Abstract
Our aim was to determine the frequency and characteristics of post-surgery prescription of opioid medication and to describe patients’ handling of discharge opioid medications. We performed a multicentre prospective cohort study of adult patients undergoing elective or emergency surgery with a postoperative stay of one or more nights, with phone follow-up at two weeks after hospital discharge. The main outcome measures included the proportion of patients prescribed discharge opioid medications, post-discharge opioid use, opioid storage and disposal. Of the 1450 eligible surgical patients, opioids were dispensed on discharge to 858 (59%, 95% confidence interval (CI) (57%–62%)), with immediate-release oxycodone the most common medication. Of the 581 patients who were discharged with opioid medication and completed follow-up, 27% were still requiring opioids two weeks after discharge. Post-discharge opioid consumption was highly variable in the study cohort. The majority (70%) of patients had leftover opioids and only a small proportion (5%) reported disposal of the surplus. In a multivariate model, patients with characteristics of age 45 years or less (odds ratio, OR = 1.78, 95% CI (1.36–2.33) versus older than 45 years), American Society of Anesthesiologists’ physical status (ASA) scores of 1 or 2 (OR = 1.96, 95% CI (1.52–2.53) versus ASA score 3 to 5), higher anticipated surgical pain (OR = 1.45, 95% CI (1.08–1.94) severe versus moderate, OR = 17.48, 95% CI (5.79–52.69) severe versus nil/mild) and public funding status (OR = 1.89, 95% CI (1.36–2.64) versus other) were more likely (P < 0.001) to receive discharge opioids. Post-surgery prescription of opioids is common and supply is often excessive. Post-discharge opioid handling included suboptimal storage and disposal.
Introduction
Opioids are frequently prescribed to manage acute pain following surgery, with oxycodone the most commonly prescribed agent in Australia. 1 Oxycodone prescription has increased significantly in the last decade leading to concerns of overdose, dependence, diversion and unintentional poisoning.1–3 Concurrently there has been a sharp upward trend of oxycodone-related deaths in Australia, adding credence to these concerns. 1 , 4 Control of oxycodone supply in the community therefore may be an important intervention to reduce harm. 4
Discharge prescriptions from acute hospitals may be a significant contributor to supply. 5 Commencement of opioid therapy for acute pain is a gateway to long-term therapy, and ultimately dependence, in some cases. 5 , 6 Furthermore, poorly targeted and excessive prescription after acute hospital care may result in a pool of oxycodone for uncontrolled use by the patient or other people. 5 Studies in the United States (US) have reported low consumption of prescribed discharge opioid medication, with patients storing opioids in unsecure locations for future unprescribed use.7–9 Strategies to optimise opioid prescribing following acute hospital discharge are therefore urgently required and of interest to hospital and community health professionals.
In this study we aimed to determine how commonly patients were being discharged following surgery with new opioid medication or a change in their usual opioid prescription. Secondary aims were to describe the duration and amount of prescription-opioid use after discharge following surgery; to determine if there is a community reservoir of opioids following hospital discharge after surgery, and to describe the handling of excess opioids by patients in this context. Also, we aimed to explore the association between patient characteristics and receiving opioids at discharge, pain since discharge and opioid use.
Materials and methods
This was a prospective cohort study investigating current practice at four hospitals in Melbourne, Victoria (The Royal Melbourne Hospital (an adult tertiary referral trauma centre); The Western Hospital, Footscray campus (an adult general hospital); The Royal Women’s Hospital (a tertiary referral obstetric and gynaecological surgery hospital) and the Peter MacCallum Cancer Centre (a specialist referral cancer centre)). Ethics and governance approvals were obtained from the Melbourne Health Human Research Ethics Committee (LNR/15/MH/252) with reciprocal approval and governance review at each of the other sites.
