Abstract
Background:
Finite element modelling has long been proposed to support prosthetic socket design. However, there is minimal detail in the literature to inform practice in developing and interpreting these complex, highly nonlinear models.
Objectives:
To identify best practice recommendations for finite element modelling of lower limb prosthetics, considering key modelling approaches and inputs.
Study design:
Computational modelling.
Methods:
This study developed a parametric finite element model using magnetic resonance imaging data from a person with transtibial amputation. Comparative analyses were performed considering socket loading methods, socket–residuum interface parameters and soft tissue material models from the literature, to quantify their effect on the residuum’s biomechanical response to a range of parameterised socket designs.
Results:
These variables had a marked impact on the finite element model’s predictions for limb–socket interface pressure and soft tissue shear distribution.
Conclusions:
All modelling decisions should be justified biomechanically and clinically. In order to represent the prosthetic loading scenario in silico, researchers should (1) consider the effects of donning and interface friction to capture the generated soft tissue shear stresses, (2) use representative stiffness hyperelastic material models for soft tissues when using strain to predict injury and (3) interrogate models comparatively, against a clinically-used control.
Introduction
The residual limb (or ‘residuum’) for a person with limb loss represents a constantly evolving interface for mechanical loading from a prosthesis, most commonly through a personalised socket. The residuum shape changes with oedema, muscle atrophy and stiffening of loaded tissue regions. 1 Anatomic and surgical considerations include bone shaping through bevelling, soft tissue coverage and retention of sensory and vascular structures. 2 Currently, prosthetic limb users often require multiple fittings to achieve an acceptable definitive socket and still report discomfort, which may limit their rehabilitation progress. 3 The need for advanced technology to assist socket design and fitting is established and recent proposals include sensors, adjustable sockets and numerical simulations, 4 but there has been limited successful translation from lab to clinic.
The finite element analysis (FEA) simulation technique is widely used in engineering, for example, to determine the distribution of structural deformations or stress. Very particular requirements arise in the robustness of finite element (FE) models when used to inform clinical practice and in extracting relevant data, due to the complexity of the biomechanical processes being modelled, patient variability across the population and appropriate parameter selection.5,6
The potential of FE to predict residuum–socket interactions is well established. A review of literature from 2000 to 2016 7 discussed how clinical implementation would enable support for prosthetists in a more evidence-based socket design process, while identifying opportunities for improving the state-of-the-art in simulation through applying dynamic loading, advances in imaging and measurements for model validation. Notable studies considered interface parameters, 8 material models,9,10 residuum morphology 10 and explicitly modelling socket donning. 11 One limitation to progress is the lack of published guidance about the effect of model construction decisions and input parameters. To develop such guidance, comparative analyses are required. 12 Clear reporting and transparency in the modeller’s decision-making process are also important, so that engineers may replicate them, and clinicians can appraise the limitations inherent due to simplifications in representing this complex, highly nonlinear system. Specifically, within residuum–socket models, there is a substantial variation in how donning is modelled and different soft tissue properties are selected, yet the effect of these decisions is unknown.
This article aimed to develop recommendations for best practice in FE modelling of lower limb prostheses, considering key modelling inputs and their biomechanical effects. The study employed a parameterised patient-specific FE model of the transtibial amputated (TTA) residuum and prosthetic socket to examine the interface pressure and tissue strain predicted using different loading methods, soft tissues stiffness and interface friction values.
Methods
An FE model was generated from magnetic resonance imaging (MRI) scans (MAGNETOM Spectra, Siemens Healthcare GmbH, Germany; 3.0-mm slice thickness, 0.5-mm in-slice resolution, T1-weighted, six-channel surface coil and 24-channel spine coil for parallel imaging, participant supine wearing a liner (6Y510, OttoBock, Duderstadt, Germany) and unrectified check-socket to minimise tissue deformation on the scanner bed). Access to imaging was obtained through ethics approval of secondary data analysis (ERGO#29927) of a patient with unilateral TTA (male prosthesis user, 65 years, 80 kg, 170 cm, peripheral arterial disease, 10-years post-amputation, with fleshy, cylindrical residual limb) who provided written, informed consent.
The MRI scan was segmented into liner, bones, patellar- and quadriceps tendons and intra-articular tissue (ScanIP, Synopsys, Inc., Mountain View, CA, USA). The remaining soft tissues were homogenised into a single body. The patella tendon was modelled as distinct from surrounding tissues due to its importance as a load-tolerant structure and the stability it lends to the limb when under tension. 8 The quadriceps tendon provides a boundary condition preventing passive knee flexion, and the intra-articular tissue facilitates numerically stable load transfer in the residuum.
