Abstract
Custom 3D printed titanium implant pelvic reconstructive surgery was implemented as a novel technique at our institutions in the last five years. It provided an option for pelvic bone malignancy patients who were previously deemed unsuitable for re-implantation of irradiated resected bone segments, as well as in revision total hip arthroplasty associated with excessive acetabular bone loss. A retrospective cohort study of the anaesthetic management of patients who underwent pelvic reconstructive surgery using custom 3D printed titanium implants from August 2013 to July 2018 was conducted. Twenty-seven patients were included in the study; 23 patients completed single-stage procedures with a mean (standard deviation) duration of surgery of 7.5 (3.3) hours (median 6.8, range 3.0–15.8 hours), and mean intraoperative blood loss of 5400 (3100) mL (median 6000, range 1400–10,000 mL). Surgery involving the sacrum (n = 7) was associated with longer intensive care stay, longer total length of hospital stay and, in three cases, unplanned two-stage procedures. The twenty procedures not involving the sacrum were successfully completed in a single stage.
The major anaesthetic challenges included massive blood loss, prolonged surgery, interventions to prevent calf compartment syndrome, and perioperative thromboembolism. Preoperative pelvic radiotherapy, malignant tumours, and procedures involving the sacrum were associated with massive intraoperative blood loss and more prolonged surgery.
Introduction
Advances in 3D titanium printing technology have allowed the creation of a customised partial or full hemipelvis. By using data from computer tomography and magnetic resonance imaging, the customised pelvis is designed with specific anchor points for re-attaching the residual structures to achieve better functional outcomes. Since 2013, our institutions implemented custom 3D printed titanium implant pelvic reconstructive surgery as a novel technique for limb preservation treatment for pelvic bone malignancy patients. This novel technique provided an alternative option for patients who were previously deemed unsuitable for en bloc resection, extracorporeal irradiation and re-implantation of the resected bone segment 1 -another limb preservation treatment performed at our institution. Indications for this pelvic reconstructive surgery have since expanded to include patients requiring revision total hip arthroplasty associated with excessive acetabular bone loss and pelvic discontinuity.
The anatomical location of excision of the pelvic bone is described by the Enneking’s system: P1-ilium, P2- peri-acetabulum, P3-pubis, and P4- sacrum (Figure 1) 2 . Each anatomical region presents different surgical challenges given the intimate proximity of major neurovascular structures and the need to consider hip and/or spinopelvic stability. Additional total hip arthroplasty (THA) is required in tumours involving the peri-acetabulur structures (P2). In tumours involving the sacrum (P4) a colostomy and a suprapubic urinary catheter may be required because some sacral nerves are sacrificed during the resection.
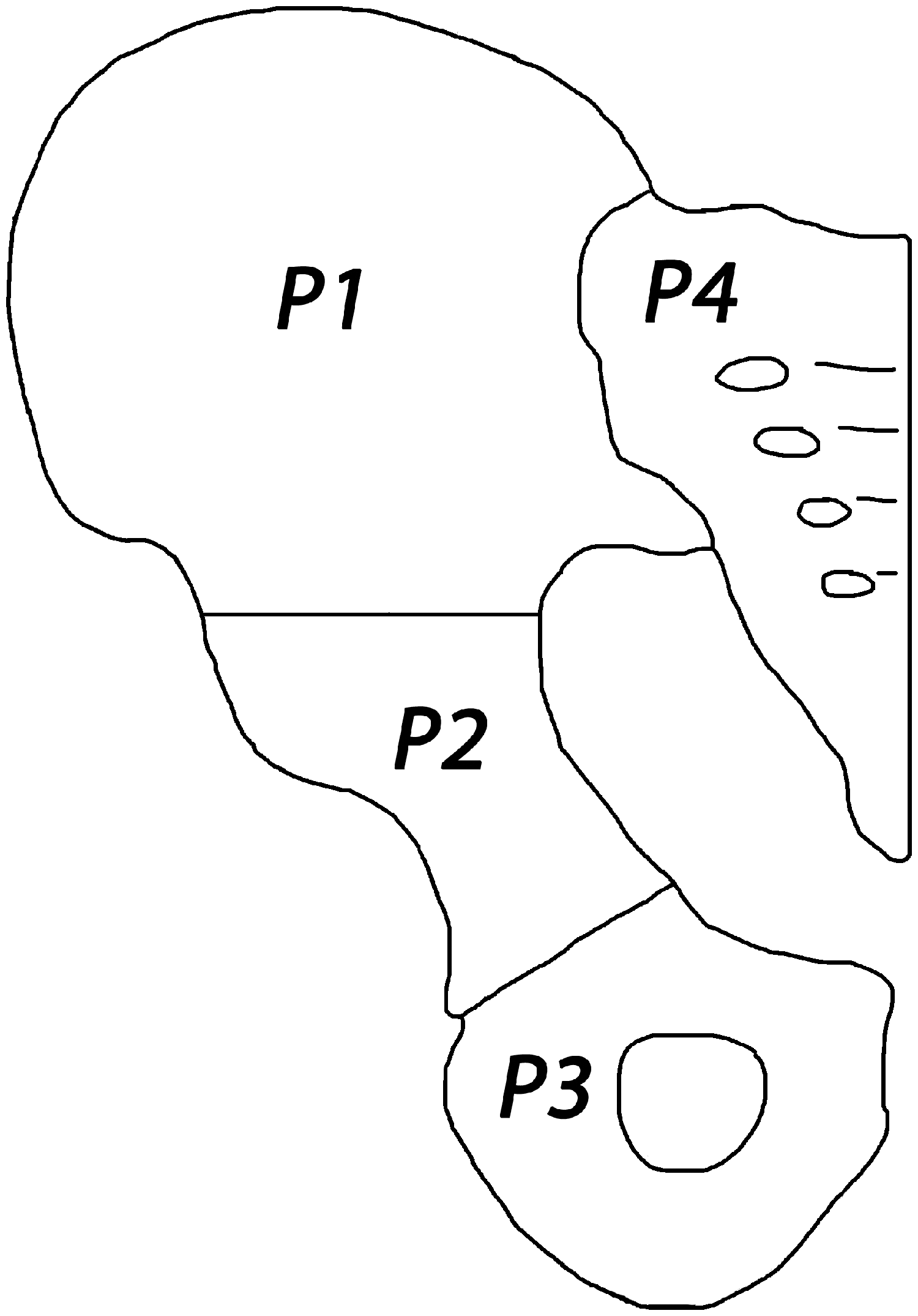
Enneking’s anatomical classification of pelvic resection: P1: the ilium; P2: the peri-acetabulum; P3: the pubis; and P4: the sacrum.