Study conduct
The audit period was for eight consecutive weeks of normal clinical activity in each centre. Patients were eligible for the study if they were aged 18 years or over, were undergoing elective or emergency surgery with a postoperative stay of one or more nights, had capacity to consent to participation and were available for phone follow-up two weeks post discharge. Patient consent was waived for collection of demographic data (age, sex, weight), surgical details (admitting unit, operation type, anticipated surgical pain (0 = nil/mild, 1 = moderate, 2 = severe), elective or emergency), pain history (chronic pain, opioid medication prior to admission including dose and type) and hospital pharmacy dispensing records for all eligible patients. Written informed patient consent was obtained for phone follow-up. Only patients who consented to participation and were dispensed opioids at the time of discharge were followed up by phone.
For each hospital the proportion of patients receiving discharge opioid medications was determined by comparison of the pharmacy records for patients who were dispensed opioids on discharge and the operating theatre lists for total admitted surgical cases during the study period.
At phone interview, participants’ current pain scores were recorded (11-point verbal numerical rating scale), along with whether they took any opioids after hospital discharge (none/less than five tablets/about half of all tablets/less than five tablets left over/all tablets taken). 7 Further questions explored whether patients were still taking opioid medication, if further supply was obtained since discharge and if they had any leftover opioids. Those with leftover opioids were questioned about storage, and if disposal of opioids had occurred, the method used. Contact after discharge was attempted on at least three occasions before determining that the patient was lost to follow-up.
Statistical analysis
Continuous variables are presented as mean and standard deviation, or median and 25th–75th percentile if data were skewed. Categorical data are presented as counts and percentages. Univariate analyses were undertaken to examine the differences regarding pre-surgery and post-surgery patient characteristics between eligible patients consenting or declining follow-up and between eligible consented patients actually followed up and not followed up. For binary and categorical variables the Chi-squared test or Fisher’s exact test (if expected count less than 5 for a cell) was used. For continuous variables, the two-sample t-test or the Wilcoxon rank-sum test (if data were considered skewed) was used.
Logistic regression was used to explore univariate associations between pre-surgery characteristics (hospital site, age (≤45 years, >45 years), weight (kg), sex, pain at hospital admission, opioid use at hospital admission, benzodiazepine use at hospital admission, American Society of Anesthesiologists (ASA) physical status score (1 or 2 versus 3 to 5), anticipated surgical pain (0, 1, 2), funding body (public, other)) and opioid dispensed at discharge (for eligible patients), severe pain in the two weeks post-discharge and opioid pain medication use at the two-week follow-up interview (for eligible consented patients who were discharged with opioid medication to the community and completed the phone interview two weeks following discharge). Variables associated (P < 0.10) with the outcome in addition to hospital site and sex were included in a multivariate logistic regression model. The adjusted odds ratio (OR), 95% confidence intervals (CI) and P-value were obtained. Pain and opioid use at hospital admission were highly correlated; therefore, it was decided to include pain in the analysis model for severe pain and opioids in the analysis models for opioid dispensed at discharge and opioid use at the two-week follow-up interview.
Missing data were removed list-wise for the analyses. All P-values are reported two-sided without adjustment for multiple comparisons. Data analysis was conducted in STATA version 12 (Stata Statistical Software Release 12, 2011, StataCorp LLC, College Station, TX, USA) and version 14 (Stata Statistical Software Release 14, 2015, StataCorp LLC, College Station, TX, USA).
Sample size
A time-based convenience sample was captured. No formal sample size calculation was undertaken for this study due to its observational nature. The duration of eight weeks across four hospitals was selected to ensure that a comprehensive snapshot of opioid handling after surgical discharge was obtained.
Results
The study recruitment and follow-up flow chart is presented in Figure 1. Our study cohort included a broad spectrum of adult surgical disciplines: general surgery (including the subdisciplines of colorectal, upper gastrointestinal, hepatobiliary, breast and endocrine, renovascular and trauma), orthopaedics, plastic surgery, neurosurgery, cardiothoracic surgery, vascular surgery, urological surgery, head and neck surgery, and obstetrics and gynaecology.
