Abstract
Background:
Research on home use of advanced upper limb prostheses is needed.
Objectives:
To describe and compare DEKA Arm usage patterns during the last 4 weeks of a home trial for participants with a personal prosthesis and those without. To compare usage patterns during home trial to those of the personal prosthesis prior to home trial. To evaluate user activity preferences for the DEKA Arm and personal prosthesis after a trial of home use.
Study design:
Quasi-experimental, time-series design.
Methods:
Data from 17 participants were analyzed. At baseline, prosthesis users reported days and hours they wore and used personal device(s). Home trial diaries documented days and hours of wear and use for the DEKA Arm and personal device(s), if applicable. Questionnaires asked prosthesis users to list activities they could do with the DEKA Arm but not with their current prosthesis and vice versa and activities they preferred doing with either devices.
Results:
The DEKA Arm was worn 81% and used 73% of functioning days, averaging 4.2 h worn and 2.4 h used on days worn. During home trial, prosthesis users used personal devices and any prosthesis for fewer hours/day than at baseline.
Conclusion:
The DEKA supplemented but did not substitute for the personal prosthesis.
Clinical relevance
Findings strongly suggest that given the limitations of the DEKA Arm and conventional prosthesis, persons with upper limb amputation would be best served and would be able to perform the widest range of activities if they had several types of devices.
Background
Prosthesis use and abandonment in persons with upper limb amputation has been studied widely.1–24 Abandonment is common, especially among those with more proximal-level amputation.18, 21–23 Varying rates of adoption (41%–94.4%) have been reported.1,2,6,14,17–19 Many studies have used survey research to characterize the amount of time an upper limb prosthesis is used or worn,2–5,7,9,13,14,24 but most do not clearly distinguish between prosthesis wear time and prosthesis use time and appear to consider wear and use synonymously. Among prosthesis wearers, between 56% and 79% of device users report “using” their device more than 8 h a day.9,14,17,24
A single type of upper limb prosthesis may not meet all the needs of a person with upper limb amputation; and many amputees use both a myoelectric as well as a body-powered device.8,11 Although it may not be universal practice, new guidelines on rehabilitation of persons with upper limb amputation explicitly recommend that amputees be provided with more than one prosthesis. 25 Yet, there are only a handful of studies that report whether amputees used more than one prosthesis.4,5,8,11,13 None report on amount of utilization of each prosthesis in persons who utilized more than one type.
With increasing availability and demand for new advanced upper limb prostheses and terminal devices, such as the DEKA Arm, 26 it is likely that more consumers will obtain multiple devices including advanced devices with specialized capabilities. Although historically, complete device abandonment has been used as a gross indicator of prosthesis prescription failure, there may be other useful markers (such as amount of prosthesis use) of successful device prescription. Although full-time prosthesis use is sometimes characterized as wear time of greater than 8 h/day 9 to our knowledge, no research has been conducted to quantify the amount of prosthesis use that constitutes a “successful” prescription. No data are available on the amount of time that users of advanced prosthesis actually use these devices or what activities they use them for. Nor do we understand how usage patterns differ for those persons who have only one type or multiple types of devices. This knowledge is needed to guide evaluation of prescription practices and prosthetic outcomes. Thus, the purposes of this study were to (1) describe and compare usage patterns of the DEKA Arm during the last 4 weeks of the home trial for participants who had a personal prosthesis (prosthesis users) and those who did not, (2) compare usage patterns during the home trial to usage patterns of the personal prosthesis prior to the home trial, and (3) evaluate user activity preferences for the DEKA Arm and personal prosthesis after a trial of home use.
Methods
The DEKA Arm
The DEKA Arm is a new advanced upper limb prosthetic device, approved by the US Food and Drug Administration in 2014 and developed under the Revolutionizing Prosthetics Program through DARPA. 26 The device is available in three configuration levels which vary in weight: radial configuration (RC) which weighs 2.8 lbs, humeral configuration (HC) which weighs 6.8 lbs, and shoulder configuration (SC) which weighs 9.8 lbs (Figure 1). A full description of the features for each DEKA configuration level has been reported elsewhere. 26 Briefly, all configuration levels include six powered hand grip patterns, powered wrist flexion/extension which is combined with radial and ulnar deviation, and powered wrist pronation/supination movements. Powered elbow flexion/extension and humeral internal/external rotation movements are available in the HC and SC DEKA Arm. The SC DEKA Arm includes powered shoulder movements (flexion/extension and abduction/adduction) which are operated separately from humeral and elbow movements. SC DEKA users operate humeral and elbow movements simultaneously via an endpoint control feature that coordinates the prosthesis into a desired position in space (e.g. up/down, right/left, or forward/back). 27 All DEKA configuration levels include light-emitting diode (LED) wrist displays that signal the mode of operation, power on/off, battery life, and what grip is currently in use. Multiple control types may be used to operate the DEKA Arm including inertial measurement unit (IMU) foot controls, pneumatic bladder controls, and surface electromyography (EMG) electrodes.
