Abstract
Summary
Opioids are often used to provide postsurgical analgesia but may cause harm if used inappropriately. We introduced an opioid stewardship program in three Melbourne hospitals to reduce the inappropriate use of opioids after patient discharge. The program had four pillars: prescriber education, patient education, a standardised quantity of discharge opioids, and general practitioner (GP) communication. Following introduction of the program, we undertook this prospective cohort study. The study aimed to describe post-program discharge opioid prescribing, patient opioid use and handling, and the impact of patient demographics, pain and surgical treatment factors on discharge prescribing. We also evaluated compliance with the program components. We recruited 884 surgical patients from the three hospitals during the ten-week study period. Discharge opioids were dispensed to 604 (74%) patients, with 20% receiving slow-release opioids. Junior medical staff undertook 95% of discharge opioid prescribing, which was guideline-compliant for 78% of patients. Of the patients discharged with opioids, a GP letter was sent for only 17%. Follow-up at two weeks was successful in 423 (70%) patients and in 404 (67%) at three months. At the three-month follow-up, 9.7% of patients reported ongoing opioid use; in preoperatively opioid naïve patients, the incidence was 5.5%. At the two-week follow-up, only 5% reported disposal of excess opioids, increasing to 26% at three months. Ongoing opioid therapy at three months in our study cohort (9.7%; 39/404) was associated with preoperative opioid consumption and higher pain scores at the three-month follow-up. The introduction of the opioid stewardship program resulted in highly guideline-compliant prescribing, but hospital-to-GP communication was uncommon and opioid disposal rates were low. Our findings suggest that opioid stewardship programs can improve postoperative opioid prescribing, use and handling, but the realisation of these gains will require effective program implementation.
Introduction
When moderate or severe postsurgical pain is expected, opioids are an integral component of adequate multimodal analgesia. However, over recent years concern has increased that intended short-term use following hospital discharge may continue beyond the expected recovery time.1–3 Prolonged opioid use may be problematic as it is associated with significant harm,4–6 and has resulted in the declaration of an opioid crisis internationally.7–11
Optimising post surgery opioid practice is a genuine challenge as the individual patient’s pain experience must be accounted for in a nuanced organisational response. Opioid stewardship programs aim to balance the need for adequate analgesia with measures to reduce potential opioid-related harm and unnecessary variations in practice. However, minimising opioid supply at the expense of adequate pain control is undesirable, and a knowledge gap exists in predicting appropriate patient-specific discharge opioid requirements following surgery.
Recent international consensus guidelines provide a framework for health services planning to implement opioid stewardship programs. 12 Academic detailing, 13 prescriber education, 14 reduction in default opioid quantities,15–17 and disposal instructions have been demonstrated to be effective in specific settings at improving opioid prescribing, use and handling.18,19 However, we, 20 and others, 1 have reported suboptimal discharge opioid prescribing and patient opioid handling, demonstrating the need for effective opioid stewardship. Currently, opioid stewardship programs that aim to improve opioid practice are variable and incomplete. 21
Assessing the success, or otherwise, of an opioid stewardship program is complex. Optimal opioid prescribing, appropriate use of opioids and safe handling of opioids are difficult to define and will vary with the clinical context. Key considerations include adequate individual opioid supply to optimise functional recovery whilst avoiding oversupply that may act as a community reservoir for diversion. Discharge prescription guidelines may assist in striking this balance for most patients.22,23 An absolute increase of 10% in the disposal rate of surplus opioids is a proposed marker of program success.19,24 The effectiveness of an opioid stewardship program can be assessed by reporting program compliance and safe opioid use and handling. Still, these data must be contextualised with the patient pain experience. Cessation of opioid therapy within the three months post surgery is desirable because of the risk of transition to chronic opioid use and increased opioid-related harm.25,26 The ability to rescue patients on non-optimal postsurgical pain management trajectories is essential, and transitional pain clinics can address this need.27–29
Following our baseline study of discharge opioid prescribing,
20
we introduced a bespoke discharge opioid stewardship program at The Royal Melbourne Hospital (RMH), Peter MacCallum Cancer Centre (PMCC) and The Royal Women’s Hospital (RWH). The program involved interventions grouped into four pillars: standardised discharge opioid prescribing, patient education, prescriber education, and continuity of care with the patient’s general practitioner (GP). Each health service adapted the components to meet its requirements. This study reports on a cohort of patients who underwent surgery at the three study hospitals after introducing the discharge opioid stewardship program. The study aims were to determine the following:
The incidence of hospital discharge following surgery with new opioid medication and the quantity of opioids dispensed. The patient demographics, pain and surgical treatment factors that impacted opioid discharge prescription. Compliance with the discharge opioid prescribing guideline. The quantity and duration of prescription opioid use by patients after surgery. The handling of excess opioids by patients. The patient demographics, pain and surgical treatment factors associated with optimal opioid management and sustained opioid use three months after hospital discharge.
Methods
This prospective cohort study was conducted in three hospitals that had previously participated in our baseline discharge opioid study (RMH, PMCC and RWH). 20 The Director of Anaesthesia or the Director of Pain Services at each site endorsed the opioid stewardship program. The program was introduced between three and five months before commencing patient recruitment.
The components of the pillars of the opioid stewardship program were:
Standardised quantity of discharge opioids – The guideline recommended standardised discharge opioid prescription (oxycodone 5 mg immediate-release quantity 10–12 tablets). The guideline did not recommend discharge opioid medication unless the patient consumed opioids in the last 24 h of their admission. The guideline recommended adjunct analgesia (paracetamol and non-steroidal anti-inflammatory drugs or cyclooxygenase type 2 inhibitor medications (anti-inflammatories)) be co-prescribed with opioids unless contraindicated. Increased quantities and other opioids, including slow-release (SR) formulations, were considered guideline-compliant if advised by the pain or palliative care team or prescribed according to a hospital pathway. Pharmacists were encouraged to query any discharge opioid prescriptions for excess quantities or omission of adjunct analgesia without clear contraindication. Patient education – Patient opioid education was delivered in the pre-admission clinic (PAC) before elective surgery to set expectations for the anticipated acute postsurgical pain trajectory and advise patients on opioid use, storage and disposal. Site investigators educated colleagues regarding the program, but the PAC anaesthetist delivered unscripted patient opioid education. In addition, site lead pharmacists distributed standardised written discharge opioid handling instructions to the dispensary. They also educated colleagues to include the written program material along with verbal opioid medication counselling at the time of discharge opioid dispensing. Prescriber education – Opioid stewardship program education was delivered to junior medical staff by pain specialists, anaesthetists or pharmacists. A standard slide set was the basis for the group education session, modified as required for site differences. In addition, all junior medical staff on surgical units, whether they attended the education session or not, received information about the stewardship program via email. Continuity of care with the GP – A standard discharge letter for all patients dispensed discharge opioids was developed and modified for site differences. The letter was provided to the nominated GP at discharge either by post or with the patient. The letter addressed opioid therapy expectations for acute postsurgical pain, including limited opioid discharge supply and the potential need for pain management review after discharge. Pain services at each hospital considered the best mechanism to deliver an opioid discharge letter to the patient’s GP, given their knowledge of ward and discharge workflows.