The segmentation masks were meshed in ScanIP with quadratic tetrahedral elements into a single continuous structure and imported into ABAQUS 6.14 FEA software (Dassault Systèmes, Vélizy-Villacoublay, France). The liner was imported as a separate body and meshed with hexahedral elements in ABAQUS (Figure 1(a)). A simplified total surface bearing (TSB) parametric socket was developed by copying and modifying the liner’s external shape (Figure 1(a), and meshed with quadrilateral shell elements. Four sockets were designed with press-fits (cross-sectional area reduction) about the principal axis of the tibia, to rectify less into the stiffer anterior surface than the fleshy posterior surface: −2% (over-sized), 0% (matched), 2% (low press-fit) and 4% (high press-fit).
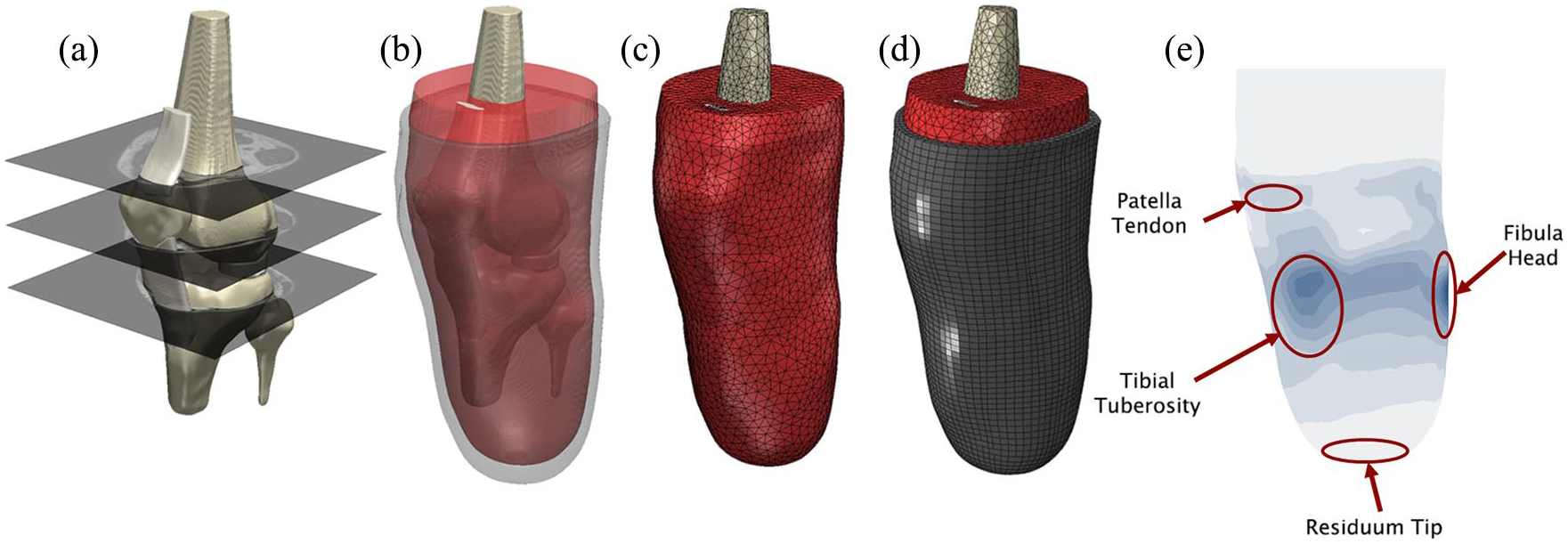
Development of the finite element (FEA) model from (a) the magnetic resonance imaging (MRI) scan, involves (b) the segmentation of bones, soft tissue and liner, (c) generation of the quadratic tetrahedral mesh of the limb and (d) hexahedral mesh of the liner. (e) Example pressure output of FE model with key analysis zones labelled.
Materials
Five soft tissue models were obtained from various literature sources,13–15 in addition to two intermediate values, and linear- and hyperelastic equivalents were calculated for each (Table 1, Appendix 1). Average flaccid muscle was selected as the baseline. The material properties of the other structures were obtained from the literature16–20 (Table 1).
Material properties of finite element (FEA) model structures.
