Abstract
Dose titration with immediate-release opioids is currently recommended for acute pain. The Australian and New Zealand College of Anaesthetists and the Faculty of Pain Medicine released a statement in March 2018 supporting their use in the treatment of opioid-naïve patients; however, the impact of this statement on clinical practice is currently unknown. This retrospective cohort study was conducted to compare opioid prescribing patterns before and after the release of the recommendations. Data were collected on 184 patients (2017, n = 78; 2018, n = 106) admitted to the Prince of Wales Hospital in November 2017 and 2018, which consisted of demographic data, opioid prescriptions and discharge opioid information. The main outcome is the number of prescriptions of slow-release opioids in 2017 versus 2018 after the recommendations were published. Confounding factors were accounted for using logistic and multiple regression as appropriate. There was a 29% decrease in slow-release opioid prescriptions during hospitalisation (n = 31, 40% versus n = 12, 11%; P < 0.001) and 17% decrease at discharge (n = 20, 26% versus n = 9, 9%; P = 0.02) post-publication. After adjusting for confounders, the odds of slow-release opioids being prescribed postoperatively and at discharge reduced by 86% and 88%, respectively (postoperative period: odds ratio 0.14, P < 0.05; discharge: odds ratio 0.12, P < 0.05). In addition, orthopaedic patients were more likely to receive slow-release opioids, consistent with existing literature. As the use of slow-release opioids has been associated with increased harm and protracted opioid use compared to immediate-release opioids, it is hoped that wider dissemination of these recommendations and a change in prescribing practice can be a step towards overcoming the opioid crisis.
Introduction
Opioids are widely used postoperatively to manage acute pain. Currently, it is recommended that when opioids are used for acute pain, dose titration with immediate-release opioids (IROs) is preferred. 1 , 2 In line with this, the Australian and New Zealand College of Anaesthetists (ANZCA) and the Faculty of Pain Medicine (FPM) released a position statement in March 2018 stating that slow-release opioids (SROs) are not recommended for acute pain management due to an association with increased adverse effects and increased conversion to long-term use. 3 Instead, ANZCA and FPM recommend the titration of IROs when necessary for moderate to severe pain in opioid-naïve patients. However, the position statement also acknowledges that SROs may be used in some patients with prior use of SROs or those with prolonged pain states, after careful consideration and on a temporary basis, and that such usage should be communicated to the general practitioner.
SROs have a longer duration of therapeutic action compared to IROs, allowing for convenient dosing intervals and uninterrupted sleep for the patient. 4 However, the evidence supporting the use of SROs in acute pain remains weak, due to a lack of head to head trials against IROs in the postoperative setting. Existing studies have compared SROs to standard therapy, whereby patient-controlled analgesia (PCA) or a different opioid is used.5–7 When slow-release oxycodone was directly compared against immediate-release oxycodone in a randomised controlled trial, the SRO treatment group was found to have a cumulative oxycodone dose four times higher than the IRO group. 8 In another study, immediate-release oxycodone demonstrated significantly better pain relief compared to slow-release oxycodone for the first seven hours post-administration, with a greater proportion of patients experiencing adverse effects in the SRO group. 4 As pain severity changes during the postoperative period, it is more difficult to titrate the minimum effective dose with SROs. 3 Moreover, any adverse effect experienced becomes more challenging to manage due to its longer duration of action. The sustained effect of SROs is also similar to a background opioid infusion, which has not been shown to confer benefit over intravenous PCA. In a study comparing PCA treatment with a concurrent opioid infusion, the infusion treatment group had a higher number of changes in therapy secondary to opioid-related side-effects and a higher total dose of morphine compared to PCA. 9
There have also been concerns of prolonged opioid use after initial prescriptions for acute pain, which may be more common with the initial use of SROs. 10 A previous study conducted on patients with acute non-cancer pain has shown that approximately 6%–14% of opioid-naïve patients continue to take opioids one year after hospitalisation, and the risk is increased to 27% if the initial prescription is a SRO. Opioid-related accidental deaths in Australia have also been on the rise, from 1877 deaths between 2002 and 2006 to 3933 deaths more recently between 2012 and 2016. 11 Mudumbai et al. (2018) 12 reported that postoperative patients given higher dose opioids or SROs at discharge were at higher risk of overdose and opioid delirium. Patients are also at risk of increased costs and healthcare utilisation from receiving SROs postoperatively due to a higher risk of healthcare encounters such as re-admissions. As such, the ANZCA/FPM position statement also encourages physicians to reassess the necessity for opioids at discharge and convey a weaning plan to the patient’s general practitioner in addition to minimising the use of SROs. 3
The release of the position statement was accompanied by an article in the ANZCA/FPM quarterly bulletin, which is distributed to all Fellows and trainees of the college and faculty. Anecdotally, there was some controversy about the content and the way in which the statement was released, and this added to awareness of the statement by Fellows. Individual hospitals varied in their response, with many departments raising it within anaesthetic departmental meetings, grand rounds, or other hospital educational activities. The effect of the release of the statement, if any, on clinical practice is unknown.
