Abstract
Background:
Environmental electromagnetic fields influence biological systems. Evidence suggests these have a role in the experience of phantom limb pain in patients with amputations.
Objectives:
This article followed a previous study to investigate the effect of electromagnetic field shielding with a specially designed prosthetic liner.
Study design:
Randomised placebo-controlled double-blind crossover trial.
Methods:
Twenty suitable participants with transtibial amputations, phantom pain at least 1 year with no other treatable cause or pathology were requested to record daily pain, well-being, activity and hours of prosthetic use on pre-printed diary sheets. These were issued for three 2-week periods (baseline, electromagnetic shielding (verum) and visually identical placebo liners – randomly allocated).
Results:
Thirty-three per cent of the recruited participants were unable to complete the trial. The resulting N was therefore smaller than was necessary for adequate power. The remaining data showed that maximum pain and well-being were improved from baseline under verum but not placebo. More participants improved on all variables with verum than placebo.
Conclusion:
Electromagnetic field shielding produced beneficial effects in those participants who could tolerate the liner. It is suggested that this might be due to protection of vulnerable nerve endings from nociceptive effects of environmental electromagnetic fields.
Clinical relevance
Electromagnetic field shielding with a suitable limb/prosthesis interface can be considered a useful technique to improve pain and well-being in patients with phantom limb pain.
Background
There are many complexities in the pathway from nociception to the appreciation of pain, 1 and this is particularly true of the experience of phantom pain where the limbs or organs in which the pain was represented no longer exist in the physical body.
Phantom limb pain (PLP) is a well-established phenomenon, occurring between 50% and 80% of patients undergoing amputation. 2 Although there is still some belief that it is a psychosomatic effect, related to a disorder of the personality or ‘grieving’ process of the sufferer, there is little empirical evidence for this. More recently, neurological changes in the brain and spinal cord have been recognised, 3 although the story is by no means complete at the present time. Numerous studies report differences in the antecedent factors for PLP4,5 and these clearly demonstrate that there are many co-existing and mutually influencing factors.
A survey by Giummarra et al. 6 in which 264 participants who had undergone amputation completed a structured questionnaire to explore the triggers of phantom phenomena including pain found that 20% reported PLP in response to changes in the weather, particularly storms. Lightning storms are known to affect the naturally occurring environmental electromagnetic fields (EMFs) which themselves are known to influence pain sensitivity and analgesia. 7
Other authors have also noted the effects of EMFs in the very low frequency range (0–100 kHz), termed VLF – atmospherics or spherics, which are generated by lightning and are thought to influence biological systems. 8 In the specific area of PLP, Pelz and Swantes 9 found a significant correlation between daily pain reports in a sample of 178 patients and spherics activity. There was also a significant correlation between the number of thunderstorms and the number of PLP complaints over a 4-year period. These findings might relate to the patients’ reports described by Giummarra et al. 6
There is no clear evidence that low-frequency EMFs are harmful to health, but sensitivity varies and thunderstorms produce low-frequency EMFs at a higher rate than are normal in the natural environment. 10 While the effects of EMFs or spherics might not be noticeable in the intact nervous system, nerve fibres in patients’ residual limbs after amputation are, by definition, compromised if not by the original injury then by the operation itself. This may change the sensitivity of the nerve endings to atmospheric variation.
The mechanism of these effects is poorly understood, but Adey suggests that environmental EMFs travel through the intercellular space in living organisms where they affect the passage of chemical messengers such as hormones and neurotransmitters. By this means, it is possible that EMFs increase the permeability of cell membranes and allow enhanced influx of calcium ions. 11 These are widely recognised to be involved in nociception and if they are enabled to penetrate neuron cells, they increase pain sensitivity by diminishing the effect of endogenous opioid activity. 12
Treatments for PLP are numerous and not all are effective in the long term. 13 It is possible that attempting to protect the nerve endings after amputation by reducing the influence of environmental EMF’s might be a useful approach. Clement and Taunton 14 describe the effect of treating PLP with Farabloc™ – a fabric made of a woven mesh of stainless steel and nylon thread that has been shown to have electromagnetic (EM) shielding properties. A double layer of this fabric covering the residual limbs of 34 patients for more than 4 h a day was reported to reduce the intensity of PLP significantly better than placebo.