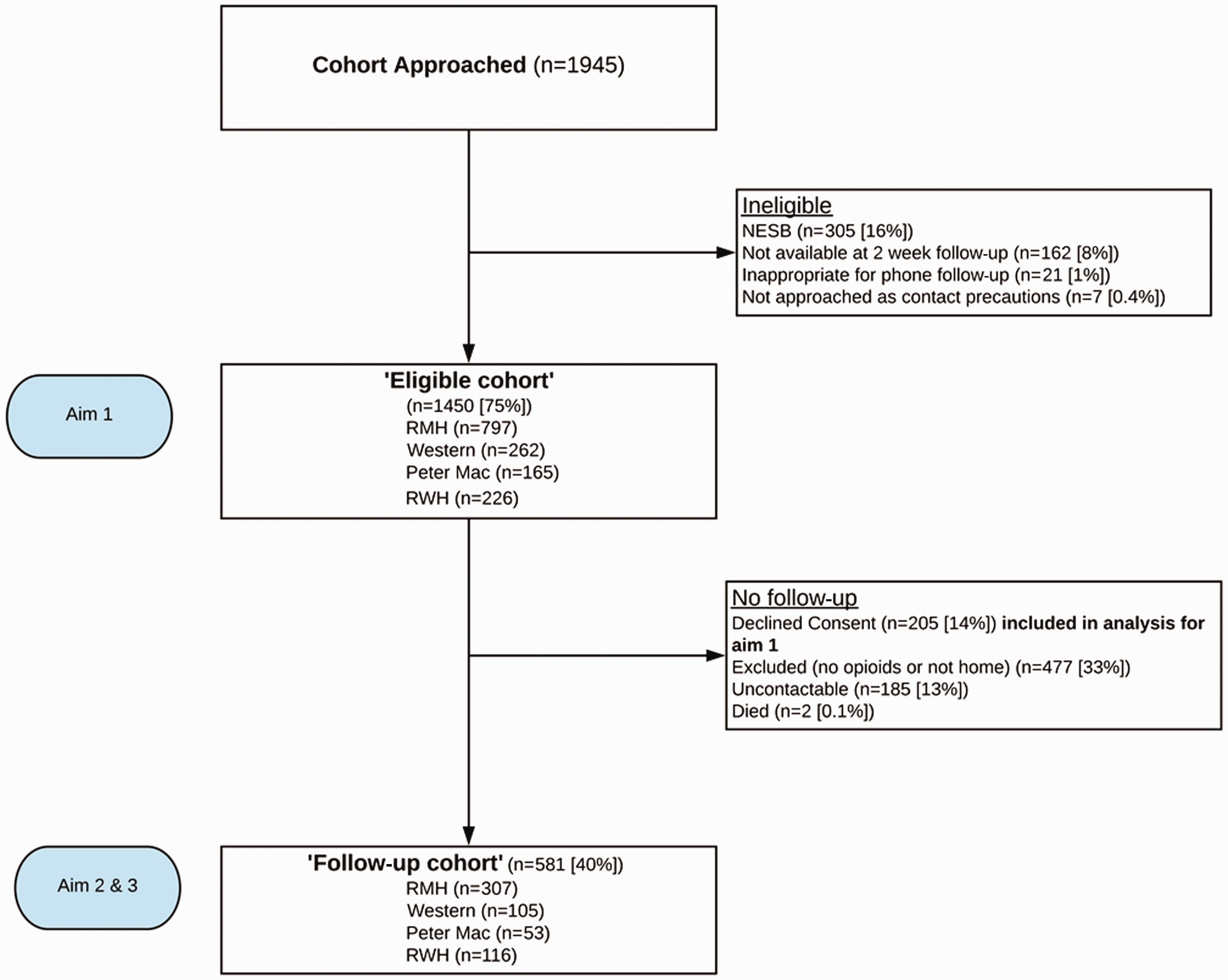
Study flow chart. The data for those declining consent were used for Aim 1 as specifically approved by the Melbourne Health Human Research Ethics Committee. Reasons for exclusion in the non-followed up group was either still in an inpatient location (e.g. rehabilitation facility, nursing home) or no opioids dispensed on hospital discharge. The ‘follow-up cohort’ was composed of those who were dispensed opioids on hospital discharge and consented to and completed follow-up at two weeks post discharge. RMH: Royal Melbourne Hospital; Western: Western General Hospital; Peter Mac: Peter MacCallum Cancer Centre; RWH: The Royal Women’s Hospital; NESB: non-English speaking background.