Although studies on the surgical aspects and short-term outcomes of custom 3D printed titanium implants have been published, the anaesthetic challenges involved have not been previously reported3–5.
The aims of this article are to report our experiences concerning anaesthetic management and outcomes of patients undergoing pelvic reconstruction with custom 3D printed titanium implant at two tertiary hospitals in Sydney.
Methods
The Royal Prince Alfred Hospital and the North Shore Private Hospital Human Research Ethics Committees approved the study (HREC number: LNR/17/RPAH/423, approved 22 August 2017). We conducted a retrospective cohort study of consecutive patients who underwent surgical reconstruction of large and complex pelvic bone defects using custom 3D-printed titanium implants (Ossis Ltd, Christchurch, New Zealand) by a single oncology orthopaedic surgeon working in two hospitals from 1 August 2013 to 30 July 2018. The Preferred Reporting Of CasE Series in Surgery (PROCESS) guidelines were followed. 6
Data collection
Perioperative data were retrieved from the anaesthetic and patient medical records. Perioperative complications were recorded using the Clavien–Dindo classification of surgical complications (Appendix 1). 7 Asymptomatic anaemia and allogenic blood transfusions were not considered a complication, as all patients received massive blood transfusion.
Anaesthetic management of specific issues during 3D reconstructive pelvic surgery
All patients received general anaesthesia with invasive monitoring (arterial blood pressure and central venous pressure), regular arterial blood gases and laboratory blood tests and measures to maintain normothermia.
Preoperative optimisation
Detailed planning to allow construction of the custom 3D printed titanium prosthesis used for pelvic reconstruction takes place approximately four to six weeks prior to surgery, thus permitting adequate time to optimise the patient. Surgery was delayed in patients who had preoperative neoadjuvant chemotherapy to enable recovery from bone marrow depression. Recovery from chemotherapy-related thrombocytopenia and anaemia takes 13 and 30 days, respectively. 8
General intraoperative issues
Access to patient from anaesthetist’s perspective
Depending on the site of the tumour and the extent of the procedure, patient positioning to provide adequate surgical exposure can limit patient access for the anaesthetist. Meticulous attention was undertaken to prevent pressure and electrical injuries. Peripheral nerves were also at risk of injury. In procedures involving the ilium, peri-acetabulum or pubis (P1, P2 or P3), the patient was placed in the supine position, and the contralateral arm was placed on an armboard at 90° to the trunk to enable access to the arm intraoperatively. When the Lloyd–Davies position was used for pelvic/sacral surgery (P4), access to the arms was not possible during the laparotomy because both the patient’s arms were placed beside the trunk. Consequently, additional intravenous cannulation in the neck (8.5 French, 16 cm long percutaneous sheath; Arrow International, Reading, PA, USA) was undertaken to facilitate intraoperative access.
Precautions against accidental displacement of intravascular access, monitoring devices and endotracheal tube
When repositioning the patient from the Lloyd–Davies position to the prone position to facilitate the posterior approach during sacral (P4) surgery, meticulous care of the vascular access, monitoring lines and endotracheal tube was undertaken. Fluid volume replacement was optimised prior to repositioning to avoid hypotension.
Prevention of deep venous thrombosis and lower limb compartment syndrome
Sequential calf compression and elastic compression stockings were used in all except one (recent history of deep venous thrombosis (DVT)) patient. In addition to the maintenance of perfusion pressure (with vasopressors), boot-styled stirrups (Allen Medical, Acton, MA, USA) were used in patients who were placed in the Lloyd–Davies position during the anterior approach for sacral (P4) surgery. We did not administer any preoperative anticoagulants because of anticipated massive intraoperative blood loss. Low molecular weight heparin was only commenced in the postoperative period when the patient’s coagulation profile was satisfactory.
