Abstract
Background:
The Ponseti method for treating congenital talipes equinovarus requires an orthosis to maintain correction after manipulation and casting, typically the ‘boots and bar’. Non-compliance with the orthosis increases the risk of recurrent deformity. This study investigates a new orthosis, the abduction dorsiflexion mechanism.
Objectives:
The aim of this study is to assess compliance of the abduction dorsiflexion mechanism when used at night and maintenance of foot morphology.
Study design:
This study is a cohort study.
Methods:
A total of 10 children with unilateral congenital talipes equinovarus previously treated with Ponseti casting were recruited to trial the abduction dorsiflexion mechanism at night for 12 weeks. Foot morphology and compliance were assessed every 4 weeks.
Results:
Participant families were pleased with the orthosis, opting to continue to use the device following conclusion of the trial. Compliance was good and no deterioration in Pirani score or dorsiflexion seen. Abduction improved during the trial. The incidence of skin problems was equivalent to that experienced with the traditional boots and bar that the children had been wearing.
Conclusion:
Good compliance and parental satisfaction, coupled with no deterioration in foot morphology, abduction or dorsiflexion present the abduction dorsiflexion mechanism boot as a feasible alternative to the traditional boots and bar, particularly in children with unilateral congenital talipes equinovarus.
Clinical relevance:
The abduction dorsiflexion mechanism is currently the subject of considerable interest as clinicians look to increase compliance and reduce the recurrence rate in Ponseti-treated congenital talipes equinovarus. The abduction dorsiflexion mechanism boot is a feasible alternative to the traditional boots and bar, particularly in children with unilateral congenital talipes equinovarus.
Background
Congenital talipes equinovarus (CTEV) is the most common limb deformity affecting newborn children in the United Kingdom. It occurs in approximately 1 in 1000 live births. 1 When untreated, it leads to permanent deformity and pain. The treatment options for CTEV have changed over the last few decades, and the Ponseti method 2 is now widely accepted as the optimum treatment for CTEV. This treatment involves serial plaster casts and often Achilles tendon release, then maintenance of the correction with an orthosis. The standard orthosis is a boots and bar device where two boots are held in a dorsiflexed and externally rotated position by connecting them to a bar. This device is worn for 23 h a day for 3 months, followed by night time use until the age of 4 or 5 years. Compliance with the boots and bar is recognised as an important factor in preventing recurrence and maintaining a good long-term result.3–5 Ponseti’s group reports a recurrence rate of 10% and found that poor compliance resulted in a higher risk of recurrence (odds ratio of 17). 6
Several different boots and bars are available, but all share a similar principle. For some families, children will not tolerate the device, especially as they become older and more mobile. The restriction imposed by the bar may be a factor in non-compliance. In addition, children with only one foot affected still have to wear boots and bars on both feet. Therefore, any new orthosis that can maintain correction of the foot position but demonstrate improved compliance would be beneficial. The economic and social costs of treatment for recurrent deformity can be considerable, as it may involve further plastering and surgery.
There have been previous reports of unilateral foot orthoses used to maintain correction of CTEV following Ponseti treatment. There is evidence that a dynamic foot abduction orthosis increases compliance and reduces the rate of recurrence of deformity requiring further surgery.7–9 However, other unilateral orthoses which do not create an abduction force have been shown to increase the rate of recurrence of deformity.10,11 This has understandably resulted in concern about the use of unilateral orthoses for maintenance of correction after successful Ponseti casting for CTEV.
In 2014, the abduction dorsiflexion mechanism (ADM) came to market. Unlike the traditional orthotic device used for CTEV (‘boots and bars’), the ADM is a dynamic foot orthosis which does not require a bar connecting the child’s feet. The aim of the ADM is to control both sub-talar and tibio-talar joint functions in a single device. It allows a range of motion from 5° inversion to 30° eversion, and 45° of plantarflexion to 30° of dorsiflexion. It contains springs so that the child can still move their foot, but when they are asleep and their muscles relax, the boot will stretch their foot away from the CTEV position. This daily manipulation acts to overcome the natural tendency of the foot to return to its original position prior to treatment with the Ponseti technique. Patients with unilateral CTEV need only to use the ADM on the affected foot. The ADM could offer a more appealing orthosis device for CTEV patients.