The characteristics of the 1450 eligible patients, both consenting to follow-up (n = 1245) and declining follow-up (n = 205), are shown in Table 1. Those who declined consent were older, more likely to be male and had higher ASA scores than those who consented. Also, patients who declined consent were more likely to have pain at hospital admission, underwent surgery with less anticipated surgical pain and were less likely to have opioids dispensed on hospital discharge than consenting patients.
Pre-surgery and post-surgery characteristics of eligible patients (n = 1450).
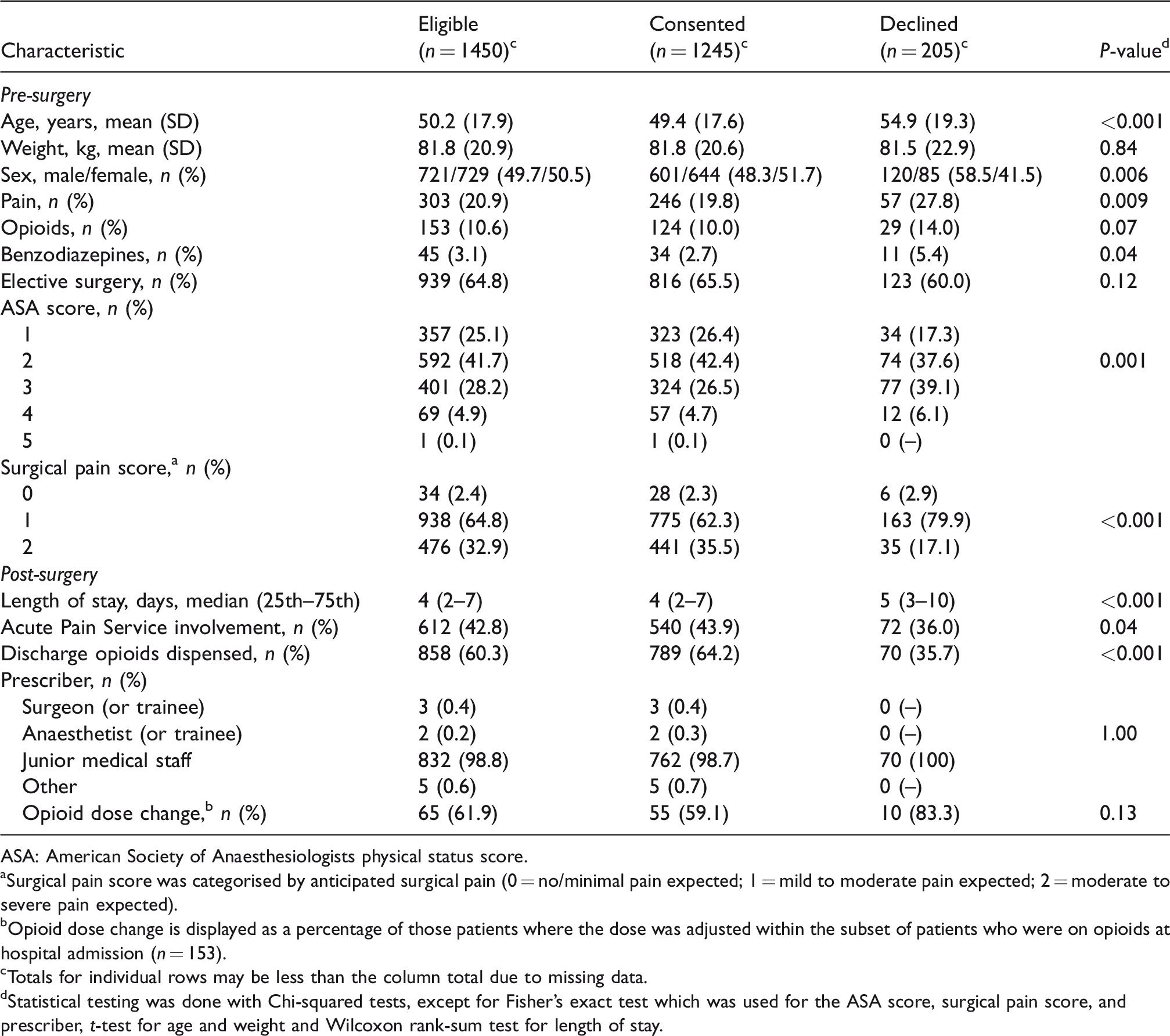
ASA: American Society of Anaesthesiologists physical status score.