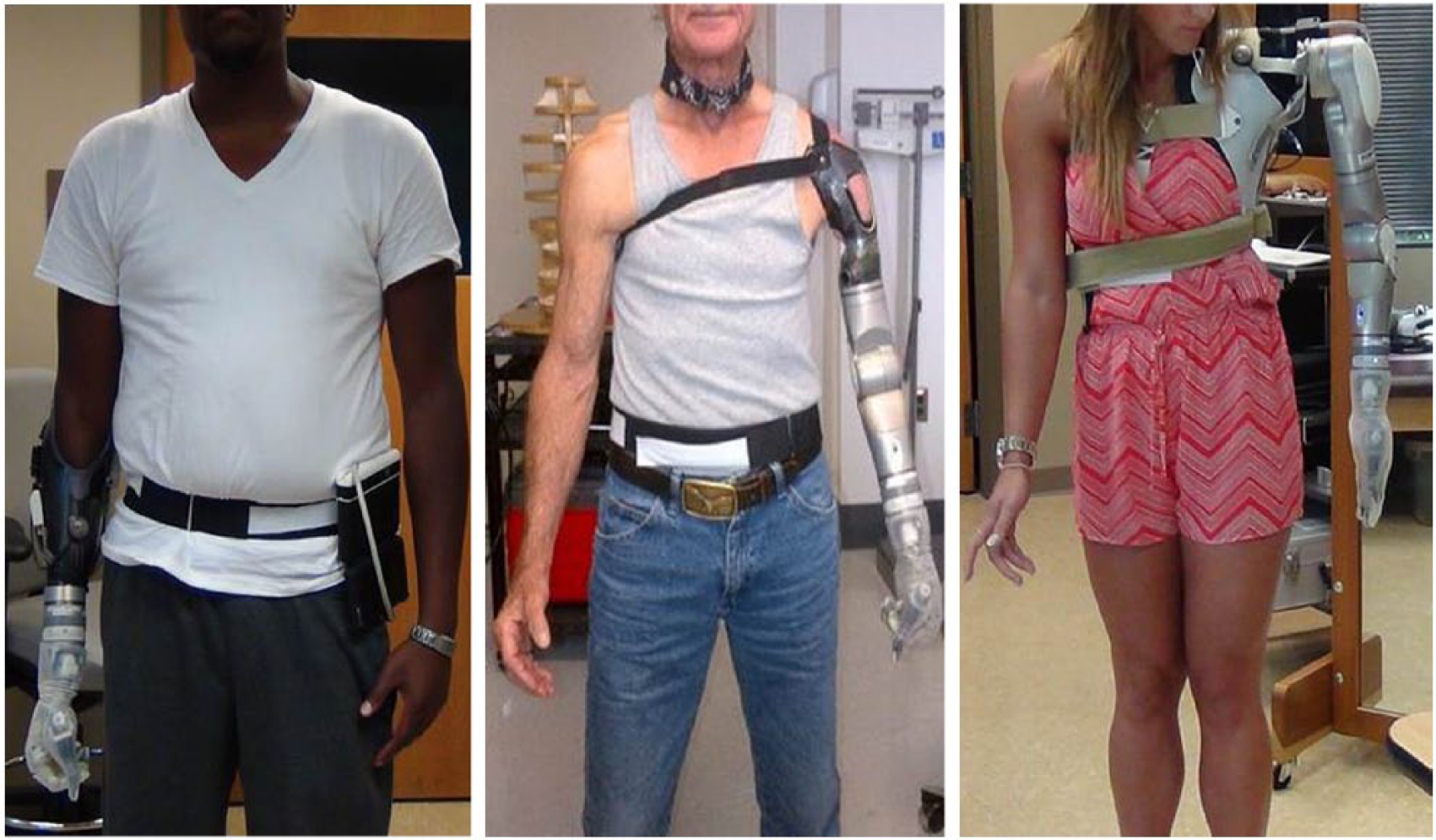
DEKA Arm configuration level: radial configuration (left), humeral configuration (middle), and shoulder configuration (right).
All DEKA configuration levels include a “standby” mode of operation, in which all prosthesis functions are deactivated. The HC and SC DEKA Arm are operated in either “hand mode” for movements of wrist and hand or “arm mode” for elbow and shoulder movements. RC DEKA Arms are powered by an external battery, while the HC and SC DEKA Arms receive power from an internal battery and an external battery. The internal and external battery life varies based on the amount of prosthesis use.
Study overview
The VA Home Study of an Advanced Upper Limb Prosthesis (Home Study) was approved by the Institutional Review Boards of the Providence VA Medical Center, the VA NYHHS, the James A Haley VA Hospital, and the Center for the Intrepid at Brooke Army Medical Center. All subjects provided voluntary informed consent. This was a two-phase study with an in-laboratory (Part A) and a home use portion (Part B) lasting approximately 3 months. During Part A, subjects were trained to use the DEKA Arm. During Part B, subjects were instructed to use the DEKA Arm for a minimum of 2 h per day during their first month of home use and then to use the DEKA Arm as much or as little as they wished during the subsequent months.
Subjects
Subjects were eligible to enroll in Part A if they were at least 18 years old; had an upper limb amputation at the transradial (TR), transhumeral (TH), shoulder disarticulation, or scapulothoracic level (Shoulder); and had sufficient control sites available to operate the DEKA Arm. Subjects were excluded if their amputation level or skin condition prohibited socket or prosthesis fitting and if they had serious health conditions that the study staff determined might limit their participation. The flow of subjects through the study is shown in Figure 2. A total of 42 subjects were enrolled in Part A and 33 completed Part A; 24 subjects began Part B and 18 completed at least 9 weeks of participation. One subject who completed Part B did not have diary data for the last 4 weeks of Part B. Thus, data for 17 subjects who completed Part B were analyzed in this study. A detailed analysis of reasons for non-participation in Part B and for withdrawal from Part B is presented in a separate paper. 28 In short, of the 9 participants who completed Part A but did not enroll in Part B, 6 (66%) declined participation and 3 (33%) were deemed ineligible by study staff because of concerns about their ability to participate in the study at home (one of whom also had declined participation). Of the 6 subjects who withdrew without completing Part B, 3 (50%) did so for personal reasons unrelated to the DEKA Arm, whereas 2 (33%) withdrew because of dissatisfaction with the DEKA Arm, and 1 (17%) withdrew for reasons both related and unrelated to the DEKA Arm.