The aim of this study was to assess this new orthosis in a cohort study. The primary outcome was compliance using the new device. Secondary outcomes were efficacy of the device in maintaining correction assessed by Pirani score, range of ankle dorsiflexion and abduction, and skin problems. As part of the study, we also sought input from paediatric orthopaedic surgeons in designing a future definitive trial.
Methods
The protocol for the study was approved by the West Midlands South Birmingham ethics committee (REC 15/WM/0142).
The parents or guardians of patients known to the Robert Jones and Agnes Hunt Hospital with CTEV who had undergone Ponseti casting and were now wearing the traditional boots and bar orthosis were offered the opportunity to trial the ADM during a routine follow-up appointment if they met the inclusion criteria. Inclusion criteria were unilateral CTEV, previous good compliance with the standard boots and bar orthosis (foot abduction orthosis, Mitchell designs) as assessed during previous clinic consultations, no concerns regarding foot position or effectiveness of current treatment with boots and bars, the parents/guardians consent to being in the trial, and are able to complete documentation and attend additional study visits to the hospital. The suitable age range was from 1 (to allow adequate fitting of the ADM) to 4 years. In our institution, we advise boots and bar usage until the fifth birthday. Exclusions to the trial were poor compliance, recurrent deformity, associated diagnoses and atypical CTEV.
The new orthosis used in this trial was the single ADM with Ponseti ADM sandal marketed by C-Pro Direct (product no. ADM001B). The 2014 version was used. An updated version has since come to market.
The initial trial period was 3 months, but with a provision that use of the ADM could continue if there were no adverse outcomes or recurrence of the deformity. The parent/guardians agreed that regular follow up and data collection could continue until the use of the device finished at the age of 5 years.