Research ethics approval was obtained from Melbourne Health (HREC/46479/MH-2018.347), and governance approvals were obtained for each study site. Based on our previous study, 20 we projected that with a ten-week study period across three centres, up to 1000 eligible patients could be recruited. Further, we anticipated that 60% of the participants would be dispensed opioids at discharge to obtain a two-sided 95% confidence interval (CI) with half-width of 3%. 20
Inclusion criteria included adult patients undergoing elective or emergency surgery with an inpatient post-operative stay of at least one night during the ten-week recruitment period. Exclusion criteria included impaired cognitive function or lack of English language proficiency to comprehend the study intent and procedures. Patients also had to be prescribed and dispensed opioids at hospital discharge to be eligible for follow-up. Written informed patient consent was obtained for study participation. Patients were recruited and had the baseline assessment performed whilst inpatients. Phone follow-up at two weeks and three months after hospital discharge utilised a standardised scripted interview. Contact was attempted on at least three occasions on different days and times before declaring the patient lost to follow-up. Failure to complete the two-week follow-up did not preclude attempting follow-up at three months.
Data were collected from the patient interview and the paper and electronic medical records. Baseline presurgery data collected included patient characteristics (age, weight, presence of pain or opioid therapy on admission, a diagnosis of depression or anxiety, Hospital Anxiety and Depression Scale (HADS), 30 American Society of Anesthesiologists physical status classification (ASA PS) and surgical characteristics (type of surgery, urgency (elective, emergency), PAC attendance and investigator-anticipated nociceptive intensity (nil to mild, moderate, severe)). The Acute Pain Service (APS) provided a consultation service of nursing and pain medicine or anaesthesia specialist teams, referred at the discretion of treating anaesthesia and surgical teams based on expected postoperative analgesia requirements. Whether APS consultation occurred during hospital admission was recorded.
Data collected at discharge included length of stay (days) and prescription details (opioid use in the 24 h before discharge, discharge opioid and adjunct analgesia prescribed (agent, formulation, quantity and oral morphine equivalents)). Data collected at the two-week follow-up included opioid use to date and ongoing, additional opioid prescription, reasons for cessation, opioid handling since discharge including storage, disposal and diversion, recall of opioid storage and disposal instructions and the modified Brief Pain Inventory short form (mBPI-sf).31,32 Data collected at the three-month follow-up included opioid use to date and ongoing, additional opioid prescription, reasons for cessation and opioid handling since discharge, including storage, disposal and diversion and mBPI-sf. Pain intensity as measured on the mBPI-sf was subcategorised as mild (score 1–3), moderate (score 4–6) or severe (score of 7 or greater). The mBPI-sf was subcategorised to assess pain intensity and interference with physical activity and affect.31–34 We investigated opioid storage at the two-week and three-month follow-ups in patients who reported that they were still taking opioids or that they had leftover opioids. Patients who said they had consumed all their opioids or had disposed of all their opioids were excluded. We investigated opioid disposal at the two-week and three-month follow-ups in patients who reported that they had ceased taking opioids and had leftover opioids. Patients who were still taking opioids were excluded.
We assessed opioid prescribing per the discharge guideline (protocol discharge opioids), reported prescriber and patient opioid education (in PAC and at discharge opioid dispensing) and GP communication to determine adherence to the opioid stewardship protocol.
