Abstract
Background:
Many people with lower limb amputation experience mobility impairment and reduced quality of life. Mobility clinics are designed to improve mobility and quality of life for people with lower limb amputation, but their effectiveness is unknown.
Objectives:
To compare changes in mobility prior to, and 12 weeks following participation in mobility clinic for people with lower limb amputation, and to explain whether changes in mobility explained changes in quality of life. To determine whether the PLUS-M™ was sensitive to the effects of participation in the mobility clinic, and to estimate the sample size required for a definitive study.
Study design:
Longitudinal observational.
Methods:
Electronic versions of the PLUS-M and SF-36v2® were completed by people living in the community with lower limb amputation prior to, and 12 weeks following participation in a mobility clinic.
Results:
There was a significant increase in mobility from baseline to 12 weeks post participation in the clinic (p = 0.012). Changes in mobility explained a significant proportion of variance in the SF-36v2 mental component summary (p = 0.024) but not the physical component summary (p = 0.804).
Conclusion:
For people with lower limb amputation, mobility increased after participation in the clinic and this explained improvements in SF-36v2 mental component summary. The PLUS-M was sensitive enough to detect a change in mobility over time.
Clinical relevance
This preliminary data indicated that participation in a mobility clinic improved mobility and the mental components of quality of life for people living with lower limb amputation. The PLUS-M™ seems sensitive to changes in mobility as a result of participation in a mobility clinic.
Background
Rehabilitation of a person with lower limb amputation (LLA) often focuses on basic mobility training, such as straight-line walking or safe stair ascent. 1 High-level mobility training that involves multi-directional activities, agility or speed is often not considered a requisite part of many rehabilitation programmes.1,2 Hence, many people living in the community with LLA have some degree of mobility impairment.3–5
Mobility impairment reduces the ability to participate in activities that bring meaning and enjoyment to life which, to some extent, explains the social isolation, depression and reduced quality of life (QoL) experienced by many people living with LLA.3,5–7 There are opportunities to reduce mobility impairment and maximise mobility potential beyond that achieved by many people at the end of rehabilitation and in doing so, improve QoL.
Össur mobility clinics (Össur Australia) have been introduced across Australia in recent years, with the goal of maximising the mobility and prosthetic capabilities of people living with LLA. 8 In Australia, these 2-day mobility clinics are typically offered once-a-year in major cities and include face-to-face tuition with a mix of lectures, peer support, physical fitness and mobility training specifically targeted to the needs of people with LLA. Activities are often multi-directional and involve balance and turning on the prosthesis as well as, running and sport-specific exercises for a variety of recreational activities.
While mobility clinics have become popular in recent years, there is no evidence to indicate that participation improves mobility and if so, whether this leads to improved QoL. If participation in a mobility clinic led to measurable improvements in mobility and, in turn, QoL, there would be a strong case to increase the availability of these clinics for people in the months and years following LLA.
Hence, the aim of this investigation was to compare mobility prior to, and 12 weeks after participation in a mobility clinic for people living in the community with LLA. Given the hypothesis that mobility would change, both 1-week and 12-weeks after participation in a mobility clinic, we also sought to explain whether changes in mobility explained any change in QoL.
Given that there has not been any previous investigation into the effects of participation in a mobility clinic, a prudent first step would be to conduct a pilot study to ensure that the choice of mobility measure was sensitive to the effects of participation in the mobility clinic, and to use the data collected to estimate the sample size required for a definitive study.
Methods
Ethics approval for this study was granted by the La Trobe University College Science, Heath and Engineering Ethics Sub-Committee (HEC18068).
Study population
Participation in the mobility clinic was open to people with LLA. The clinic was advertised on social media, the Ossur website and amputee support organisation Limbs4Life (https://www.limbs4life.org.au/), as well as notice boards in prosthetic clinics. In response to an advertisement for the mobility clinic, those interested in participating registered directly with the clinic organisers. Registered participants who were over 18 years of age were contacted through email by the clinic organisers on behalf of the investigators and invited to participate in the study. The invitation explained the purpose of the study, included contact details for enquiries regarding the study and an electronic link to an online survey. Consent to participate was implicit by survey submission.