Risks were discussed with the parent/guardians as part of the consent process. The risks of the study were problems with the new device relating to fitting and pressure areas, recurrence of deformity and difficulty in returning to the boots and bar regimen if problems occurred.
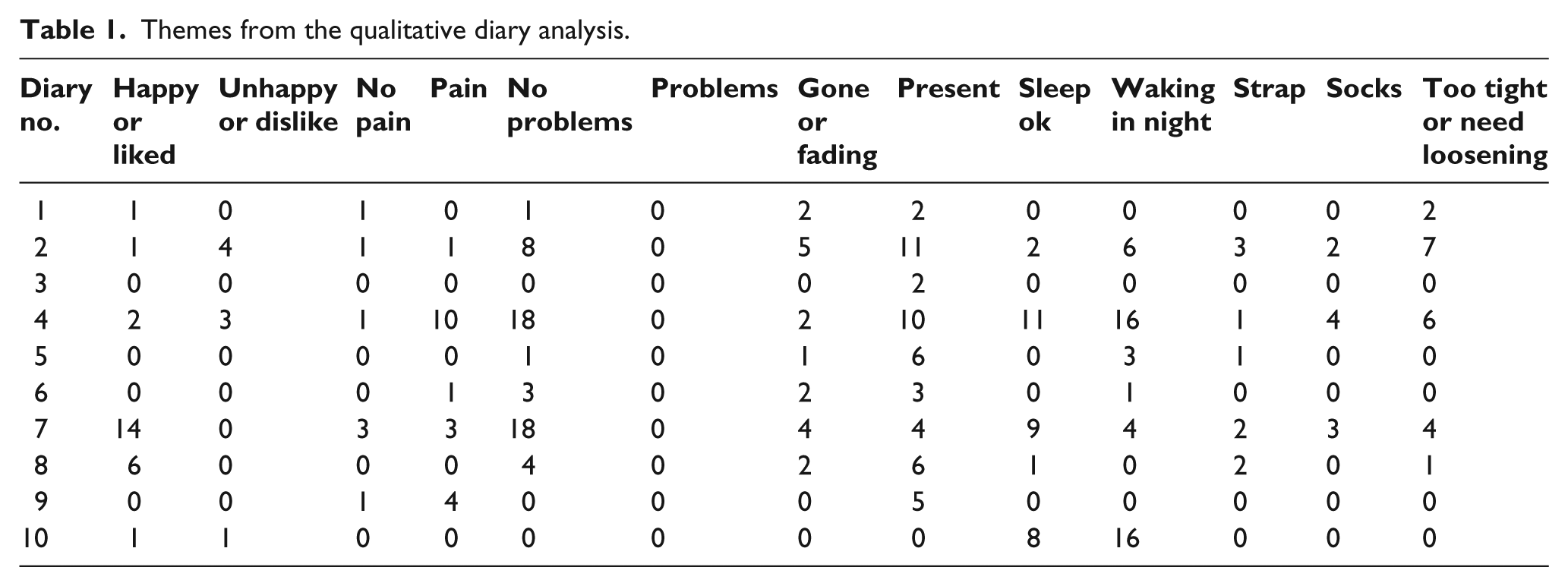
The parent/guardians were asked to complete a daily diary of time the orthosis was put on and taken off and comments related to the ADM. The diary was completed 2 weeks before the study using the boots and bar device and subsequently for 12 weeks using the ADM. Qualitative analysis was performed, with the statements in the diaries grouped into categories of opinion, pain, problems, red mark, sleep quality and ADM straps/padding. Each was divided into positive and negative comments, and the number of comments in each diary was counted.
The ADM was measured and fitted by a specialist paediatric physiotherapist, and a training session was provided. After fitting of the ADM, reviews were arranged at 1 week to check for initial problems, and then at 4, 8 and 12 weeks. The outcome measures for foot anatomy were dorsiflexion at the ankle joint and forefoot abduction. Passive range of movement was measured using a goniomenter. The Pirani score was also recorded. 12 These assessments were carried out by a single specialist paediatric orthopaedic physiotherapist.
Figure 1 shows the patient journey through the study.
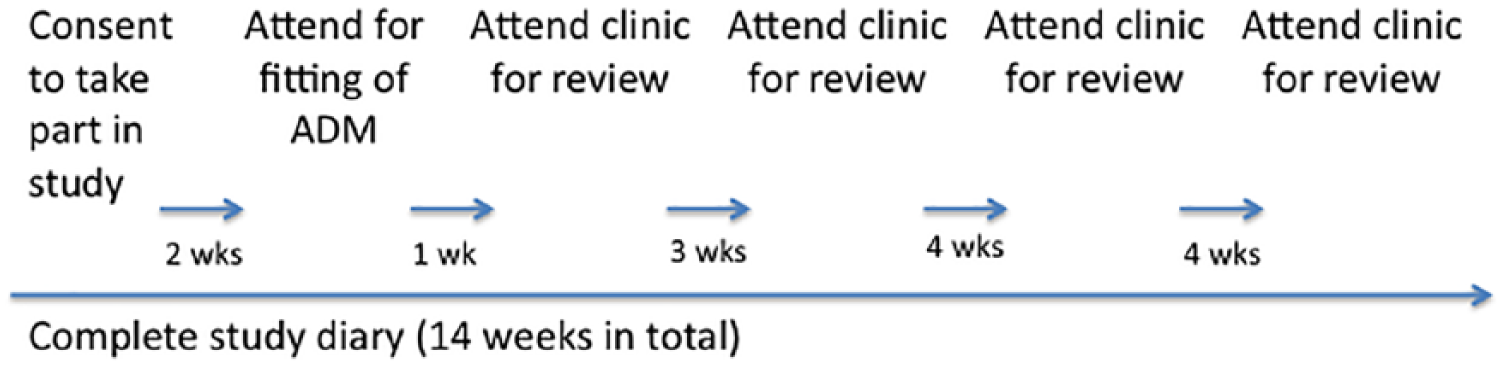
Flowchart of the patient’s journey through the trial.
All paediatric orthopaedic surgeons who are members of the British Society of Children’s Orthopaedic Surgery (BSCOS) were contacted by email with a link to an online survey asking them to provide input to the design of a larger trial to follow on from this small cohort study.