Intraoperative blood transfusion strategy
Rapid infusion devices and maintenance of normothermia were employed in anticipation of massive blood transfusion. Intraoperatively, we maintained our target haematocrit (>30%) and coagulation parameters (platelet count >100 × 109/L, International Normalized Ratio <1.5, fibrinogen >1.5 g/L and ionised calcium >1.1 mmol/L) and normothermia. Administration of blood products was guided by frequent arterial blood gas analysis, laboratory measurements of blood count and coagulation and occasionally thrombelastography. To minimise intraoperative massive blood loss, preoperative interventional radiological embolisation was performed in three patients with pelvic metastatic renal cell carcinoma (one patient underwent embolisation twice). Tranexamic acid was administered based on the experience of its use in total joint arthroplasty. 9 Intraoperative cell salvage (IOCS) was used only in non-malignancy patients.
Postoperative pain management
Perioperative multimodal analgesia utilised various combinations of gabapentin, ketamine, lidocaine, buprenorphine patch, intrathecal morphine, peripheral nerve block and rectus sheath catheters as part of an opioid-sparing strategy.
Results
Demographic data
Twenty-seven patients (16 (59%) males, 11 (41%) females; mean (standard deviation) age 47.8 (17.6) years, median 53 years, range 20–76 years) underwent surgical pelvic reconstruction using custom 3D printed titanium implants (1 August 2013 to 30 July 2018). The mean (SD) body mass index was 25.7(6.2) kg/m2 (median 25.4 kg/m2, range 17.0–40.5 kg/m2). The majority of patients (81%) were American Society of Anesthesiologists (ASA) physical status 3. The indications for surgery were classified into four groups: malignant tumour (n=14), benign tumour (n=4), failure of THA (n=5) and other (n=4) (Table 1). Preoperative pain was the most common symptom: 14 (52%) patients were on opioids and 10 (37%) patients on simple analgesics, with three (11%) patients requiring no analgesics.
Indications for surgery and use of preoperative chemotherapy, radiotherapy and/or embolisation in all patients (n=27).
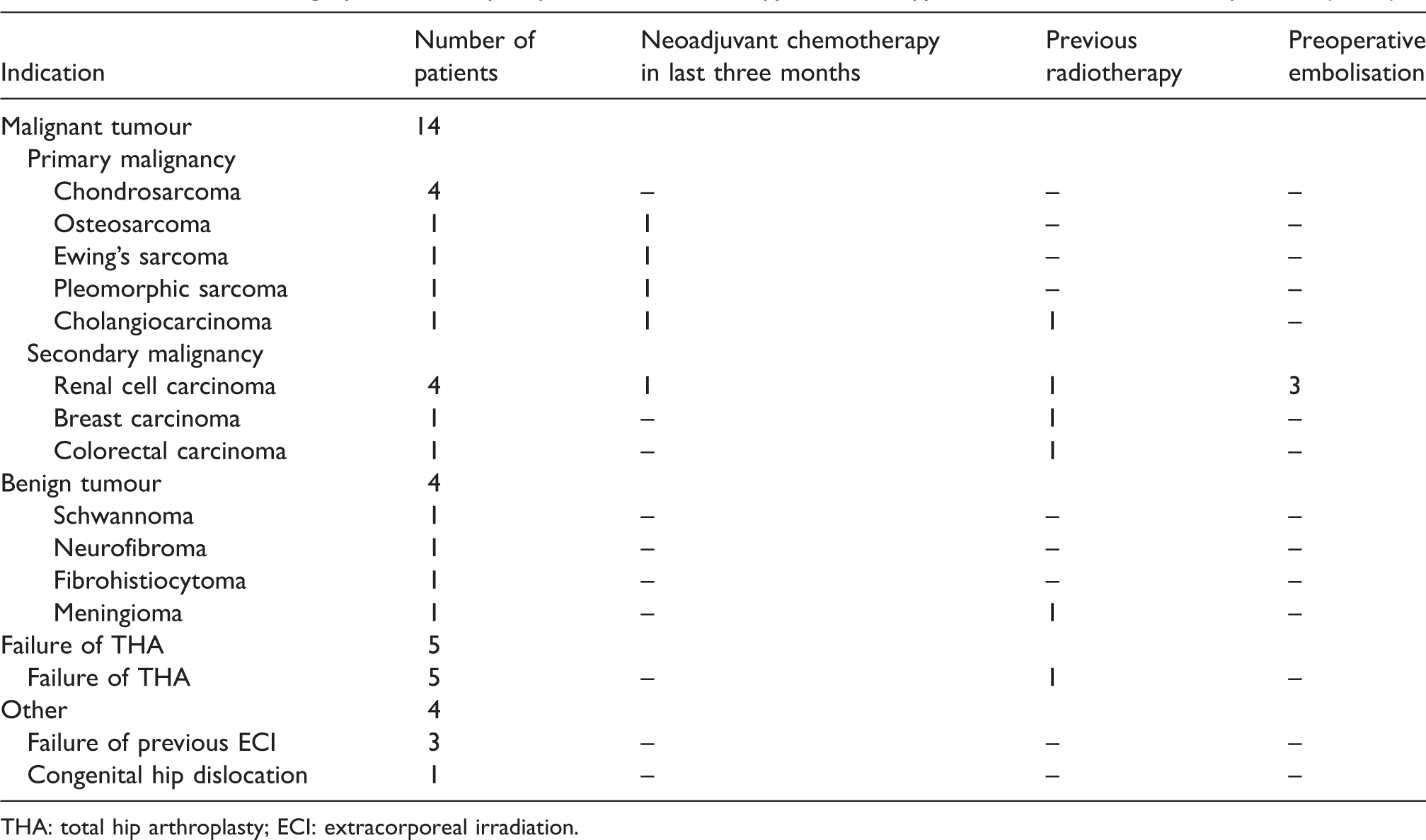
THA: total hip arthroplasty; ECI: extracorporeal irradiation.
