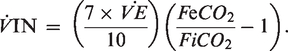Abstract
Powered air-purifying respirators (PAPR) are a high level of respiratory personal protective equipment. Like all mechanical devices, they are vulnerable to failure. The precise physiological consequences of failure in live subjects have not previously been reported. We conducted an observational safety study simulating PAPR failure in a group of nine healthy volunteers, wearing loose-fitting hoods, who were observed for a period of ten minutes, or until they requested the experiment be aborted, with continuous monitoring of gas exchange. Relative to baseline, participants demonstrated median reductions in peripheral oxygen saturation of 3.5% (95% confidence interval (CI) –4% to –2%; P = 0.0039) and fraction of inspired oxygen of 0.045 (95% CI –0.05 to –0.04; P = 0.0039), and median increases in inspired partial pressure of carbon dioxide of 27 mmHg (95% CI 23.5–32 mmHg; P = 0.0039), end-tidal partial pressure of carbon dioxide of 11 mmHg (95% CI 7–16 mmHg; P = 0.0039) and minute ventilation of 30 l/min (95% CI 19.4–35.9 l/min; P = 0.0039). Median collateral entrainment of room air into the hood was 17.6 l/min (interquartile range 12.3–27.0 l/min). All subjects reported thermal discomfort, with two (22.2%) requesting early termination of the experiment. Whilst the degree of rebreathing in this experiment was not sufficient to cause dangerous physiological derangement, the degree of reported thermal discomfort combined with the consequences of entrainment of possibly contaminated air into the hood, pose a risk to wearers in the event of failure.
Keywords
Introduction
Emerging evidence regarding the novel beta-coronavirus, SARS-CoV-2, and the disease it causes, coronavirus disease 2019, suggests that infection may be transmitted via an airborne route. 1 , 2 This in turn has highlighted requirements for an appropriate level of personal protective equipment (PPE) for the healthcare workers who treat these patients, particularly during aerosol-generating procedures. 3 Generally speaking, airborne precautions imply a high level of protection for the oral and nasal mucosa, provided by an appropriate disposable or reusable facial covering that is capable of filtering 95% of particles measuring ≤5 μm (commonly described as an N95 or P2 respirator) or a partial, full-face or loose-fitting air-purifying respirator with a dedicated, positively pressured and filtered air supply. 4 These latter devices are commonly grouped together as powered air-purifying respirators (PAPR).
PAPR have a number of postulated advantages over disposable N95 respirators. 5 While they are more expensive as a single unit relative to other devices, PAPR do not require formal fit testing, 4 are largely protected from supply-chain failure once obtained 6 and are more comfortable when worn for extended periods. 7 Simulation studies have demonstrated that relative to N95 respirators, PAPR do not compromise the ability of the wearer to manage a difficult airway 8 or perform cardiopulmonary resuscitation safely. 9
Like all devices, however, PAPR has an inherent, albeit low, risk of mechanical failure. Common advice from manufacturers in the event of device failure is to exit the contaminated area as quickly as possible. However, this may not always be feasible if the wearer is performing a time-critical clinical task, as (a) abandoning the task suddenly may put the patient at risk (i.e. after administering neuromuscular blockade but prior to intubation), and (b) based on our experience, it takes an experienced operator approximately five minutes to doff their PAPR and full PPE safely. The immediate respiratory consequences of attempting to persist with clinical tasks following such a failure are unknown. Accordingly, we undertook a prospective, observational safety study in healthy volunteers, where we continuously measured gas exchange and other respiratory parameters in a simulation of sudden complete PAPR failure, such as might be experienced if the battery supply to the motor unit were inadvertently disrupted. We hypothesised that such a simulated failure would result in either (a) progressive rebreathing of expired gas, with an associated risk of hypoxia (and hypercapnia), or (b) unacceptable entrainment of potentially contaminated air into the hood from the external environment.