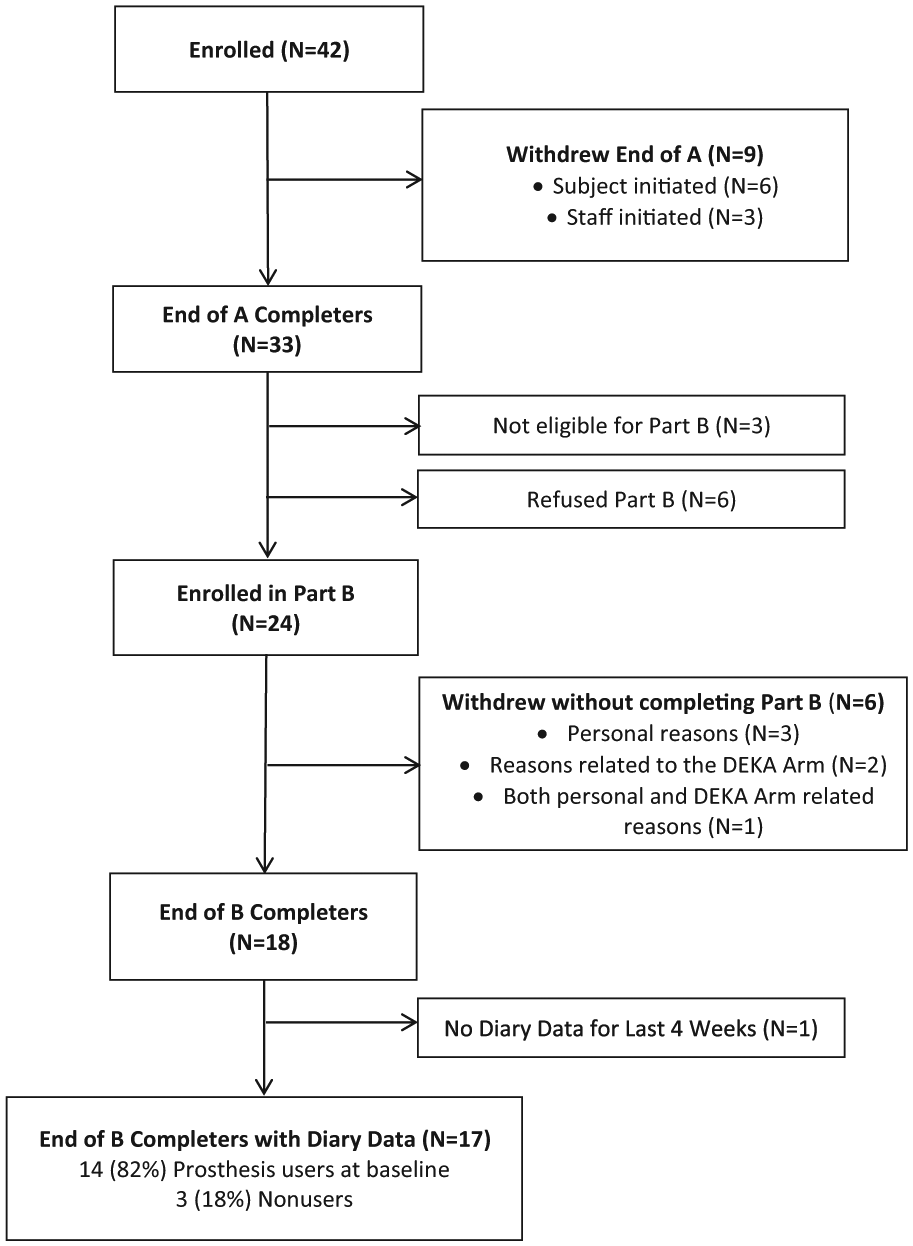
Flowchart of study completion.
Data collection
At study baseline, data were collected on the types of prostheses owned and used. Before beginning Part A, subjects who were prosthesis users were asked to complete a 2-week diary documenting the number of days and hours that they wore their personal prosthesis/prostheses. Given that we believe that some people wear a prosthesis but do not actively use it, we also asked that they estimate the number of hours each day that they used their prosthesis. During participation in Part B, participants were asked to maintain daily diaries to record whether they wore and used the DEKA Arm, and if so, the hours of each day that they wore and used it. Subjects could add comments on each diary day to document whether the DEKA Arm was functioning or broken. If they did not wear the DEKA Arm, they could explain why. The diaries were also used to record whether they wore and used their personal prosthesis, as well as the hours of wear and use of that device. Subjects were called by study staff weekly and reminded to complete their diaries. At the end of the study, participants who were prosthesis users at baseline were asked to list up to five activities they could do with the DEKA Arm but not with their current prosthesis, as well as up to five activities that they could do with their personal prosthesis but not with the DEKA Arm. They were also asked to list up to five activities that they preferred doing (1) with the DEKA Arm and (2) with their personal prosthesis.
Data analysis
Key variables
Weekly diary data were used to create the following key variables.
Baseline. The number of days and number of hours that any personal prostheses were (1) worn and (2) used over the baseline period was used to calculate the proportion of days that a personal prosthesis was worn and used and average hours per day of wear and use at baseline.
Part B. Diary data from the last 4 weeks of Part B were used for this analysis. Subjects were provided with blank dairies for each week and asked to record dates and use time. However, there were occasions where data were not entered for specific days, dates were duplicated on diaries, and/or that subjects added extra days to a diary. Thus, we counted number of days documented in the last 4 weeks of dairies (Diary Days).
Given that we did not expect subjects to wear or use the DEKA Arm if it was broken, the number of days that the DEKA Arm was functioning (DEKA Functioning Days) during the last 4 weeks of diaries was calculated. The number of days and hours that the DEKA Arm was worn (DEKA Days Worn and DEKA Hours Worn, respectively) during the last 4 weeks was counted. Similarly, the number of days and hours that the DEKA Arm was used (DEKA Days Used and DEKA Hours Used, respectively) was counted. We calculated the proportion of DEKA Days Worn and the proportion of DEKA Days Used per Diary Day and per DEKA Functioning Day. We also calculated the average DEKA Hours Worn and the average DEKA Hours Used per Diary Day and per day in which the DEKA Arm was worn and per DEKA Functioning Day. Reasons for not using the DEKA Arm on DEKA Functioning Days were classified as due to user preference or residual limb problems. Number of days the DEKA Arm was not used for either of these reasons was counted. Whenever possible, given subject availability, the study time was increased to enable as much home use time as possible (in keeping with the study protocol).
