Abstract
TEG6s® is a new device introduced by the Haemonetics Corporation and designed to provide the same information as TEG® 5000 (Haemonetics Corporation, Braintree, MA, USA) but with much greater ease of use. We tested whether using citrated TEG6s gave reaction time, maximum amplitude and percentage of clot that had lysed at 30 minutes values similar to a non-citrated TEG5000, to allow clinical interchangeability using our current thrombelastography management algorithm for cardiac surgery. We also examined the agreement between the alpha-angle and functional fibrinogen maximum amplitude in our cardiac surgical patients.
In total, 243 paired arterial blood samples in 99 patients were tested, using TEG5000 (non-citrated) and TEG6s (citrated) after induction of anaesthesia (prior to heparin administration), following protamine administration at the end of the cardiac bypass and whenever a TEG5000 was requested after this by the attending anaesthetist. Bland–Altman plots and Lin’s concordance coefficient were used to compare agreement whereas modified Bland–Altman plots and McNemar’s test were used to illustrate the differences in management recommendations between the two thrombelastography devices.
All 243 samples were compared for reaction time and alpha-angle; 239 samples were compared for maximum amplitude; 136 samples were compared for the percentage of clot that had lysed at 30 minutes; 16 samples were compared for functional fibrinogen maximum amplitude. Lin’s concordance coefficient for these parameters was: reaction time 0.63, alpha-angle 0.39, maximum amplitude 0.5, percentage of clot that had lysed at 30 minutes 0.09 and functional fibrinogen maximum amplitude 0.31. Differences between the two devices became more marked at more abnormal values. Significant differences in median values, suggesting a fixed bias, were found for maximum amplitude and functional fibrinogen maximum amplitude. Differences in treatment recommendation could only be calculated for reaction time and maximum amplitude. Maximum amplitude was found to have a significant difference in treatment recommendation between the two devices using our current thrombelastography management algorithm for cardiac surgery with TEG6s recommending treatment in 11.5% more patients than TEG5000.
Using the TEG6s with our current TEG5000–based thrombelastography management algorithm for cardiac surgery would result in a change in treatment recommendation in at least 10% of our cardiac surgical patients. Agreement between the two thrombelastography devices appears to decrease with increasing patient coagulopathy. New algorithms will need to be developed and tested to validate TEG6s for cardiac surgical patients in our institution.
Keywords
Introduction
Thrombelastography (TEG) is a commonly used viscoelastic haemostatic assay (VHA) that measures the properties of whole blood clotting. The ability to detect disorders of coagulation not adequately detected by conventional coagulation tests and a comparatively fast turnaround time make TEG useful in clinical settings such as cardiac surgery and trauma. VHAs are recommended in the National Institute for Health and Care Excellence guidelines during and after cardiac surgery to monitor coagulation. 1
The TEG®5000 (Haemonetics Corporation, Braintree, MA, USA) measures whole blood clotting by calculating increasing torsion on a wire suspended in a small cup of clotting blood that rotates backward and forward between 0 degrees and 45 degrees. 2 The use of this as a true point-of-care test has several limitations. These include the need for the TEG5000 machine to be calibrated daily, the need for standard techniques to be used, the need for trained skilled personnel to perform these tests, and the need for the machine to be placed in a low-vibration environment where movement of the machine will not occur. TEG6s® (Haemonetics Corporation, Braintree, MA, USA) is a newly released device that uses the resonant frequency of a clotting blood sample to produce the same clotting measures and visual representation produced by the TEG5000 device. Its purported advantages are ease of use, less frequent calibration requirements, ability to perform four tests simultaneously on a single cartridge and less sensitivity to movement.
The parameters used by both TEG5000 and TEG6s to specify initiation, amplification, propagation of clot and its breakdown include reaction time (R-time), kinetic time (K-time), alpha-angle (α-angle), maximum amplitude (MA) and percentage of clot lysed at 30 minutes (LY 30). Protocols derived from these values are used to guide product transfusion. The current TEG5000–based TEG management algorithm for cardiac surgery (TMACS) used at our institution can be found in Figure 1.
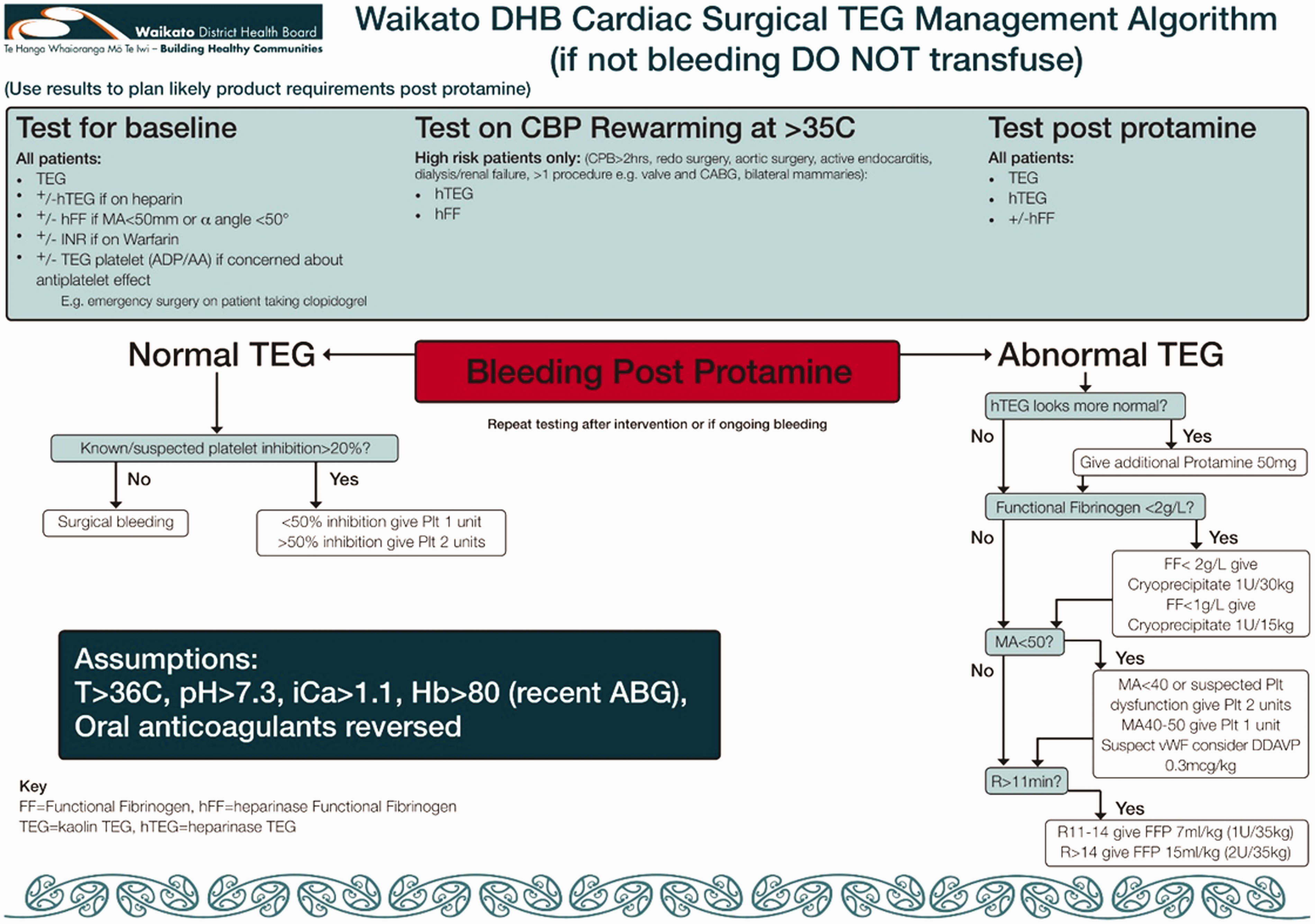
Waikato District Health Board cardiac surgical thrombelastography management algorithm. TEG: thrombelastograph; INR: International Normalized Ratio; CBP/CPB: cardiopulmonary bypass; CABG: coronary artery bypass surgery; ADP/AA: adenosine diphosphate and arachidonic acid; Plt: platelets; vWF: von Willebrand Factor; DDAVP: desmopressin; MA: maximum amplitude; FFP: fresh frozen plasma; Hb: haemoglobin; ABG: arterial blood gas.