Kern et al., 15 working on the same principle, reported the results of a double-blind randomised crossover trial in which 22 patients with lower limb amputations used a specially designed silicon liner as the interface between their skin and the artificial limb. This liner was manufactured to include a woven fabric containing conductive filaments, which provided EM shielding. A placebo liner (PL) was also specially made to resemble the experimental liner (termed verum) in all respects except that the interwoven material was nonconductive and therefore could not provide the EM shield. After 2 weeks’ use of each liner, the results were strongly in favour of the experimental fabric in reducing phantom pain and improving general well-being.
This study aimed to replicate this experimental design in a new cohort of patients with PLP who did not regularly use a silicon liner to investigate the specific effect of the EM shielding material relative to placebo.
Methods
National and local ethics approval were sought and granted. The study design was a randomised placebo-controlled double-blind crossover trial in which three 2-week periods of recording of relevant parameters were collected during (1) baseline (participants’ existing prosthetic technology); (2) while using liner A and (3) while using Liner B, which were randomly assigned by the manufacturer to be the experimental or PLs.
The liners were supplied by the German company medi and utilised Umbrellan® technology, which provides electromagnetically shielding properties. 15 Two liners were used, one involving the magnetic fibre woven throughout. This was the verum liner (VL). The other – the PL – consisted of nonconductive material, which did not provide the EM shielding effect. However, in order to maintain blindedness between the VL and PL, the extreme distal end of the PL was made of Umbrellan as without this, the difference would have been noticeable. 15
The measures used were printed on daily diary sheets and were 10-point Numerical Rating Scales (NRSs) with anchor points 1 (no pain) and 10 (worst pain). Participants were asked to circle a number on the scale five times a day (early morning, mid morning, afternoon, evening and night) to record their level of PLP.
In response to the question ‘How did you feel in yourself today?’, participants recorded well-being once a day on an NRS with anchor points 1 (well) to 10 (poorly). A question ‘How much did pain limit your activity today?’ was answered with three response options – ‘less than usual’ (scored 1), ‘same as usual’ (scored 2) and ‘more than usual’ (scored 3). Participants were also asked to record for how many hours they had worn the liner each day. The mean number of hours per day was used for data analysis. Participants were recruited from routine medical or prosthetists’ clinics in a Prosthetic Rehabilitation Unit attached to a specialist Orthopaedic Hospital.
In order to be eligible for inclusion, participants must
- have been over 16 years of age,
- have had an amputation at the transtibial level,
- have had phantom pain, resistant to any other treatment, which had persisted for at least 1 year,
- have had no open wounds or unhealed skin lesions,
- have had no treatable reason for the pain (e.g. neuroma),
- not have been entered into any other treatment trial,
- have been on stable doses of any relevant medication, such as anticonvulsants for neuropathic pain or psychotropic medication,
- have been able to comprehend sufficient English and able to give consent,
- have been willing and able to fill in the pain diary sheets and to attend prosthetic clinics for monitoring and provision of liners as necessary.
All participants were examined by a physician and a senior prosthetist. They were given a sheet of information including their right to withdraw at any time without jeopardising their future care and were asked to provide written consent before being admitted to the trial. Measurements of their residual limbs were made so that the appropriate size and thickness of liners were ordered, custom made from the manufacturer.
The participants then spent 2 weeks filling in their daily diaries while using their existing artificial limbs. After this period, and when the liners had arrived, a cast was made for each participant for a new socket that would accommodate the medipro® silicon liner, as these were not routinely used.