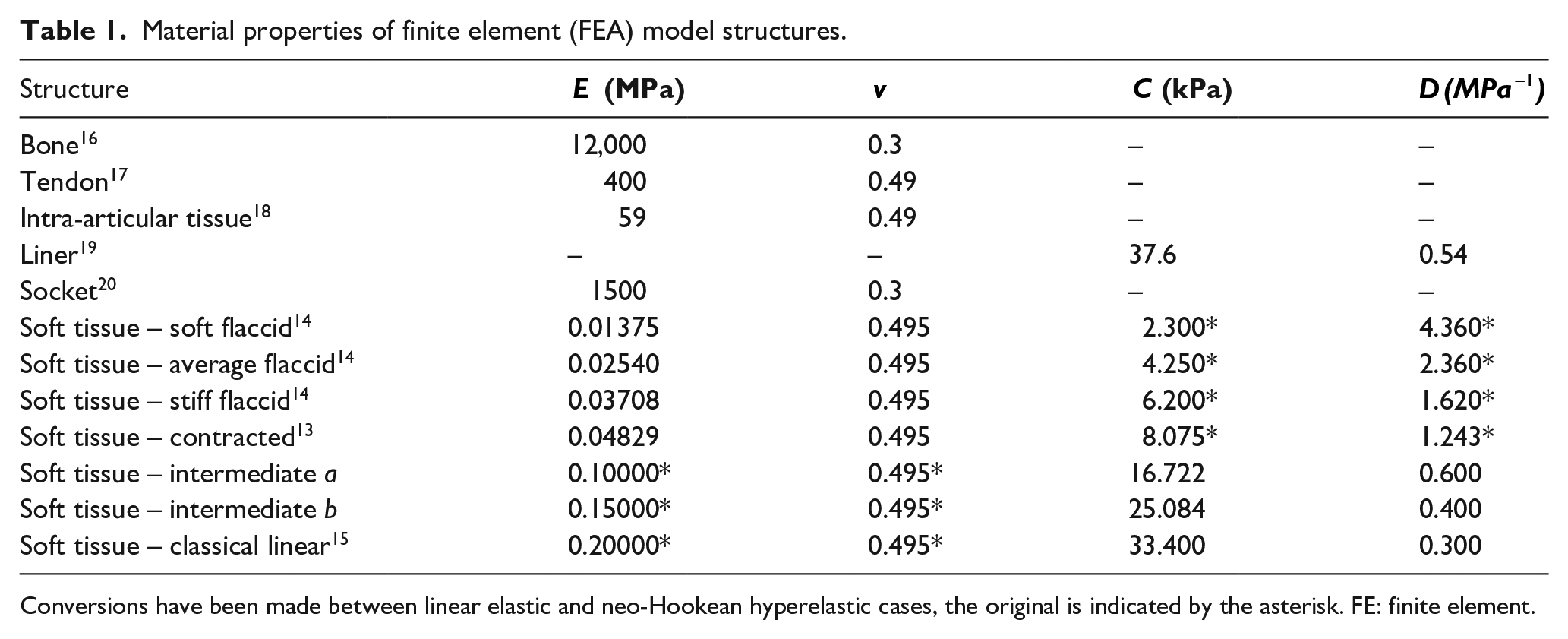
Conversions have been made between linear elastic and neo-Hookean hyperelastic cases, the original is indicated by the asterisk. FE: finite element.
Socket loading
Socket donning applies pre-stresses to the residuum tissues. Simulating this process presents a complex, nonlinear problem to reconcile the difference in shape between the limb and the press-fit socket. For modelling simplicity and computational stability, most studies have used a simplified ‘overclosure’ method where the socket is fixed in an assumed position and residuum’s external surface is displaced until it contacts the internal surface of the socket.
20
Alternatively, explicit donning can be simulated. The socket is moved into small initial contact with the limb, a small overclosure step is solved to give numerical stability, followed by sliding the socket onto the limb and calculating the generated interface stresses and press-fit strains.