Haemonetics Corporation (Braintree, MA, USA) states their data shows substantial equivalence between TEG6s and TEG5000, with results being translatable to clinical decision points. 3 A study looking at agreement between TEG5000 and TEG6s using samples from healthy patients with various doses of clopidogrel showed good levels of agreement using TEG platelet mapping between TEG6s and TEG5000 results. 4 The first technical study was published in 2016 5 and produced normal reference ranges for TEG6s (see Table 1). Recently a study involving ten patients undergoing liver transplant compared TEG5000 non-citrated and citrated samples against TEG6s and found poor agreement particularly with values in the abnormal range, 6 whereas another study involving 25 intensive care patients found ‘near perfect’ agreement comparing citrated TEG values between TEG5000 and TEG6s with almost all values in the normal range. 8
Normal thrombelastography values. 5

R-time: reaction time; K-time: kinetic time; α-angle: alpha-angle; MA: maximum amplitude; LY 30: percentage of clot lysed at 30 minutes.
To investigate a ‘real-world’ situation using an actual treatment algorithm, we undertook paired blood sample analysis of 99 patients undergoing cardiac surgery at baseline, post-bypass following reversal of heparin, and whenever the attending anaesthetist asked for a TEG5000 test post protamine to treat coagulopathy as per our established TEG5000–based TMACS.
Methods
This was a single-centre prospective trial conducted at Waikato Hospital, New Zealand, which is a tertiary referral hospital performing 500–600 cardiac surgical cases requiring cardiopulmonary bypass (CPB) annually. Local District Health Board ethics approval and HDEC National Ethics approval was obtained on 15 June 2016 (reference 16/STH/82) and informed consent was obtained from every patient. Patients were recruited between 27 October 2016 and 30 June 2017.
A TEG6s machine and the necessary global cartridges were provided free of charge by the Haemonetics Corporation for the purposes of the study. The Haemonetics Corporation had no input into the study design, data collection, analysis, interpretation or drafting of the manuscript. The TEG5000 machines are owned by the hospital and testing was only performed according to established hospital protocols as a standard of care for cardiac surgical patients.
The primary outcome of the study was to determine whether using TEG6s and a global cartridge gave results similar enough to a non-citrated TEG5000 to allow clinical interchangeability of the two devices using the current TMACS at our institution. Secondary outcomes were to investigate the agreement between parameters produced on the TEG6s global cartridge and those available on TEG5000 in a cardiac surgical population.
Inclusion criteria were adult patients booked for elective or semi-urgent (in-hours surgery but admitted acutely) cardiac surgery with CBP. Patients were excluded if they had received an adenosine diphosphate receptor antagonist, warfarin, low molecular weight heparins, Xa inhibitors or IIa inhibitors in a time frame consistent with the drugs still having a clinical effect at time of surgery. Other exclusion criteria included emergency surgery, inability to speak English, age less than 18 years or an inability to provide informed consent.
The calculation of sample size was hampered by a paucity of published studies. We took a pragmatic approach where we considered a 10% difference in MA to be clinically important. Assuming TEG6s values could be higher or lower than the TEG5000 value and an alpha of 0.05, this gave a sample size of 97 patients. A total of 99 patients were recruited. This may have underpowered the study for other parameters evaluated; however, each included patient had at least two paired samples taken at different times during surgery, more than doubling the paired samples for comparison for most parameters. See Figure 2 for details.
Blood was taken from patients after the start of anaesthesia and prior to heparin administration (baseline) and nine minutes after the reversal of heparin with protamine after CPB (post-protamine). Following removal of a minimum of 7 ml of blood from the radial arterial line (to remove approximately five line dead spaces), sample blood was taken from the arterial line into two separate 3 ml syringes. Subsequently, 2.7 ml of blood from one of the syringes was then immediately transferred into a 0.109 M citrated blood tube (BD Vacutainer® 9NC by Becton, Dickinson and Company, USA) and inverted five times. After inversion of the citrated blood tube, blood from the tube was allowed to sit for four minutes then used to perform TEG6s testing using a global cartridge as per manufacturer instructions. The other syringe was used to immediately perform TEG5000 testing. The specific tests run on the TEG5000 (kaolin, kaolin with heparinase (KH) and functional fibrinogen (FF)) were performed as per manufacturer recommendations and according to our institutional TMACS (Figure 1). All testing was performed by one of seven cardiac anaesthetic technicians, all of whom were hospital approved regular users of the TEG5000 and were all trained to use the TEG6s by Haemonetics Corporation prior to study commencement.