The first liner was then issued and recorded as liner A. For a further 2 weeks, the participants filled their daily diary sheets and then returned to hand in their diaries and be issued with liner B. After 2 weeks with this, the trial was complete. When all the participants’ data had been entered for analysis and no more people could be recruited since the available supply of placebo material had been allocated, the manufacturers were asked for the code that indicated which participants had received liner A or B as the VL.
Data analysis was undertaken by SPSS. 16 Improvement from baseline was defined as one or more NRS points of reduction in mean and maximum pain, one or more NRS points of increase in well-being, one point reduction in activity limitation and at least 1 h of increase in the mean time the prosthesis was worn.
Statistical analyses were performed by paired t-tests that were used for the difference between baseline and VL, baseline and PL and VL and PL. An order effect to test whether there was a difference depending on the order of presentation of VL and PL was analysed by calculating difference scores between baseline and VL and between baseline and PL for all variables. These scores were then treated with independent samples t-tests between group 1 (VL first) and group 2 (PL first). Larger or smaller percentages of participants who improved from baseline under VL than PL were analysed by calculating the difference in change scores in both conditions.
Results
All variables were assessed for skewness and the results were found to lie between −1 and +1, suggested by Bulmer 17 to be the acceptable limits of a normal distribution. The data from repeated scoring of ordinal measures approximate to continuous measures 18 so parametric analysis was deemed appropriate.
In this exploratory series of patients, 20 participants (21 limbs), who all had amputations at the transtibial level, were available for recruitment. All participants experienced continuous pain that varied to a greater or lesser extent throughout the day but never disappeared. In the end, however, five could not tolerate the effects of the silicon on their skin. They discontinued the trial while using liner A.
According to the manufacturer’s randomisation code, these were two cases in which the liner was VL and three in which it was PL. One participant died during the baseline period. These data are shown in Figure 1.
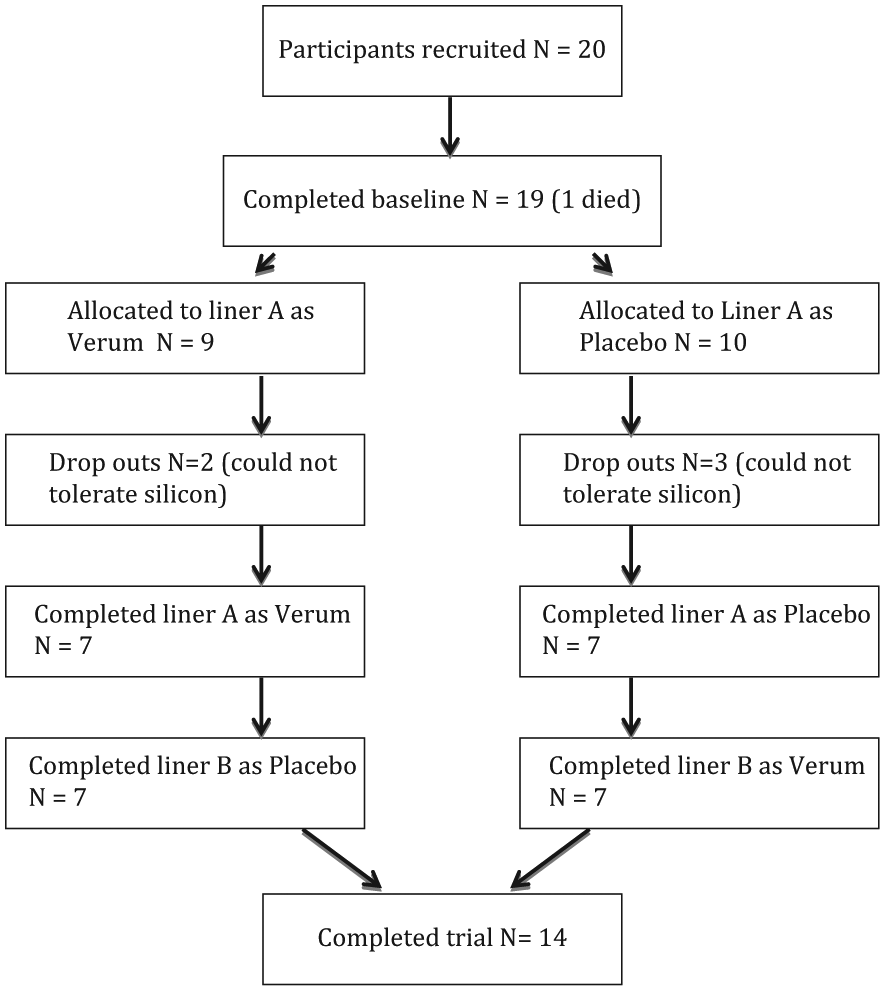
CONSORT diagram of participant allocation.