Patient opioid groups
The three-month follow-up cohort was examined to assess the factors associated with patient opioid use and handling. The opioids three-months group comprised patients who were still taking opioids. The optimal three-months group included patients who had ceased opioid therapy and reported no diversion and no leftover opioids (either due to finishing their supply or disposing of any excess opioids). Patients who were not taking opioids but reported diversion or leftover opioids were not included.
Data analysis
Data were entered into the study database and exported to STATA (Version 15: StataCorp. 2017 and version 16: StataCorp. 2020; Stata Statistical Software: StataCorp LP, College Station, TX, USA), where summaries and analyses were prepared.
Continuous data were summarised using mean and standard deviation (SD) or median and 25th to 75th percentile if skewed, and categorical data as counts and percentages. Two-sided 95% CIs for proportions were calculated using the Clopper–Pearson method.
Logistic regression was used to explore the associations between (a) pre-surgery characteristics and opioids dispensed at discharge; and (b) pre-surgery, discharge and post-discharge characteristics and inclusion in a patient opioid group. Pre-surgery characteristics included hospital site, age (≤50, >50 years; dichotomised due to nonlinearity), weight, sex, pain at hospital admission, opioid use at hospital admission, baseline morphine equivalents, surgical urgency, ASA PS (1–2 =‘healthy’ 3–5 = ‘comorbid’), diagnosis of anxiety or depression, HADS scores for anxiety and depression, and investigator-anticipated nociceptive intensity. Discharge characteristics consisted of the length of hospital stay, APS review, post-discharge characteristics of the mBPI-sf score at three months post-surgery, and discharge opioid prescription guideline adherence.
Characteristics (i.e. covariates) with a
Results
A total of 884 patients were recruited in this study from three hospitals (RMH (
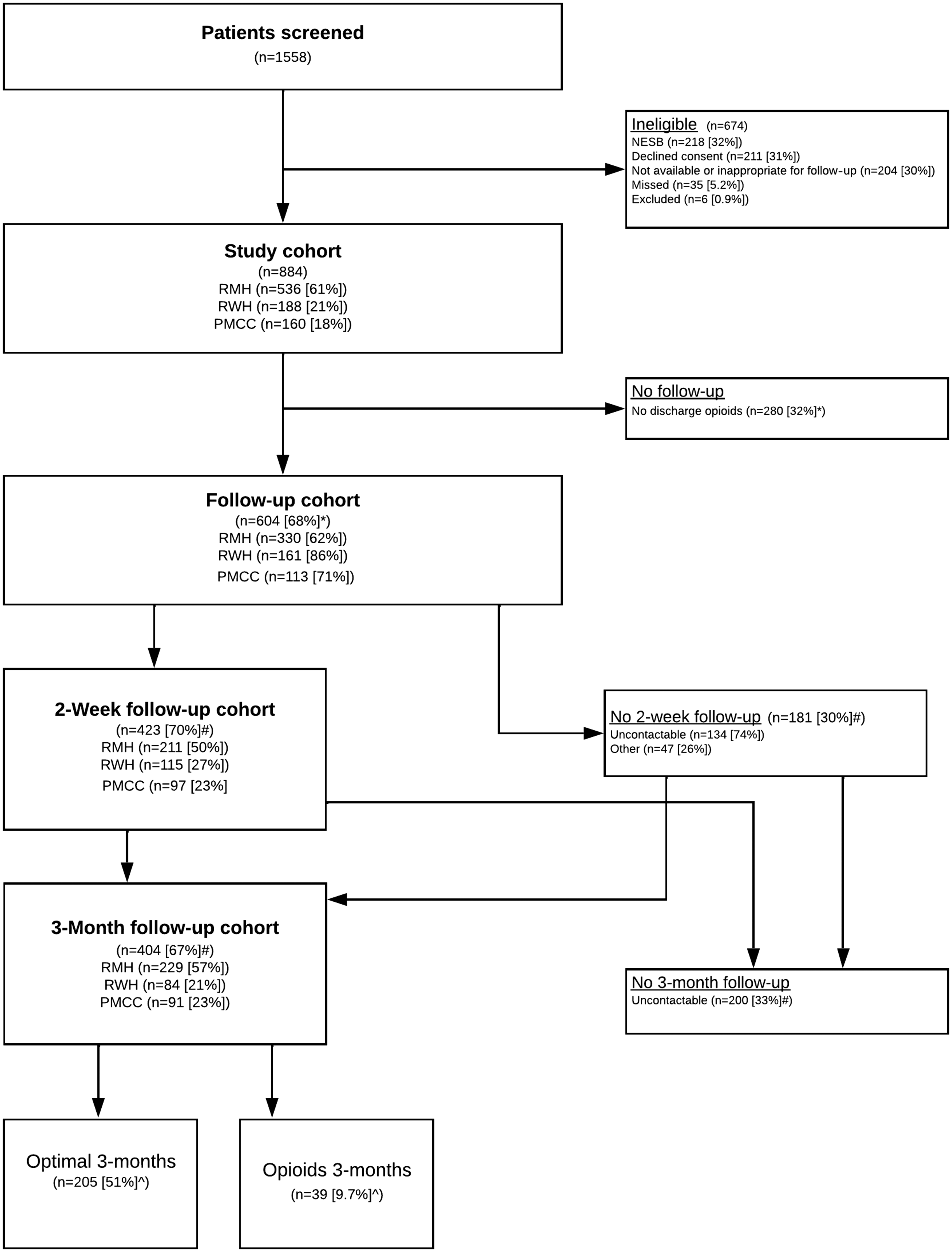
Recruitment flow chart. Follow-up cohort is the subset of the study cohort that received discharge opioids. Other reasons for two-week no follow-up were either still in an inpatient location (e.g. rehabilitation) or not managing opioids dispensed on hospital discharge (e.g. nursing home, hospital in the home medication management). Note that failure to follow-up at two weeks did not preclude attempt to follow-up at three months. Optimal three-months group = Had ceased opioid therapy and either had no leftover opioids or had disposed of the excess. No diversion. Opioids three-months group = ongoing opioid use at the three-month follow-up. *Sites expressed as percentage of study cohort; #percentage of follow-up cohort; ^percentage of three-month follow-up cohort. NESB, Non-English speaking background; PMCC, Peter MacCallum Cancer Centre; RMH, Royal Melbourne Hospital; RWH, the Royal Women’s Hospital.
The mean age of participants was 50 years (SD 17.1 years; range 18–94 years). Of those who consented, 488 (55%) were female and 396 (45%) were of male biological sex. This study covered elective (
Patient pain and surgical treatment characteristics.
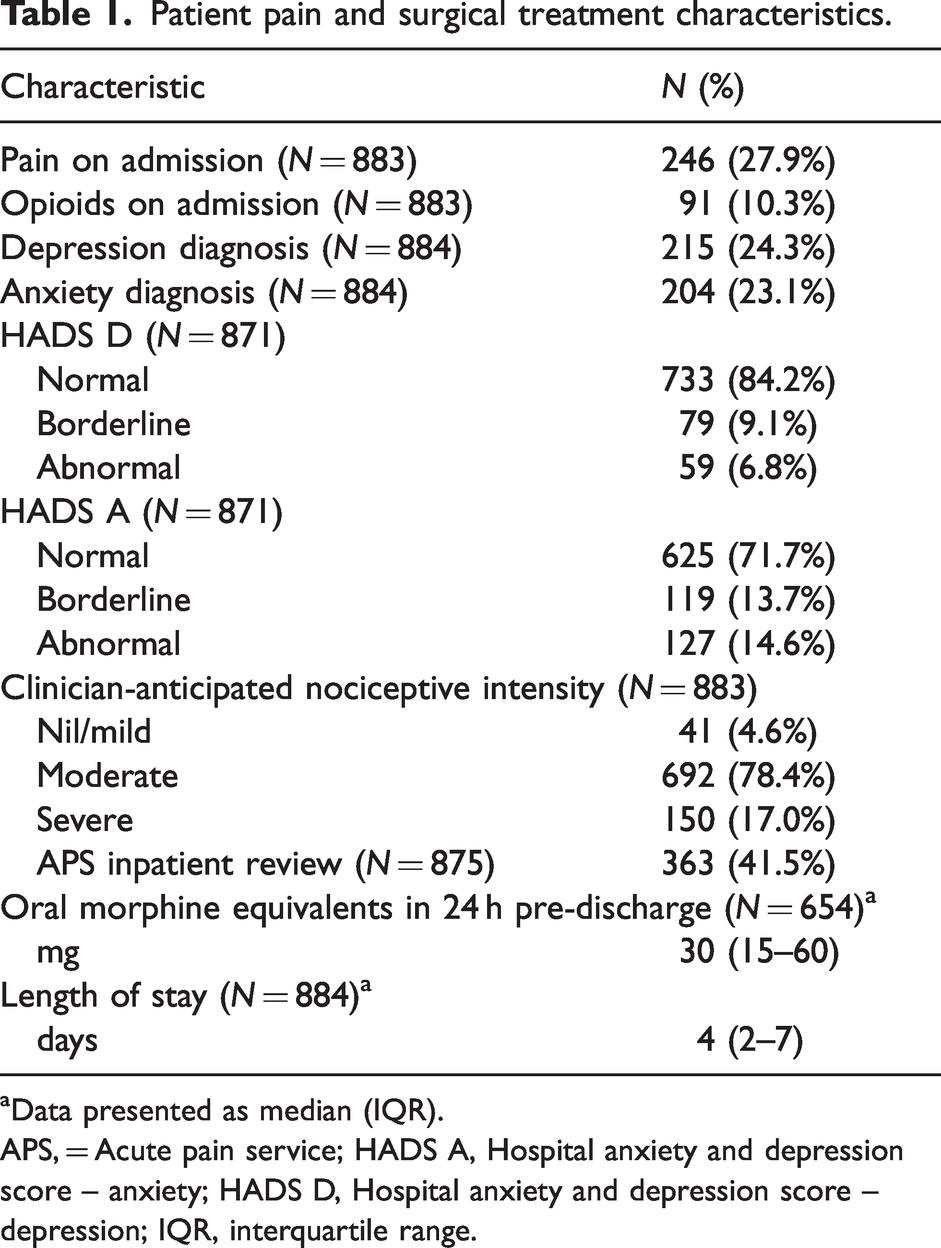
aData presented as median (IQR).
APS, = Acute pain service; HADS A, Hospital anxiety and depression score – anxiety; HADS D, Hospital anxiety and depression score – depression; IQR, interquartile range.
Paracetamol was prescribed to 97% of participants during their inpatient stay and anti-inflammatories to 52%. There was APS input for 42% of patients.
Discharge prescribing and opioid stewardship measures
At the time of hospital discharge, opioids were prescribed to 604 patients (74%) (95% CI 71% to 77%) (Table 2).
Discharge opioid prescribing and stewardship protocol compliance.
