moderate or minor harm was most commonly reported for substitution errors and dosage errors (see Table 2). The impact of a substitution error depends considerably on the medication actually administered, although it may also depend on the lack of effect of the intended medication (as in the case in errors of omission) (see Table 3). In theory, substitutions within the same class of medication are likely to be less problematic than substitutions between classes. From Figure 3 it can be seen that the range of classes substituted for each other in these reports is remarkably large. Relatively common combinations that were potentially harmful and signal areas for special care can also be identified. The substitution of an ND-NMBA for midazolam is one such example, and has considerable potential to result in awareness, or, if this occurs in a pre-anaesthetic holding area and passes unnoticed, may create considerable jeopardy for the patient (see Table 3). Another example is the substitution of a sympathomimetic agent for an opioid, which might well result in an unwanted physiological response that might harm a patient (see Table 3). On the other hand, substituting one opioid for another or one sympathomimetic agent for another (which was also both commonly reported) would probably have little consequence. The impact of a dosage error depends both on the medication involved and on the magnitude of the error. It is disconcerting to see 26 reports in which the dose administered was five or more times greater than the dose intended (see Table 5). Underdoses may be as problematic as overdoses, notably in relation to awareness (see Table 3, which contains one example of an underdose of volatile agent and two of omission of volatile agent).
Figure 4 shows a range of combinations between intended and actual routes. The reports of IV administration being given extravascularly speak to the importance of care over the integrity of IV lines (see Table 2 for an example of harm from this type of error). Injections intended for IV administration given intra-arterially (again, see Table 2) or epidurally (and conversely, re the epidural route) have considerable potential for harm, and these reports underline the care that is needed in labelling of lines and in the choice of injection site. There is also an important role for route-specific connectors in reducing these errors 27 .
This study has the limitations of incident reporting in general: there is no denominator (i.e. the number of cases from which these reports have originated, which will have been in the millions) 28 and reporting is voluntary so no inference can be drawn about the rate of these errors. Soberingly, we know that voluntary incident reporting identifies only a small proportion of the medication errors that actually occur; the frequency of medication error identified by facilitated incident reporting7,12,29–31 and direct observation8,9 is much higher. Some pragmatism was involved in assigning errors to particular categories (e.g. two errors categorised as overdoses via the intrathecal route could alternatively have been categorised as substitution errors or wrong route errors). Despite efforts to identify all reports of medication errors in the database, some may have been missed (as evidenced by those identified through means other than our formal searches). These data cannot be easily extrapolated to anaesthesia practice beyond the borders of Australia and New Zealand. In 46.3% of the reports of dosage errors, the actual dosage was not specified (see Table 5). It is also disappointing that (as is often the case in incident reporting) there are relatively few reports of near misses, and thus limited opportunity for Safety-II learning. One can only speculate that the motivation to report a near miss may be less strong than when something has actually gone wrong. Conversely, the study’s strengths lie in webAIRS’ broad base of reporting hospitals across two countries and its well-established web-based platform, its relatively standardised format for reporting and its provision for free text narratives.
In the light of these limitations and strengths, care is needed in the interpretation of the factors identified as being more or less likely to be associated with reports of medication errors. It is plausible that trainee anaesthetists in years one and two would be more prone to making medication errors than more experienced anaesthetists. It is also plausible that this observation may simply reflect differences in the conscientiousness with which errors are reported when they occur. It is possible that medication errors are less common in patients ASA PS 4–5, aged one to 16 years and over 80 years, or with a BMI greater than 40, but this seems unlikely. A more plausible explanation might be that the lower proportion of reports related to medication errors in these potentially challenging groups of patients actually reflects their greater vulnerability to other types of incidents (and hence incident reports). A similar explanation could apply to the lower proportion of medication errors reported from the PACU, but it could also be that the nature of the environment in the PACU, including the role of nurses in administering and checking the administration of medications there, is conducive to medication safety. The total number of errors reported from the PAC or reception areas was only six, so their lower proportion is probably just a reflection of the low number of medications administered in this setting. It is not surprising that the proportion of medication errors considered preventable is higher than that for incidents in general—by definition, all errors (including medication errors) are at least theoretically preventable, despite their statistical inevitability. 32 However, this fact emphasises that more could and should be being done to prevent them.