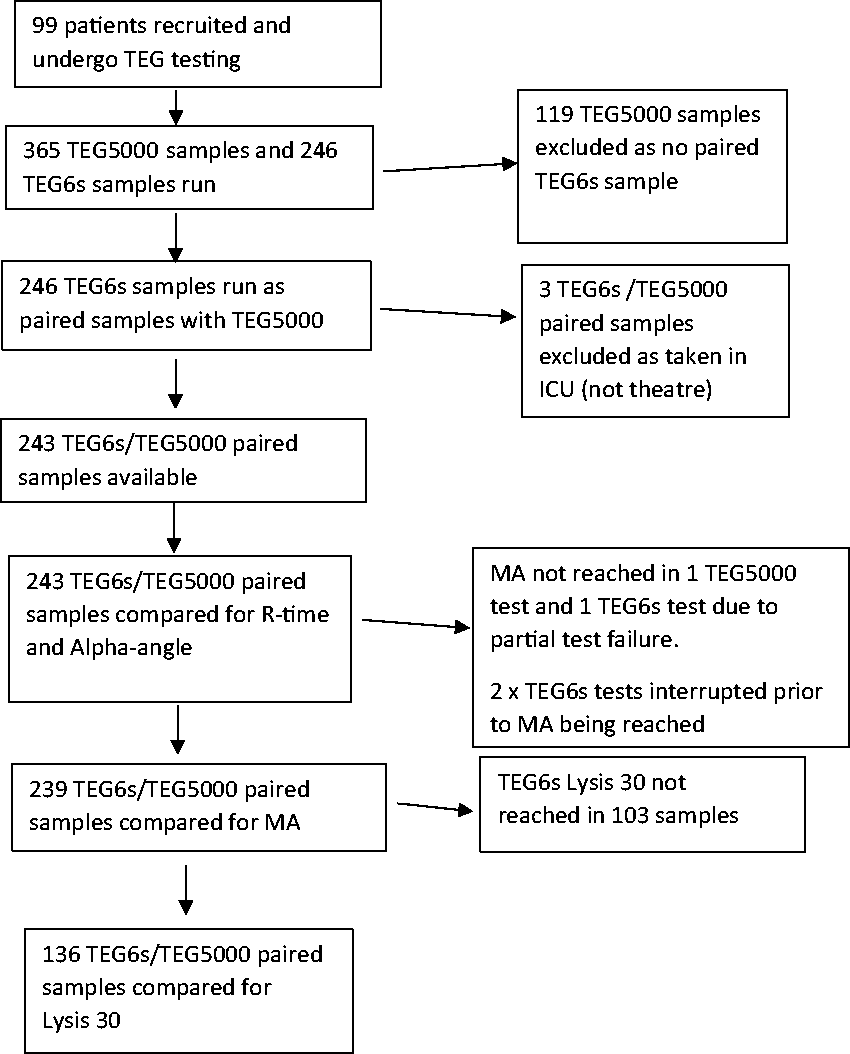
Exclusion flow chart. TEG: thrombelastography; TEG5000 and TEG 6s (Haemonetics Corporation, Braintree, MA, USA); ICU: intensive care unit; R-time: reaction time; MA: maximum amplitude; Lysis 30: percentage of clot lysed at 30 minutes.
Results of the TEG5000 machine were available to the anaesthetist in real time via a monitor in theatre as per standard practice. Transfusion was in accordance with the institutional TMACS based on the TEG5000 results (Figure 1).
Both the TEG machines recorded each test using the patient’s National Health Index number, age, sex, date and time and results were copied from the devices into a Microsoft Excel® (Microsoft Corp, USA) spreadsheet by the Hospital Point of Care Coordinator.
Non-paired tests were excluded from analysis: for example, where a TEG5000 test was performed but no matched TEG6s test performed. RapidTEG™ (Haemonetics Corporation, Braintree, MA, USA) is not routinely done on the TEG5000 machines at our institution so was not available for comparison.
Comparisons were made between equivalent test types. For example, the R-time on TEG5000 using a KH test was compared with the R-time on TEG6s using citrated KH (CKH). Analyses were performed using Microsoft Excel and Social Science Statistics (https://www.socscistatistics.com). Lin’s concordance was calculated using the New Zealand National Institute of Water and Atmospheric research online calculator (https://www.niwa.co.nz/services/statistical). Combining both pre-CPB and post-protamine data, Bland–Altman plots were created for each of the following type of paired TEG test parameters: R-time, α-angle, MA and LY 30 and for FFMA. Clinical interpretations were determined using the TMACS for R-time, MA and lysis, and proposed treatment decisions determined by the TEG5000 and TEG6s were recorded as a binary of whether treatment was indicated for that measurement. The number of patients for whom treatment (for example platelet administration) was recommended and not recommended for a given TEG parameter (for example MA) were compared between the two machines using McNemar’s test (paired chi-squared).
Lin’s concordance coefficient compares agreement between two methods for measuring the same variable. Values near +1 indicate a strong concordance between the two methods, values near −1 a strong discordance, whereas values near 0 indicate no concordance. Intermediate values are less clearly defined, although several authors have defined values >0.8 representing excellent concordance and those <0.2 poor concordance. Several previous studies have also used Lin’s concordance coefficient to compare values returned by TEG5000 and TEG6s.6,8
Results
In total, 99 patients were recruited and had blood taken for testing, with a total of 365 blood samples. Male patients accounted for 267 samples. Patients were aged between 26 and 85 years with a mean patient age of 66.2 years.
All 365 samples were tested using TEG5000 but only 246 were tested using the TEG6s, giving 246 paired samples for comparison. This difference was due almost entirely to unavailability of a study approved technician to run the TEG6s sample. Three of these 246 paired samples were rejected from analysis because the timing of the sampling was outside of study protocol. Results from kaolin-TEG, KH-TEG and FF were available for comparison.
In total, 243 measurements were compared for the R-time and α-angle and 239 measurements were compared for MA. This difference was due to failure of the test in two cases (1x TEG6s, 1x TEG5000) and interruption of the test in two cases (TEG6s).
LY 30 data from the TEG6s were only available in 136 paired samples because 103 of the TEG6s tests were terminated prior to LY 30 occurring. This was either to allow for further TEG6s testing on another patient or as part of the cardiac technician resetting all equipment in readiness for the next cardiac case. Unfortunately, only one TEG6s machine was available at a time for up to two concurrent patients.
Only 16 paired samples were available for FFMA. This was because of a difference in the FF run in the TEG6s global cartridge (which is non-heparinase) and the FFs run as part of our TEG5000 bleeding management algorithm (which is a heparinase FF).
The TEG parameters were assessed using a Bland–Altman plot. For R-time there was a large cluster of results within the normal range (<11 min) (Figure 3). This was expected as many of the samples come from pre-CPB and should be normal. There were fewer patients with abnormal TEG results (R>11 min). Despite this, at a mean R-time of >15 min there is an appreciable increase in the variation in value returned between the two machines. The Bland–Altman plot shows that R-time agreement between the two machines decreases with longer (more abnormal) median R-time.