The remaining sample consisted of 14 people, of whom 10 (71%) were men. Six people (43%) underwent amputation as a result of vascular pathology, including diabetes, five (36%) because of infection and three (21%) because of trauma. The mean age was 60.07 years (standard deviation (SD): 12.05 years). The mean duration of PLP was 4.29 years (SD: 3.29 years). Characteristics of the participant sample are summarised in Table 1.
Characteristics of participants with phantom pain following transtibial amputation.
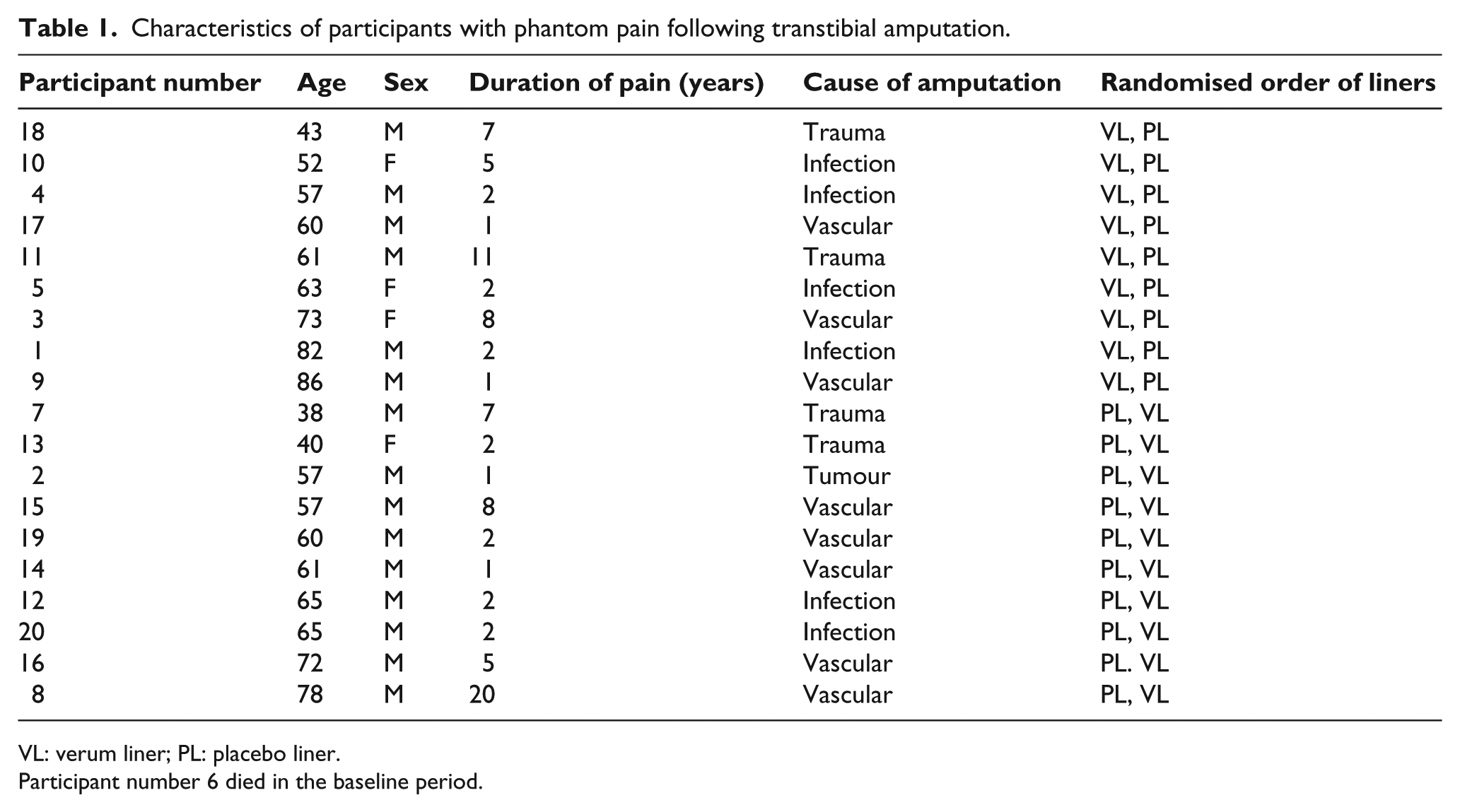
VL: verum liner; PL: placebo liner.