Days and hours of wear and days and hours of use of the personal prosthesis (if any) were also counted. These variables were used to calculate the average hours that the personal prosthesis was used and worn per diary day. Data from Diary Days were used to count the number of days that either the DEKA Arm or the personal prosthesis was used and the proportion of Diary Days that both devices were used.
Statistical analyses
Demographics of the full sample, as well as the prosthetic user and non-user subsamples, were examined. DEKA Arm usage patterns were described using frequencies and central tendencies. The daily hours of prosthesis wear and use were compared for baseline (personal prosthesis) and the last 4 weeks (personal prosthesis and DEKA hours combined) using Wilcoxon signed-rank tests. The usage patterns of the DEKA Arm were compared for participants who were users of a personal prosthesis and those who were not via the Wilcoxon rank-sum tests. Given the small sample sizes, we consider p values of ≤0.10 to be evidence of statistical significance. SAS Version 9.1 and STATA Version 11.2 were used for statistical analysis of the data.
Qualitative analysis
The proportion of respondents who reported that they could only perform specific activities with one type of device but not the other (DEKA Arm or personal prosthesis) as well as the proportion who expressed a preference for using one device over the other to perform specific activities was calculated. Responses to survey questions about the type of activities that could only be performed as well as the type of activities for which users preferred using one type of device were categorized. Results were stratified by type of personal prosthesis to examine patterns of responses associated with device type.
Results
Data from 17 of the 18 subjects who completed at least 9 weeks of the home portion of the study were used (Table 1). One subject was excluded due to missing diaries. In all, 82% used a personal prosthesis at baseline, 29% used a body-powered device, and 71% used a myoelectric device; 82% of participants were male, 88% were white, 59% had TR amputation, 35% had TH, and 7% had amputation at the shoulder level. All participants reported wearing their personal device daily at baseline and used it for 90% of days. On average, they wore their prostheses for 10.0 h/day (10.4 h/day body-powered and 9.8 h/day myoelectric) and used it for 6.3 h/day (7.7 and 5.7 h/day body-powered and myoelectric, respectively; Table 2). Wilcoxon signed-rank comparisons of usage patterns of prosthetic users show a statistically significantly decrease in total hours of wear (z = 2.98, p = 0.0029) and use (z = 2.06, p = 0.0392) of any prosthesis (DEKA Arm or personal) in the last 4 weeks of the home study as compared to personal prosthesis wear and use at baseline (Table 2). DEKA Arm use in the last 4 weeks is shown in Table 3. Diary data were available on average for 27.9 days (28 days for prosthesis users and 27.3 days for non-prosthesis users). The DEKA Arm was functioning during 98% of diary days. Overall, participants wore the DEKA Arm for 81% of functioning days and used it 73% of functioning days. The DEKA Arm was worn and used an average of 3.5 and 1.9 h, respectively, across all functioning days. However, it was not worn daily. Subjects elected not to wear the DEKA Arm an average of 6.1 days even when it was functioning and were unable to wear it because of residual limb problems an average of 0.6 days. On days that it was worn, wear time was an average of 4.2 h and use time was 2.4 h.
Characteristics of 17 participants.
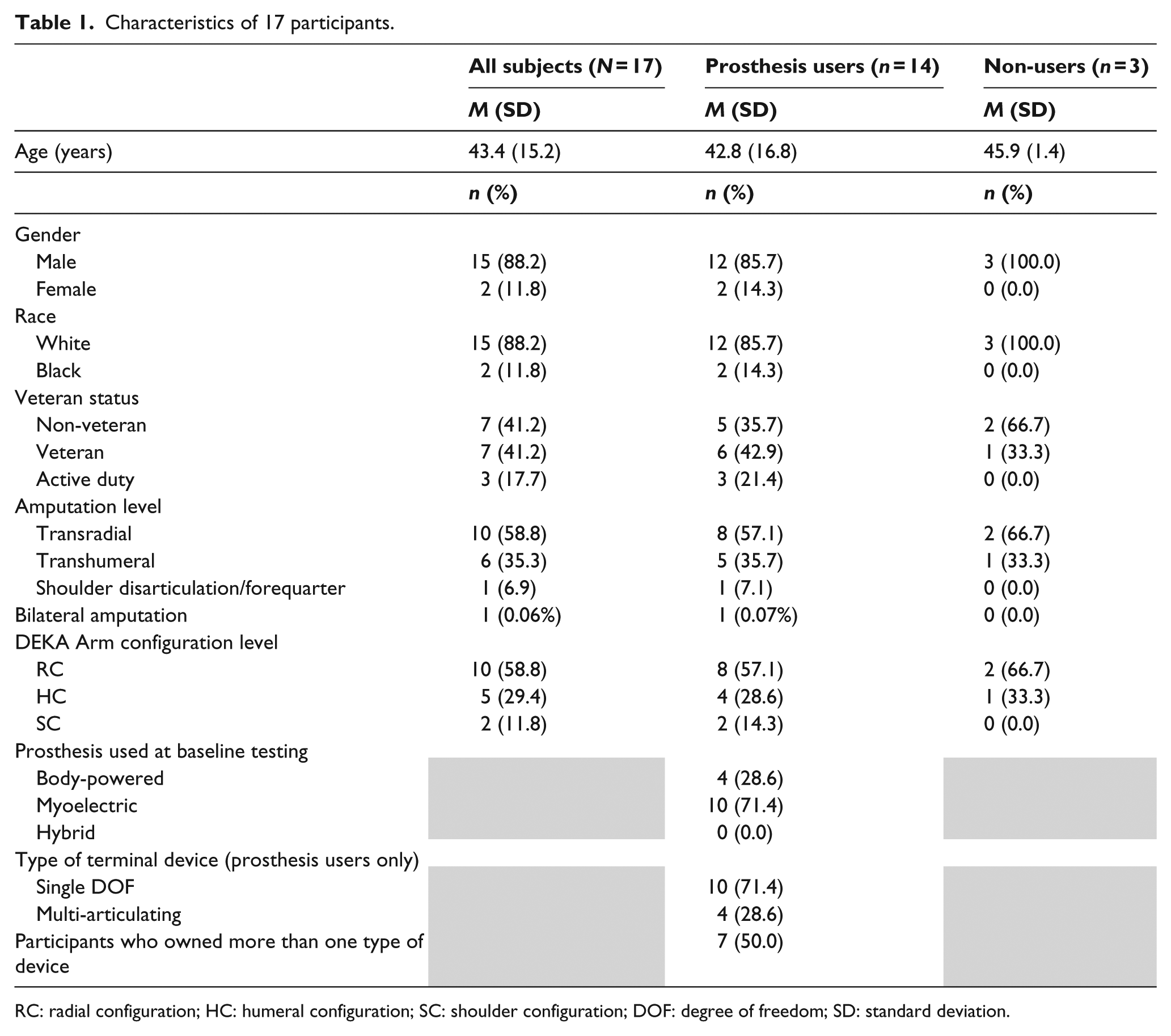
RC: radial configuration; HC: humeral configuration; SC: shoulder configuration; DOF: degree of freedom; SD: standard deviation.