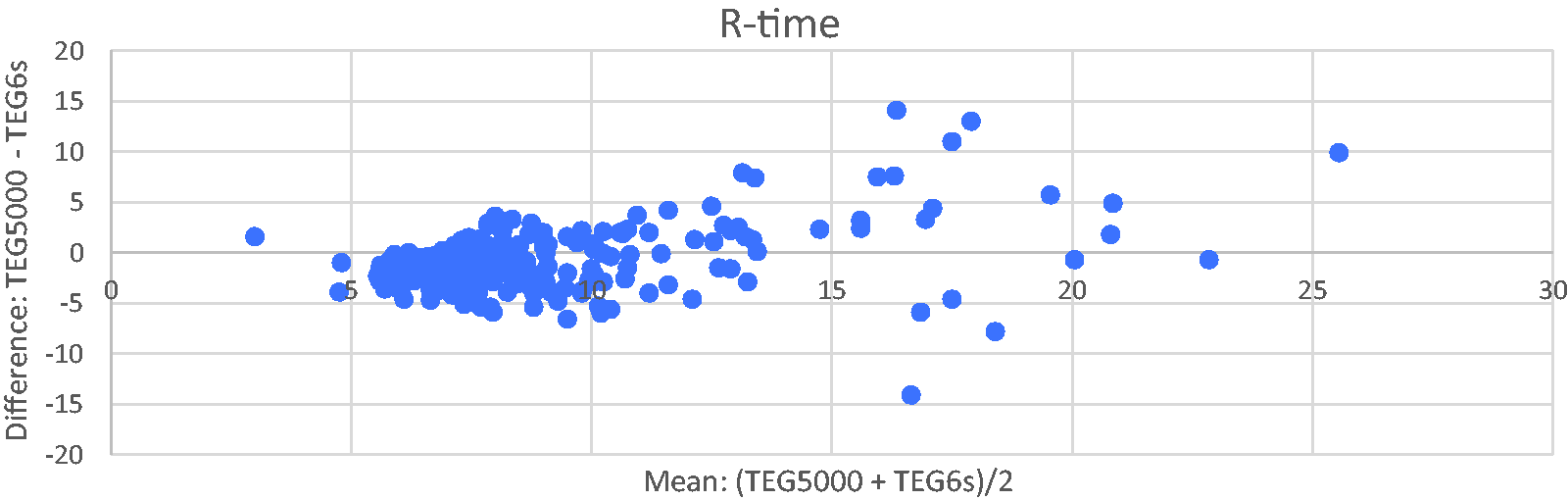
Bland–Altman plot of reaction time (R-time) for TEG5000 vs TEG6s (Haemonetics Corporation, Braintree, MA, USA).
Results for the α-angle were similar to R-time, with a large number of results clustering above 65 degrees (normal range) having differences of less than 10 degrees (Figure 4). With values less than 65 degrees the agreement between the two machines decreased, and at an α-angle mean of <48 degrees, TEG6s appeared to return much higher α-angle values than the TEG5000. However, there are only seven data points with a mean α-angle of <48 degrees.
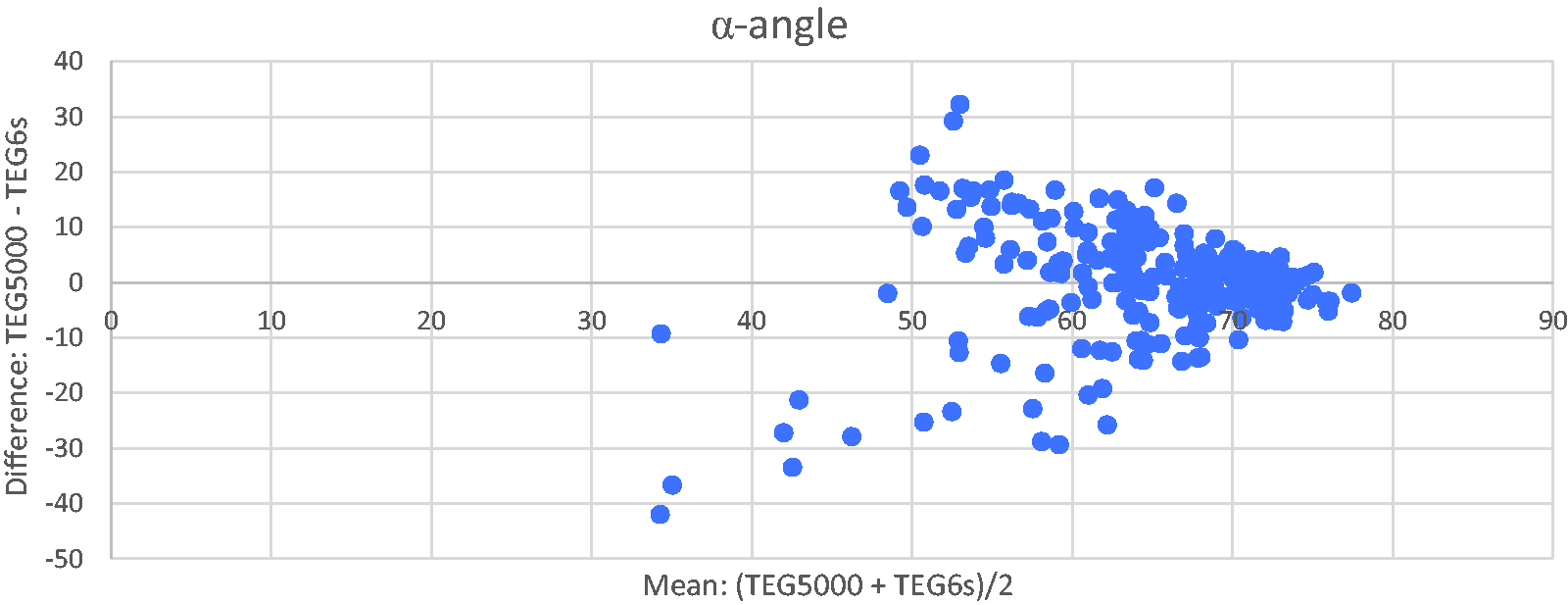
Bland–Altman plot of alpha-angle (α-angle) for TEG5000 vs TEG6s (Haemonetics Corporation, Braintree, MA, USA).
Results for MA show a cluster of results with mean MA >50 mm with very few data points below this (Figure 5). Once again, this is in keeping with most of the patients in the study having normal clotting. The Bland–Altman plot suggests a bias (median difference 5.8, 95% confidence intervals 5.1 to 6.7) between the two devices for MA, with the TEG5000 giving a higher MA than the TEG6s. This is visually represented on the Bland–Altman plot by the centre of the cluster being above the zero-difference line.