Surgical pain score was categorised by anticipated surgical pain (0 = no/minimal pain expected; 1 = mild to moderate pain expected; 2 = moderate to severe pain expected).
Opioid dose change is displayed as a percentage of those patients where the dose was adjusted within the subset of patients who were on opioids at hospital admission (n = 153).
Totals for individual rows may be less than the column total due to missing data.
Statistical testing was done with Chi-squared tests, except for Fisher’s exact test which was used for the ASA score, surgical pain score, and prescriber, t-test for age and weight and Wilcoxon rank-sum test for length of stay.
Opioids were dispensed on discharge following surgery to 858 eligible patients (59%, 95% CI (57%–62%)). For those dispensed opioids on hospital discharge, the median oral morphine equivalent provided was 150 mg (25th–75th percentile 75–225 mg, range 37.5–1760 mg). Immediate-release oxycodone was the most commonly dispensed agent (88% of patients who were dispensed opioids) with a median strength of 5 mg (range 5 to 10 mg) and median quantity of 20 tablets (range 5 to 20 tablets). Of the patients who were on opioid medications at the time of hospital admission (n = 105, 7%) an opioid dose change occurred in 65 (62%, 95% CI (52%–71%)) of these patients. In a multivariate model including hospital site, age, sex, opioid use at hospital admission, ASA score, anticipated surgical pain, and funding body, patients with characteristics of age 45 years or less (OR = 1.78, 95% CI (1.36–2.33) versus more than 45 years), ASA scores 1 or 2 (OR = 1.96, 95% CI (1.52–2.53) versus ASA score 3 to 5), higher anticipated surgical pain (OR = 1.45, 95% CI (1.08–1.94) severe versus moderate, OR = 17.48, 95% CI (5.79–52.69) severe versus nil/mild) and public funding status (OR = 1.89, 95% CI (1.36–2.64) versus other) were more likely (P < 0.001) to receive discharge opioids.
Those eligible and consenting to follow-up (n = 581) were compared to those without follow-up (n = 664) (Table 2). Those without follow-up were older, had higher ASA scores, were more likely to have pain at hospital admission, had longer hospital length of stay and were less likely to have a discharge opioid dose change if they were taking prescribed opioids at hospital admission.
Pre-surgery and post-surgery characteristics of consented patients (n = 1245).