More than a third (38%) of the medication errors in this study were associated with moderate or severe harm. The limitations discussed above apply to the interpretation of this point. The actual proportion of medication errors that cause harm is simply not known, although 12.5% of respondents to a survey of New Zealand anaesthetists in 1995 reported having harmed a patient by a medication error, 10 and 33.3% of medication errors in a recent observational study in the US led to an observed AME. 8 It could be that the reporting of an incident is more likely when harm occurs. Conversely, the link between a medication error and consequent harm is not always made (e.g. between a failure to administer a prophylactic antibiotic and a subsequent postoperative infection). The fact that no deaths were associated with these reports is interesting. Deaths do sometimes occur after medication errors, and have sometimes been followed by serious legal repercussions.32–34 A reluctance to report a medication error known to have contributed to the death of a patient would be understandable, although unedifying and also contrary to current expectations in relation to both open disclosure and incident reporting. At any rate, there is more than enough serious harm to patients associated with these reports (see Table 5) to underline the clinical importance of this problem.
What then can we take from these reports to improve the safety of anaesthesia practice in the future? We have already acknowledged the challenges faced by anaesthetists in managing the very large numbers of medications they administer. We do not doubt that the vast majority of anaesthetists are trying to do this successfully. Over recent decades there has been a growing understanding that healthcare is a complex system within which human error is an inevitable component,35,36 and we recognise that efforts have been made in many departments to improve the systems by which medications are managed. 12 However, it has also been pointed out that violations, often apparently minor, have an important role to play in the genesis of unintended harm from healthcare, and this fact has rather different implications for the improvement of patient safety;32,35 notably, greater effort to be careful is unlikely to eliminate error, but it may reduce violations. 37 As already indicated, there is a growing emphasis on Safety-II in which guidance for increasing the resilience of healthcare is sought from the many things that go well rather than the (arguably) fewer things that go wrong.22,23 In this regard, there is evidence of the value of focusing on reducing minor failures in patient care, at least in some settings. 38 Unfortunately, some of our reports do suggest that there is still room for greater care in contemporary anaesthetic practice; descriptions of opioid ampoules left in the beds of patients, the administration of medications to the wrong patient, regional blocks on the wrong side, and medications given by wrong routes are depressing, if not chilling. The fact that these errors are continuing to occur as they have always done suggests that more still needs to be done. This is a matter of culture and of priorities for limited resources—the call must be taken up by those who lead departments and hospitals, and by the specialty collectively, as well as by individual practitioners. It must be backed by investment in implementing reasonable and known recommendations to improve medication safety, drawing from the relevant professional documents of the Australian and New Zealand College of Anaesthetists, 39 the Anesthesia Patient Safety consensus statement on a new paradigm for medication safety, 18 and the recent expert consensus statement of Wahr et al. 21
Of course, medication error is not confined to anaesthesia, and we can learn from other fields as well as our own. The third global patient safety challenge of the World Health Organization (WHO), Medication Without Harm, 40 is perhaps the latest authoritative call to arms on medication safety in general. The WHO has identified three key areas for action: transitions of care, high risk situations, and polypharmacy. These all apply to a greater or lesser extent to many patients undergoing anaesthesia. Such action requires a multifaceted approach that recognises the nature of complex systems. 41 To this end the WHO has identified four domains for action: patients and the public; healthcare professionals; medicines and systems; and practices of medication. Efforts to improve medication safety in anaesthesia would certainly be supported by patients and the public, and should ideally be aligned with this global initiative to improve medication safety in general. The starting point should be with explicit hospital and departmental plans for improving medication safety backed by investment in technology and training. However, these will not be effective unless embraced by individual practitioners. A cornerstone of the success of the well-known Keystone project to reduce infection associated with central venous lines was a non-negotiable expectation that, after receiving a reasonable explanation of the importance of the problem and the evidence to support the initiative, clinicians would participate. 42 We suggest that a similar expectation is overdue in relation to safer medication practices. 43