A total of 30 procedures were performed on 27 patients because three patients required an unplanned two-stage procedure. Successful completion of custom 3D implant reconstructive surgery was achieved in 25 patients (23 patients: one-stage procedure; two patients: two-stage procedure). The procedures were abandoned in two patients because of technical difficulties, resulting in excessive life-threatening blood loss (one abandoned during a single procedure; another abandoned during the two-stage procedure) (Figure 2). These two abandoned procedures involved the sacrum (P4) and were excluded from the study.
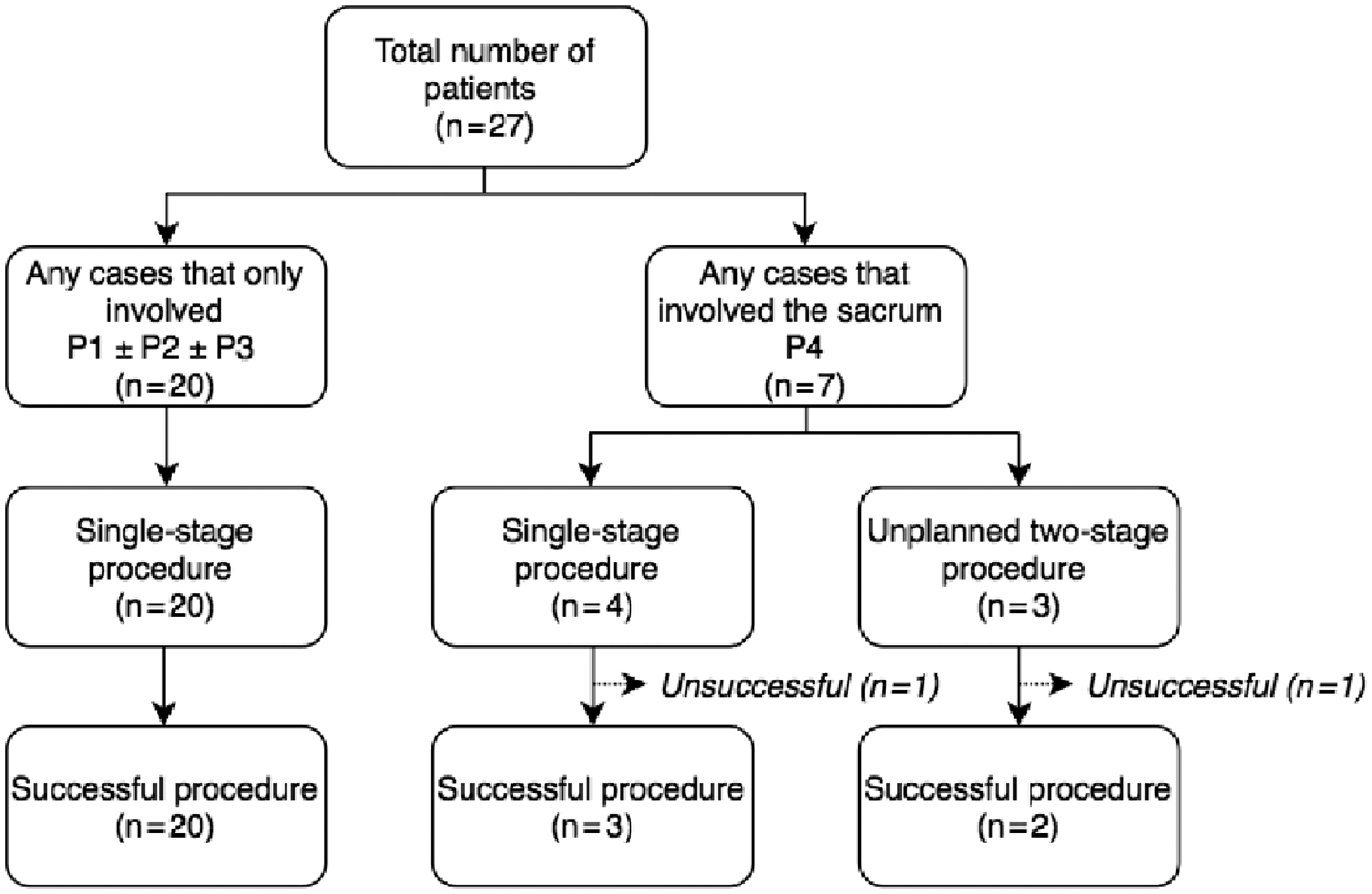
Flow chart for study participants.
Intraoperatively data of patients with completed procedures (n=25)
Tables 2 and 3 summarise the intraoperative details of the patients who had their surgical procedures completed.
Intraoperative details of patients who had a successful single-stage procedure (n=23).