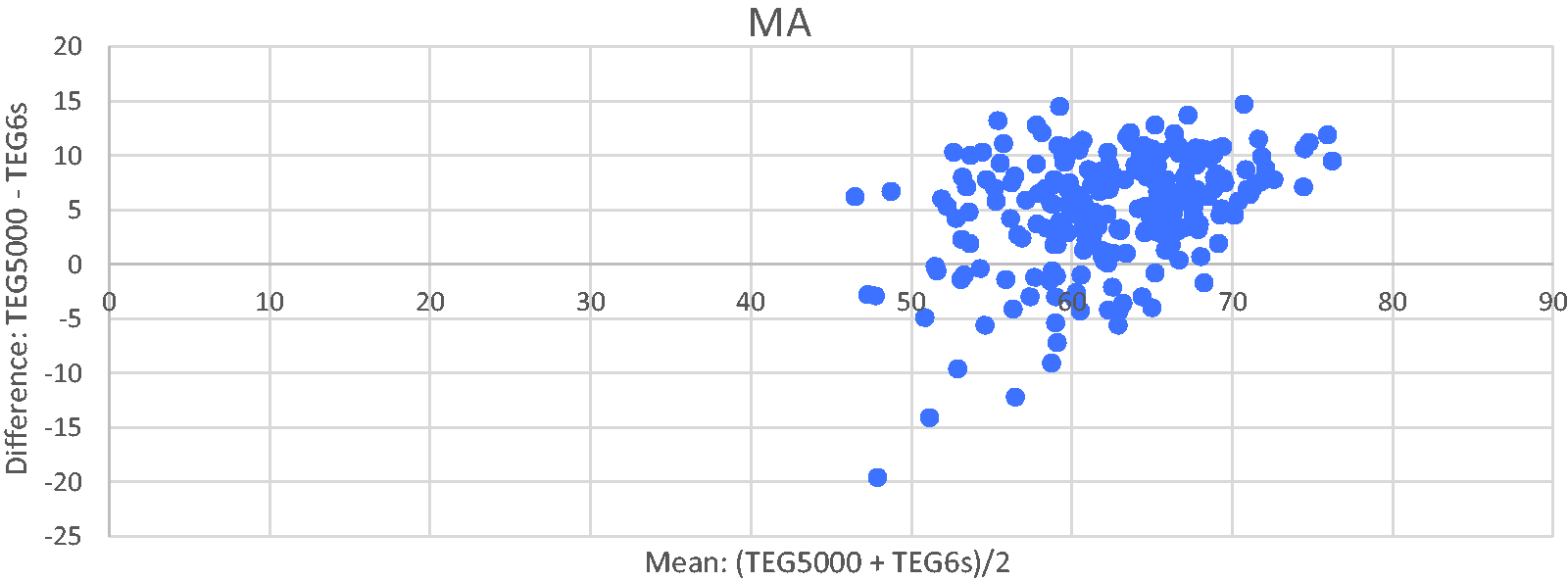
Bland–Altman plot of maximum amplitude (MA) for TEG5000 vs TEG6s (Haemonetics Corporation, Braintree, MA, USA).
There were 136 paired samples available for LY 30. In the majority of samples there was no lysis. In samples where lysis occurred, the Bland–Altman plot shows an increasing difference between TEG5000 and TEG6s values with increasing mean LY 30 (Figure 6). LY 30 with mean values >2% demonstrate an almost linear relationship between TEG5000 and TEG6s with TEG5000 LY 30 increasing whereas TEG6s LY 30 remains largely unchanged.
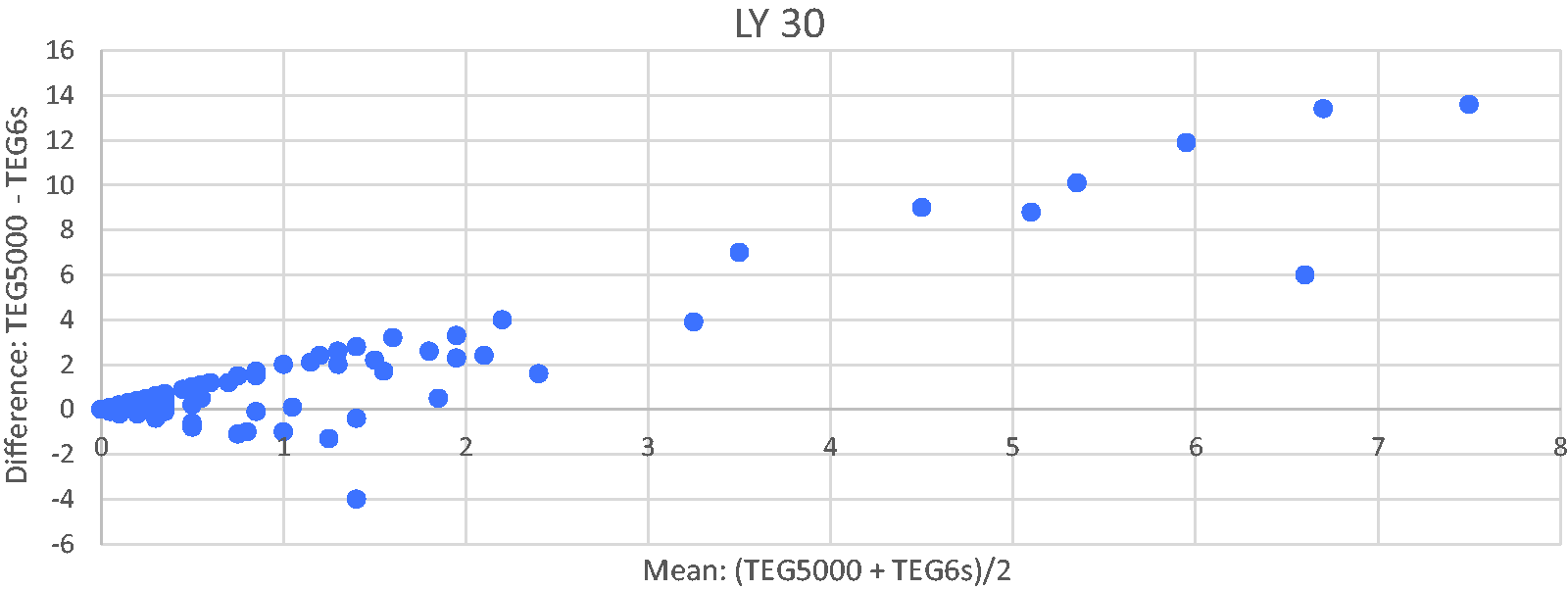
Bland–Altman plot of percentage of clot lysed at 30 minutes (LY 30) for TEG5000 vs TEG6s (Haemonetics Corporation, Braintree, MA, USA).