The primary outcome of this retrospective study is to compare postoperative prescribing patterns of SROs before and after the release of the position statement. In addition, we aim to identify changes in opioid prescribing at discharge and the provision of opioid cessation plans before and after the release of the position statement. Differences in pain scores will also be recorded as a secondary outcome.
Methods
Study sample and data sources
Ethics approval was obtained from the Human Research Ethics Committee of Prince of Wales Hospital (POWH) (HREC 17/223). All adult patients admitted into POWH surgical wards in November 2017 and November 2018 were identified from the electronic medical records. Patients admitted in 2017 were classified as the pre-intervention group whereas those admitted in 2018 were the post-intervention group. Time periods of six months before and after the release of the ANZCA/FPM statement were chosen to allow time for dissemination of the publication. Sample size was calculated with the G*Power software using an estimated reduction of SRO prescriptions from 40% to 10% post-publication. 13 Based on this assumption, a sample size of 58 patients per study group was required (P = 0.05, 95% power). Patients were included if they were admitted for more than 24 hours, had a surgical procedure during their stay and were prescribed an opioid postoperatively. Exclusion criteria were those with the use of SROs pre-admission, significant missing data, interhospital transfers, unplanned discharge and those who died during their stay. Data on patients meeting the inclusion criteria in the first 12 days of both months were collected to ensure the timely collection of data. Data collection was limited to 14 days of postsurgical stay for each patient. Data from electronic medical records and paper charts (e.g. anaesthetic charts and PCA charts) were captured into research electronic data capture (REDCap) software. 14 , 15 REDCap is a secure, web-based application designed to support data capture for research studies, providing: (a) an intuitive interface for validated data entry; (b) audit trails for tracking data manipulation and export procedures; (c) automated export procedures for seamless data downloads to common statistical packages; and (d) procedures for importing data from external sources.
Data collection and measures of outcome
Demographic data and medical information (age, gender, type of surgery, type of specialty, length of stay, American Society of Anesthesiologists Physical Status (ASA PS) classification 16 and creatinine clearance (using the Cockcroft–Gault equation)) were collected. Medication administration data were recorded including the use of opioids given by any route of administration but did not include preoperative and intraoperative opioid use. Information on the type of opioid used, the doses and number of administrations in a day, adjuvant analgesics used, opioids given at discharge, the number of pills dispensed at discharge and whether an opioid cessation plan was provided was also collected. To assess efficacy, pain intensity scores using the 0–10 verbal numeric rating scale (VNRS) were recorded, with ‘0’ being no pain and ‘10’ being the worst pain imaginable, 17 and then mean scores for each postoperative day for each patient were calculated.
Statistical analysis
The total opioid use was calculated for each postoperative day and at discharge by converting opioid doses into oral morphine equivalent daily doses (OMEDDs). 18 Demographic variables and outcomes of the pre-intervention and post-intervention study groups were compared using chi-square analysis, t-test or the Mann–Whitney test depending on the normality of the data. 19 Similarly, demographic variables were compared between the patient groups receiving SROs with or without IROs (SRO ± IRO) and those receiving only IROs. However, due to prescribers’ perceptions of the relative safety of tapentadol, patients prescribed modified-release or immediate-release tapentadol were analysed separately from the SRO ±IRO and IRO study groups. Means and standard deviations (SDs) were reported for normally distributed data while median and interquartile range (IQR) were preferred for non-parametric data. 19 Univariable and multivariable regression was conducted against the outcomes of interest (SROs prescribed postoperatively and SROs prescribed at discharge) for significance, adjusted for clinically relevant variables (e.g. renal function, age and ASA PS class). Orthopaedic prescribers may be influenced by local protocols for analgesic regimens within this institution; therefore, prescribing practice for orthopaedic admissions may not be a true reflection of the effect of the ANZCA publication. To account for this, the results were adjusted for orthopaedic admissions using multivariate regression. Odds ratios (ORs) were reported for all multivariate logistic regression analyses (for binary outcomes), whereas standardised beta coefficients (B) were reported for all multivariate linear regression analyses (for continuous outcomes). All tests were conducted at the 95% significance level and 95% confidence intervals (CIs) were reported. IBM SPSS statistical software version 24.0 was used for all statistical analyses in this study.