Methods
Study design and participants
Approval was granted by the Austin Health Human Research Ethics Committee (HREC/63489/Austin-2020). All participants gave written consent prior to any study-related procedure. Potential participants were eligible for inclusion if they were medical or nursing members of the Austin Health Department of Anaesthesia Coronavirus Intubation Team and had received prior practical training in the safe use of PAPR. There were no specific exclusion criteria. An invitation to participate in the study was distributed electronically to all eligible participants. Those who responded positively were enrolled into the study until the predefined sample size was achieved, after which the study was closed to recruitment.
Equipment
Participants were asked to don the Austin Health Department of Anaesthesia Coronavirus Intubation Team standard PPE. This consisted of a Sundström SR700 PAPR fan unit and a SR530 loose-fitting PAPR hood (Sundström Safety Ab, Stockholm, Sweden), over which were worn a disposable polypropylene surgical gown and hood with inbuilt Perspex visor. A mouthpiece connected via a Pro-Tec® PF30S microbiological filter (Pall Corporation, Port Washington, NY, USA) to a Fleisch pneumotachograph was inserted under the hood, with sampling tubing connected to a pressure-flow transducer (Validyne Engineering, Northridge, CA, USA) for monitoring of minute ventilation, and a rapid gas analyser (Datex Capnomac Ultima; General Electric, Inc., Boston, MA, USA) for recording of tidal gas concentrations via standard sidestream gas sampling tubing. A nose clip was used to ensure that the mouthpiece would capture all inspired and expired gas. As the hood was loose-fitting, the sampling tubing resulted in minimal disruption of gas flow in and out of the hood and was considered to represent a negligible change in hood performance. A pulse oximeter was placed on the participant’s finger and connected to an anaesthesia machine (Maquet/Getinge Flow-i, Stockholm, Sweden) with integrated monitor (Philips IntelliVue MP70, Eindhoven, The Netherlands). The anaesthetic machine also served as an emergency source of oxygen and resuscitation equipment if required. All monitoring devices were interfaced for data download and storage in real time to a notebook computer (Apple Corp., Cupertino, CA, USA) running a custom-made programme constructed using LabVIEW 2011 software (National Instruments, Austin, TX, USA) via an analog-digital converter (USB 6009; National Instruments). A 20G intravenous cannula was placed in the cubital fossa to allow repeat sampling of venous blood at pre-specified timepoints during the study procedure.
Study procedure
Each study procedure was supervised by two members of the study team. Following correct application of monitoring, participants were asked to sit, and a three-minute period of rest (with the PAPR fan active) commenced to allow baseline steady-state conditions to be achieved. Venous blood was sampled during this period and stored on ice in a heparinised syringe for subsequent blood gas analysis. Recording of baseline respiratory flow rates, tidal gas concentrations and pulse oximetry was also commenced. Data were collected on patient characteristics, specifically age (years), weight (kg), height (cm) and sex. Recorded respiratory variables were pulse oximetry (SpO2) (%), fraction of inspired oxygen (FiO2), partial pressure of inspired CO2 (PiCO2; mmHg), partial pressure of end-expired CO2 (P

Example physiological variable output from a sample experiment over time.  : partial pressure of CO2 (mmHg);
: partial pressure of CO2 (mmHg);  : minute ventilation (l/min);
: minute ventilation (l/min);  : peripheral oxygen saturation (%);
: peripheral oxygen saturation (%);  : partial pressure of O2 (mmHg);
: partial pressure of O2 (mmHg);  : heart rate (beats/min). T=0 and T=1 are shown by
: heart rate (beats/min). T=0 and T=1 are shown by  .
.
Following this period of rest, the participant was asked to deactivate the PAPR fan unit, and filtered gas flow into the hood ceased. The point of deactivation was defined as T = 0. Participants were asked every 60 seconds if they wished to continue the study procedure. Participants were required to respond immediately in the affirmative or the experiment was terminated. Recording of study parameters then continued until one of four criteria to end the experiment were satisfied: (a) SpO2 fell below 90% for ten seconds, (b) SpO2 fell below 88% at any point, (c) the participant failed to respond in the affirmative at any of the predefined assessments (as above) or otherwise indicated that they wished to terminate the test or (d) ten minutes had elapsed from T = 0.