Wahr et al. 21 note that, to date, there has only been one randomised clinical study of an intervention to improve medication safety, 9 although there has also been a randomised study based on high-fidelity simulation 44 and at least two prospective before and after studies based on facilitated incident reporting.12,45 However, Wahr et al. 21 make the point that ‘The dearth of evidence from randomized controlled trials, however, is not permission for us to do nothing, or to view the current state as acceptable.’ Similarly, Webster et al. 46 have emphasised the value of evidence from other study designs, and there is good justification from a wide range of studies for the initiatives identified by the consensus statement of Wahr et al., which are largely in alignment with other recommendations.18,39,47
Investment in technology is one common recommendation, and there is evidence that this can have at least some benefit. 12 However, as can be seen from our data, medication errors reflect many varied forms of failure contributed to by a wide variety of factors. It follows that there are also many other discrete things that can and should be done within an overall systematic approach to improving medication safety that make sense and are likely to reduce the rate of the various types of error reported here. For example, the repeated substitution of an NMBA for midazolam pre-induction could be addressed by providing midazolam in clearly labelled prefilled syringes (thus differentiating their presentations). This approach has had anecdotal success in Auckland City Hospital after a series of such events some years ago. In fact, selected clearly labelled prefilled syringes have considerable potential to reduce many substitution errors, and would incidentally at least partially ameliorate the problem of contamination of IV medications during the process by which they are drawn up. 48 There is a clear message in our data in relation to double checking, and we suggest that with low-volume, high-risk routes of administration (notably spinal and epidural), this should be mandatory. 39 Renewed advocacy is required in relation to look-alike medications, and much can be done in relation to the arrangement of medications in drug drawers. The theme of communication failures comes through strongly in our data, and this can be addressed by training in teamwork and communication, in concert with promoting the importance of these skills within departments and institutions.25,26,49 The problem of flushing residual medication from IV lines is simply a failure in technique, and (again anecdotally) has been addressed in some institutions by an interdepartmental focus on this point of practice and the use of checklists at handover to the PACU. Somewhat similar comments apply to wrong-sided regional blocks. 50 Concealment of IV (and other) cannulae by surgical drapes is again a matter of technique and should either be avoided or should promote double checking to ensure the patency of all connectors. The emerging challenge of computer interfaces with various anaesthesia delivery systems requires a combination of improved design and specific user training. Administering medications intended for a previous patient is surely something that could be eliminated by strict procedural rules in relation to keeping such medications beyond the end of a case or preparing medications too far in advance. Similarly, a failure to have suxamethonium available for urgent use if needed is surely testament to poor departmental policies and procedures. The management of fatigue is complex, but again much can be done given appropriate departmental and institutional culture and commitment.
In conclusion, the incident reports summarised in this paper suggest that medication error in anaesthesia is an ongoing problem which continues to result in avoidable patient harm. The wide variation in the nature of the errors and contributing factors underlines the need for increased systematic and multifaceted efforts underpinned by a strengthening of the current focus on safety culture to improve medication safety in anaesthesia. This will require the concerted and committed engagement of all concerned, from practitioners at the clinical workface, to those who fund and manage healthcare.
Footnotes
Author Contributions
Acknowledgements
The authors would like to thank ANZTADC for their ongoing support of webAIRS.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Alan Merry has financial interests in Safersleep LLC, and is a co-author of a book on medication safety in anaesthesia and the perioperative period which is to be published in 2021, and also of several of the publications referenced in this paper. Craig Webster is a minor shareholder in SaferSleep LLC and a co-author of a number of publications referenced in this paper. Martin Culwick is the medical director of ANZTADC and Alan Merry is the chair of the ANZTADC Publications Group. The remaining authors have no conflicts of interest to declare.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