Results
Of 545 patients admitted into surgical wards during the time period studied in 2017 (n = 270) and 2018 (n = 275), 230 patients met the inclusion criteria (2017, n = 103; 2018, n = 127). In total, 184 patients were included in the final analyses while 46 were excluded. Of the patients that were excluded, 18 were due to the use of SROs pre-admission, 18 due to interhospital transfers, five due to a substantial lack of information, three due to death during hospitalisation (unrelated to opioids) and two due to unplanned discharge. Surgeries encompassed in this study in descending order of magnitude were joint replacement surgeries (15%), other orthopaedic surgeries (13%), intrathoracic surgeries (13%), open (11%) and minimally invasive intra-abdominal surgeries (11%) and others. No significant differences were noted between the pre-intervention (2017, n = 78) and post-intervention (2018, n = 106) groups (Table 1). Study groups who were prescribed only IROs (n = 123) during hospitalisation and those who were prescribed SRO ± IRO (n = 43) were also compared (Table 2). Notably, there were more orthopaedic patients in the SRO ± IRO group (n = 26, 60%) compared to the IRO group (n = 18, 15%, P < 0.001). There was also a higher proportion of patients with an ASA PS class of 1 or 2 in the SRO ± IRO group (n = 21 (72%) in the SRO ± IRO group versus n = 39 (46%) in the IRO group), as opposed to the IRO group, which had a higher proportion of patients with a ASA PS class of 3 or 4 (n = 45 (54%) in the IRO group versus eight (28%) in the SRO ± IRO group). These differences were later accounted for in multivariate regression. Although the use of IROs pre-admission and pre-existing chronic pain were also recorded, the low number of results obtained suggests inaccuracies, possibly due to poor record keeping and thus were not presented.
Demographic characteristics of patients before and after publication of ANZCA guidelines.
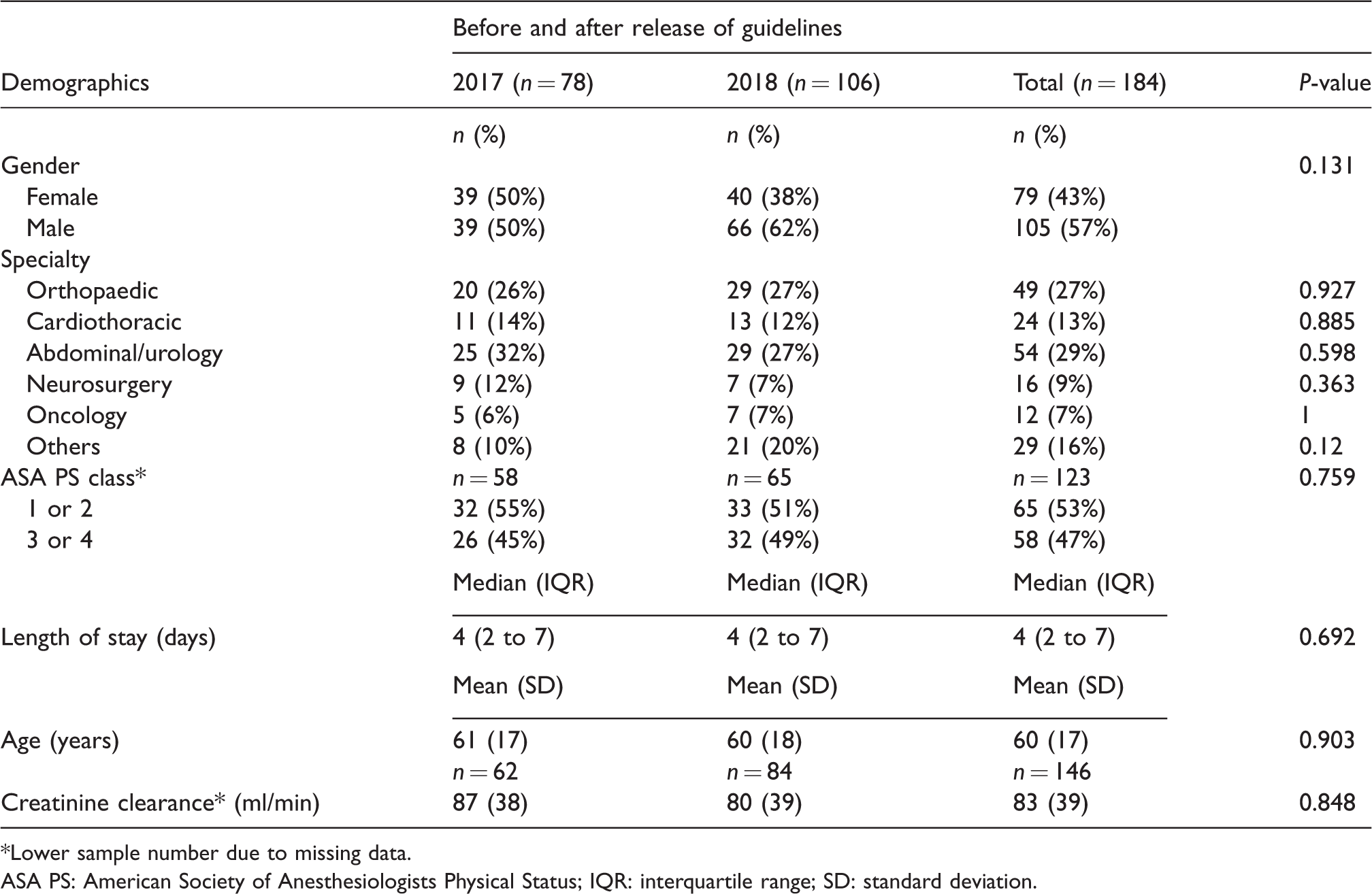
*Lower sample number due to missing data.