Equipment
Self-report data were collected electronically using the SurveyMonkey platform. The online survey comprised the Prosthetic Limb Users Survey of Mobility 12-item Short Form (PLUS-M™), 9 the 36-item Medical Outcome Short Form Health Survey Version 2 (SF-36v2®), 10 and demographic questions to characterise the age, sex, cause and level of amputation, comorbid health conditions, annual income and education level of the sample.
The PLUS-M9,11 is a self-report instrument for measuring lower limb prosthetic users’ mobility, defined as the ability to move from one place to another independently and intentionally. The PLUS-M was developed using focus groups 12 and cognitive interviews 13 with people with LLA. The PLUS-M performs equally well in the paper-based and electronic forms, and in both the short form and long form, 14 demonstrates no evidence of floor or ceiling effects, 14 has good construct validity 15 and is reliable for both group and individual comparison with test–retest reliability interclass correlation coefficient (ICC) ⩾ 0.9. 14 The PLUS-M 12-item Short Form comprises a sub-set of 12 calibrated questions from the PLUS-M item bank. The PLUS-M results in a T-score ranging from 17.5 to 76.6, with an average of 50 and a standard deviation (SD) of 10. A T-score of 50 represents the mean mobility reported by the development sample, with higher scores indicating greater mobility. The PLUS-M 12-item Short Form takes approximately 2–3 min to complete.
The SF-36v2 is a widely used generic health survey that measures perceived health-related QoL. 10 It has been shown to be valid and reliable across a range of populations16–18 and its wide-spread use in studies of people with LLA19–23 suggests its acceptance as a QoL measure for this population. The SF-36v2 comprises 36 questions across the following health domain scales: Physical Functioning, Role-Physical, Bodily Pain, General Health, Vitality, Social Functioning, Role-Emotional, Mental Health. Results from the SF-36v2 are reported as a physical component summary (PCS) and a mental component summary (MCS). Similar to the PLUS-M, the SF-36v2 uses norm-based scoring with T-scores higher than 50 indicating greater than average QoL. However, SF-36v2 scores are based on norms established from among the United States general population. The SF-36v2 takes approximately 10 min to complete.
Procedures
Those who completed the online survey, and thereby consented to participate, were emailed an online survey at three time points: in the week prior to the mobility clinic (T1), as well as 1 week (T2) and 12 weeks (T3) following the mobility clinic. Reminder emails were sent to those who had not completed the survey at 3 and 7 days after each of these three time points.
The mobility clinic took place at a sports and recreation facility which utilised both an indoor sports stadium and outside grassed ovals in Adelaide, a major metropolitan city in Australia. The 2-day mobility clinic was designed for people with LLA of all abilities with a focus on walking, running and sports. Over a 6-h period, each day, physiotherapists and athletic coaches gave lectures and led activity sessions appropriate to the varying levels of mobility of the clinic’s participants. In addition to participation in a variety of group-based sports and recreational activities, participants were taught strengthening exercises specifically developed for people with LLA and heard from motivational speakers and peer support groups.
Data analysis
Upon completion of data collection, demographic data were entered into a Microsoft Excel spreadsheet. The PLUS-M data were also entered into the spreadsheet and raw scores were converted to a T-score according to the user manual. 11 The SF-36v2 data were entered into the QualityMetric Incorporated Health Outcomes Scoring software (QualityMetric Incorporated, Lincoln, RI) to derive the PCS and MCS scores. Data were described using measures of central tendency and variability appropriate to the data type.