Results
Mean time recorded in the diaries spent in the boots and bars was 10.3 h (standard error of mean: 2.9) and in the ADM was 11.6 h (standard error of mean: 1.3). This is not a significant difference between length of time boots and bar or ADM were worn for (t(8) = −0.061, p = 0.953). Patient 9 was excluded as they had not recorded any data for time spent in boots and bar. Patient 5 was an outlier but there was no significant difference even if this outlier was excluded (t(7) = 1.396, p = 0.205). Normality was confirmed by a visual inspection of a normal Q-Q (quantile–quantile) plot.
The themes of the qualitative analysis were opinion, pain, problems, red mark, sleep quality and ADM straps/padding. None of these were repeatedly mentioned in the diaries in either a positive or negative sense, as demonstrated by Table 1.
Themes from the qualitative diary analysis.
Fifty percent of the boots (5/10) broke during the 12-week trial. However, all children completed the trial and all families preferred to continue in the ADM after the trial rather than returning to the boots and bar.
There was no deterioration in Pirani score although 1 of the 10 children had a change in Pirani score. This was an improvement from 0.5 to 0 in the hindfoot. Of the 10 children, 8 had skin problems with their existing boots and bars. Three of these improved in the ADM. Of the 10 children, 6 developed new skin problems with the ADM, all of which were red marks other than 1 blister. Abduction improved by a mean of 24.9° (range: 6–42°, standard deviation: 12.3°). Dorsiflexion improved by 5.4° (range: −4 to 16°, standard deviation: 5.9°) (Figure 2).
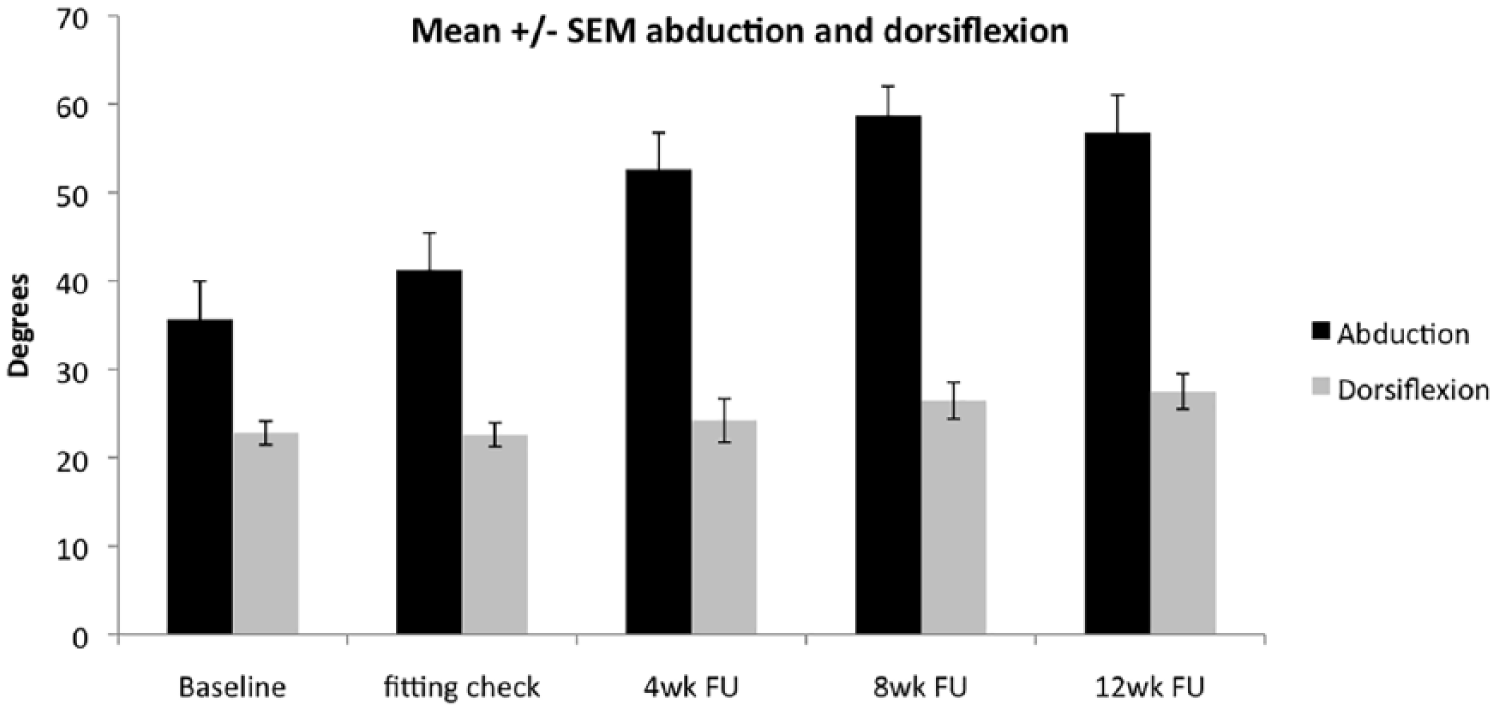
Passive range of abduction and dorsiflexion during the time wearing ADM.