ASA PS: American Society of Anesthesiologists Physical Status; IQR: interquartile range; SD: standard deviation.
Demographic characteristic of patients prescribed only immediate-release opioids (IRO) and those prescribed slow-release opioids with or without immediate-release opioids (SRO±IRO).*
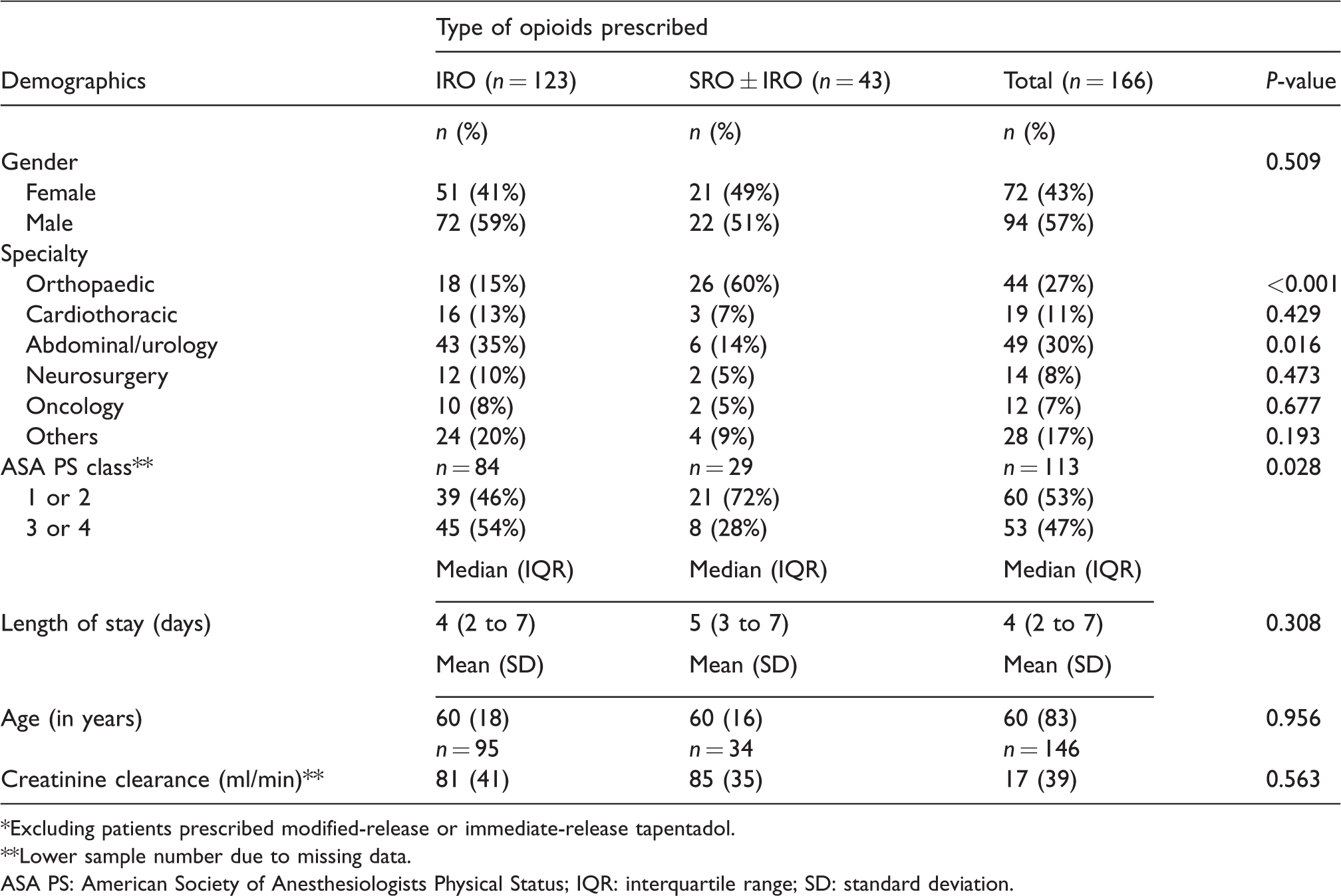
*Excluding patients prescribed modified-release or immediate-release tapentadol.
**Lower sample number due to missing data.
ASA PS: American Society of Anesthesiologists Physical Status; IQR: interquartile range; SD: standard deviation.