11
For this study, the methods were compared (Figure 2).Following calculation of the limb-liner-socket press-fit, the socket was loaded. Load cases used were a uniaxial 400-N load case representing double leg stance, and three quasi-static load points from gait, defined from six-axis load cell data
21
, corresponding to heel-strike, mid-stance and toe-off (Table 2). A coordinate system was constructed from the residual tibia’s principal axes. Only forces and moments in the sagittal plane which are the dominant forces during gait, were modelled
8
. The
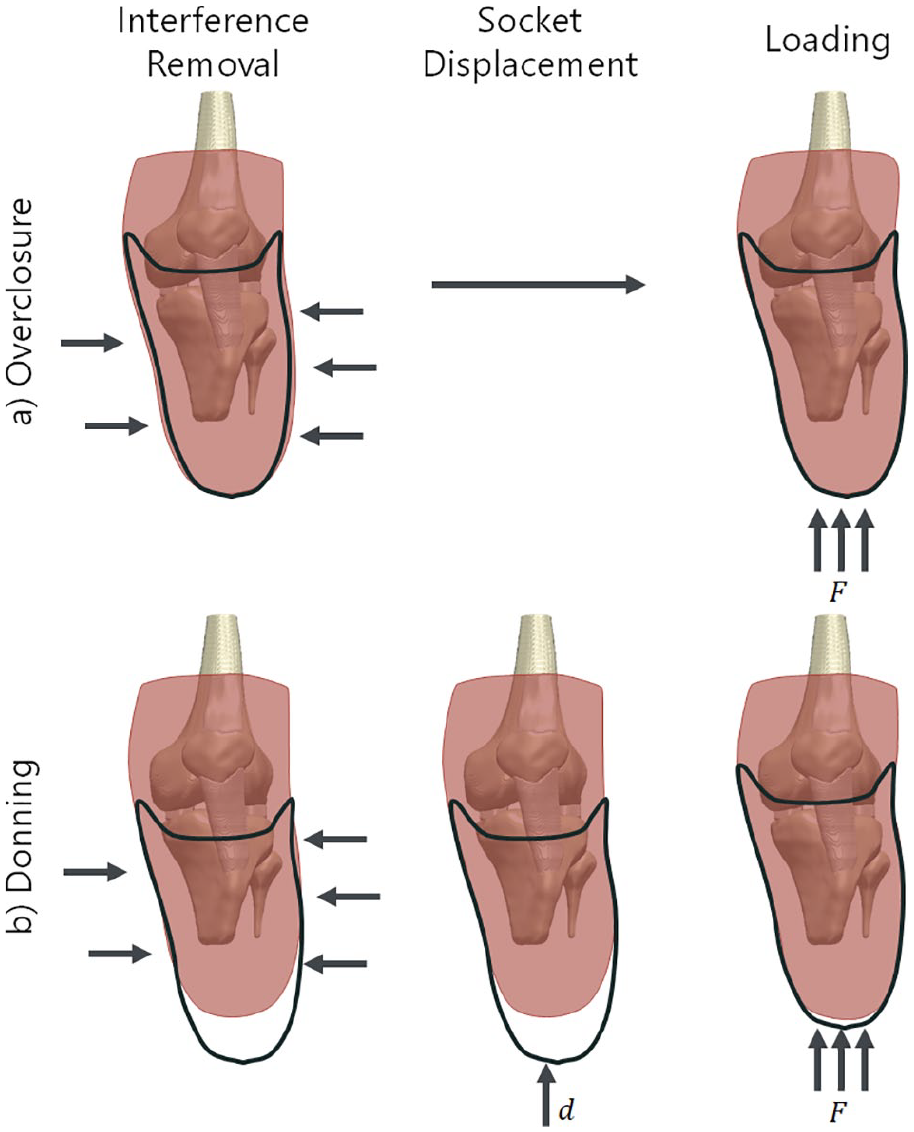
Comparison of the two socket donning methods. (a) The overclosure method removes radial interference (lateral and medial arrows indicate the displacement of the limb) and then loads the socket, while (b) the explicit donning method uses a small overclosure step for stability and then slides the socket onto the residuum and liner.
Applied load cases, 21 scaled to this study participant’s 80 kg bodyweight.

Interface properties
The large pressure gradients and frictional sliding at the liner–socket interface require sophisticated automated contact analysis within the FE solver. 22 Experimental studies have aimed to quantify the interface properties between these bodies, specifically the static coefficient of friction (COF), in order to predict the shear stress generated under a given pressure. 23 Cagle et al. 8 studied the effect of varying the COF at the liner–skin interface between 0.5 and fully bonded, but did not investigate the socket–liner COF, keeping it constant at 0.5. In this study, a baseline value of COF = 0.5 was chosen and varied in increments of 0.1 between 0.3 and 0.7 under double-leg stance loading. The residuum–liner surface was fully bonded, representing a sticky gel liner.
Results parameters
The model was interrogated by reading the 95th percentile pressure and shear stresses on the liner surface in locations associated with socket fit and comfort: 24 30-mm circular zones around the fibular head and tibial tuberosity, a 40-mm circular zone at the residuum distal tip and a 20 × 10 mm elliptical zone at the patella tendon. The 95th percentile minimum principal (compressive) strain in a 20-mm spherical volume of interior soft tissue near the residual tibia’s distal tip was used, as strain is the most accepted indicator of deep tissue injury risk. 25 These results were compared between models. These output parameters were used to inform a mesh convergence analysis using mesh sizes between 2.0 and 12.0 mm (Supplementary Material). Mesh convergence of less than 3 kPa pressure and 1% strain was achieved with the 5.0 mm element edge length or 2.5 mm node-node distance, giving 71.5k elements, and this was selected as an appropriate trade-off between accuracy and computational resources. 26
Results
Except where otherwise stated, the results are presented for baseline models using average flaccid soft tissue stiffness, liner–socket COF = 0.5, 2% press-fit socket, explicitly donned and two-leg stance loading.