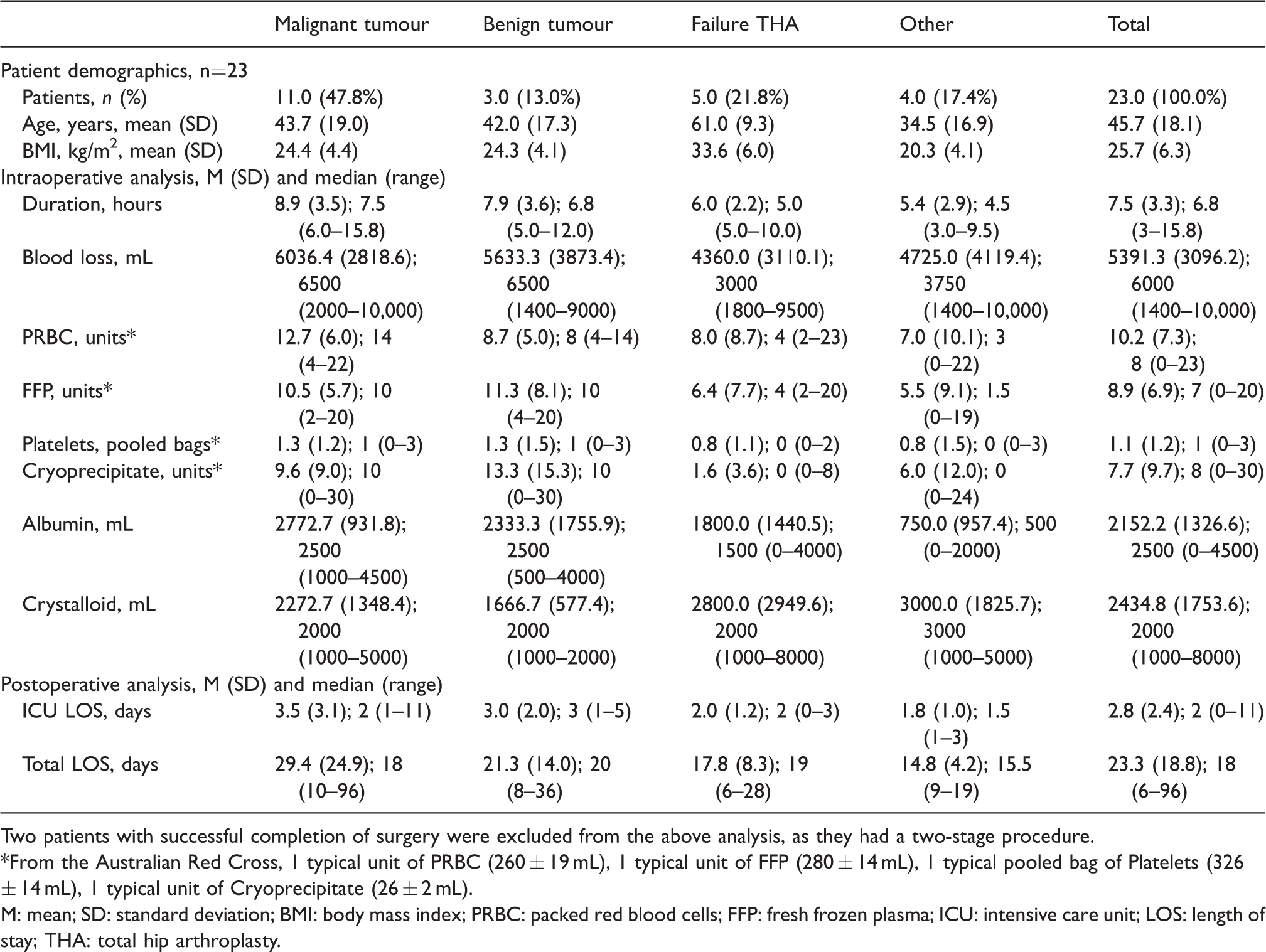
Two patients with successful completion of surgery were excluded from the above analysis, as they had a two-stage procedure. *From the Australian Red Cross, 1 typical unit of PRBC (260 ± 19 mL), 1 typical unit of FFP (280 ± 14 mL), 1 typical pooled bag of Platelets (326 ± 14 mL), 1 typical unit of Cryoprecipitate (26 ± 2 mL).
M: mean; SD: standard deviation; BMI: body mass index; PRBC: packed red blood cells; FFP: fresh frozen plasma; ICU: intensive care unit; LOS: length of stay; THA: total hip arthroplasty.
Intraoperative details of successful operations (n=25) for procedures involving Enneking’s classification P1/2/3 without P4 versus any case with P4 involvement.