Primary outcomes
A significant decrease in the prescribing of SROs was found in the post-intervention group postoperatively (n = 31, 40% versus n = 12, 11%; P < 0.001) (Table 3) and during discharge (n = 20, 26% versus n = 9, 9%; P = 0.02) (Table 4). However, tapentadol prescriptions were analysed separately due to common perceptions among prescribers of a better safety profile compared to other opioids. These results remained significant even after clinical factors such as age, renal function and ASA PS class were accounted for through multivariate regression (postoperative period: P = 0.006, OR 0.14, 95% CI 0.03 to 0.56; during discharge: P = 0.019, OR 0.12, 95% CI 0.02 to 0.71) (Table 5).
Prescribing practices with regard to opioids during the postoperative period before and after publication of recommendations.
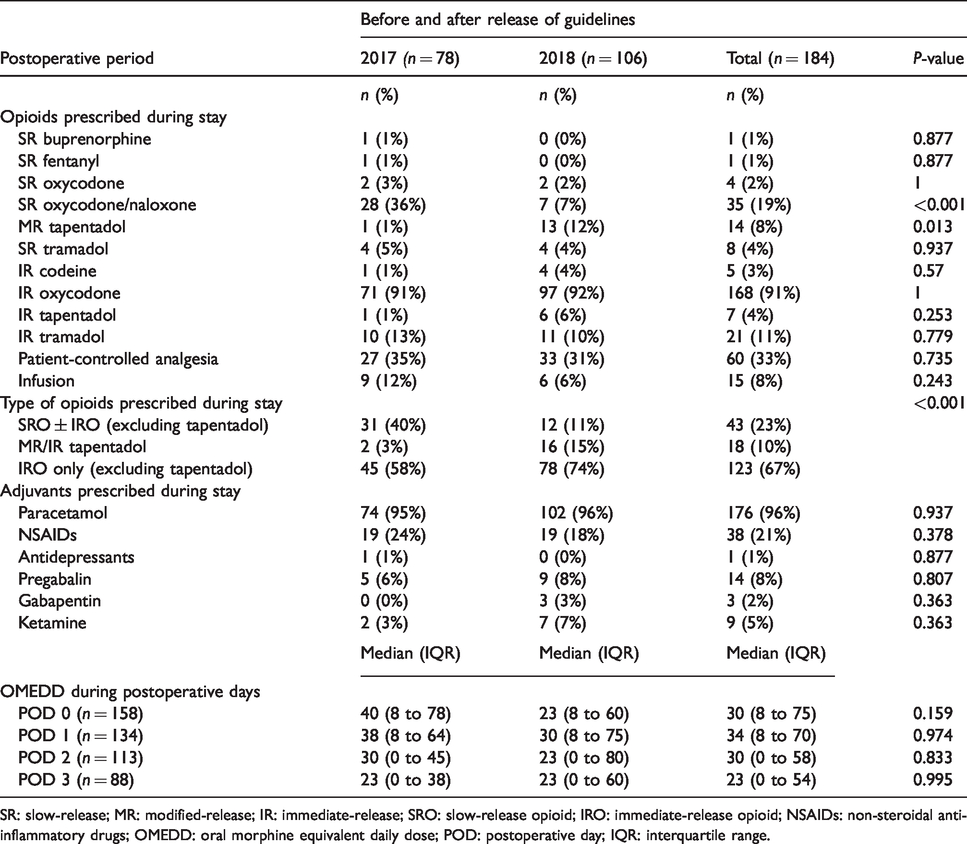
SR: slow-release; MR: modified-release; IR: immediate-release; SRO: slow-release opioid; IRO: immediate-release opioid; NSAIDs: non-steroidal anti-inflammatory drugs; OMEDD: oral morphine equivalent daily dose; POD: postoperative day; IQR: interquartile range.
Prescribing practices and opioid use at discharge.
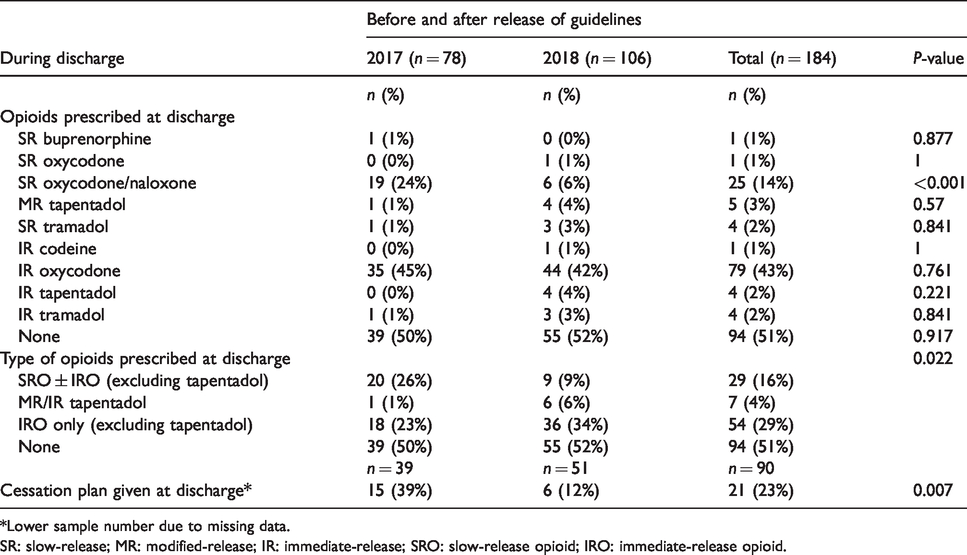
*Lower sample number due to missing data.