Effect of socket donning and COF on interface pressure and shear
Substantial differences in pressure and shear were observed between the two socket press-fitting methods, notably at the residuum tip (Figure 3(a)). Inclusion of socket donning increased proximal interface shear (22 kPa in donning vs. 4 kPa in overclosure), whereas the residuum tip pressure was predicted to decrease (0 kPa in donning vs. 41 kPa in overclosure). This effect of increased longitudinal shear on the residuum tip pressure was observed with an increase in the socket–liner COF for the same baseline model (Figure 3(b)). Comparing between COF values of 0.3 and 0.7 for the low press-fit 2% socket, the peripheral shear was predicted to increase from 12 to 21 kPa accompanied by a subsequent drop in residuum tip pressure from 14 to 0 kPa, indicating that the residuum tip did not reach the socket after donning and loading.
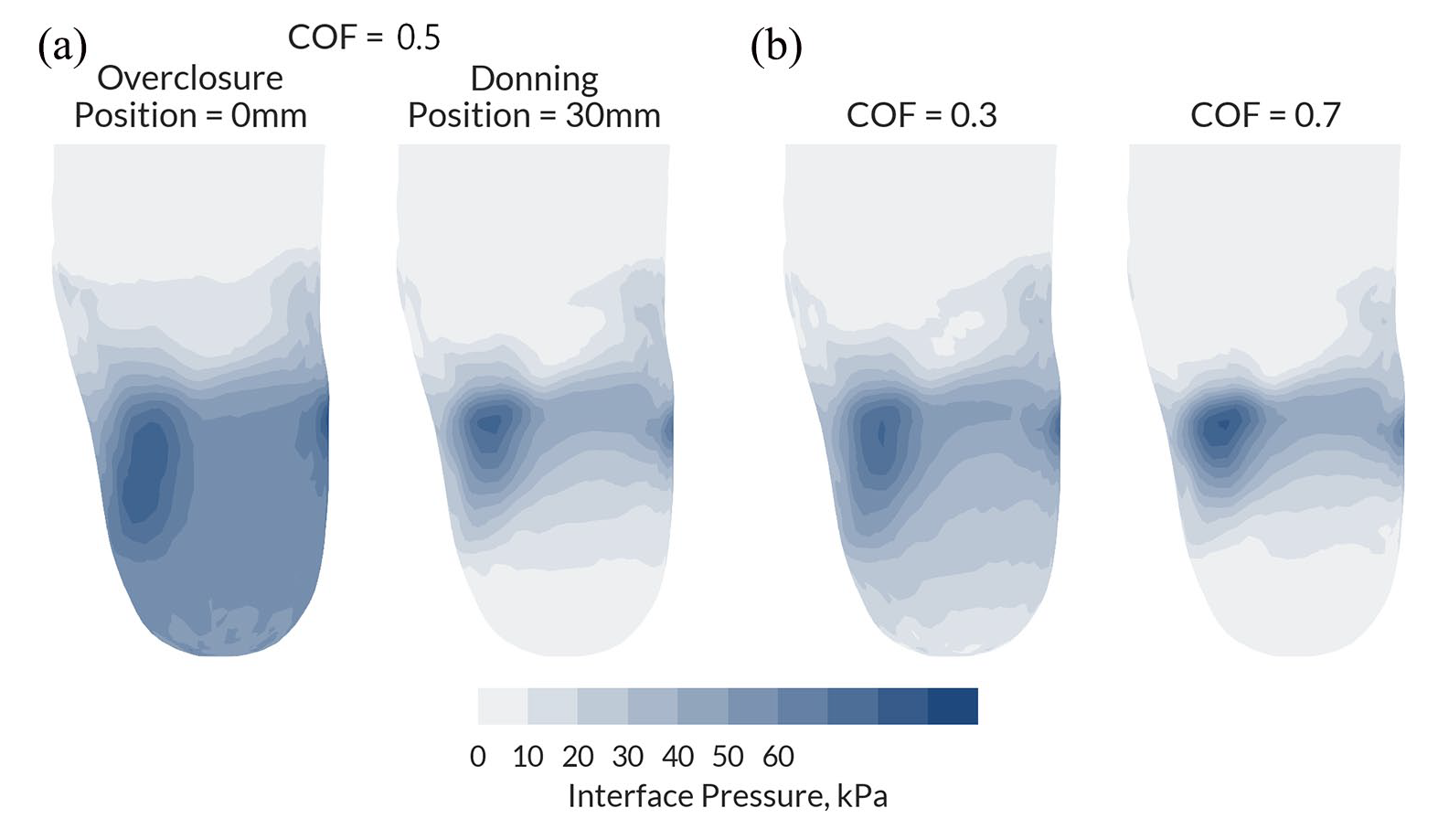
Interface pressure predictions under stance loading for different socket press-fitting methods and interface coefficient of friction (COF) parameters: (a) overclosure method with 0-mm initial distance compared to 30-mm initial distance in donning for the 2% socket design under stance loading, both with baseline COF of 0.5. (b) Effect of changing the COF from 0.3 to 0.7 on the predicted pressure distribution for the 2% socket under stance loading. View rotated by 35° around the vertical axis from the anterior coronal plane to better view the lateral aspect of the residuum.