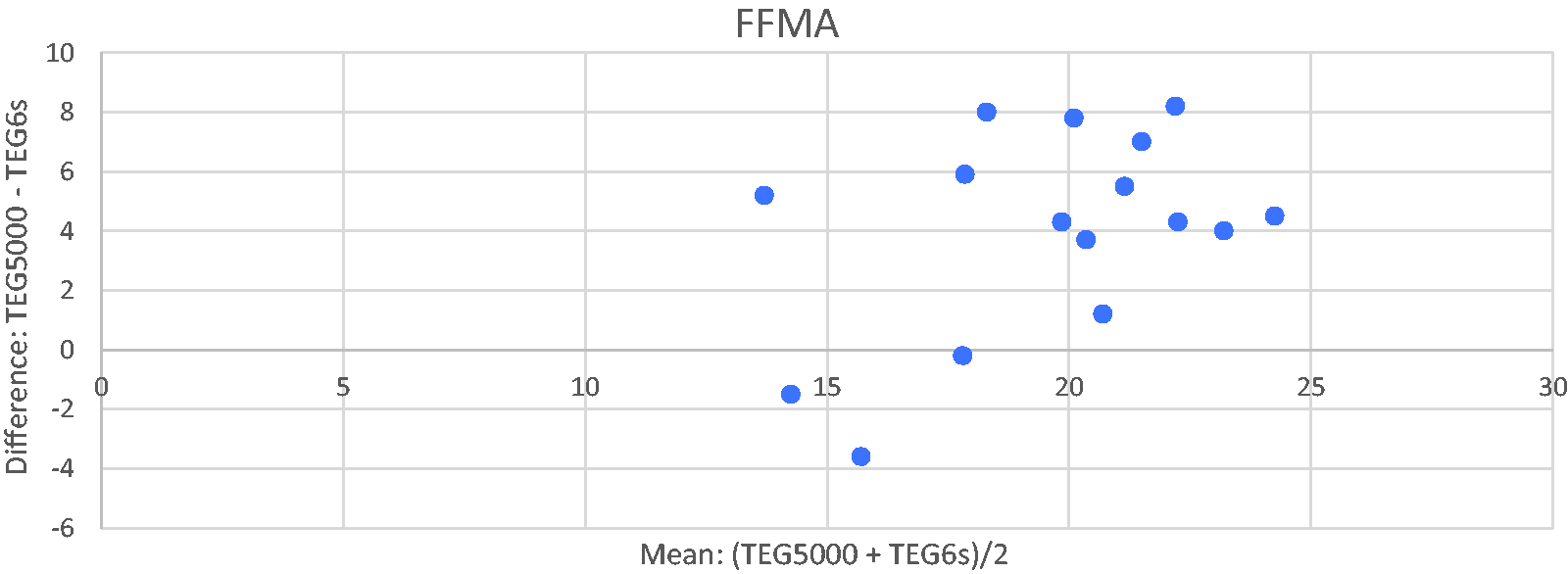
Bland–Altman plot of functional fibrinogen maximum amplitude (FFMA) for TEG5000 vs TEG6s (Haemonetics Corporation, Braintree, MA, USA).
Only 16 paired data points were available for FFMA (Figure 7). It is difficult to draw any firm conclusions from this although there was a statistically significant difference with TEG5000 returning higher FFMA values than TEG6s (median difference 5.7 mm, 95% confidence intervals 4–8 mm) visualised on the Bland–Altman plot with most of the data points lying above the zero-difference line.
Lin’s concordance coefficient was calculated for R-time, α-angle, MA, LY 30 and FFMA (Table 2). McNemar’s test was used to compare whether there was a significant difference in the number of patients in whom treatment was or was not recommended (for example platelets for a low MA) by the two machines.
Median, median difference, 95% confidence intervals for the median difference, McNemar’s P statistic test (for differences in treatment recommendations) and Lin’s concordance coefficient for R-time, α-angle, MA and LY 30.
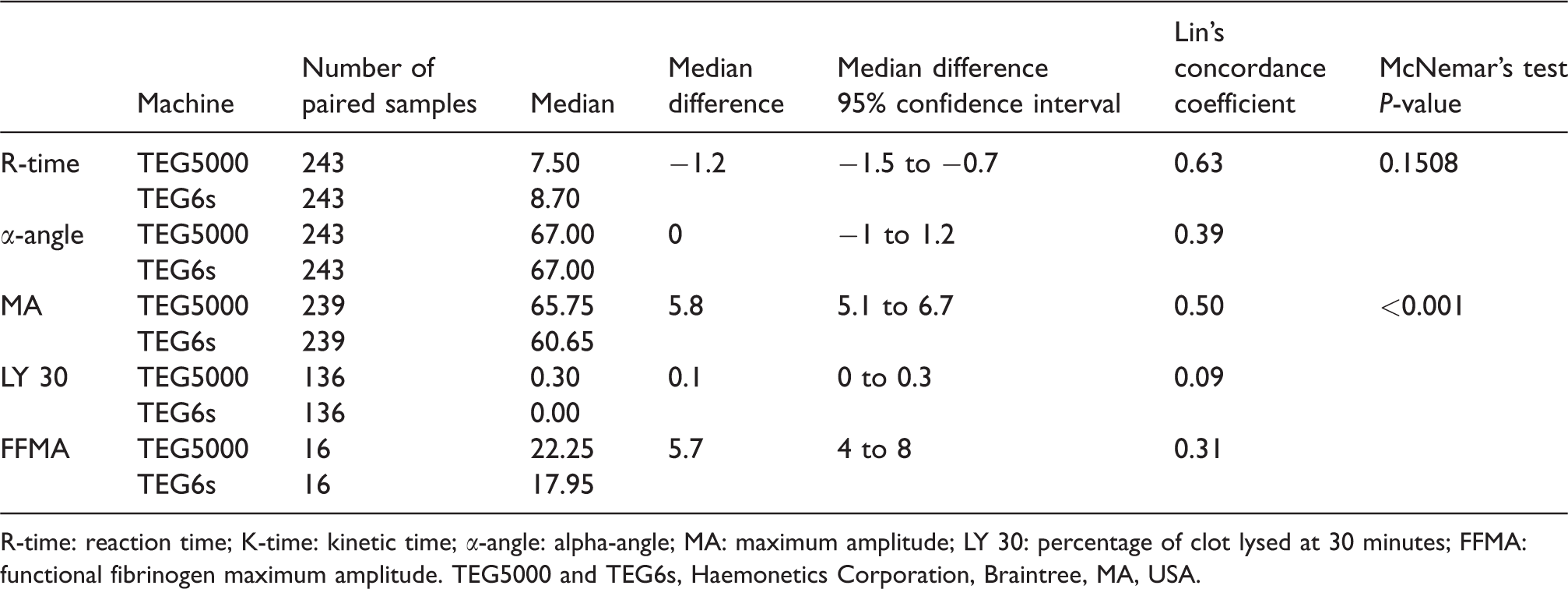
R-time: reaction time; K-time: kinetic time; α-angle: alpha-angle; MA: maximum amplitude; LY 30: percentage of clot lysed at 30 minutes; FFMA: functional fibrinogen maximum amplitude. TEG5000 and TEG6s, Haemonetics Corporation, Braintree, MA, USA.