SR: slow-release; MR: modified-release; IR: immediate-release; SRO: slow-release opioid; IRO: immediate-release opioid.
Multivariable logistic regressions for the use of slow-release opioids after surgery and at discharge.
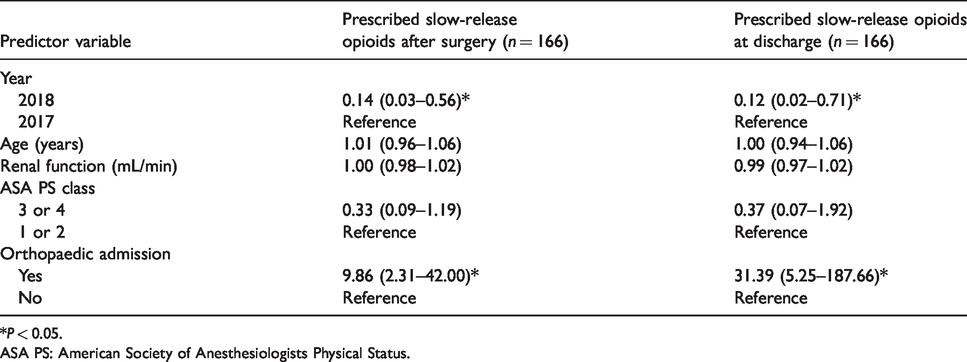
*P < 0.05.
ASA PS: American Society of Anesthesiologists Physical Status.
The overall decrease in the use of SROs appears to correspond to the decrease in slow-release oxycodone/naloxone (n = 28, 36% versus n = 7, 7%; P < 0.001) (Table 3). On the other hand, there was a significant increase in the use of tapentadol modified-release in the 2018 group (n = 1, 1% versus n = 13, 12%; P = 0.013). There were no other significant changes with regard to the prescribing practices for other opioids.
Secondary outcomes
A significantly higher proportion of patients were not given an opioid cessation plan to be communicated to the patient's general practitioner post-intervention (patients given an opioid cessation plan pre-intervention, n = 15, 39%, versus post-intervention, n = 6, 12%; P = 0.007), contrary to the ANZCA/FPM guidelines (Table 4). After accounting for the type of opioid prescribed at discharge, this difference was still found to be significant (P = 0.02, OR 0.24, 95% CI 0.07 to 0.80) (Table 6).
Multivariable logistic regression for opioid cessation plans given at discharge.
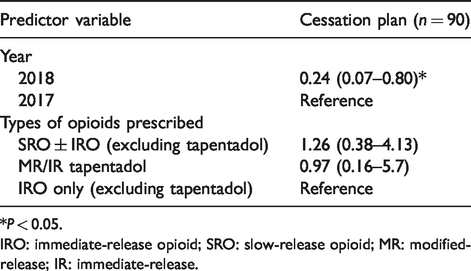
*P < 0.05.
IRO: immediate-release opioid; SRO: slow-release opioid; MR: modified-release; IR: immediate-release.
Those who were using SROs during their stay were also more likely to have a higher median OMEDD throughout postoperative days (POD) 0 to 3 (POD 0: 53 versus 20; POD 1: 48 versus 15; POD 2: 38 versus 8; POD 3: 30 versus 8; P < 0.001) (Table 7). After adjusting for age, renal function and pain scores, only OMEDD for the first postoperative day (POD 0) was significantly higher in the SRO ± IRO group (POD 0, P = 0.046, B = 42.9 ± 21.3) (Table 8).
A comparison of opioid use and pain scores during stay between those prescribed immediate-release opioids and those prescribed slow-release opioids.*
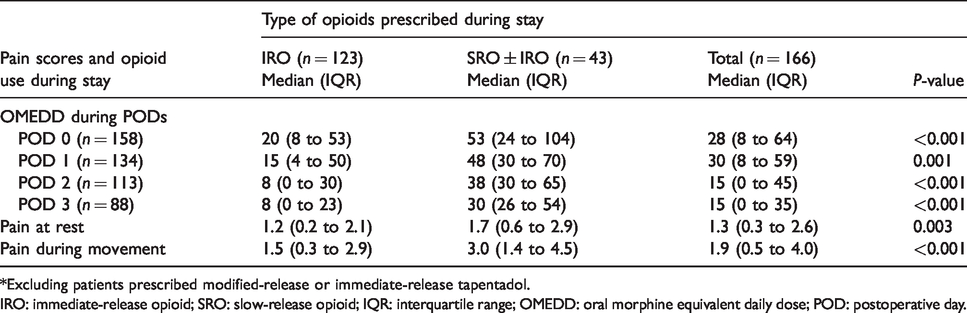
*Excluding patients prescribed modified-release or immediate-release tapentadol.