Loading conditions
Substantial differences were predicted between the quasi-static load cases of heel-strike, mid-stance and toe-off (Figure 4). Fibular head and tibial tuberosity pressure were predicted to be highest at heel-strike or mid-stance (70.7–117 and 51.5–76.7 kPa, respectively). Differences in pressure distribution were also observed between socket designs. The high press-fit 4% socket was predicted to reduce residuum tip pressure to zero in all load cases, and therefore, reduce soft tissue strain in this region. However, it also generated high fibular head and tibial tuberosity pressure. Toe-off loading was predicted to generate high pressure over the patella tendon in all socket designs (116–145 kPa). Distal tissue strain was highest during toe-off loading for looser sockets (76.2%–89.2%) and during mid-stance for tighter sockets (44.1%–48.5%).

Full field pressure data for the four socket designs (−2% clearance, 0% line-to-line fit, and 2% and 4% uniform press-fit) during heel-strike, mid-stance and toe-off. View rotated by 35° around the vertical axis from the anterior coronal plane. The two-legged stance load case results are given for comparison.
Soft tissue material model
Strain was evaluated at heel-strike in the 0% press-fit socket, representing the worst-feasible cases of distal tip strain) (Figure 5). However, negligible differences were noted between linear and hyperelastic models of equivalent initial stiffness. The most notable difference between the linear and hyperelastic models was that the hyperelastic models were more numerically stable (able to converge) than the linear elastic models at lower, physiological-range stiffnesses. More flexible FE models are more likely to fail to converge (reach a numerical solution) under full load. The lowest soft tissue stiffness achieving numerical convergence at 100% loading was 100 kPa for the linear model and 25.4 kPa for the hyperelastic model. Increasing the soft tissue stiffness resulted in lower compressive strain around the distal tibia. For the matched 0% socket, the lowest stiffness tissue resulted in a predicted strain of 99% compared to 14% for the stiffest tissue of 200 kPa. Similar trends were observed for the stance loading condition, and pressure predictions were affected more than the strain predictions by the elasticity model, with the linear elastic model predicting lower pressures at 150 and 200 kPa stiffnesses (Supplementary Material).
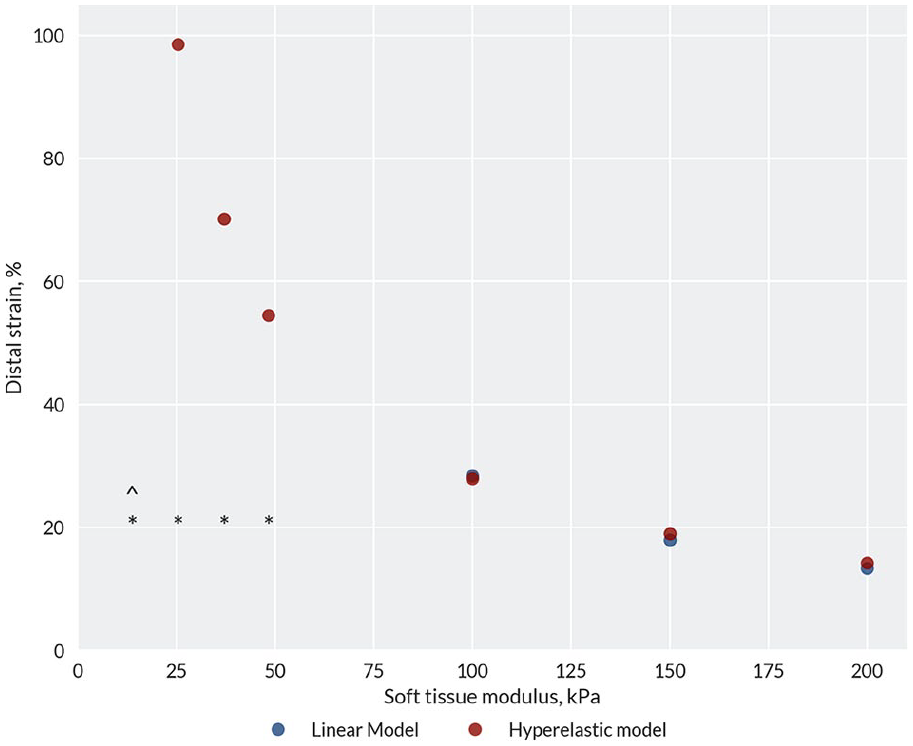
Distal soft tissue strain for hyperelastic and infinitesimal-strain equivalent linear elastic soft tissue material models of different moduli. The model was evaluated under heel-strike loading with a 0% press-fit socket design, representing the worst-feasible case of this model’s input scenarios, and coefficient of friction (COF) of 0.5. The asterisk indicates that the linear model did not converge, while ‘^’ indicates that the hyperelastic model did not converge.