A one-way repeated measures analysis of variance (ANOVA) with the Bonferroni post-hoc was used to test for significant different in the PLUS-M T-scores over the three time points in accord with the techniques described by Lund and Lund. 24 Accordingly, violations of the assumptions of normality and sphericity were tested, and so too were the data inspected to identify outliers. To determine the extent to which changes in mobility explained variation in the SF-36v2 PCS and MCS scores, simple linear regression models were built according to the techniques described by Lund and Lund 25 which included assumption testing for linearity, independence of residuals, homoscedasticity and normality. Parameter estimates including standardised regression coefficients (β), p-values, and part correlation coefficients were reported.
Results
Study population
Of the 11 people who participated in the study, two were lost to follow-up as they did not complete either of the two post-clinic surveys. These two people were demographically similar to the participants included in the study (mean age was 45.5 years, both were male, one unilateral and one bilateral transtibial amputation). Of the nine participants who completed the study, the majority were female with unilateral transtibial amputation many years prior due to trauma (Table 1).
Clinical and demographic characteristics of the sample.
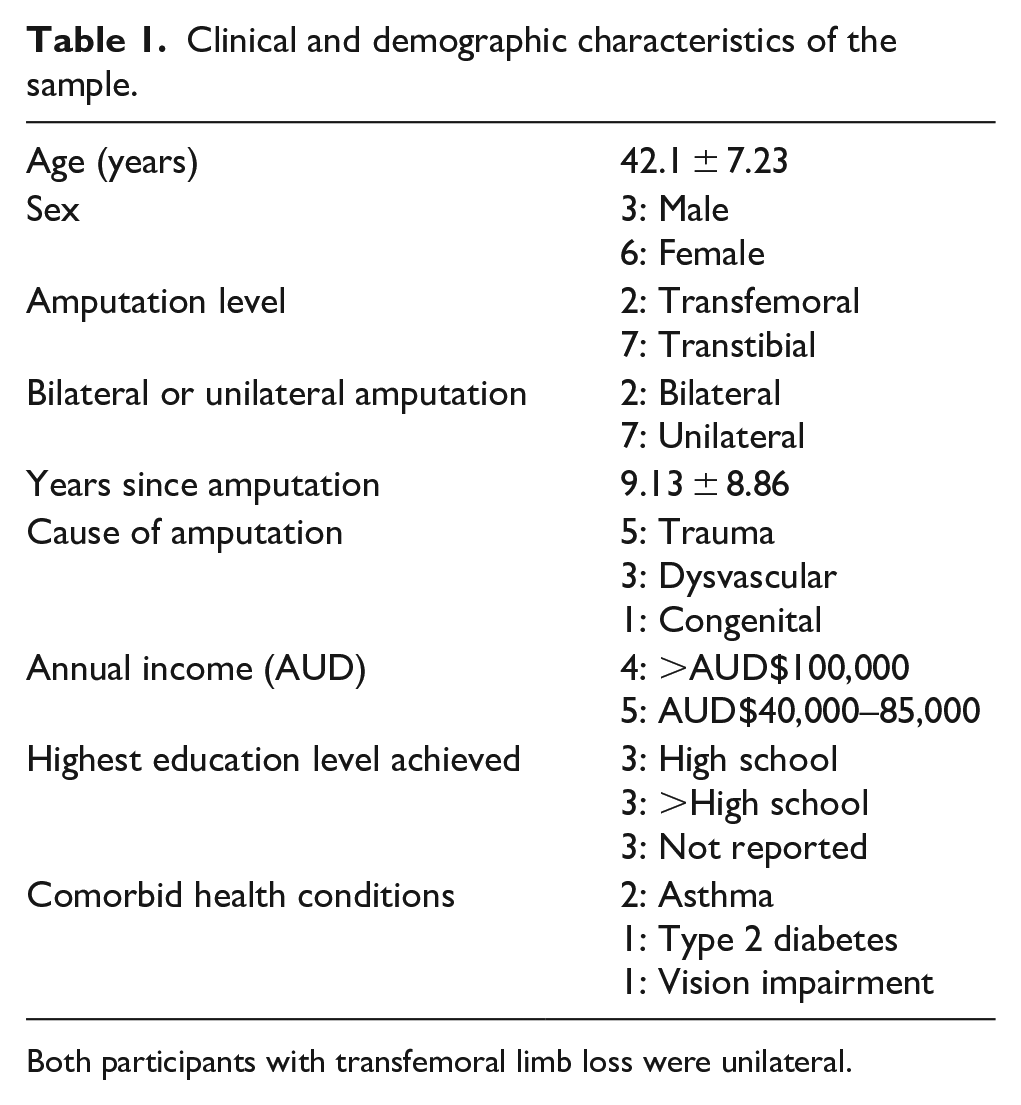
Both participants with transfemoral limb loss were unilateral.