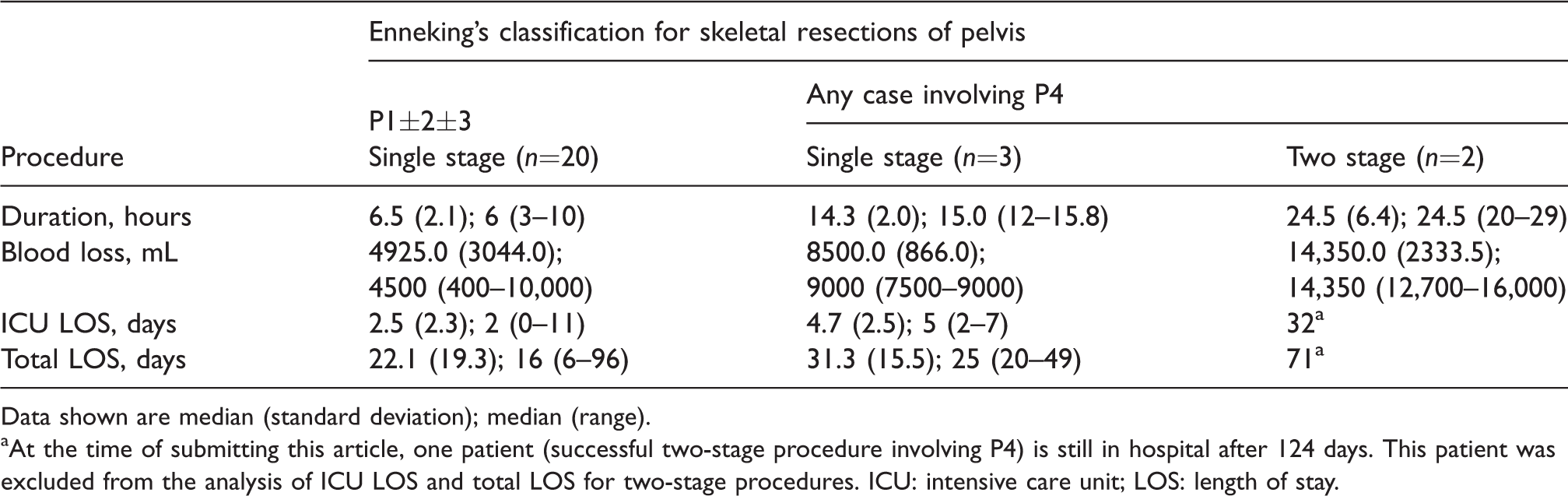
Data shown are median (standard deviation); median (range).
At the time of submitting this article, one patient (successful two-stage procedure involving P4) is still in hospital after 124 days. This patient was excluded from the analysis of ICU LOS and total LOS for two-stage procedures. ICU: intensive care unit; LOS: length of stay.
Intraoperative positioning
Patient positioning during surgery was dictated by the site of the pelvic pathology which influenced surgical exposure. In procedures that involved only the ilium, peri-acetabulum and/or pubis (Enneking’s classification P1, P2 and P3, respectively), the patients (n=20) were placed in the supine position with a pelvic tilt (provided by a sandbag placed under the buttock on the operative side). In the procedures that involved the sacrum (Enneking’s classification P4), patients (n=5) were placed first in the Lloyd–Davies position for the anterior laparotomy, and were then moved to the prone position for the posterior approach to complete pelvic/sacral resection and reconstruction.
Duration of surgery
The mean (SD) duration of single-stage procedures was 7.5 (3.3) hours (median 6.8 hours, range 3.0–15.8 hours). Procedures involving malignant tumours were associated with a prolonged duration (8.9 (3.5) hours; median 7.5 hours, range 6.0–15.8 hours Table 2). Two patients required unplanned two-stage procedures because of surgical difficulties resulting in excessive bleeding and prolonged operating time (a total of 20 and 29 hours for the two stages).
Blood transfusion and issues
Observed factors that appeared to be associated with intraoperative massive blood loss included (a) site of lesion, (b) underlying pathology and (c) preoperative pelvic radiotherapy. Resection of sacral tumours (P4) was associated with higher blood loss in single-stage procedures (Mean blood loss 8500 mL versus 4925 mL; Table 3). Massive blood loss (6036 (2818)mL, median 6500 mL, range 2000–10,000 mL; Table 2) occurred in procedures for malignant tumours. Preoperative radiotherapy also appeared to be associated with a higher blood loss (8125 mL versus 4816 mL; Table 4). Intraoperative volume replacement therapy is summarised in Table 2. Tranexamic acid was administered to 18 (72%) patients. IOCS was used in four patients.
Intraoperative details of successful single-stage procedures (n=23) for procedures that involved previous radiotherapy versus no previous radiotherapy.

Data shown are mean (standard deviation); median (range).
Two patients with successful completion of surgery were excluded from the above analysis, as they had a two-stage procedure.
Acute hypersensitivity reactions
Intraoperative acute hypersensitivity reactions occurred in three patients. One patient required resuscitation with adrenaline infusion, and surgery was suspended for 90 minutes. The operation was completed without further problems after haemodynamic stabilisation. The second patient developed severe intraoperative hypotension (successfully treated with ephedrine and metaraminol) and urticaria. Urticaria detected at the end of surgery with no intraoperative haemodynamic disturbances in the third patient was treated with intravenous hydrocortisone. As no allergies to the drugs administered were found during the investigations of the three patients at the Anaesthetic Allergy Clinic, these reactions were attributed to ‘plasma proteins’ because large volumes of fresh frozen plasma were administered intraoperatively.
Intraoperative analgesia
Fourteen (56%) patients received gabapentin preoperatively. Intrathecal morphine was administered to 10 (40%) patients, and peripheral nerve blocks were performed in four (16%) patients. Other intraoperative analgesics used included ketamine infusions (n=21 (84%) patients), lidocaine infusions (n=6 (24%) patients), transdermal buprenorphine (n=4 (16%) patients), tramadol (n=6 (24%) patients) and tapentadol (n=1 (4%) patient). Bilateral rectus sheath catheters were placed in all patients (n=5) who had laparotomy for sacral (P4) resection, for local anaesthetic infusions (ON-Q® PainBuster®, B. Braun Melsungen AG, Hessen, Germany) over three days postoperatively.