Discussion
The aim of this study was to critically appraise different methods and input variables for developing FE models of the TTA leg and prosthetic socket. The motivation was to provide informed guidelines for future model developments and assist clinicians in appraising the FE research they read in the literature. This comparative analysis tested a range of inputs, in addition to different socket designs and loading scenarios. The benchmark model predicted pressures and shears under stance loading in the order of 50 and 20 kPa, respectively, with peak gait pressure of 180 kPa at toe-off, comparable to experimental pressure and shear sensor data27,28 and skin–liner interface stresses from previous computational models.8,29
Effect of shear at residuum–socket interface
A key finding of this study is the influence of the donning method and liner–socket interfacial properties on shear stresses generated at the residuum–socket interface. Explicitly modelling donning was predicted to introduce shear stresses that reduce pressure over the residuum tip, protecting this relatively load-intolerant area, a key part of the socket design’s intention. Conversely, the pure overclosure method of introducing the socket was unrealistic in that it did not generate any peripheral shear stresses, thereby causing elevated pressure at the residuum tip once stance loading was applied. These predictions are consistent with the benchmark report by Lacroix and Patiño in a transfemoral case. 11 This study predicted a similar effect in a TTA model and went on to add stance loading to the donned sockets, showing the relative contributions of these load cases.
The longitudinal shear stresses were also influenced by the liner–socket interfacial properties, where higher friction resulted in reduced residuum tip pressure. The increase in longitudinal shear stresses with COF is also consistent with previous studies of the liner–skin interface. 8 Prosthetists aim to achieve limited socket–residuum shear load transfer to protect the residuum distal tip from end-bearing, while avoiding excessive shear that may increase the risk of skin breakdown. 30 As such, accounting for the different sources of shear loading in these models is of particular importance for clinical translation.
Comparative analysis of socket designs versus load cases
The modelled loading conditions produced substantially different interface pressure predictions. Peak pressures were observed at the patella tendon during the toe-off load case. The socket design had a greater effect upon the peak pressure location under heel-strike loading. Tighter sockets (2% and 4% press-fit) loaded the tibial tuberosity and fibular head, whereas looser sockets (exact fit or 2% over-sized) demonstrated end-bearing throughout heel-strike, mid-stance and toe-off and elevated pressure at the distal anterior tibia.