IRO: immediate-release opioid; SRO: slow-release opioid; IQR: interquartile range; OMEDD: oral morphine equivalent daily dose; POD: postoperative day.
Multivariable linear regression for opioid intake during postoperative days 0 to 3.
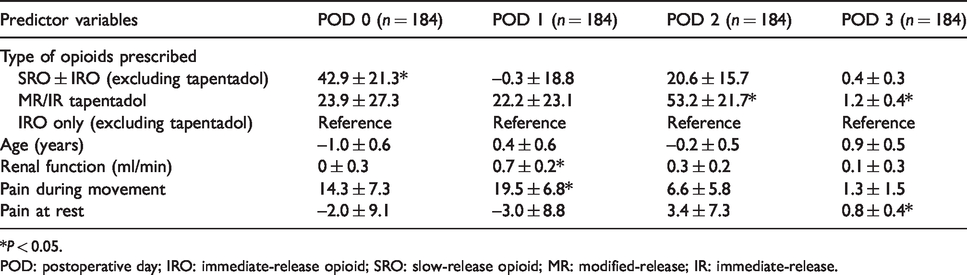
*P < 0.05.
POD: postoperative day; IRO: immediate-release opioid; SRO: slow-release opioid; MR: modified-release; IR: immediate-release.
Median pain scores at rest and during movement across POD 0 to 3 were also significantly higher for patients administered SROs (at rest: 3.0 versus 1.5, P = 0.003; during movement: 1.7 versus 1.2, P < 0.001) (Table 7). After adjusting for the use of adjuvants and orthopaedic specialty, pain scores during movement remained significantly higher when SROs were used (P = 0.004, B = 1.1 ± 0.4) (Table 9).
Multivariable linear regressions for pain at rest and during movement.
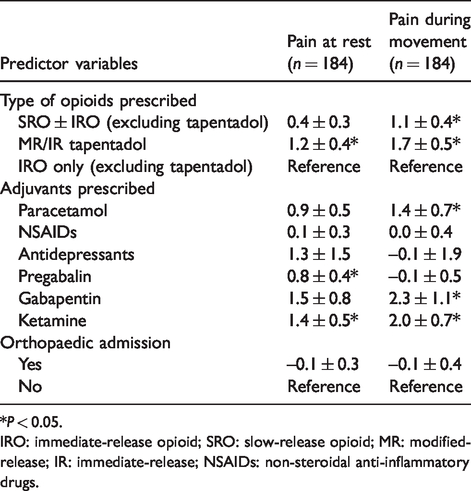
*P < 0.05.
IRO: immediate-release opioid; SRO: slow-release opioid; MR: modified-release; IR: immediate-release; NSAIDs: non-steroidal anti-inflammatory drugs.
Discussion
The primary outcome of this study is to compare the prescribing rates of SROs before and after the publication of the ANZCA/FPM position statement. A significant decrease in the prescription of SROs in hospital and at discharge was found between 2017 and 2018 after these recommendations were released. In particular, the odds of SROs being prescribed postoperatively and at discharge reduced by 86% and 88%, respectively, after adjusting for potential confounders (postoperative period: OR 0.14, P < 0.05; discharge: OR 0.12, P < 0.05) (Table 5). There was a significant increase in tapentadol modified-release prescriptions, which is likely to be due to tapentadol modified-release and immediate-release being approved for use for acute pain in this institution in August 2018 during the study period. Prescribers may also be more inclined to prescribe tapentadol due to some evidence of its superior safety profile compared to other opioids. 20 , 21 Therefore, tapentadol prescriptions have been analysed separately to avoid introducing a skew to the results.
Orthopaedic surgery is thought to result in a higher level of postoperative pain, and orthopaedic surgical patients are prescribed higher levels of postoperative opioid medication compared to patients of other surgical specialties. 22 In this study, orthopaedic patients were ten times more likely to be prescribed SROs while in hospital, and 31 times more likely to be prescribed SROs for discharge medication, than patients of other specialties (Table 5). Specialty-specific guidelines used by orthopaedic surgeons at our institution may have contributed to this, such as recently published recommended analgesic regimens for orthopaedic procedures such as hip and knee arthroplasty, as part of enhanced recovery after surgery protocols. 23 , 24 These protocols included the recommendation to use SROs postoperatively; however, the evidence supporting this recommendation was lacking.