Postoperative aspects of patients who had procedures completed (n=25)
Length of stay in intensive care unit and hospital
All patients except one were admitted to the intensive care unit (ICU) immediately following surgery for postoperative management. Thirteen (52%) patients required elective postoperative ventilation. The mean length of stay (LOS) in the ICU was 2.8 (2.4) days (median 2 days, range 0–11 days) and total hospital LOS was 23.3 (18.8) days (median 18 days, range 6–96 days) in the patients who had successful single-stage procedures. Surgery for malignant tumours was associated with a longer ICU LOS (mean 3.5 (3.1) days, median 2 days, range 1–11 days) and hospital LOS (mean 29.4 (24.9) days, median 18 days, range 10–96 days; Table 2). Procedures involving the sacrum (P4) were associated with longer ICU LOS (4.7 (2.5) days, median 5 days, range 2–7 days versus 2.5 (2.3) days, median 2 days, range 0–11 days) as well as hospital LOS (31.3 (15.5) days, median 25 days, range 20–49 days versus 22.1 (19.3) days, median 16 days, range 6–96 days; Table 3).
DVT and thromboprophylaxis
Preoperatively, six patients had a past history and one patient had a recent history of DVT. Elastic compression stockings and sequential calf compression were employed in all the patients except the patient with a recent history of DVT. There was no intraoperative pulmonary embolism. DVT occurred in one patient (no past history, Enneking’s classification P4, and two-stage procedure) post surgery.
Other complications
Relevant anaesthetic complications included intraoperative acute hypersensitivity reactions related to massive blood product transfusion in three patients. Overall, a total of 38 surgical complications occurred in 25 patients (Table 5). Neuropraxia associated with the surgical resection occurred in five patients (four patients with malignant tumours, and one patient who had a failed previous extracorporeal irradiated implant reconstruction). There was one death as a result of intra-abdominal sepsis and multi-organ failure after a 96-day hospital stay, including more than two weeks in the ICU.
Perioperative complications classified according to Clavien–Dindo classification.

Surgical classifications were classified using the Clavien–Dindo classification, which was based on the type of therapy necessary to treat the complication. 7
CT: computed tomography; IV: intravenous.
Discussion
The major anaesthetic challenges in reconstructive pelvic surgery using custom 3D printed implants included massive blood loss, prolonged surgery, prevention of calf compartment syndrome, and perioperative thromboembolism. Preoperative pelvic radiotherapy, malignant tumours and procedures involving the sacrum appeared to be associated with more massive intraoperative blood loss and prolonged surgery. Preoperative pelvic irradiation was associated with surgical difficulties resulting from radiation-induced fibrosis. Renal cell carcinoma metastases are highly vascular and a risk factor for increased blood loss.10,11
There is a high risk of DVT (e.g. secondary to prolonged surgery, pelvic surgery and/or malignancy) in this cohort of patients. Lower limb compartment syndrome is associated with the lithotomy and steep Trendelenburg position as a result of diminished lower limb perfusion in prolonged surgery (i.e. more than four hours).12,13 In addition to the maintenance of perfusion pressure (e.g. administration of vasopressors), boot-styled stirrups which exert pressure over the heels rather than the calves were essential in minimising this serious complication in patients placed in the Lloyd–Davies position. Although the use of intermittent pneumatic compression devices is considered a risk factor for calf compartment syndrome, both sequential calf compression and elastic compression stockings were employed, as the risk of DVT (incidence of 1.8%, 10% and 11.6% in pelvic uro-oncological surgery, pelvic and acetabular trauma surgery and gynaecological surgery, respectively) is greater than that of calf compartment syndrome (incidence of 0.029% in the Lloyd–Davies position).13–17
Intraoperative pulmonary tumour embolism during hemipelvectomy for pelvic chondrosarcoma, a rare but catastrophic complication, has been reported. 18 Intra-operative transoesophageal echocardiography could be considered for the detection of tumour emboli.
In addition to preoperative radiotherapy and renal cell carcinoma metastases as risk factors associated with massive blood transfusion, the total blood loss is correlated with the operative time in musculoskeletal tumour surgery. 19 The use of rapid infusion devices and avoidance of hypothermia are essential. The use of IOCS should be considered in anticipation of massive blood transfusion. Intraoperatively a ‘liberal’ (trigger for transfusion: haematocrit ≤30%) rather than ‘restrictive’ (haematocrit ≤24%) transfusion strategy was adopted, as it has been suggested to improve perioperative survival in adult patients. 20 The aim was to maintain critical coagulation profiles (platelet >80 × 109/L, International Normalized Ratio <1.8 and fibrinogen >1.0 g/L) intraoperatively to ensure normal haemostasis. 21
Surgical temporary occlusion of the aorta using an intravascular balloon has been reported by Liang et al. in their experience of 3D printed titanium pelvic reconstruction surgery for malignant bone defects. 5 Their prostheses were modular, with each component having three different sizes. Their mean surgical duration was 4.3 (0.6) hours, with their mean balloon occlusion duration 1.8 (0.6) hours. An intra-aortic balloon was not incorporated into our practice for several reasons. First, insertion of a balloon in the hybrid/radiological suite introduced logistic issues. Second, our mean surgical duration of single-stage procedures was longer at 7.5 (3.3) hours, which might render the intra-aortic balloon less effective. The long duration of surgery was in part related to the careful and precise resection of the pelvic bone required to fit the custom-made single-component prosthesis. Third, the surgery could be suspended to reassess the clinical conditions, and if necessary, the surgery could be abandoned during the period of soft tissue resection. The use of an intravascular balloon could mask the potential for uncontrolled bleeding in patients with inoperable pathology, resulting in intraoperative mortality. In this study, two patients were considered ‘inoperable’ during the procedure because of life-threatening massive intraoperative blood loss.