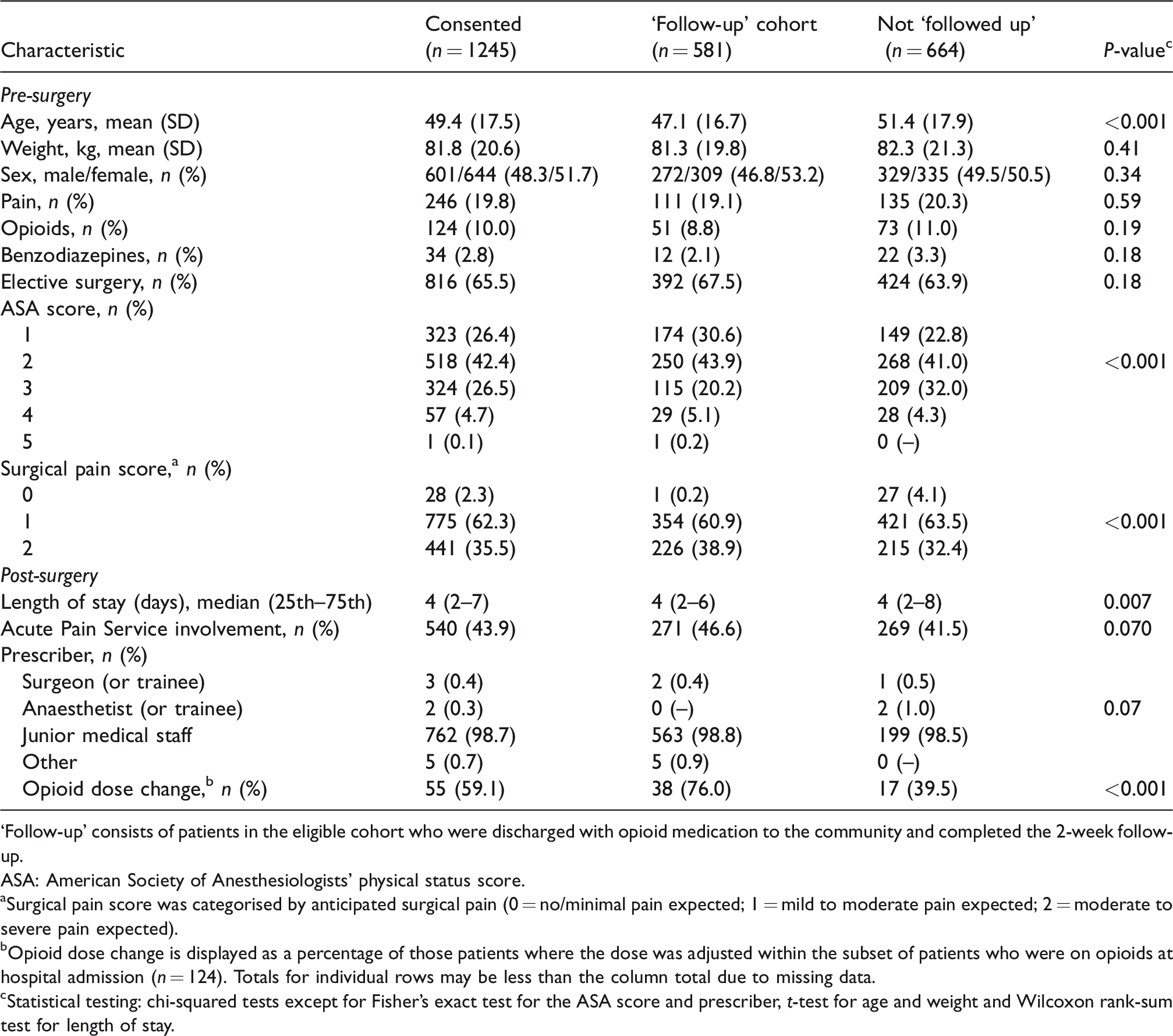
‘Follow-up’ consists of patients in the eligible cohort who were discharged with opioid medication to the community and completed the 2-week follow-up.
ASA: American Society of Anesthesiologists' physical status score.
Surgical pain score was categorised by anticipated surgical pain (0 = no/minimal pain expected; 1 = mild to moderate pain expected; 2 = moderate to severe pain expected).
Opioid dose change is displayed as a percentage of those patients where the dose was adjusted within the subset of patients who were on opioids at hospital admission (n = 124). Totals for individual rows may be less than the column total due to missing data.
Statistical testing: chi-squared tests except for Fisher’s exact test for the ASA score and prescriber, t-test for age and weight and Wilcoxon rank-sum test for length of stay.