Usage patterns at baseline and last 4 weeks of home use as recorded in diary days.
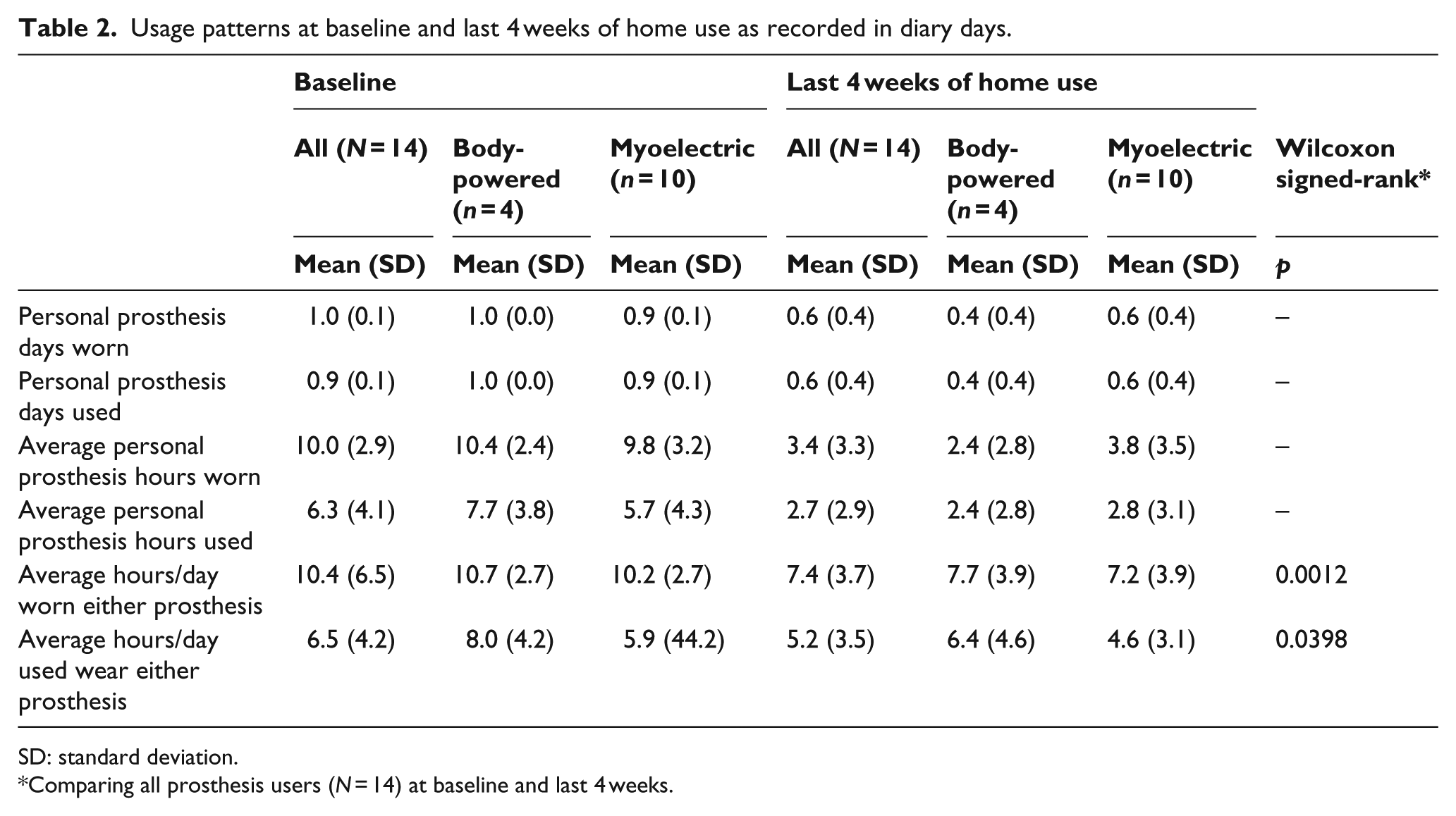
SD: standard deviation.
Comparing all prosthesis users (N = 14) at baseline and last 4 weeks.
Usage patterns of the DEKA Arm during last 4 weeks of home use.
SD: standard deviation.