Participant number 6 died in the baseline period.
Mean daily pain
The test for change in mean pain compared to baseline did not show a statistical significance difference compared to baseline in either VL (p = 0.10) or PL (p = 0.11) conditions.
Maximum pain
The reduction in maximum pain from baseline was statistically significant different in VL (p = 0.03). This was not statistically significant in PL (p = 0.13).
Well-being
The test for increase in well-being showed a statistically significant difference from baseline for VL (p = 0.02). This was not statistically significant for PL (p = 0.06).
Activity limitation
The test for improvement in activity limitation was not statistically significantly different from baseline in either VL (p = 0.09) or PL (p = 0.20).
Time worn
This variable did not reach a statistically significant difference from baseline in either VL (p = 0.38) or PL (p = 0.61). These data are summarised in Table 2.
Means and standard deviations, t scores and values of p for differences from baseline for all variables.
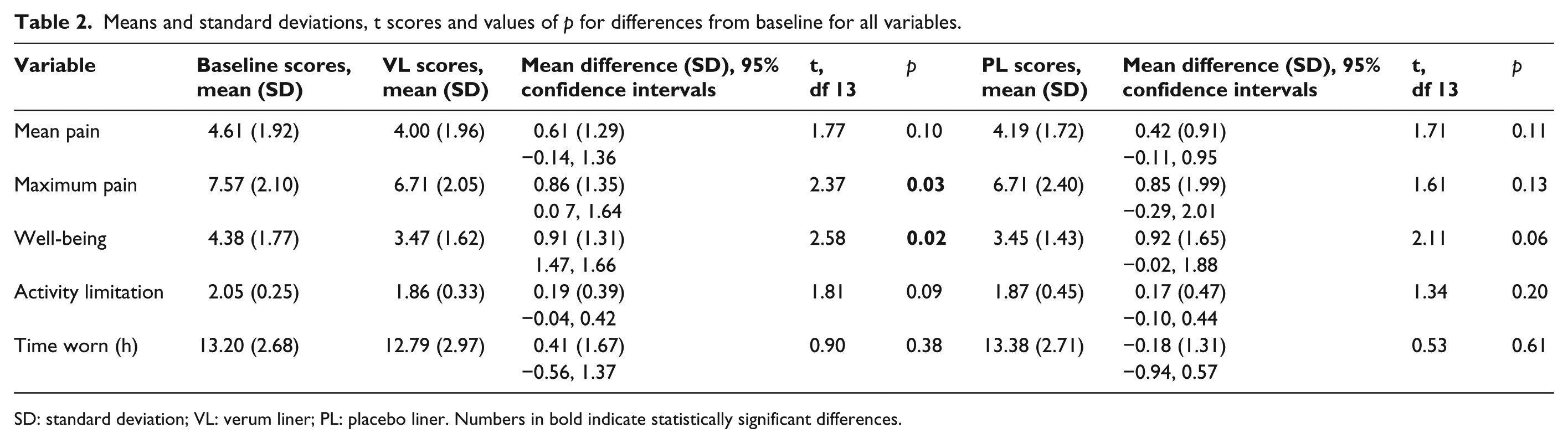
SD: standard deviation; VL: verum liner; PL: placebo liner. Numbers in bold indicate statistically significant differences.