There is debate regarding the safety of IOCS for autologous blood transfusion in surgery for malignant tumours. It was used in four non-malignant patients in this study. Although IOCS is traditionally not advocated in cancer surgery because of the risk of disseminating cancer cells, it has been advocated in some gynaecology, urology, gastrointestinal and hepatobiliary oncological surgery.22–27 A brief review of the literature suggested that there may a place for IOCS in cancer surgery. Catling et al. reported that no viable tumour cells could be detected after IOCS and a leucocyte depletion filter (LDF) in 50 consecutive gynaecological oncology patients. In that study, blood collected after IOCS was used only for analysis and was not reinfused back to the patient. 28 This observation is supported by another ‘non-reinfusion’ study by Kumar et al. who could not detect any viable malignant cells after IOCS and LDF in 50 patients undergoing metastatic spine tumour surgery. 29 Utilising flow cytometry studies in 11 patients, Kumar et al. concluded that IOCS and LDF in combination were effective in removing and significantly reduced the number of tumour cells in eight patients. 30 These findings are consistent with other studies by Ashworth and Waters who concluded that the use of IOCS was not contraindicated in cancer surgery and reinfusion of IOCS-LDF treated blood was safe in metastatic cancer surgery.31,32 A systematic review evaluating IOCS and LDF in cancer surgery by Kumar et al. found that the clinical outcomes were equal or better in patients who received IOCS compared to those who did not. 33 However, no randomised controlled trials are available to address this question.
Various combinations of gabapentin, ketamine, lidocaine, buprenorphine patch, intrathecal morphine, peripheral nerve block and rectus sheath catheters were used as part of an opioid-sparing strategy.34–39 We avoided sympathetic block with spinal local anaesthetic agent, as it could worsen hypotension during major blood loss. We also avoided using epidural catheters because of the risk of perioperative coagulopathy.
Despite successful pelvic reconstruction with custom 3D printed titanium implants, two patients in this series returned for hindquarter amputation within 12 months of their initial surgery: one as a result of infection, and another as a result of recurrent pelvic chondrosarcoma. Although the initial pelvic resections did not involve the sacrum, their implants were surgically fixed onto the sacrum to provide pelvic stability. As a result of this and post-surgical scarring, subsequent hindquarter amputation in these two patients was technically difficult and was associated with massive intraoperative blood loss of >13,000 mL. In our experience, the average intraoperative blood loss for a hindquarter amputation without previous custom 3D printed titanium implant surgery was around 2000–4000 mL. Anaesthetists involved for such subsequent procedures should anticipate massive blood loss.
In conclusion, in this retrospective cohort study of our early experience involving 27 patients undergoing pelvic reconstruction with custom 3D printed titanium implants, 25 patients had successful completion of surgery, and only two patients had abandoned operations. All procedures were associated with massive blood loss, prolonged surgery and prolonged hospital LOS. Previous pelvic radiotherapy, malignant tumours and surgery involving the sacrum appeared to be associated with a higher intraoperative blood loss and longer duration of surgery.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Clavien–Dindo classification of surgical complications.
7
Includes anti-emetics, antipyretics, analgesics, diuretics, electrolytes and physiotherapy Includes antibiotics, transfusions or total parenteral nutrition Did not include blood transfusions IIIa: operation not under general anaesthesia IIIb: operation under general anaesthesia IVa: single-organ dysfunction IVb: multi-organ dysfunction
Grade
Clavien–Dindo classification of surgical complications
I
Any deviation from the normal postoperative course without the need for pharmacological treatment or operative treatment (surgical, endoscopic, radiological)
Examples in our study
Adverse drug reaction/drug allergy, bladder retention, pancreatitis, chyle leak, delirium/postop confusion, electrolyte imbalance, ileus (conservative management), neuropraxia, pressure injury
II
Requiring pharmacological treatment (drugs other than allowed in Grade I)
Examples in our study
Acute pulmonary oedema, additional antibiotic treatment (urinary tract infection, respiratory infection), deep surgical site infection, deep vein thrombosis, anaphylaxis
III
Requiring operative treatment (surgical, endoscopic, radiological)
Examples in our study
Abdominal sepsis requiring computed tomography–guided drainage, hardware issues, hip dislocation requiring open reduction, peri-prosthetic fracture, pseudo-aneurysm requiring radiological embolisation, wound necrosis requiring surgical debridement
IV
Life-threatening complication (including central nervous system, excludes transient ischaemic attack) requiring intensive care unit/high dependency unit (unplanned)
Examples in our study
Intracranial haemorrhage requiring bilateral burrholes, cerebrospinal fluid leak requiring retie of thecal sac
V
Mortality (death)