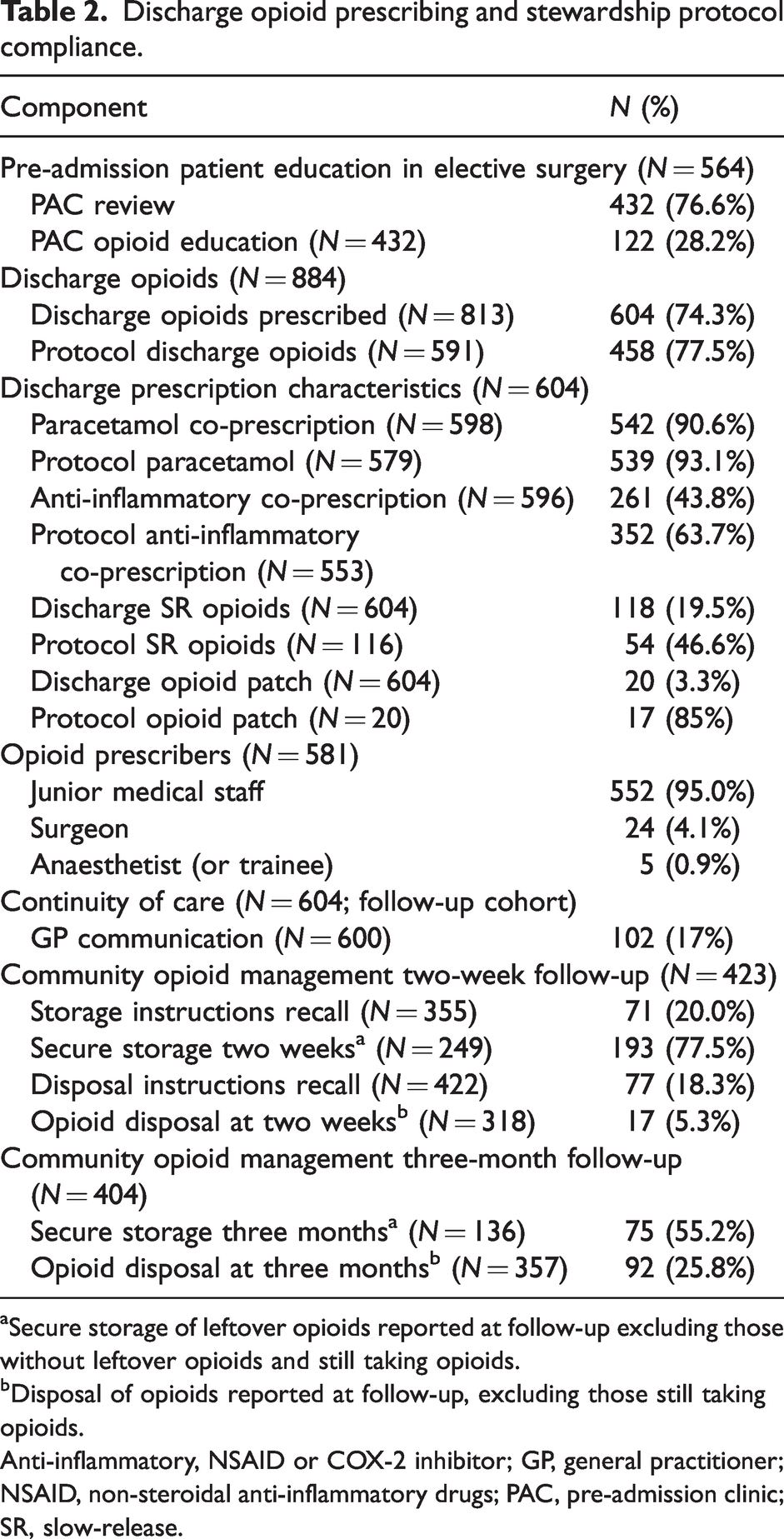
aSecure storage of leftover opioids reported at follow-up excluding those without leftover opioids and still taking opioids.
bDisposal of opioids reported at follow-up, excluding those still taking opioids.
Anti-inflammatory, NSAID or COX-2 inhibitor; GP, general practitioner; NSAID, non-steroidal anti-inflammatory drugs; PAC, pre-admission clinic; SR, slow-release.
The median prescribed oral morphine equivalent dose for discharge opioids was 80 mg (interquartile range (IQR) 75–150 mg). Junior medical staff were responsible for 95% of discharge prescribing. Discharge opioid guideline prescription compliance was 78% (Table 2) and guideline adherence for adjuvant co-prescription of paracetamol and anti-inflammatories was 91% and 64%, respectively. Immediate-release oxycodone was the most commonly prescribed discharge opioid (510/604 = 84%). Prescription of SR opioids was less common (118/604 = 19.5%) and guideline compliance for SR opioids was lower at 47%. Amongst the patients that attended PAC before elective surgery, 28% (
A multivariable logistic regression model was constructed for patients prescribed opioids on hospital discharge. See Table 3 for univariable and multivariable analysis.
Factors associated with prescription of discharge opioids (study cohort).
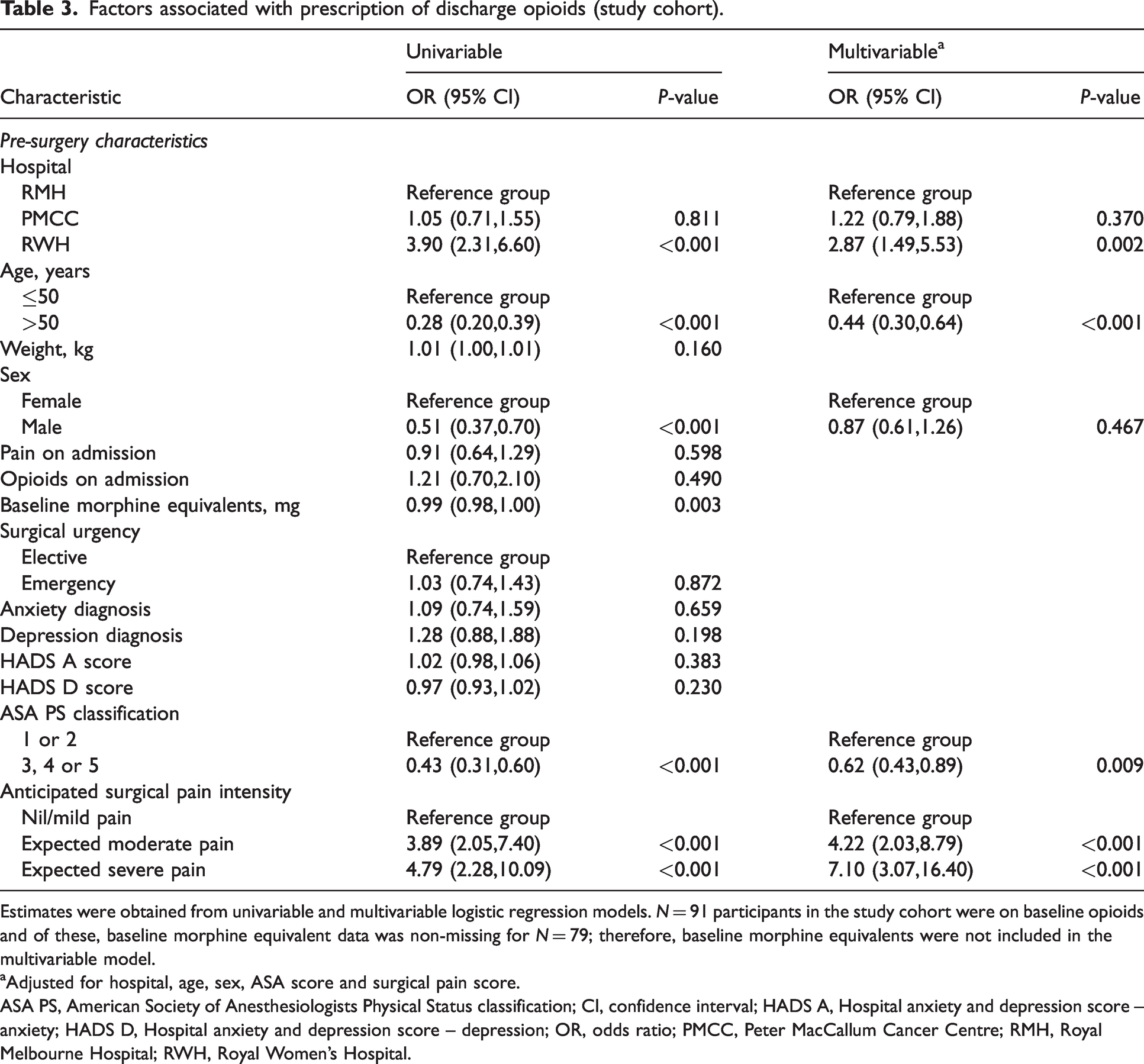
Estimates were obtained from univariable and multivariable logistic regression models.