The plots of individuals’ scores for maximum pain and well-being (after Senn 19 ) are shown in Figures 2 and 3.
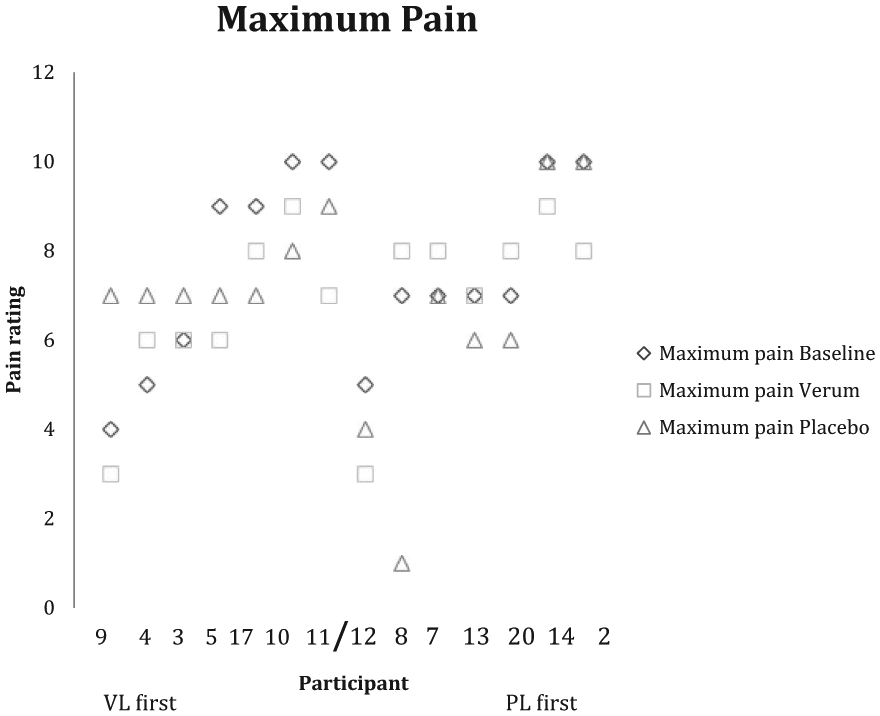
Maximum pain. Baseline, verum and placebo scores of individual participants.
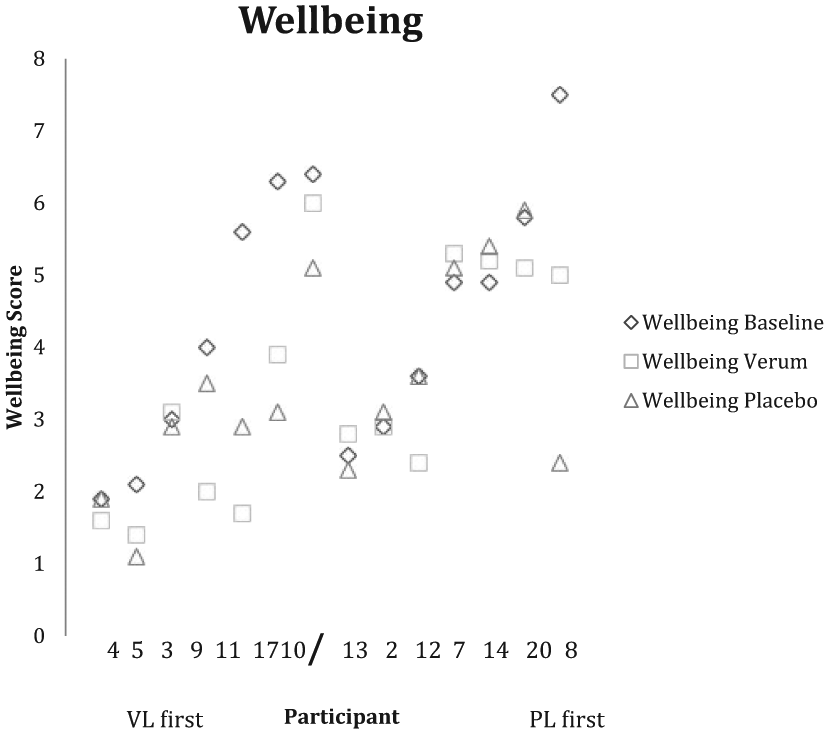
Well-being. Baseline, verum and placebo scores of individual participants.