A one-way repeated-measures analysis of variance (ANOVA) test of abduction revealed that there were no outliers in the data, as assessed by inspection of a boxplot for values greater than 1.5 box-lengths from the edge of the box. Abduction was normally distributed, as assessed by Shapiro–Wilk’s test (p > 0.05). Mauchly’s test of sphericity indicated that the assumption of sphericity had not been violated (χ2(2) = 5.775, p = 0.774). Abduction was statistically significantly different at different time points (F(4, 28) = 14.359, p < 0.0005).
At most recent follow up (mean: 16 months), four children had completed their treatment, two had experienced skin problems which had resolved and one had returned to the traditional boots and bar due to a recurrence of deformity. No treatment other than return to the boots and bar was required. The other nine children had no recurrence and the increase in abduction has been maintained.
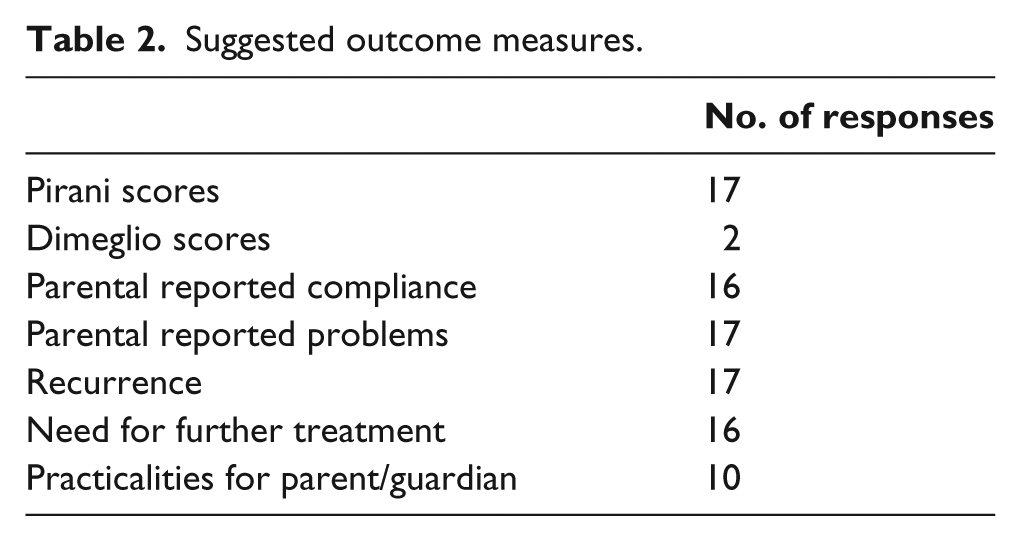
The BSCOS survey had 21 responses. The main concerns about the new ADM were efficacy of the ADM boot, cost of the boot (particularly for bilateral patients) and lack of evidence. Suggested outcome measures for a study aiming to provide evidence about the new orthosis were measures of recurrence of deformity such as Pirani scores, 12 Dimeglio scores, 13 recurrence and need for further treatment; and assessment of compliance using the ADM such as parental reported compliance, problems and practicalities (see Table 2). Suggested inclusion criteria were age, aetiology of the CTEV and previous compliance (see Table 3).