aAdjusted for hospital, age, sex, ASA score and surgical pain score.
ASA PS, American Society of Anesthesiologists Physical Status classification; CI, confidence interval; HADS A, Hospital anxiety and depression score – anxiety; HADS D, Hospital anxiety and depression score – depression; OR, odds ratio; PMCC, Peter MacCallum Cancer Centre; RMH, Royal Melbourne Hospital; RWH, Royal Women’s Hospital.
The variables included were hospital, age, sex, ASA PS and investigator-anticipated nociceptive intensity based on 773/884 (87%) complete cases. Factors identified as being associated with a higher likelihood of opioid prescribing on discharge were 50 years of age or less (OR 2.29, 95% CI 1.56 to 3.35,
Post-discharge opioid use and handling
Of the patients prescribed opioids on discharge, follow-up was completed by 70% (
At two weeks following hospital discharge, participants reported variable use of their supplied discharge opioids. No opioid use since discharge was reported by 20% (
At the two-week follow-up, 67% of patients reported having leftover opioid medication, and 78% of this group reported storing the excess in a secure location. Only 5.3% of respondents reported disposing of their leftover opioids at the two-week follow-up, and ten participants (2.4%) reported diversion of prescribed opioids.
Of the patients followed up successfully at three months, 9.7% (95% CI 7% to 13%) reported ongoing opioid use. In patients who were opioid naïve at hospital admission, the rate of opioid use at three months was 5.5% (95% CI 3.4% to 8.4%). The proportion of patients reporting ongoing opioid use at three months was 14.0% (95% CI 9.8% to 19.2%) at RMH, 6.6% (95% CI 2.5% to 13.8%) at PMCC and 1.2% (95% CI 0.03% to 6.5%) at RWH. Reasons for ceasing opioid therapy (participants could nominate more than one) included no pain (82%), running out of opioid medication (24%) or experiencing a side-effect (12%). Leftover opioids were reported by 36% of those followed up, and 55% were storing these in a secure location. Disposal of excess opioids was reported by 26%, with methods including returning to a pharmacy (40%), household garbage (37%) or washing down the sink or toilet (8.6%). At the three-month follow-up, a further eight participants (2%) reported diversion in addition to those reported at two weeks.
Pain intensity
At follow-up two weeks after hospital discharge, 16% (
Opioid outcomes at three months
Table 4 shows the demographics of the participants who received discharge opioids and of the patient opioid groups optimal three-months and opioids three-months.
Patient opioid group demographics.
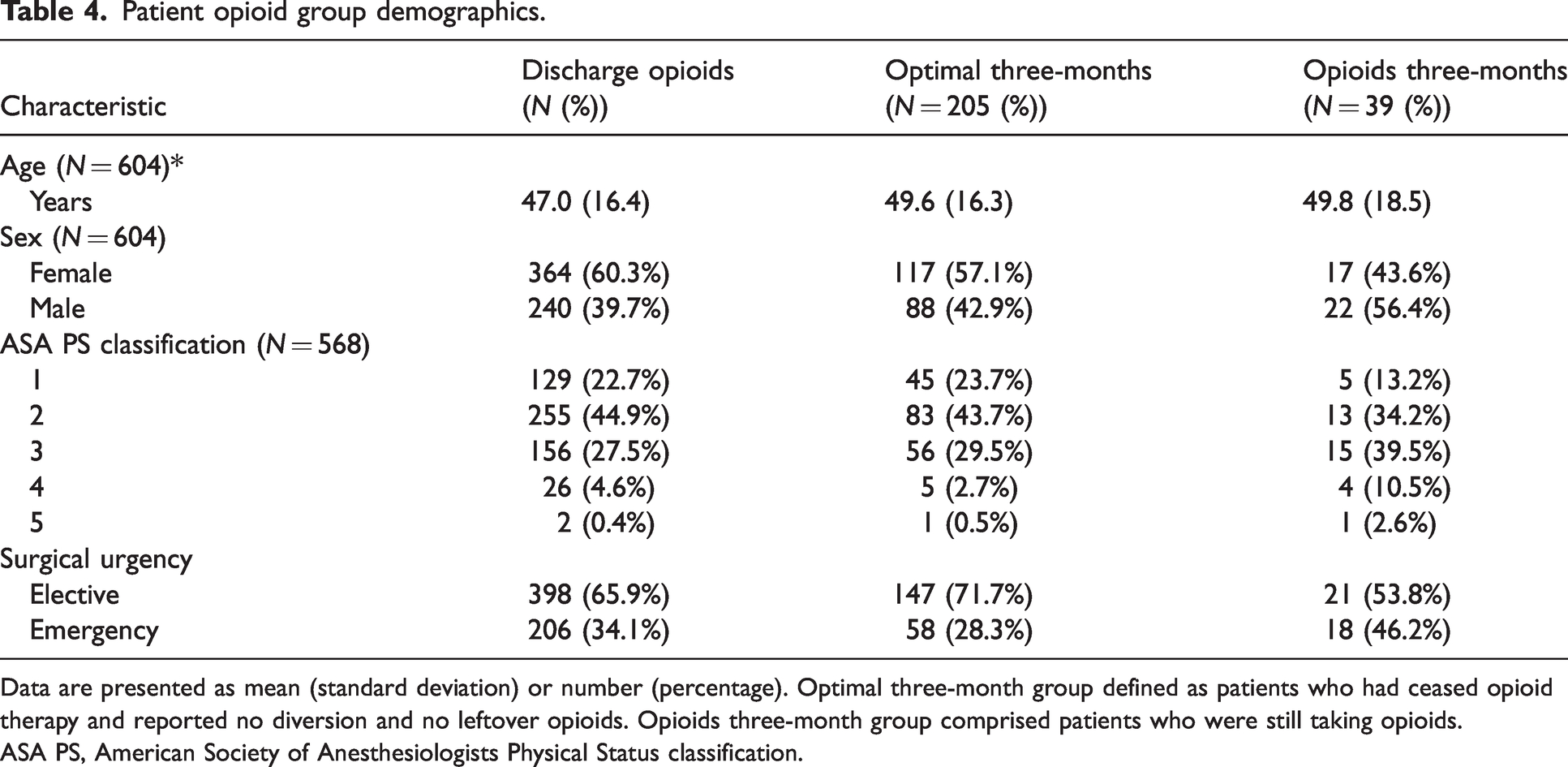
Data are presented as mean (standard deviation) or number (percentage). Optimal three-month group defined as patients who had ceased opioid therapy and reported no diversion and no leftover opioids. Opioids three-month group comprised patients who were still taking opioids.