Bias was suggested for R-time, MA and FFMA. The magnitude of this bias was greatest for MA and FFMA with median differences of 5.8 mm and 5.7 mm respectively.
Lin’s concordance coefficient values were all <0.8 so the TEG machines did not have excellent agreement for any parameter. LY 30 gave a value of 0.09, suggesting poor concordance. All other values were in the intermediate range, with lowest to highest concordance being LY 30, FFMA, α-angle, MA and R-time.
McNemar’s test did not show a significant difference in treatment recommendations for R-time but did show a significant difference in treatment recommendations for MA when the TEG6s values were applied directly to the current TMACS. Using the TEG6s MA values would recommended treating 28 patients (11.5%) with platelets, which the TEG5000 would not have recommended. There were insufficient non-zero data points to perform a McNemar’s test for LY 30 and too few data points to calculate one for FFMA. The α-angle does not form part of our current management algorithm so McNemar’s P statistic could not be calculated.
Discussion
The primary outcome of the study was to determine whether using TEG6s and a global cartridge gave results similar enough to a non-citrated TEG5000 to allow clinical interchangeability of the two devices using the current TEG management algorithm for cardiac surgery at our institution. The study examined three major parameters to answer this question: R-time, MA and LY 30. We found that R-time had the greatest agreement between the two machines with a Lin’s concordance coefficient of 0.63 (intermediate concordance). There was no significant difference in terms of a treatment recommendation if R-time values from the TEG6s were used in our current TMACS. This was not the case for the MA parameter. Lin’s concordance coefficient for MA was 0.5, representing an intermediate level of concordance. However, there appeared to be bias between the two machines for MA. The TEG5000 returned significantly higher values than the TEG6s (median difference 5.8 mm, 95% confidence interval (CI) 5.1–6.7 mm). This bias also affected the treatment recommendations as indicated by the significant P statistic for McNemar’s test (P<0.0005) where 11.5% of patients would have received platelets based on the TEG6s but not with the TEG5000. TEG6s results applied to the current TMACS would have resulted in significantly more patients receiving platelets. LY 30 showed poor concordance with a Lin’s concordance coefficient of 0.09. The Bland–Altman plot demonstrates an almost linear relationship between the two TEG devices for this variable with TEG6s often returning 0% lysis at 30 minutes despite increasing values of LY 30 on the TEG5000. The large number of LY 30 results with 0% lysis in our samples from both machines made it impossible to calculate a McNemar’s P statistic for this variable. This may be due to the routine administration of tranexamic acid in this study population. Despite the lack of this statistic, it appears that using TEG6s values in our current TMACS would potentially undertreat lysis in some patients in our study population.
Secondary outcomes were to investigate agreement between other parameters produced on the TEG6s global cartridge and those available on TEG5000 in a cardiac surgical population. In addition to R-time, MA and LY 30 discussed above, this study looked at α-angle and FFMA. Lin’s concordance coefficient for the α-angle was 0.39. There was no apparent bias between the two machines for this parameter with identical median values and a CI of –1.2 to 1 degree. However, at lower levels of α-angle (<65 degrees), agreement becomes very poor with the suggestion that TEG6s returns higher values of α-angle than the TEG5000 with α-angle <48 mm. Unfortunately, there were very few data points in this range to draw any conclusion. FFMA had only 16 paired samples for comparison. However, even with this limited number of data points there appeared to be a significant degree of bias with TEG6s returning lower values than the TEG5000 (median difference 5.7 mm, 95% CI 4–8 mm). The limited number of data points precluded calculation of a McNemar’s P statistic but if this bias is real, one would expect that TEG6s results applied to our current TMACS would result in more patients receiving cryoprecipitate.
Taken as a group, it appears that values returned by the TEG6s using a global cartridge are different from those returned from a non-citrated TEG5000.
The results of our study support the findings from a smaller recent TEG5000/6s comparison study in liver transplant patients. 6 This study, which involved samples from ten patients, demonstrated increasingly poor agreement with abnormal TEG values with overall only moderate Lin’s concordance coefficient values for R-time, K-time, α-angle and MA. LY 30 was not reported. Another recent study of 25 intensive care patients, very few of whom had abnormal TEG values, found ‘near perfect’ agreement between TEG5000 and TEG6s 8 with Lin’s concordance coefficient values for R-time, K-time, α-angle and MA all being greater than 0.78. LY 30 reported in this group was the outlier with a Lin’s concordance coefficient of 0.34. One potential reason for the difference in findings between these two earlier studies is a difference in the number of abnormal TEG values in these studies.
Our study, which had a significant proportion of samples with abnormal coagulation, shows that agreement between TEG5000 and TEG6s deteriorates with increasing coagulopathy. Once these values are included in any calculation of Lin’s concordance coefficient, the level of concordance deteriorates rapidly. This is an important finding because clinical decisions regarding whether to treat are based on TEG results in and near the abnormal range. It is, for example, an abnormal TEG value that will guide how much fresh frozen plasma to administer, not a normal value, and so the degree of abnormality becomes important in dose calculations.
A previous TEG5000/6s comparison study also found a bias in MA values (mean difference 5.2 mm), with TEG5000 returning a higher MA than the TEG6s for the same sample. 8 Our study supported this finding, with a median difference in MA values of 5.8 mm. It is interesting because it runs counter to what would be expected from the reported reference ranges for MA on the two devices. The reference range for MA for TEG5000 is 50–70 mm whereas that for TEG6s is 63–78 mm. 5 This finding warrants further investigation.
Our study suggests TEG5000 and TEG6s do not return the same results for a given blood sample, particularly when coagulopathy exists. This has important implications for the application of TEG6s to any pre-existing TEG5000 algorithms as well as the development of future algorithms based on TEG6s. At present there is no gold standard so it is unknown whether TEG5000 or TEG6s results better represent the actual state of a patient’s blood. However, TEG5000 has been used for many years and is well validated in a number of studies in terms of its ability to reduce blood product usage by guiding appropriate product and volume selection. Previous work at our institution also supported this. 7 TEG6s has, to our knowledge, not yet been similarly validated in any large-scale study. It is likely that TEG6s will detect significant coagulopathy but patient management will require TEG6s specific treatment algorithms to guide therapy rather than simple adoption of those developed for the TEG5000 and validation will be needed.