A total of 581 (40%) eligible patients were discharged with opioid medication to the community and completed follow-up at two weeks (Table 2). Two hundred and seventy-two (47%) of these patients reported severe post-surgery pain at some stage since hospital discharge but the median pain score at the time of interview was 1 (25th–75th percentile: 0–3). In a multivariate model including hospital site, age, sex, pain at hospital admission, ASA score and expected surgical pain score, patients with baseline pain (OR = 2.30, 95% CI (1.43–3.70) versus none) and ASA scores 1 or 2 (OR = 2.98, 95% CI (1.89–4.69) versus ASA score 3 to 5) were more likely (P < 0.001) to report having experienced severe pain since discharge.
At two weeks after hospital discharge 155 (27%) of patients contacted were still consuming opioid medication. In a multivariate model including hospital site, age, sex, weight, opioid therapy at hospital admission, benzodiazepine therapy at hospital admission, ASA score and funding body, patients who were taking prescribed opioids at baseline were more likely to still be requiring opioids at follow-up (OR = 3.66, 95% CI (1.54–8.70), P = 0.003 versus no), as well as patients >45 years (OR = 1.77, 95% CI (1.08–2.91), P = 0.024 versus ≤45 years) and having higher weight (OR = 1.01, 95% CI (1.00–1.02), P = 0.035 for each 1 kg increase).
Wide variability in post-discharge prescription opioid use was reported at follow-up (Table 3). At follow-up, 142 (24%) patients reported requiring an additional prescription of opioids after hospital discharge, but 28% (n = 40) of these had subsequently ceased opioid therapy by the two-week follow-up phone interview. The majority (n = 399, 70%) of patients in our cohort had leftover opioid medications at follow-up. Of those who reported storing opioid medications at the time of follow-up (n = 403), 88% reported that this was in an unlocked location. Only a small proportion who had ceased opioid medication therapy reported disposing of the medications (n = 19, 5%). The methods of disposal were variable (household garbage n = 12, sink or toilet n = 4 and returned to pharmacy n = 1). This is consistent with only 49 (9%) patients who were followed up after hospital discharge reporting that they received instructions for how to dispose of their opioid medications once their use was complete. Three patients (0.5%) admitted to giving their prescription opioid medications to someone else.
Post-discharge opioid use in the cohort of patients followed up.
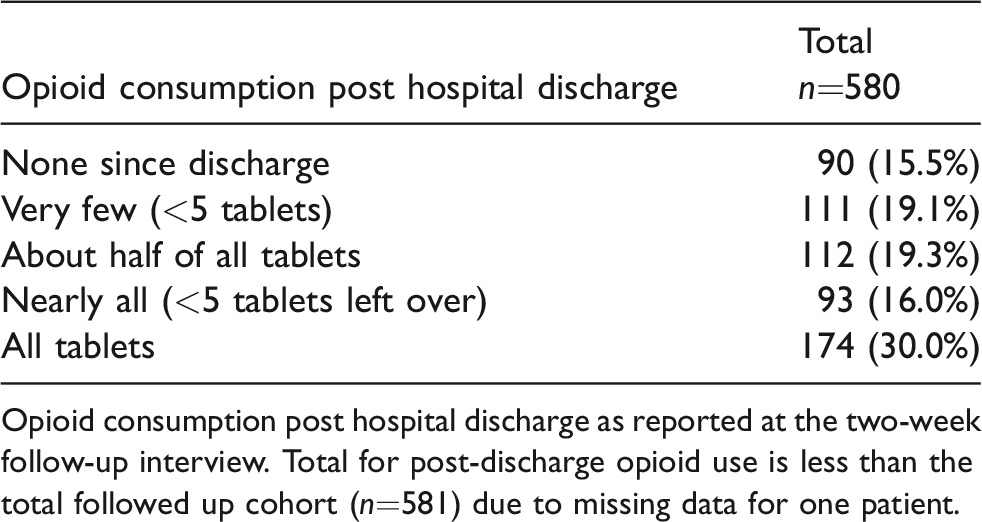
Opioid consumption post hospital discharge as reported at the two-week follow-up interview. Total for post-discharge opioid use is less than the total followed up cohort (n=581) due to missing data for one patient.