Comparison of VL and PL
There were no statistically significant differences between VL and PL on any variables (p > 0.05 in all cases).
Order effect
Calculations of change scores from baseline to VL and baseline to PL were made and analysed by independent t-tests between group 1 (VL first) and group 2 (PL first). This test was not statistically significant for any variable (p > 0.05 in all cases), showing that the order of use of the liners did not affect the outcome.
A higher percentage of participants improved from baseline with VL compared to PL on all variables except time worn. These data are shown in Table 3.
Percentage of participants who improved from baseline, VL and PL conditions compared.
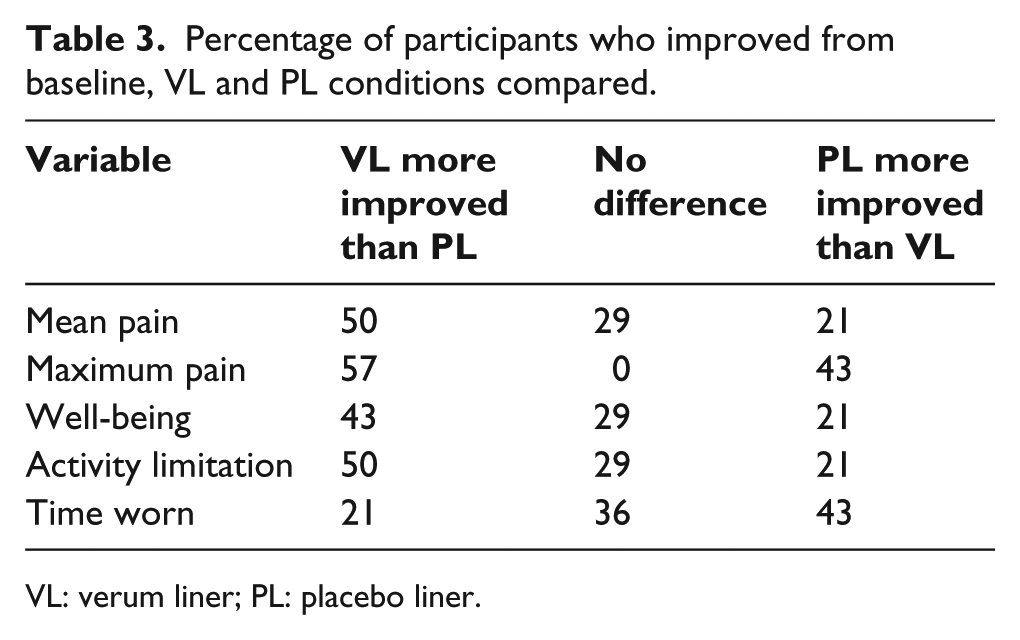
VL: verum liner; PL: placebo liner.
Discussion
The results of this analysis show results in favour of the magnetic shielding liner versus the placebo, and although this is not as strong an effect as found by Kern et al., 15 there are some notable findings. Maximum pain and well-being were statistically significantly better than baseline in the VL condition. It is also clear that a higher percentage of participants improved from baseline on pain, well-being and activity limitation using VL than PL.