Change in mobility over time
There was a significant main effect for changes in mobility over time (F2,16 = 8.798, p = 0.003, ηp2 = 0.524). Post hoc analysis showed that PLUS-M scores increased from T1 (55.23 ± 4.73) to T3 (60.57 ± 6.30), which was statistically significant (Δ5.33, 95% confidence interval (CI): 1.31–9.36, p = 0.012). Changes in PLUS-M scores from T1 (55.23 ± 4.73) to T2 (58.10 ± 4.11) were not significant (Δ2.87, 95% CI: 0.44–6.17, p = 0.092). Similarly, differences in PLUS-M scores from T2 (58.10 ± 4.11) to T3 (60.57 ± 6.30) were also not statistically significant (Δ2.467, 95% CI –1.66 to 6.60, p = 0.328, Figure 1).
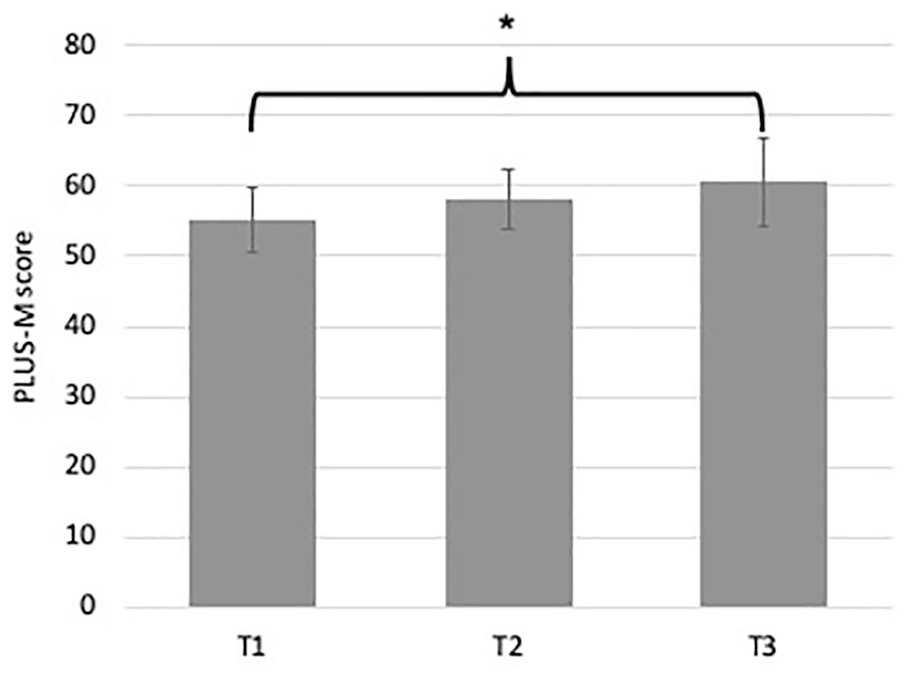
PLUS-M™ scores for T1, T2 and T3.