Discussion
Our study shows that opioids are commonly dispensed following acute surgical care and that the quantities dispensed are excess to need in most cases. Furthermore, our study confirms that there is a community reservoir of opioids originating from acute hospital care. Whilst only a quarter of patients were still taking opioid medication two weeks following hospital discharge, over two-thirds reported having leftover opioid medication at the time of follow-up.
Post-discharge opioid use was highly variable, with a quarter of patients requiring further opioid dispensing after hospital discharge and a third of patients consuming no or less than five tablets once they left hospital. This suggests that both under- and over-prescribing are occurring on hospital discharge after surgical care. The vast majority of post-surgery prescribing was for immediate-release oxycodone in a quantity corresponding to the standard pack size in Australia (20 tablets). This suggests that protocolised rather than individualised prescribing is occurring.
Uniform postoperative opioid prescribing has also been shown to be a problem in the USA. 10 When opioid dispensing is matched to a standard pack size or with the intention that no re-supply will be needed even for patients with high use, it is likely that most patients will receive an excess of opioid medication.7–9 This may have consequences as it has been demonstrated that the larger the quantity of opioids supplied on initial prescription, the greater the probability of continued opioid use. For opioid-naïve patients, the rate of ongoing opioid use at one year was 6% for those who received a 1- to 7-day opioid supply, increasing to 13.5% for an 8- to 30-day supply and 29.9% for a more than 30-day supply. 11
Prescribers have to balance optimal management of acute post-surgery pain with avoidance of excessive supply of opioid analgesia. One study sought to reduce variability of opioid prescription for the same operations by dosing based on meeting the opioid needs of 80% of patients with the first opioid prescription, therefore reducing the amount of opioids actually dispensed by over 40%. 8 Other studies report development of prescription guidelines that resulted in decreased opioid supply (proportion of patients and quantity) and increased simple analgesia adjunct use with no increase in pain scores or repeat opioid prescription requirements. 12 , 13 These approaches are encouraging at the single institution level, but much larger-scale and Australia-specific studies are required.
The majority of patients in our study were storing their opioid medication in an unlocked location. This is consistent with work from the USA which has shown that opioid storage is generally suboptimal. 7 , 14 Furthermore, only 9% of patients in our study recalled receiving instructions for opioid medication disposal, with even fewer patients (5%) actually disposing of opioids by the time of follow-up. Appropriate education, in the form of a brochure about the importance of proper opioid disposal, has been demonstrated to be effective at increasing compliance with disposal post discharge. 15 The reported diversion rate in our study was very low at 0.5%, but it is likely that this was underreported.
Limitations
Practice was captured during a short time period at a limited number of health services. We did not collect data on individual inpatient opioid consumption and so we cannot make any inferences about inpatient consumption and after-discharge use. Our study involved a single timepoint of phone follow-up at two weeks after hospital discharge which may have been insufficient to truly reflect how patients intended to handle their opioid medications. As being at home in the community was a requirement for follow-up, trauma patients may be under-represented in our study, as inpatient rehabilitation following acute care is common. As use, storage, disposal and diversion was necessarily self-reported, accuracy cannot be guaranteed.
Future directions
Our findings suggest that an intervention to better target opioid prescription following a surgical admission is needed. Such an intervention would require development of an opioid use prediction model and an evidence-based surgery-specific guideline, and engagement with hospital and community health professionals, patients and their families. Robust assessment of the introduction and impact of this opioid stewardship programme would be vital. We plan to develop such a programme for use in Australia.
Conclusion
Current opioid prescription, storage and disposal following acute surgical care was suboptimal when measured across four different practice contexts. This snapshot is alarming for the situation in Australia given the current global concern about opioid-related harm. This presents an opportunity for an intervention, with the intention of improving the care of our patients following surgery and safety for our communities.
Footnotes
Acknowledgements
This project involved medical students from The University of Melbourne and their contribution to data collection was substantial (Kelsey Turner, Samuel Thorburn, David Heelan, Justin Er Wenn Lim and Malcolm Moss).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by a project grant from the Australian and New Zealand College of Anaesthetists.