There are important reasons for the difference between these findings and those of Kern et al. 15 In the first place, because of the skin problems in response to the silicon encountered by more than a quarter of our sample population, the study ended up underpowered. With a larger sample, it is likely that our results might have shown a difference between the performance of VL and PL more clearly. This could not be achieved because of the availability of the fabric for the PL.
It is also true that the participants in the original study were already using liners produced by the manufacturer and so did not require the creation of new, specially produced sockets to accommodate the silicon liners as was the case with our study. This also resulted in a limitation of suitable participants.
Senn 19 states that a problem with crossover trials may be ‘carry over’ of the effect of a treatment in one period that may influence the effect of the next treatment. As Kern et al. 15 state that no such problem was demonstrated in their study after using logistic regression to test for crossover effects, we did not specifically test for this between the results of the VL and PL conditions so we cannot be certain that there was no interaction.
Nevertheless, in spite of our small N, we were able to demonstrate the potential benefit of EM shielding beyond what would be expected from a purely placebo effect as VL showed statistically significant improvement from baseline in maximum pain and well-being, whereas PL did not show a significant difference in any variable. More participants improved from baseline in pain, well-being and activity limitation under VL than PL conditions. Colloca et al. 20 have recently reviewed the psychological and neurobiological mechanisms of placebo analgesia and conclude that there is a learning phenomenon in which people learn to produce a benefit by verbally induced expectations, contextual conditioning or social learning. Since our study was specifically designed to minimise the chances of participants discriminating between VL and PL, there would have been no reason to identify any differences between the liners if a placebo effect explained the overall results. This hypothesis was not upheld. These authors further state that placebos given after pre-exposure to active analgesics mimic the actions of the pharmacological agent by associative learning and these are enhanced by the presence of a relevant clinician. 20 Our participants had the same treating team throughout their trial period and showed pain reduction and well-being improvement irrespective of the order of presentation of the liners. Therefore, no associative learning from VL to PL could be said to have taken place. While it is true that placebo responses probably contribute to the overall effect of medical interventions, this study has attempted to control for these and to demonstrate the added effect of the active EM shield.
The overall explanation as to why EM shielding is effective in reducing PLP is still unclear and requires greater specific experimental investigation. The effect of calcium ions in the nervous system is known to influence nociceptive responses and there is evidence to suggest that EM environmental effects modulate membrane permeability. 11 Animal experiments have shown that mice exposed to a time-varying magnetic field displayed significantly enhanced nociceptive sensitivity and reduced levels of morphine-induced analgesia. 21 This effect was also produced during a geomagnetic storm – a disturbance of the ambient EMF by increased levels of charged atmospheric particles. 22 Prato et al. 23 demonstrated that mice exposed to magnetic shielding in a specially constructed metal alloy box showed evidence of an increased pain threshold.
Given the findings of Giummarra et al. 6 and Pelz and Swantes 9 on the increased incidence of PLP during disturbances of EMFs in thunderstorms, the evidence might suggest that by reducing this effect and preventing the influx of extra calcium into the nerve cells, EM shielding can render the pain threshold less vulnerable and improve the experience of patients with PLP.
Conclusion
This study has shown that the use of an electromagnetically shielding liner worn with the prosthesis achieved a statistically significant reduction in maximum pain and an increase in well-being in patients suffering PLP, whereas the PL did not. On four of five variables, more participants improved from baseline under verum than placebo conditions.
Footnotes
Acknowledgements
The authors acknowledge with gratitude the company medi who supplied the verum and specially manufactured placebo liners. Thanks are due to the prosthetists who meticulously followed the treatment protocol for all the participants and to the participants themselves for conscientiously filling in their daily diaries and returning them promptly. Also to Erica Cook, statistician, for advice on the analysis of the results and to helpful anonymous reviewers of a previous draft of this report.
Author contribution
All the authors contributed equally in the preparation of this article.
Declaration of conflicting interests
The authors report no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