Suggested outcome measures.
Suggested inclusion criteria.
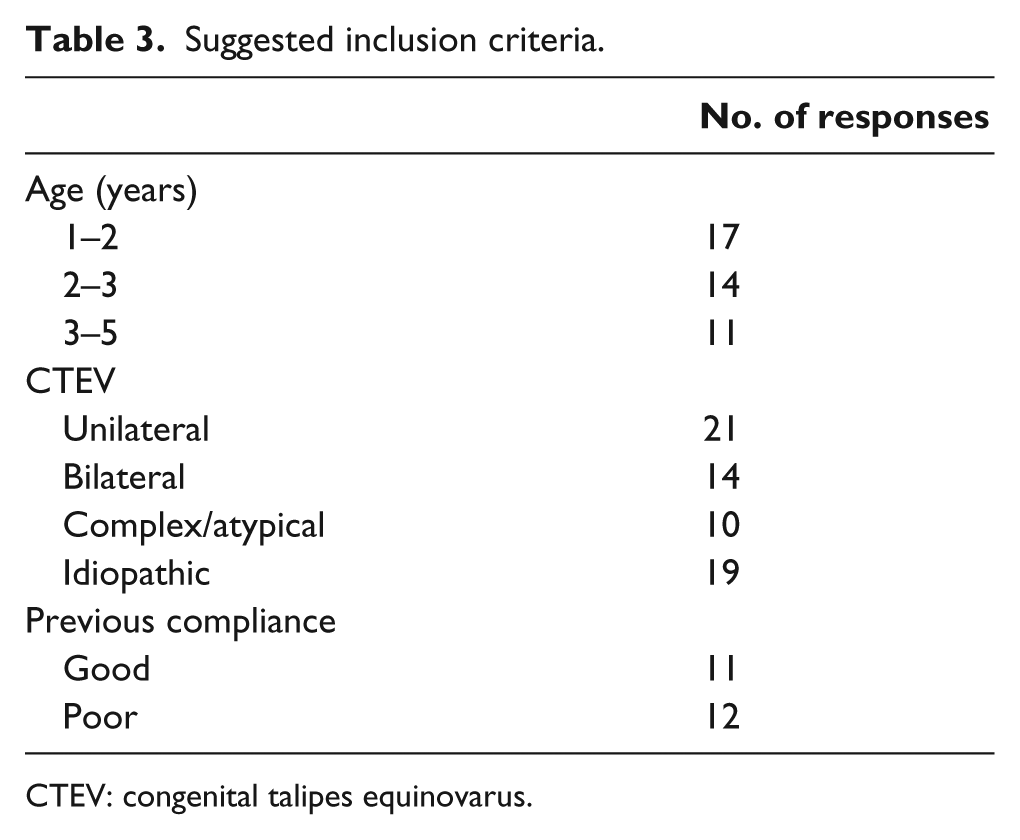
CTEV: congenital talipes equinovarus.
Discussion
The diaries and verbal feedback at appointments suggest that the ADM device is well tolerated by children and parent/guardians. In this group, the dorsiflexion remained largely unchanged, but abduction has significantly increased. Since the passive range of movement was not measured in the boots and bars, this apparent increase in abduction warrants further investigation. However, if reproducible, it could have implications for future treatment.
The parent/guardians reported some problems with fitting and rubbing, but these were able to be resolved and were reported to be less than the problems the families had been having with the traditional boots and bars.
The device demonstrated breakages due to manufacturing problems. There was a weakness at the spring hinge connecting the foot to the calf on the original ADM used for this study and all broken boots failed at the same point. The manufacturer has released a new version of the ADM which is reinforced at the part which was failing to reduce the risk of this problem occurring in future.