The time at which the experiment termination criteria was achieved was defined as T = 1. Once T = 1 was reached, repeat venous blood sampling was performed, following which the PAPR fan unit was then reactivated. Respiratory data continued to be recorded until SpO2 returned to baseline, defined as T = 2. Participants were allowed to return to work as soon as oxygenation and other vital signs were within normal limits and they felt able to do so.
The primary outcome was time to achieve a SpO2 of <90%. Secondary outcomes were time to recovery of SpO2 >95% after T = 1, minimum recorded FiO2, maximum PiCO2 and P
Derivation of this equation on mass balance principles can be found as the Supplemental Material to this manuscript.
We did not undertake a formal sample-size calculation. A predetermined sample size of nine participants was used for the study. It was recognised that the confidence intervals (CI) for this small sample size could be relatively wide (standard error ±30% of the mean) if considerable inter-individual variance was present for certain variables. If the inter-individual variance was not large, then a wide CI would not be expected, irrespective of sample size. Regardless of what the observed variance might be, we expected the standard deviation (SD) and range of values encountered would be sufficient to indicate the limits of safety of the device during simulated fan failure.
Data are presented as mean (SD) or median (interquartile range (IQR)) for continuous variables, depending on normality. Statistical comparisons were performed for recorded physiological data between T = 0 and T = 1. Data were examined for normality. Parametric variables were analysed using the two-tailed Student’s t-test. Non-parametric variables were analysed using the Wilcoxon signed-rank test. The median of differences and their 95% confidence intervals were calculated using the Hodges–Lehmann estimator. Statistical analyses were performed using Stata v16.1 (StataCorp LLC, College Station, TX, USA).
Results
Descriptive data for all nine participants in the final study cohort are outlined in Table 1. One patient was not able to have a venous sampling performed at T = 1 due to cannula failure. There were no other missing data. The median age of the study cohort was 36 years (IQR 34–43 years), the median weight was 72 kg (IQR 61–88 kg) and the median height was 176 cm (IQR 167–183 cm).
Description of participants.
No participant achieved the primary endpoint of a SpO2 of <90%.
Changes in physiological variables between baseline (T = 0) and peak/nadir values (generally correlating with T = 1) are outlined in Table 2. Statistically significant increases were noted in HR,
Physiological variables.
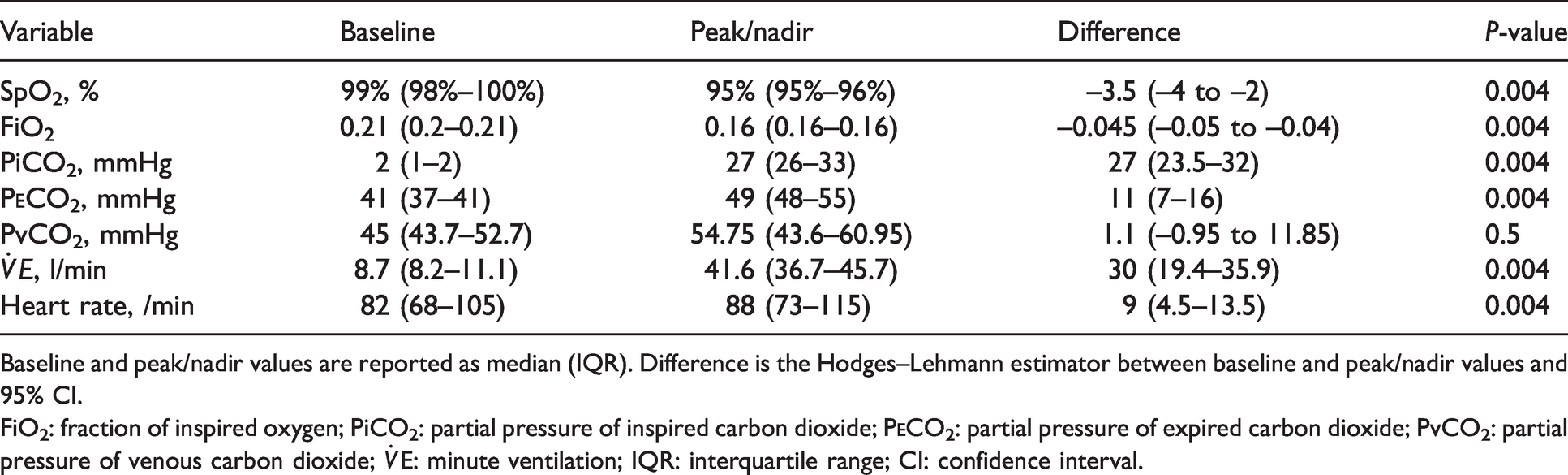
Baseline and peak/nadir values are reported as median (IQR). Difference is the Hodges–Lehmann estimator between baseline and peak/nadir values and 95% CI.