Our study attempted to quantify the clinical implications of using TEG6s results with our current TEG5000 TMACS. We found a significant difference in treatment recommendation with regard to platelet transfusion (11.5% increase in recommendation for platelet transfusion) when TEG6s results were used with the TEG5000 TMACS for MA, and a non-significant difference for R-time. Other parameters could not be calculated. This difference for MA can be seen graphically in Figure 8. This figure is a Bland–Altman plot with the addition of treatment thresholds lines. These treatment threshold lines centre on the treatment value in question, for example an MA of 50 mm. A difference in result between the two tests at this point will result in a different treatment decision. As we move further away from this decision point, for example an average MA of 52 mm, a difference of greater than 2 mm between TEG5000 and TEG6s is required to cause a different treatment decision. Addition of the treatment threshold lines divides the Bland–Altman plot into four quadrants, which encompass the four possible outcomes: both machines would recommend treatment, both machines would not recommend treatment, one machine would recommend treatment whereas the other would not and vice versa.
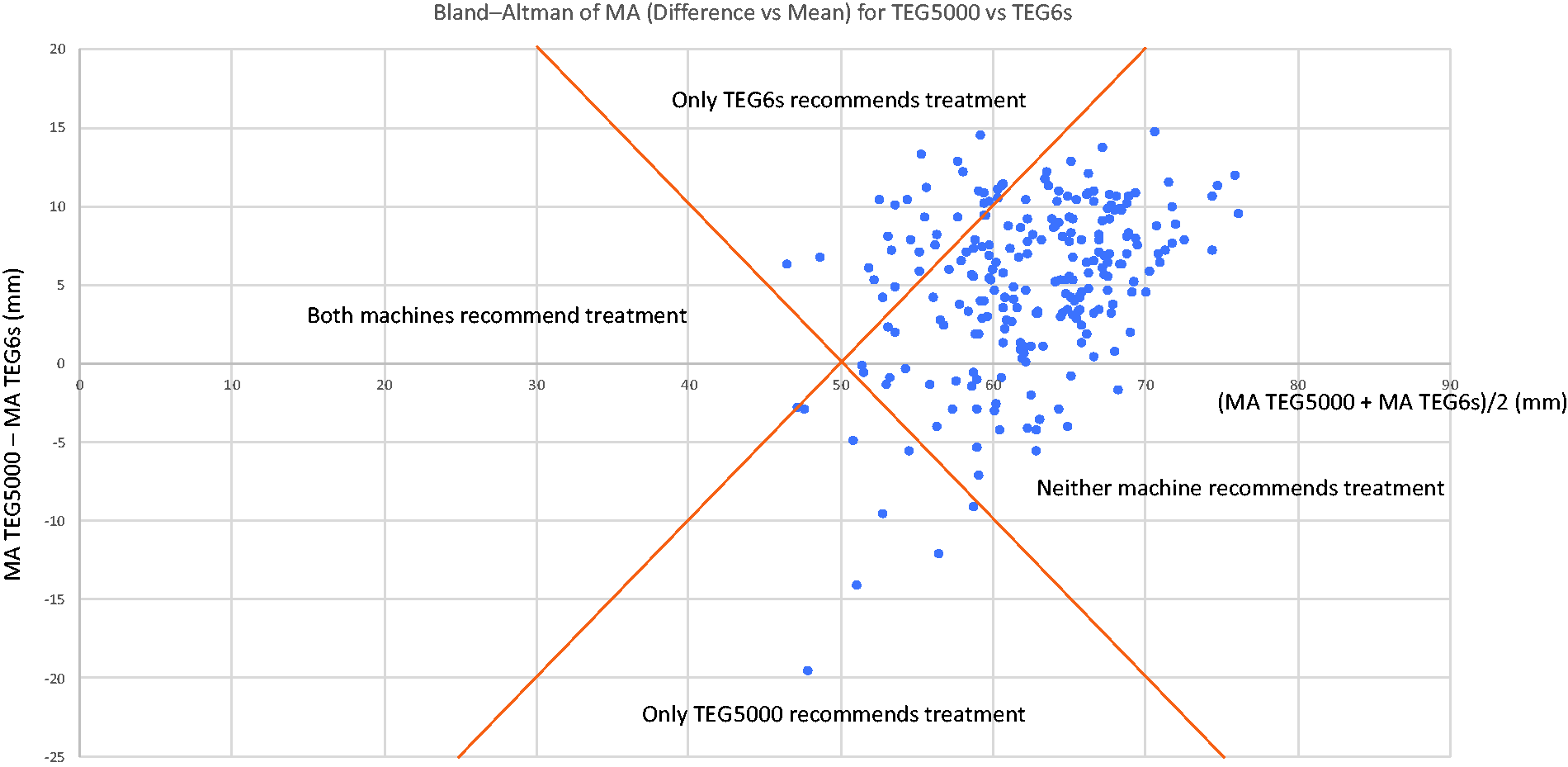
Bland–Altman plot of maximum amplitude (MA) with treatment threshold lines shown. Differences in treatment recommendation between the TEG5000 and the TEG6s (Haemonetics Corporation, Braintree, MA, USA) when using the same algorithm can be visualised.
The strengths of this study are that it contains the largest patient sample to date using cardiac surgical patients, that data were prospectively collected, treating clinicians were blinded to TEG6s results and had no influence over TEG6s testing, and that comparison is made between non-citrated TEG5000 and citrated TEG6s. Non-citrated TEG5000 is used by many operating theatres and cardiac units where testing is performed at (or very close) to point of care so this study uses a real-world situation when comparing the two TEG machines. Finally, the large number of coagulopathic results was also a strength because it allowed comparison of the TEG machines in this context.
Limitations of the study are the large number of patients who had more than two samples taken, that a large number of tests on the TEG6s were interrupted prior to completion to allow for further testing and the very limited number of FF tests performed on TEG5000. This was because non-heparinase FF is not part of our current TMACS but these data would have been very useful given the growing interest in fibrinogen replacement in coagulopathy.
In conclusion, TEG6s results have poor concordance with TEG5000 results, particularly when a coagulopathy exists. For MA and possibly FFMA there appears to be a fixed bias between the two machines with TEG6s returning lower values than TEG5000. This is in contrast to the reported normal ranges and warrants further investigation. The TEG6s cannot be used interchangeably with the TEG5000 using our current TEG5000 TMACS. Although it is entirely feasible that TEG6s could act as a better diagnostic tool, especially given the ease of operation, its utility still requires independent prospective validation.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