We conclude from this small cohort study that a larger trial of the ADM is warranted. Our aim is to develop a randomised controlled trial to compare the ADM to the conventional boots and bar device, to investigate the role of the ADM in the treatment of CTEV including any effect of the ADM on compliance and relapse rates. The difference in mean change in Pirani score with the traditional boots and bar and the new ADM will be used to perform a power calculation in planning further work. The opinions of paediatric orthopaedic surgeons will also be taken into account, as it is important that any evidence we produce is accepted as reliable, sufficient and as conclusive as possible.
It is important to gather evidence as the ADM could offer improved clinical outcomes through increased compliance and greater parent/guardian satisfaction. Changes in forefoot abduction warrant further investigation, and if they are repeatable and true, then a difference in need for further surgery for recurrence could be identified on long-term follow up. However, our main concern is to avoid the adoption of a new device without evidence of its efficacy.
This study has highlighted an issue of how much evidence surgeons and parent/guardians need on the effectiveness of an orthosis before it is routinely used in practice. The authors are aware of hospitals where this ADM is already used in clinical practice, despite current lack of evidence of its effectiveness. This is contrast to the high-strength evidence that we have supporting the use of boots and bars. In designing a full trial, it is important that the outcomes used in the full trial are outcomes which those who prescribe the device deem important. We have attempted to understand which outcomes are seen as important through our survey of consultant paediatric surgeons. There are a number of outcomes that have been used in previous studies such as recurrence rate, which is defined as a need for further intervention (surgical or casting). However, there are some weaknesses with this as an outcome measure in that further intervention is often decided subjectively and there is potential for wide variation in practice to exist between surgeons. The Pirani score is also used as an outcome in studies. Again, this as an outcome measure has some difficulty associated with it because some deterioration might be accepted as long as function is maintained. However, studies have shown that there is good inter-rater reliability for Pirani scoring.12,14,15
In this study, patients with good compliance were recruited to ensure that any compliance problems with the new device were due to the orthosis itself and not related to social, domestic or psychological factors that are related to poor compliance. A future study could look at patients who have just finished casting and never used traditional boots and bars, and compare them to patients who only use boots and bars. Otherwise, a recurrence noted while wearing one orthosis could have in fact begun while wearing the other, but only become notable with advancing age and growth of the child. Or perhaps, the change in mean Pirani score for the same patient in different orthoses is in fact a more significant method of assessing efficacy. The link between non-compliance and recurrence is well established,3–5 so perhaps, measures of compliance using a combination of the diary and modern GPS technology attached to the boot to detect movement when the boot is being worn or applied and removed would be sufficient evidence to support the use of ADM.
The study has generated much discussion, and work is ongoing to design a trial that would provide meaningful evidence about this new ADM. Long-term follow up of the 10 children trialling the ADM continues, and to date there has been one recurrence of foot deformity which required a return to boots and bars but no other intervention. This suggests that although initial results are encouraging, further work to investigate the clinical effectiveness of the device should be undertaken before the device is introduced into routine clinical practice.
Conclusion
Our hypothesis that the new ADM would be acceptable to families has been supported by this work. The overall time that the children wore the boot did not change compared to the time these children with good compliance spent in the traditional boots and bars. All the families wanted to continue with the new ADM at the end of the trial period. Furthermore, no deterioration in foot morphology was noted during the trial period, although subsequently one child has returned to the traditional boots and bar due to a recurrence of deformity. We have concluded that a further larger study of the ADM is warranted. Work is ongoing towards this with patients, their families and paediatric orthopaedic surgeons across the United Kingdom involved in the design of the trial.
Footnotes
Author contribution
This paper was written by Sarah McCartney with revision by Nigel Kiely. All the authors have approved the final version. Nigel Kiely and Sarah McCartney are orthopaedic surgeons (consultant and trainee, respectively) who designed and set up the study; Jan Morris and Claire Sproston are extended scope practitioners in paediatric orthopaedics who reviewed the patients and collected data; and Kirsty Davies and Sarah Turner are research project managers who administrated the trial and analysed the data.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This study was funded by the Orthopaedic Institute Limited (charity no. 104496, funding no. RPG 148).