Wilcoxon rank-sum tests indicated that prosthetic users did not significantly differ from non-users (p > 0.05) in the amount of time that they reported wearing or using the DEKA Arm in the last 4 weeks of the home (Table 3). Prosthesis users reported that they wore at least one type of device on 91% of Diary Days on average as compared to non-prosthesis users who used the DEKA Arm on 64% of Diary Days (data not shown, W = 10.5, z = –2.13, and p = 0.03). Prosthesis users used both the DEKA Arm and their own personal prosthesis on 46% of Diary Days (data not shown).
Qualitative results
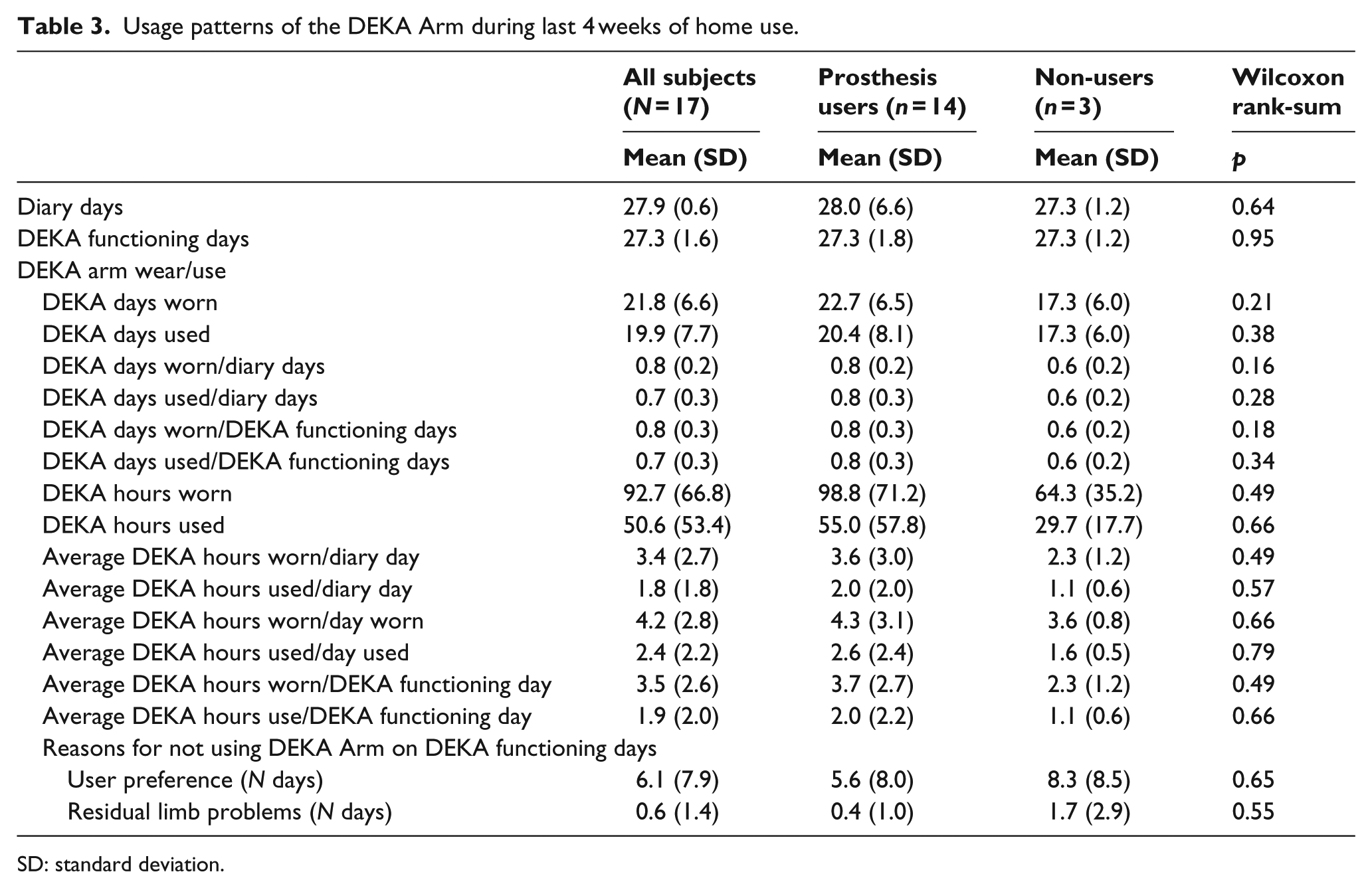
Of the participants, 86% reported new activities that could be done with the DEKA Arm, 100% of body-powered users, and 80% of myoelectric users; 36% reported that they could do activities with their personal prosthesis that they could not do with the DEKA Arm, 100% of body-powered users, and 10% myoelectric users (Figure 3(a)).