Multiple studies have also reported overprescribing during orthopaedic discharge.25–28 Ruder et al. (2017) 25 found that orthopaedic patients are more likely to be prescribed SROs during discharge and reported that discharge prescriptions of slow-release oxycodone are more likely to be for a longer duration and of a higher OMEDD. On the contrary, Kim et al. (2016) 26 reported a utilisation rate of discharge opioids of only 34% after upper extremity procedures, while Saini et al. (2018) 27 reported a rate of 50% after foot and ankle surgery. A three-part review recently published in the Lancet discussed efforts carried out in a few studies to curb unnecessary prescribing at discharge while still maintaining adequate pain relief and patient satisfaction. 29 Some of the methods discussed were to personalise the number of pills dispensed according to opioid use 24 hours before discharge and deciding on the number of pills at discharge in discussion with the patient, both of which have yielded a lower number of pills prescribed at discharge while maintaining similar patient satisfaction scores. 30 , 31
After accounting for potential confounders, there was no significant difference in median pain scores between the SRO ± IRO and IRO groups for pain at rest throughout the hospital stay. In addition, pain during movement throughout hospitalisation only differed by a score of one (Table 9), which may not reach clinical significance. This may be due to higher OMEDDs prescribed in the SRO ± IRO group during postoperative day 0 (Table 8), possibly due to a need for a higher opioid dose in the SRO ± IRO group. Alternatively, the use of local anaesthetic blocks, cyclooxygenase-2 inhibitors and anti-inflammatories given intraoperatively may account for these differences. Another contributor to this may be that SROs do not confer much benefit over IROs, which would be consistent with the findings of Sunshine et al. (1996). 4
The provision of cessation plans decreased by 27% in the post-intervention group and only 23% of the total number of patients received a plan (Table 4). After adjusting for potential confounders, the odds of providing a cessation plan was four-fold less likely in the post-intervention group (OR 0.24, P < 0.05) (Table 6). The ANZCA/FPM position statement recommends that the initial prescriber be responsible for the weaning and cessation of the opioid prescribed. 3 However, the initial prescriber may not prepare discharge summaries and this may play a role in the lack of weaning instructions. This is concerning as first prescriptions of opioids often occur in the hospital setting and postoperative opioid use increases the risk of overdose and protracted opioid use. 32 , 33 Zhang et al. (2018) 33 showed that three major predictors of opioid use 18 months after first prescriptions of opioids were prescriptions of SROs, OMEDD of 50 mg or more and a supply exceeding seven days. This suggests that those discharged with SROs without cessation plans are at risk of the long-term use of opioids.
This study had a few limitations. It is based on a single institution, includes a heterogeneous surgical population and consisted of only two timepoints. The lack of a control group not exposed to the statement meant that while we could show a change in prescribing patterns during this time, we are unable to ascribe it to the release of the statement. These two timepoints may have been part of a broader continuum of change related to the awareness of opioid-related harms. In the period between the two measured timepoints, there has been one education session in July 2018 at hospital grand rounds regarding the ANZCA/FPM position statement and the distribution of the bulletin to ANZCA/FPM members regarding this recommendation. Besides that, information dissemination is largely reliant on word of mouth, with no official guidelines distributed to all prescribers. As the study is retrospective, the data collected are limited to those in medical records, paper charts and discharge prescriptions as well as being reliant on the reliability of recorded data. Information on the use of IROs pre-admission and pre-existing chronic pain were poorly recorded, and thus were not presented, which may have contributed to the confounding of study results. Preoperative and intraoperative use of opioids was not recorded as the focus is on the postoperative prescriptions of opioids. Further studies can include multiple timepoints or a follow-up period post-discharge to confirm the findings of this study. Besides that, future studies can also explore the perceptions of prescribers on acute pain management in order to curb the rising trend of inappropriate opioid prescribing. Notably, poor discharge management of opioids such as the lack of cessation plans can be addressed by using discharge templates and educational activities for medical officers.
Conclusion
There has been a decrease in the use of SROs for acute pain during hospitalisation and at discharge for postoperative patients between 2017 and 2018, which may be associated with the release of the ANZCA/FPM position statement. Notably, this reduction in the use of SROs was not accompanied by an increase in daily pain scores postoperatively, which may mean SROs provide minimal advantage over IROs. There is evidence in the literature of an association between the use of SROs and unintended long-term use as well as related harms. Therefore, these findings may be in the direction of more appropriate acute pain management. Specialty-specific guidelines on opioid use may have an impact on encouraging SRO prescriptions, especially for orthopaedic surgery. Moreover, a significantly low number of patients were found to have received an opioid cessation plan on hospital discharge and there was a decrease of cessation plans provided between 2017 and 2018. As such, further and wider dissemination of the guidelines is necessary.
Footnotes
Acknowledgements
The author(s) would like to thank Joanne Rimington for acting as the research supervisor for this study and Monica Gerges for assisting with the data collection.
Declaration of conflicting interests
The author(s) declare that there is no conflict of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding was provided by the School of Pharmacy, University of Sydney (Sydney School of Pharmacy Summer Scholarship 2018/19).