ASA PS, American Society of Anesthesiologists Physical Status classification.
Of the 404 patients who completed follow-up at three months, 205 (51%, 95% CI 46% to 56%) met the criteria for allocation to the optimal three-months group. There were 39 patients (10%, 95% CI 7%, 13%) who met the criteria for allocation to the opioids three-months group.
Table 5 shows the patient pain and surgical treatment characteristics of the participants who received discharge opioids along with the characteristics of those in the patient opioid groups optimal three-months and opioids three-months.
Patient opioid group pain and surgical treatment characteristics.
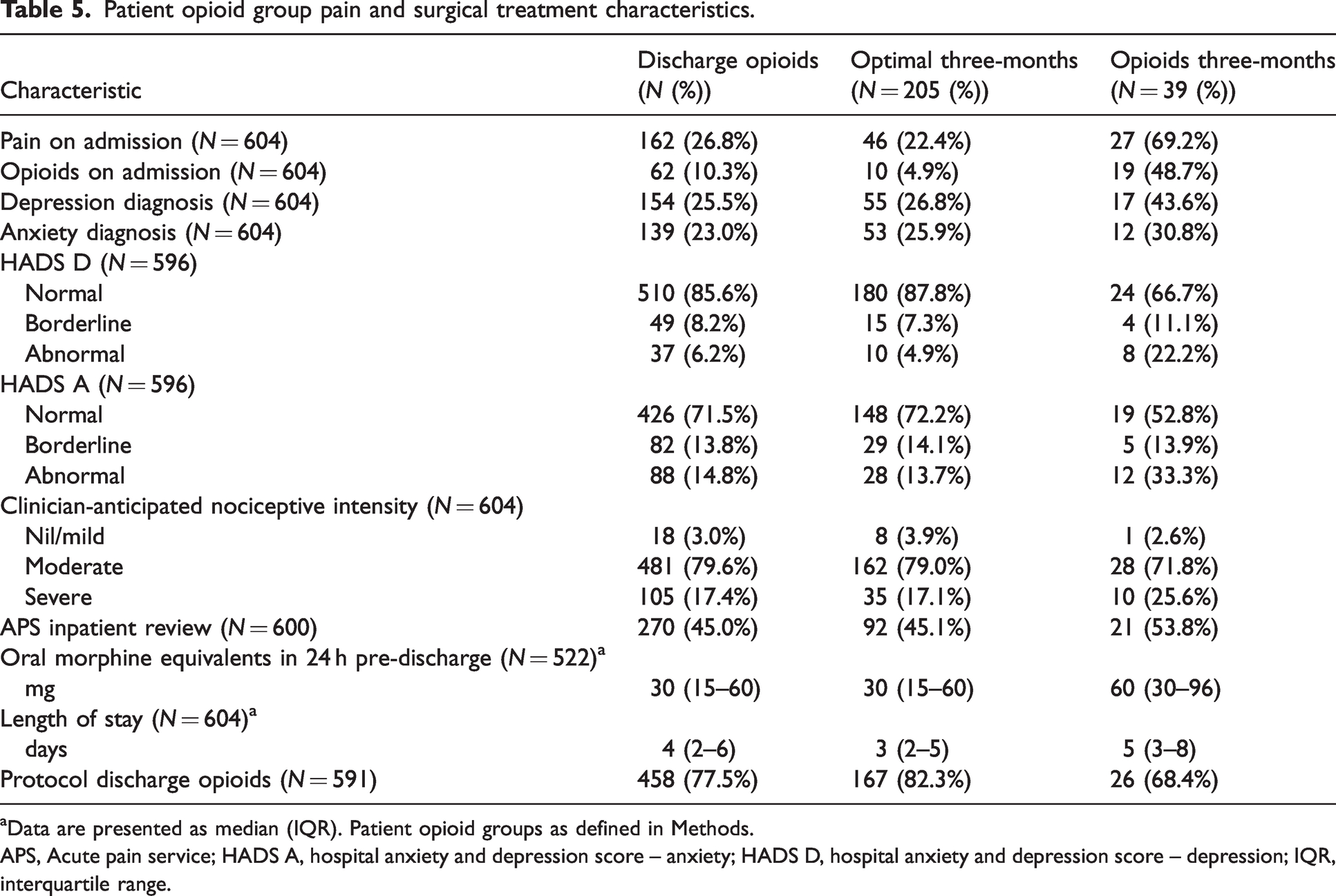
aData are presented as median (IQR). Patient opioid groups as defined in Methods.
APS, Acute pain service; HADS A, hospital anxiety and depression score – anxiety; HADS D, hospital anxiety and depression score – depression; IQR, interquartile range.
A multivariable logistic regression model was constructed to identify the variables associated with the inclusion of patients in either the optimal three-months or opioids three-months groups. See Table 6 for the univariable and multivariable analysis.
Factors associated with allocation to the patient opioid groups Optimal three-months and Opioids at three months.