Effect of soft tissue stiffness and elasticity models upon predicted strain
Variation of the soft tissue stiffness caused substantial differences in the predicted strain (Figure 5). There was minimal difference in the strains predicted for equivalent linear- and hyperelastic models; however, the hyperelastic models were more computationally stable at physiological stiffness levels: the equivalent hyperelastic models produced solutions under considerably higher loads. Increasing the soft tissue stiffness increased the model’s stability, as a stiffer material distorts less. This effect has been observed previously, and one study selected a 300-kPa stiffness linear model on the basis of its numerical stability, 8 although this is considerably stiffer than indicated by experimental tests. 7 FE models have been proposed as tools to predict soft tissue damage based upon strain thresholds, 31 reported between 40% and 60%.25,32 Based on the presented results, if such a model was used for prediction of soft tissue damage risk, this would be heavily influenced by the material stiffness selected and, critically, underestimated if an unrealistically high stiffness was used. Clinical assessment of soft tissue damage risk should, therefore, be based upon carefully selected, representative soft tissue material properties, and these would ideally be patient specific. 29
Limitations
Considering the model itself, simplifications were made in the use of a single individual’s imaging data. The soft tissue, fat and skin layers were all consolidated into a single body, apart from the two tendons, and no sliding was allowed at the knee or between the bulk tissues and tendons. This is in contrast to some previous studies that segmented MRI scans of multiple participants into tissue, fat, skin and scar layers. 10 A further enhancement in the model’s geometric biofidelity can be captured using diffusion-tensor MRI. 33 Such models would likely facilitate more realistic load transfer than one in which the soft tissues are homogenised. Advances in medical imaging will present opportunities for models to capture the complex structures of the residuum. Furthermore, to best critique the current literature, only quasi-static loading was simulated. Models which use explicit modelling may better capture the dynamic residuum-socket interaction during gait. For the four socket designs presented, no experimental data were collected, thereby preventing full model validation. Instead, the predicted pressure response of the residuum was corroborated by comparison to experimental measurement data from the literature. These four sockets represent a simplification of true socket designs for illustrative purposes only, which might limit the study’s generalisability in comparison to FE research that uses more realistic sockets. However, similar generalisability limitations exist for all such research that does not apply large population analysis. 34 Finally, the effects of liner donning were not explicitly included, so the pressure and strain reported is additive and analysed comparatively, as it is acknowledged to be safer than absolute analysis using such models.
Clinical relevance
Any clinically implemented modelling technologies must provide sufficiently reliable and representative results. This study demonstrated that the choice of modelling approaches (i.e. donning vs. overclosure) and selected input parameters (i.e. material properties) will have substantial and potentially significant effects on the predicted biomechanical response of the limb under prosthetic loading. The relationship between peripheral shear stresses and residuum tip pressure is one such effect, due to the risks associated with residuum end-bearing. 35 Therefore, any reliance on modelling predictions to inform clinical practice must be used in light of the mentioned limitations. Indeed, models will be best used as an adjunct to clinical reasoning and experience. Without this appreciation, reliance on model outputs could result in poor fit for the prosthesis user and, in extreme cases, development of skin and sub-dermal tissue damage from adverse pressure and shear gradients. 30
High-quality input data from experimental studies, in addition to good practice in model development, are necessary for reliable FE modelling. Many studies have sought to characterise patient anatomy, 34 gait analysis, 36 soft tissue material models 37 and interface properties. 23 More recently, a framework has been presented to incorporate population variability, to enhance this evidence base without large computational resources, FE training and experience. 34 Such methods have applications for lower limb prosthetics to move away from single-case research studies and towards a more general understanding of how differences in residuum shape and prosthetic socket design affect clinical outcomes. This approach would also enable prosthetists to access rapid, comparative predictions of a wide range of socket designs.
Recommendations
Future studies in the field should:
Simulate the effects of donning, with appropriate interface friction properties, to initiate interface shear stresses. The simplified ‘overclosure’ loading method will neglect these shear strains, and therefore, markedly change the limb-socket load transfer and suspension predictions;
Ideally employ a comparative approach in analysing different socket designs under a representative range of loading conditions, as even intra-patient variability (e.g. volume fluctuations) will influence the models’ absolute validity;
Use hyperelastic material models to increase computational stability, with appropriate stiffness, especially when using strain to predict soft tissue damage risk; and
Clearly justify their modelling decisions and research questions within a clinical reference frame.
Finally, as there is no consensus regarding the data required for model generation, continued effort to add to the evidence base of soft tissue properties, gait analysis and patient anatomy would benefit the community.
Supplemental Material
10.1177_0309364620967781_Supplemental_material – Supplemental material for Key considerations for finite element modelling of the residuum–prosthetic socket interface
Supplemental material, 10.1177_0309364620967781_Supplemental_material for Key considerations for finite element modelling of the residuum–prosthetic socket interface by JW Steer, PR Worsley, M Browne and Alex Dickinson in Prosthetics and Orthotics International
Footnotes
Appendix 1
Author contribution
All authors contributed equally in the preparation of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: J.W.S. was funded by the University of Southampton’s EPSRC Doctoral Training Programme (Ref No. EP/M508147/1) and EUROSTARS project (Ref. No. 9396), P.R.W. was funded by the EPSRC-NIHR ‘Medical Device and Vulnerable Skin Network’ (Ref. No. EP/M000303/1), and A.S.D. was funded by the Royal Academy of Engineering, UK (Ref. No. RF/130). Herr Ganter and other members of the ImpAmp consortium under EUROSTARS project (Ref. No. 9396) were funded for technical support and data collection.
Data accessibility statement
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