FiO2: fraction of inspired oxygen; PiCO2: partial pressure of inspired carbon dioxide; P

Study population median (IQR) trends in minute ventilation over time ( ;
;  : median;
: median;  : IQR), and partial pressures of end-tidal CO2 (
: IQR), and partial pressures of end-tidal CO2 ( ;
;  : median;
: median;  : IQR) and inspired CO2 (
: IQR) and inspired CO2 ( ;
;  : median;
: median;  : IQR). IQR: interquartile range.
: IQR). IQR: interquartile range.
Importantly, no participant recorded increases or decreases in measured physiological values that were considered dangerous for the time period examined in this study (ten minutes). No experiment was terminated due to hypoxaemia. The median time to lowest SpO2 after T = 0 was 245 seconds (IQR 151–317 seconds). SpO2 recovered rapidly after reactivation of the PAPR fan, with a median recovery time of 36 seconds (IQR 34–43 seconds). Two (22%) participants terminated the test due to intolerable thermal discomfort. Both of these tests were ended at the six-minute mark. The remaining experiments ended at the planned ten-minute limit. However, significant thermal discomfort was reported by all participants, despite the experiments being conducted effectively at rest. The subjective experiences voiced directly by the study participants immediately after the experiment ended are outlined in Table 3. All participants believed their clinical performance would be impaired if they experienced a similar PAPR failure in a real-life situation.
Direct commentary from participants on the subjective experience of the described experiment.

Collateral ventilation entrained externally into the PAPR hood was derived from the equation outlined in the methods. Using this technique, median collateral ventilation at peak
Discussion
We conducted a prospective, observational safety study of simulated PAPR failure in a cohort of nine healthy volunteers. We found that such a simulated failure, with the participant at rest, was unlikely to result in sufficient rebreathing of expired gas (and associated hypoxaemia and/or hypercapnia) to incapacitate or harm the wearer. However, we did show a marked increase in minute ventilation over the course of the experiment, likely a due to multifactorial stimulation of the respiratory centre (reflective of hypercapnia, thermal distress and anxiety), which appeared to be supported by substantial collateral entrainment of room air from outside the hood. These findings imply that were the failure to occur in an aerosol-contaminated environment, entrainment of viral particles into the hood could theoretically lead to the inadvertent infection of the wearer. Additionally, thermal distress forced the early termination of the experiment in a minority of participants, and the remaining participants reported an increase in thermal distress sufficient to impair subjective clinical performance.