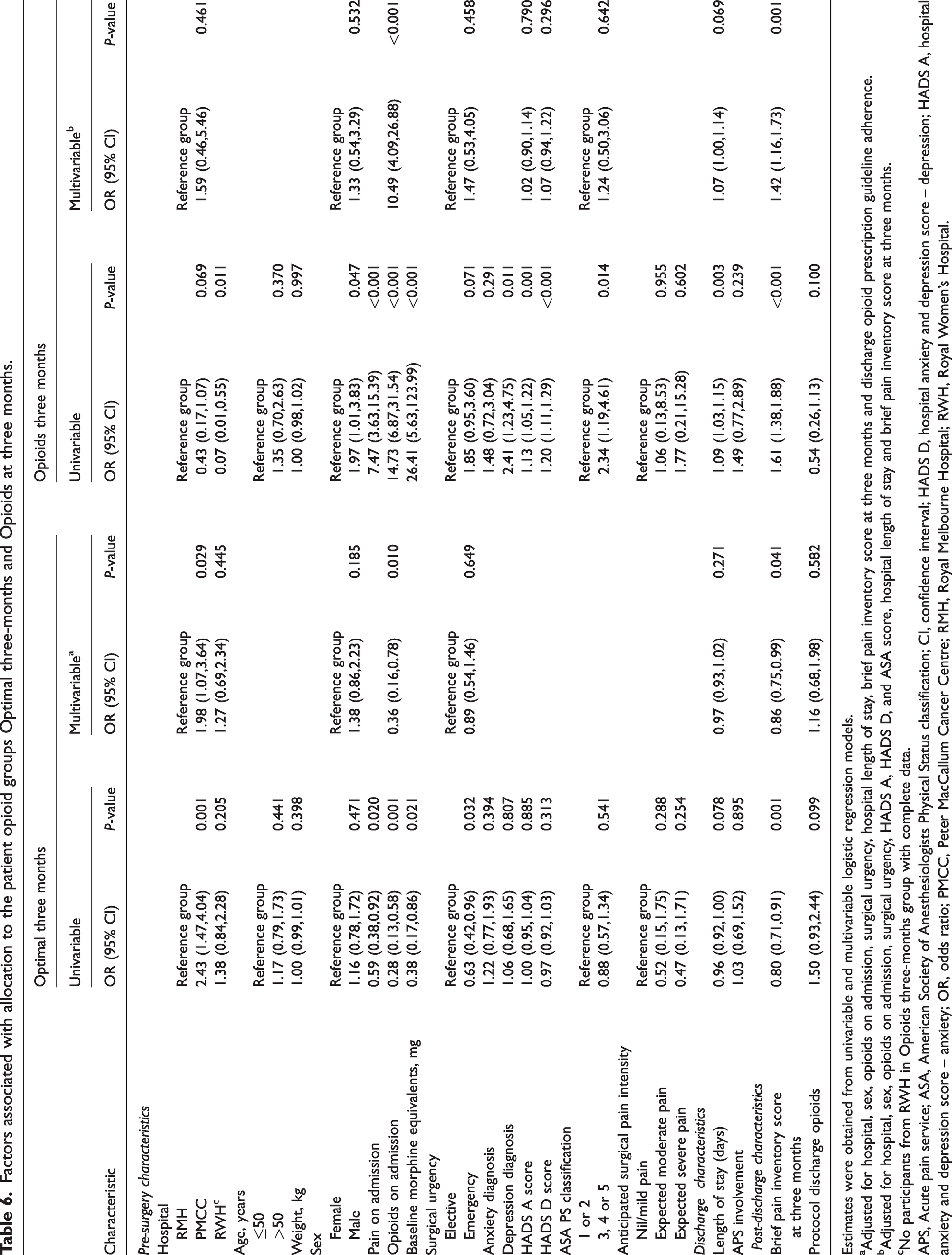
Estimates were obtained from univariable and multivariable logistic regression models.
aAdjusted for hospital, sex, opioids on admission, surgical urgency, hospital length of stay, brief pain inventory score at three months and discharge opioid prescription guideline adherence.
bAdjusted for hospital, sex, opioids on admission, surgical urgency, HADS A, HADS D, and ASA score, hospital length of stay and brief pain inventory score at three months.
cNo participants from RWH in Opioids three-months group with complete data.
APS, Acute pain service; ASA, American Society of Anesthesiologists Physical Status classification; CI, confidence interval; HADS D, hospital anxiety and depression score – depression; HADS A, hospital anxiety and depression score – anxiety; OR, odds ratio; PMCC, Peter MacCallum Cancer Centre; RMH, Royal Melbourne Hospital; RWH, Royal Women’s Hospital.
Factors identified as being associated with belonging to the optimal three-months group were being on opioids at hospital admission (OR 0.36; 95% CI 0.16 to 0.78;
Discussion
We conducted this study of discharge opioid prescribing for surgical patients after introducing an opioid stewardship program in three Melbourne hospitals. In our previous study, before the opioid stewardship program, we reported that discharge opioid prescribing was often for more than patient requirements. 20 In addition, patients were not storing their opioids securely, and disposal of excess opioids was uncommon. 20 In this study, we report fewer opioids were dispensed on hospital discharge, that guideline compliance was high and there was improved opioid storage but ongoing low rates of early opioid disposal. Ongoing opioid therapy three months after hospital discharge was consistent with other reports.
Opioid prescribing, use and handling
Patients who were more likely to be dispensed opioids at the time of hospital discharge were younger, healthier and had undergone surgery that was anticipated to be more painful. Therefore, no modifiable patient demographic, pain or surgical treatment factors were associated with the rate of discharge opioid prescribing in our study cohort. The pre-surgery characteristics included in the logistic regression analysis are listed in Methods, and the associations are displayed in Table 3. These were similar to the factors included in the regression model from our pre-stewardship program cohort, 20 and modelling discharge opioid prescribing in other practice contexts.35,36 In trauma patients, factors such as injury severity score have been associated with discharge opioid prescribing. 36 In our study, the investigator-anticipated nociceptive intensity was used instead as it applied to elective surgery and both trauma and non-trauma emergency surgery. Comparing the results from this study with data from our previous cohort, 20 the proportion of recruited patients prescribed opioids on hospital discharge increased (74% in this study versus 60%). However, the median oral morphine equivalent dose dispensed on discharge was reduced from 150 mg to 80 mg. Junior medical staff were the discharge opioid prescribers in 95% of cases, and opioid prescribing was guideline-compliant in 78% of cases.
The prescribing of SR opioids for acute pain is controversial due to the potential for opioid-related harm, including the transition to chronic opioid use. In 2018, the Australian and New Zealand College of Anaesthetists and its Faculty of Pain Medicine released a position statement cautioning against such use. This document was updated in 2022. 37 There is provision for using SR opioid formulations in acute pain where there is an identified need, close monitoring and cessation planning. In our study, a minority (19.5%) of patients were prescribed SR opioids on discharge, and protocol compliance in this group was 47% compared with 78% overall compliance. Prescribing of SR opioids was considered compliant if advised by pain services or as part of the RMH arthroplasty pathway. Prescription of paracetamol and anti-inflammatories as adjunct analgesia was consistent with the discharge opioid prescription guideline in 91% and 64%, respectively.
A recent study in the USA identified difficulty accessing appropriate pain assessment instruments and technological support, lack of awareness of guidelines and time pressure as barriers to opioid prescribing guideline adherence. 38 However, opioid prescribing was perceived as important by the prescribers, pharmacists and nursing staff in the study focus groups. 38 It is unknown whether these same barriers existed in our study hospitals. The study hospitals used conventional paper medical records, and the GP letter was hard copy. GP communication regarding the hospital plan for discharge opioids was uncommon at 17%.
The rate of continued opioid therapy two weeks after hospital discharge was 17% in this study (compared with 27% in our previous cohort). 20 Opioid consumption at the two-week follow-up (Table 3) was variable, with 20% of patients not using any of the dispensed opioids and 37% consuming all opioids. This suggests that discharge opioid prescribing could be better targeted, especially for patients at these extremes. One potential strategy would be to determine discharge opioid prescription by the opioid consumption in the 24 h before discharge. The proportion of patients requiring extra supply was similar (25% in this study versus 24% in our previous cohort), 20 despite the lower median oral morphine equivalents dispensed after implementing the opioid stewardship program. The stable rate of rescue opioid prescribing after the opioid stewardship program introduction is reassuring that a ‘burden shift’ from hospital prescribers to GPs was avoided. However, further improvement in community opioid prescribing and use is possible. Secure opioid storage at two weeks increased to 78% in this study from 12% in our previous study. 20 This improvement was possibly due to the opioid counselling in PAC, and medication counselling along with written opioid handling information at dispensing. The rate of opioid disposal at the two-week follow-up was low in our previous study at (5%), 20 and the incidence was unchanged in this study (5%). The three-month follow-up in this study revealed that the disposal rate was 26%; there was no three-month follow-up in our previous study for comparison. At three months, the increased disposal rate could reflect the expected further recovery from surgery, including resuming errands like visiting the pharmacy facilitating opioid disposal. Alternatively, the two-week follow-up phone call may have prompted patients to dispose of their opioids.