Change in mobility and their effect on QoL
A simple linear regression was conducted to understand the extent to which changes in PLUS-M explained changes in SF-36v2 MCS and PCS over time (Table 2).
Mean (standard deviation) SF-36v2® MCS and PCS scores at T1, T2 and T3.
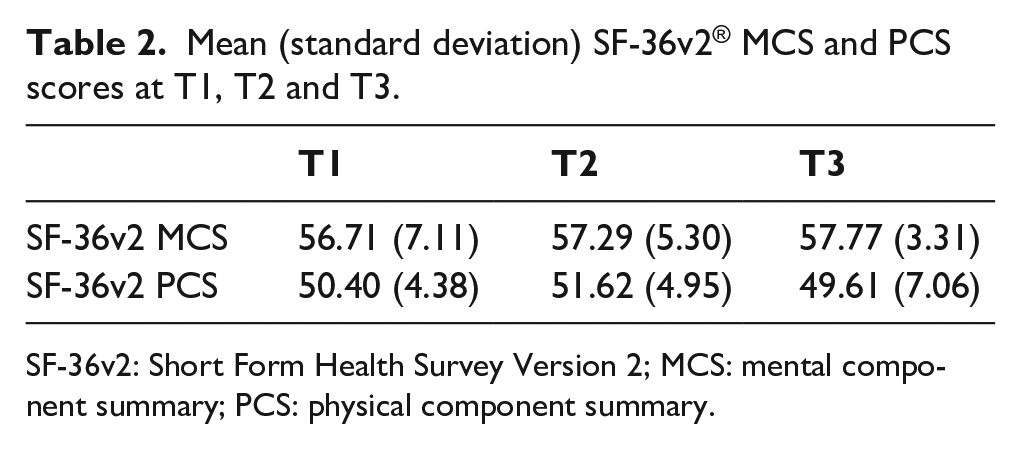
SF-36v2: Short Form Health Survey Version 2; MCS: mental component summary; PCS: physical component summary.
Change in PLUS-M scores from T1 to T3 explained 54% of the change in the SF-36v2 MCS scores across the same time period (R2 = 0.54) which was statistically significant (F1,7 = 8.25, p = 0.024, Table 3).
Linear regression for the predictor variable: change in PLUS-M™ scores from T1 to T3.

PLUS-M: Prosthetic Limb Users Survey of Mobility 12-item Short Form; SF-36v2: Short Form Health Survey Version 2; MCS: mental component summary; PCS: physical component summary.
Changes in PLUS-M scores from T1 to T3 explained <1% of the change in the SF-36v2 PCS scores across the same time period (R2 = 0.009) which was not statistically significant (F1,7 = 0.67, p = 0.804, Table 3).
Discussion
The aim of this study was to compare mobility prior to, and 1 and 12 weeks after participation in a mobility clinic, for people living in the community with LLA. Given the hypothesis that mobility would change after participation in a mobility clinic, we also sought to explain whether changes in mobility explained changes in QoL.
In comparison to a pre-clinic baseline measure, it is interesting to consider why mobility did not improve 1 week after the clinic but did significantly improve at 12 weeks. We hypothesise that at 1-week post-clinic, participants were unlikely to have had the opportunity to practice their newly acquired skills. In contrast, during the weeks that followed, participants would have had repeated exposure to the broad range of community mobility activities that the PLUS-M seeks to measure: carrying a shopping basket in one hand (PLUS-M, question 6) or walking on an unlit street or sidewalk (PLUS-M, question 8). We suggest that participation in the clinic, and the subsequent practice, led to improved mobility 12-week post-clinic.