To our knowledge, few studies in the infection control or occupational health literature have attempted to examine the ventilatory consequences of sudden PAPR failure. In particular, collateral ventilation, and the associated risk of aerosol entrainment, has not previously been quantified in a live subject. A previous study using a manikin head and torso and a breathing simulator was undertaken by Bostock in 1985. The author demonstrated that at a simulated
The relative risk of wearer contamination in the event of sudden PAPR failure is unknown, although various factors could foreseeably increase this risk. These include high wearer
Perceived thermal discomfort was reported by all participants in this study, and resulted in early termination of two of the experiments. A steady state was reached for P
Increased thermal distress in the setting of simulated failure has been previously described for tight-fitting PAPR. In an observational study by Johnson et al., participants were asked to engage in physical activity (defined as 80–85% of maximum oxygen consumption
We acknowledge some limitations. First, the derived mass balance equation for collateral inflow (Appendix 1) assumes steady-state gas exchange. Dynamic collateral flow could be derived (and has been in manikin studies) if the volume of the enclosed hood and the head of the participant were known. As not all of this information was available to us, we elected not to present data on change in collateral inflow over time, instead presenting maximum collateral inflow in isolation. We do not believe that not being able to derive dynamic collateral flow detracts much from our main findings, specifically that, based on these results and other simulation studies, PAPR failure results in rapid entrainment of external gas into the hood, and there is a risk of user contamination. Whilst it is not possible using this study protocol and a live subject to measure particulate count inside the hood, we note that some centres wear loose-fitting PAPR hoods, with the hood hem not covered by either an isolation gown or another layer of disposable PPE, 19 and that entrainment of air into the hood from the external environment, unfiltered by additional PPE (a) could result in direct exposure of the wearer to infectious particles, and (b) based on the results of Bostock et al., such exposure could occur almost immediately after PAPR failure. 10 Second, the study was performed with the participants at rest, and therefore would not be a direct representation of the physiological changes that would occur if the participants had been engaging in simulated clinical tasks. However, accurate measurement of gas exchange whilst the participant was engaging in physical activity was found to be unfeasible while we were developing the study protocol, and the central thesis of this study remains unchanged—that whilst physiological derangement was successfully limited by collateral inflow, subjective thermal distress was still reported in all participants, and it was felt that this degree of discomfort would substantially affect clinical performance. Finally, temperature inside the hood was not measured as part of the study, as thermal distress was not included in our hypothesis. Consequently (and unfortunately), we did not incorporate temperature measurement inside the hood into the study design. It is possible that many of the phenomena reported by the participants as thermal discomfort may have been due to acute hypercapnia, which is recognised to cause increased heart rate, cardiac output, dyspnoea and peripheral blood flow. 20 However, this is a moot point. Whatever the cause of the reported thermal discomfort—whether it be subjective (due to hypercapnia) or objective (due to an actual increase in temperature inside the hood)—it was reported by all participants. It is the subjective impression of thermal discomfort that impairs clinical performance, rather than an objective temperature.
In summary, we have undertaken a prospective, observational safety study of ventilatory parameters in a simulated model of unexpected complete PAPR failure in healthy volunteers at rest. While our results do not show that rebreathing sufficient to cause dangerous physiological derangement occurs under these test conditions, we have shown that the rebreathing of expired gas which does occur is offset by considerable collateral entrainment of room air, exposing the wearer to possible aerosol contamination. Additionally, a marked increase in subjective thermal distress is reported, sufficient to terminate the experiment in some participants and a self-evaluated deterioration in likely clinical performance by others. On the basis of these findings, we urge departments that have recently incorporated PAPR into their practice to develop protocols for wearers to follow in the event of sudden or anticipated PAPR failure, as well as defining criteria for potential exposure in the event of PAPR failure a priori to inform subsequent wearer furloughing if required.
Supplemental Material
sj-pdf-1-aic-10.1177_0310057X20978982 - Supplemental material for Ventilatory parameters measured during a physiological study of simulated powered air-purifying respirator failure in healthy volunteers
Supplemental material, sj-pdf-1-aic-10.1177_0310057X20978982 for Ventilatory parameters measured during a physiological study of simulated powered air-purifying respirator failure in healthy volunteers by Lachlan F Miles Philip J Peyton in Anaesthesia and Intensive Care
Footnotes
Acknowledgements
The authors would like to acknowledge the medical and nursing members of the Austin Health Department of Anaesthesia Coronavirus Intubation Team who volunteered to participate in this study.
Author contribution(s)
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Supplemental Material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.