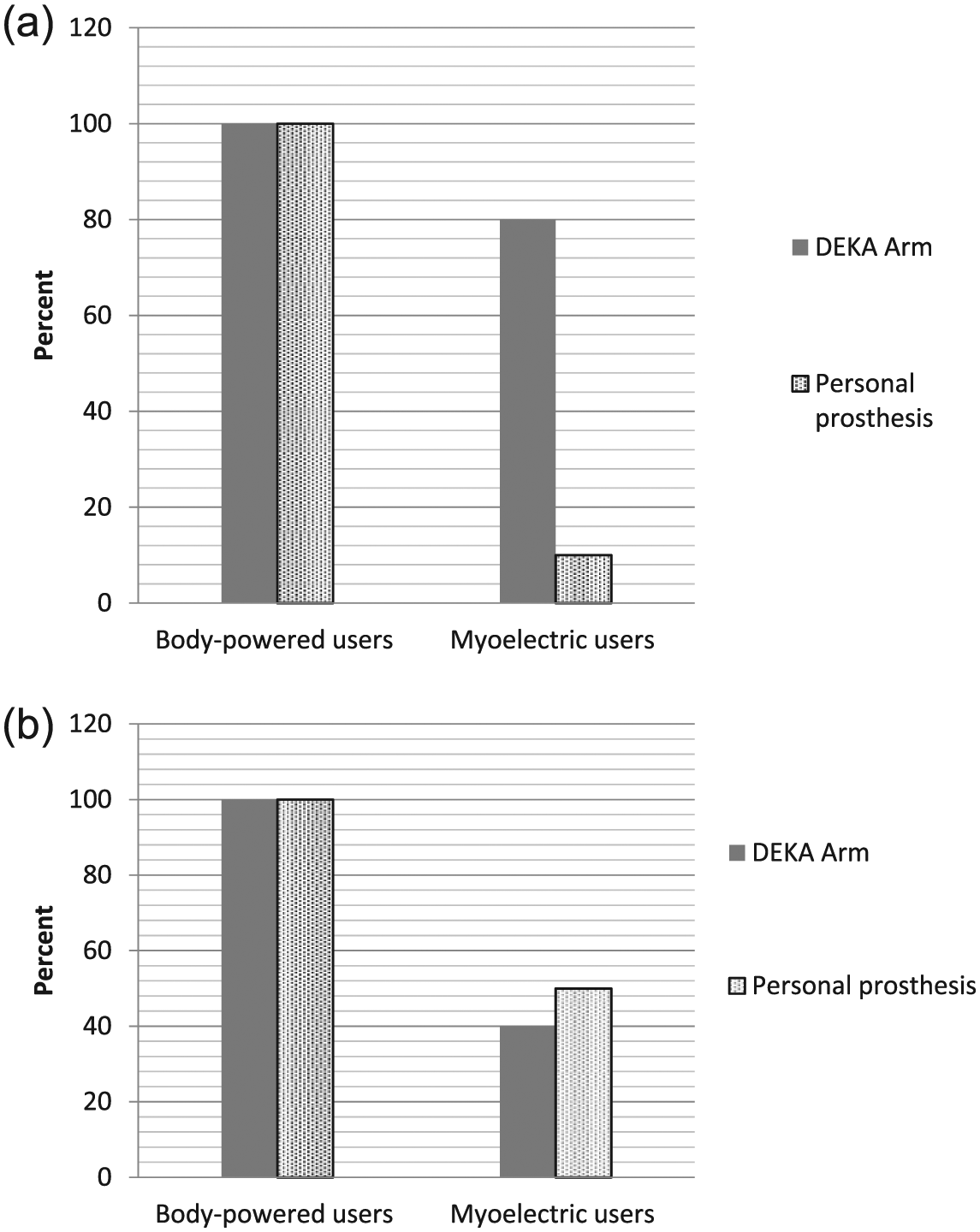
(a) Specific activities that could be performed with one type of prosthesis only and (b) preference for type of prosthesis for specific activities.
Activities that participants reported that they were able to do with the DEKA Arm but not with their personal prosthesis and vice versa were grouped into 10 categories. Activities that could be done exclusively with the DEKA Arm were categorized as self-care, eating/drink, and food preparation. Activities that could be done only with the personal prosthesis were classified as using powered/dangerous equipment and activities that would get the prosthesis wet (examples in Table 4).
Examples of activities participants could do with one prosthesis but not the other.

When asked whether there were any activities they preferred to do with their personal prosthesis rather than the DEKA Arm, 64% said “yes,” 100% of body-powered users, and 50% of myoelectric users (Figure 3(b)). The percentage who said “yes” they preferred to do specific activities with the DEKA Arm was 57%, 100% of body-powered users, and 40% of myoelectric users.
All body-powered users listed some activities that they preferred doing with the DEKA Arm and some activities that they preferred doing with their personal prosthesis. Whereas about half of myoelectric users expressed a preference for each type of device. The most frequent types of activities for which the DEKA Arm was preferred were eating, drinking, food preparation, and using tools. The most frequently listed activities for which the personal prosthesis was preferred were using powered/dangerous equipment, yard/farm work, and activities that would get the prosthesis wet (examples in Table 5). All body-powered users, but only 10% of myoelectric users, listed activities that they preferred doing with each type of device (Figure 4). Of the myoelectric users, 30% only listed activities that they preferred doing with their own device, while 20% only listed activities that they preferred doing with the DEKA Arm.
Example of prosthetic preference for specific activities (amputation level and DEKA Arm configuration level).

RC: radial configuration; HC: humeral configuration; SC: shoulder configuration; TR: transradial; TH: transhumeral.
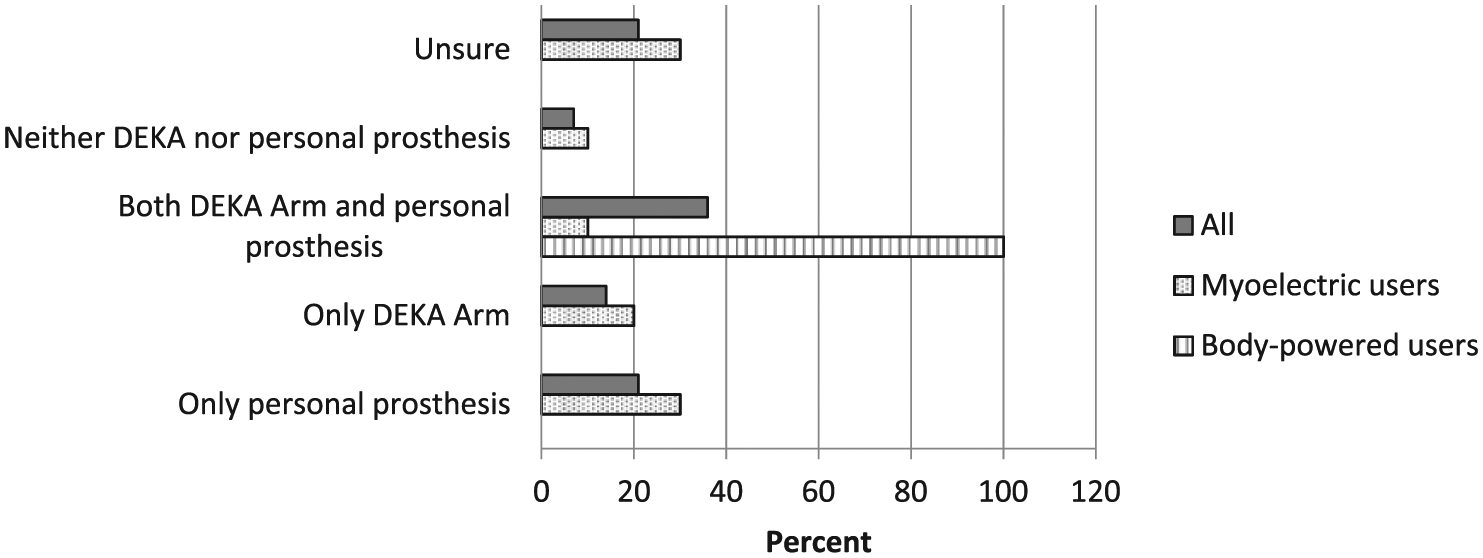
Preferences for one type of prosthesis for specific activities by type of personal prosthesis used.