It is also interesting to consider why changes in mobility over time explained more than half of the variance in SF-36v2 MCS, but not the PCS. We suggest that improved mobility may have led to an improved ability to participate in activities that bring joy and meaning to life that, in turn, support social engagement and improve mental health. However, this hypothesis falls short when you consider that improved mobility should have been reflected in improved SF-36v2 PCS scores over time, which was not the case. Mobility is but one aspect of physical function; yet many of the SF-36v2 questions in the physical function domain focus on the ability to mobilise. An alternative hypothesis is that we were unable to explain significant parts of the variance in the SF-36v2 PCS scores using mobility as the only independent variable, given the likely interaction that exists between mobility and the many other factors known to influence QoL, such as pain, level of social support or presence of comorbidities.
To the best of our knowledge, this is the first study to describe changes in mobility as a result of participating in a mobility clinic and the extent to which these, in turn, influence QoL. As a pilot study, there are opportunities to use what we have learnt through this work to inform the design of future research in this area.
Given the recruitment rate (52%) from this mobility clinic, investigators can anticipate the number of mobility clinics needed to obtain the required sample size. Using the group means and SDs to determine the effect size, a total of 42 participants would be needed to detect a significant between-group difference based on error probability (α = 0.05), Power (1 – β) = 0.8, effect size (dz = 0.45) with a two-tailed repeated measures test (G*Power v. 3.1.9.4; Kiel, Germany). A sample of 42 people would be sufficient to power a simple linear regression with just a couple of independent variables assuming typical error probability (α = 0.05), power (1 – β) = 0.8 and a small effect size (f2 = 0.15). Hence, we suggest that future investigations will need to consider the sample size requires given an understanding of the number of independent variables required and the strength of the association between the dependent and independent variables.
Results from this study suggest that the PLUS-M was sensitive to changes in mobility that resulted from participation in a 2-day mobility clinic and as such, it could be reasonably be used in future investigations of this kind.
Limitations
The results of this pilot should be considered in light of the limitations of the study. Unfortunately, we were unable to use an intention-to-treat approach in this investigation given that both participants who were lost to follow-up did not complete the first post-clinic surveys, thus preventing post-intervention data from being carried forward.
Ideal study design for a research project such as this would be to conduct a randomised controlled trial to reduce the potential for selection bias. However, in using such a design, it would be very difficult to establish a meaningful control that does not involve participation in peer discussion or activities to improve mobility. As such a stronger design could include a time-series approach with multiple before and after intervention evaluations, and in this way engender confidence that the pre-intervention measure was stable.
Given the small number of participants in this investigation, there is a risk that the small differences in mobility observed between baseline and 1-week post mobility clinic would have been statistically significant in a larger sample. Given the heterogeneity of the sample, it is also likely that the variance in mobility would be less in samples stratified by cause and level of amputation, and that with this stratification, differences over time may have been easier to detect.
We acknowledge that there are many factors known to influence QoL in people with LLA, and that future investigations should look to recruit sufficiently large cohorts to be able to control for the confounding influence of older age, comorbid health conditions and other factors as part of their regression analysis. In this way, the likely interactions that exist between mobility and these other factors can be controlled so as to explain the variance in QoL.
While the participants included in this research may be typical of those who may be interested in participating in a mobility clinic, they are not representative of the broader population of people living with LLA in the community. As such, care should be taken to generalise the outcomes to the broader population of people living with LLA, particularly those who are older with amputation due to dysvascular causes and multiple comorbid conditions.
Conclusion
Results from this investigation suggest that while participation in a mobility clinic improved mobility, significant changes were not observed until 12 weeks after participation in the mobility clinic. Changes in mobility explained significant parts of the variance in the SF-36v2 MCS scores but not the PCS scores. The PLUS-M seems sensitive to changes in mobility as a result of participating in a clinic and is likely suitable for use in similar investigations. Results from this pilot have been used to suggest the sample size requirements for future investigations. Although further investigations are required to corroborate these findings, our results suggest that mobility clinics may improve mobility potential for people living with LLA and this may have a positive effect on QoL.
Footnotes
Author contributions
All authors contributed equally in the preparation of this manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: Funding for this project was provided by Össur ehf, R&D, Medical Office, Iceland.