Pain experience
Despite a significant proportion of patients self-reporting substantial pain, scores on the mBPI-sf for the cohort engaged at two weeks and three months reported low intensity, interference with activity and interference with affect. The finding that the pain severity was strongly associated with interference in physical and affective function domains is consistent with the previous research. 39 Thus, pain management impacts physical functional and emotional activity with worse pain and greater opioid use associated with reduced function. 40
Our cohort included a substantial urgent or semi-emergent caseload (36% emergency surgery; trauma, cancer and obstetric cases also captured). The prevalence of postsurgical pain was similar to that in previous studies.41–43 Another study involving orthopaedic trauma patients at RMH found the rate of persistent pain at three months was 65%, and 31% of those with pain were on opioids, with or without non-opioid adjunct analgesia. Concerningly, 11% of participants reported ongoing opioid use without persistent pain at three months. 44 Persistent postsurgical pain is commonly neuropathic,45,46 and opioids may not be the most suitable treatment modality given their potential harm.25,26,47 Furthermore, ongoing opioid use after surgery may not be due to nociception, but rather physical or psychological dependence. The predictors of patients continuing opioid therapy beyond the acute post-surgical phase are partially understood.46,48 Factors include patient characteristics such as distress and anxiety, and system factors such as local prescribing and dispensing practices. Surgical patients on opioids at hospital admission require careful assessment, planning and education regarding post-discharge opioid use and management if the cessation of opioid therapy at three months post–hospital discharge is a therapeutic goal. There is an evolving role for transitional pain clinic assessment to differentiate neuropathic and nociplastic pain from nociceptive pain, and to institute optimal management for the different pain trajectories.27–29,49 Ideally, the patient’s GP would also be capable and willing to reassess and adjust management.
Optimal opioid therapy post surgery
Postoperatively, our concept of ideal opioid use includes optimal postsurgical pain relief with multimodal analgesia to minimise side-effects and promote functional recovery. Ideally, opioid use should be time-limited, corresponding with patients’ clinical improvement, and there should be minimal residual opioids that patients safely dispose of upon cessation of opioid therapy. A program that supports this ideal opioid use would include preoperative patient education, expectation setting and opioid weaning where possible. At discharge, such a program would advocate for adequate, not excessive, opioid prescription, provide clear direction on opioid use and handling, and communicate hospital pain management with the GP to promote continuity of care. Ideally, patients identified as high risk or following non-optimal pain management or opioid use trajectories would receive a transitional pain clinic review.
We undertook multivariable logistic regression analysis to determine the factors associated with belonging to the opioids three-months group or the optimal three-months group within our study cohort. This exploratory analysis sought to identify the characteristics that may impact surgical patients’ post-discharge opioid use and handling. In our study, patients admitted to the hospital on opioid therapy were more likely to be included in the opioids three-months group and not in the optimal three-months group. In addition, the higher reported mBPI-sf pain scores in the opioids three-months group support a more intense pain experience in this group. Therefore, the characteristics associated with belonging to the optimal three-months group or the opioids three-months in our study require evaluation in a separate cohort if predictive inference is to be made.
The pre-surgery and discharge characteristics assessed for association with opioids or optimal three-month group membership are listed in Methods, and the univariable associations are displayed in Table 6. The hospital site was included due to the inherently different clinical populations served. The pre-surgery characteristics of age,35,50 sex,35,50 pain at presentation to hospital,35,50 comorbidity, 36 depression and anxiety,35,50 were included in other studies exploring the factors associated with progression to persistent opioid use.46,48 Opioid use at hospital admission may be considered an extension of pain on admission and was also included in the univariate analysis. The discharge and post-discharge characteristics of APS review, adherence to discharge opioid prescription guidelines and pain reported at follow-up were included because a larger initial opioid discharge prescription is associated with persistent opioid use.48,50
That 5.5% of opioid naïve patients were still on opioid therapy three months after hospital discharge is consistent with some local and international literature.1–3 A single optimum rate of persistent postsurgical opioid use is overly simplistic. A construct including the nuance of patient demographic and psychological, surgical, pain experience and social factors would be more meaningful. It remains unclear what proportion of the opioids three-months group continuing opioid therapy is consistent with optimal opioid use and whether ongoing opioid consumption is due to physical or psychological dependence rather than persisting nociception.
Limitations and future directions
This study has some limitations. Firstly, patients that consented but did not receive discharge opioids (
Given the limited number of patients in the opioids three-month group, all study findings are exploratory.
This study has revealed incomplete opioid stewardship program implementation across health services. The opioid prescription guideline showed high compliance, but patient opioid education and GP communication had lower adherence rates. In addition, assessment of the extent of patient opioid education in the PAC and upon dispensing discharge opioid medications was limited by the patient recall, likely under-reporting the extent of opioid education.
This study was conducted across three hospitals servicing different patient populations–a general adult trauma hospital, an obstetric hospital and a specialist cancer centre. Therefore, the variable patient presentations and clinical practices should be considered when interpreting our study findings. Further, our work has focused on metropolitan tertiary referral centres. Therefore the study patient population and specialist hospital services may not represent the general surgical population, hospital care and perioperative opioid management in other hospitals around Australia.
There is a demonstrated gap in comprehensive opioid stewardship programs in many health services throughout Australia and New Zealand. 21 Reports of the situation in smaller health services, those in rural settings, in the private sector and for individual surgical specialty populations would add to our understanding. With the introduction of an electronic medical record at the study hospitals, the opportunity exists to include the postsurgical analgesia plan, including opioid tapering to cessation, in the electronic discharge summary. This transition has the potential to improve the low rate of hospital to GP communication in our study. Future directions include a comprehensive exploration of the factors promoting and hindering the implementation of the opioid stewardship program to provide context to the compliance reported in this study. Other areas that warrant investigation include the impact of prescriber education and guideline awareness on discharge opioid prescribing and the reasons for the low rates of PAC opioid education and opioid handling instruction recall.
Conclusion
The introduction of the opioid stewardship program resulted in a lower quantity of opioids dispensed than in our pre-stewardship study and in prescribing that was highly guideline compliant. Additionally, patients reported high rates of secure opioid storage. These are encouraging signals of improved opioid prescribing, use, and handling after introducing the opioid stewardship program. The rate of ongoing opioid use at three months in our study was consistent with the local and international literature. However, hospital to GP communication was uncommon and opioid disposal rates were low. These findings suggest that there remains potential to improve the post-discharge patient opioid handling to mitigate longer-term opioid-related harms. Future directions include exploring the health service factors impacting opioid stewardship program implementation and post-discharge opioid management. Opioid stewardship programs can optimise opioid prescribing, use and handling, but the attainment of these benefits relies on effective program implementation.
Footnotes
Author Contributions
Acknowledgements
The authors thank Gabriel Segal, Harriott Maixner, Jamie Dagher and Nurile Joffe (University of Melbourne medical students) for data collection and data entry and Christina Tourville, Sally Tsang, Daniel Travena and Austin Lee (Royal Melbourne Hospital) for data collection and data entry.