Discussion
We reported on hours of wear and use of the DEKA Arm during a device home trial and compared patterns of usage for participants who did and did not use a personal prosthesis. Participants who had a personal prosthesis continued to use it during the home trial, albeit for fewer average hours per day, and for only 60% of days. These results suggest that the DEKA Arm supplements, but does not substitute for, a personal prosthesis.
The DEKA Arm was worn on average for 81% of the days that it was functioning. We found that the average hours that any prosthesis (DEKA or personal prosthesis) was worn and used during the study was significantly less than the personal prosthesis at baseline. The average hours per day that participants wore a functioning DEKA Arm was only 3.5 h, while average use was 1.9 h. This finding suggests that for some, the wear time tolerance was lower for the DEKA Arm as compared to their personal prosthesis. This may be attributable, in part, to fatigue from the weight of the DEKA Arm. Prior analysis showed that over 80% of study participants trained in the laboratory preferred the weight of their own devices to the DEKA Arm. 29
We did not observe differences between prosthesis users and non-users in days of DEKA Arm use, average hours per day that the DEKA Arm was worn or used on days that it was used. Although we found that participants chose not to wear their DEKA Arm an average of 6 days during the last 4 weeks and could not wear a prosthesis due to residual limb problems 0.6 days, these figures are consistent with reports that only 50% of prosthesis users use their device daily 15 and that prosthesis users used their devices only 71%–80% of days/month.8,17 Our sample’s prosthesis use at baseline (average 10 h/day) was within the ranges reported in earlier studies.7,8,14,17
Despite the modest amount of DEKA Arm wear and use, our findings suggest that while there are functional advantages of the DEKA Arm, there are functional advantages of other types of devices as well. This finding is consistent with earlier reports on myoelectric and body-powered device owners that found that those with more than one type of device use them selectively to meet specific functional needs. 5 Together, these findings strongly suggest that given the limitations of each type of prosthesis, persons with upper limb amputation would be best served and would be able to perform the widest range of activities if they had several types of devices.
Limitations
There are limitations to the use of self-reported data used in our analyses. Self-report diaries may be subject to recall bias and thus may not be accurate for either the personal prosthesis or the DEKA Arm. Although we have no method to evaluate the accuracy of participant self-report for the personal prosthesis use, a separate analysis using data logged from the onboard data logging feature in the DEKA Arm from a subset of the participants analyzed in this study corroborates the finding in this study that participants used the DEKA Arm for only short periods per day. That study reported that the DEKA Arm was powered on for a median of 1.1 h/day (interquartile range: 0.3–2.1). Median hours of powered on time as gauged from data logs was substantially lower than the hours of use reported in diary data. This finding is not entirely surprising, given that hours powered on and hours of use (which may include passive use) are different metrics and would not be expected to be fully consistent.
Participants were only asked to report the hours of use in their diaries, but were not asked at the end of their home study participation if they thought that they wore their prostheses more or less than they did prior to beginning the study.
Findings, particularly those comparing prosthesis users to non-users, should be interpreted cautiously due to limitations of very small sample size. Additional analyses with larger sample sizes are warranted. Our study examined patterns of wear and use during the last month of a home trial; however, we do not know whether similar patterns would continue given additional months of home use. Another limitation is that we did not explore all factors related to wear time. We recognize that other factors related to device satisfaction (e.g. perceived weight, ease of donning/doffing, and appearance) are likely to affect usage. 29 Additional studies are needed to examine the relationships between aspects of device satisfaction and hours of use.
Conclusion/implications
This study reported on the pattern of usage of the DEKA Arm and the personal prosthesis during a study of home usage. For those with a personal prosthesis, we conclude that the DEKA Arm supplements, but does not substitute for that device. Those who had a personal prosthesis continued using it but for fewer days and hours/day, and the overall hours/day of any prosthesis (DEKA or personal prosthesis) wear and use were reduced as compared to baseline. Participants wore the DEKA Arm during 81% of the days that it was functioning. On days that it was worn, it was worn and used an average of 4.2 and 2.4 h/day, respectively. Most participants reported activities that could be done only with the DEKA Arm. Findings have implications for prescription and support the practice of prescribing multiple types of devices to maximize activity and participation.
Footnotes
Acknowledgements
The view(s) expressed herein are those of the author(s) and do not reflect the official policy or position of the US Government.
Author contribution
All authors contributed equally in the preparation of this manuscript. If the authors provide any alternative wording, it is fine to keep that instead. Please let me know if you have any questions.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The information in this manuscript does not necessarily reflect the position or policy of the government; no official endorsement should be inferred.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by Rehabilitation Research and Development Service (VA RR&D A9226-R).
