Abstract
Background:
Phantom limb pain is reported in 50%–85% of people with amputation. Clinical interventions in treating central pain, such as mirror therapy, motor imagery, or virtual visual feedback, could redound in benefits to amputee patients with phantom limb pain.
Objectives:
To provide an overview of the effectiveness of different techniques for treating phantom limb pain in amputee patients.
Study design:
Systematic review.
Methods:
A computerized literature search up to April 2017 was performed using the following databases: PubMed, Scopus, CINAHL, MEDLINE, ProQuest, PEDro, EBSCOhost, and Cochrane Plus. Methodological quality and internal validity score of each study were assessed using PEDro scale. For data synthesis, qualitative methods from the Cochrane Back Review Group were applied.
Results:
In all, 12 studies met our inclusion criteria, where 9 were rated as low methodological quality and 3 rated moderate quality. All studies showed a significant reduction in pain, but there was heterogeneity among subjects and methodologies and any high-quality clinical trial (PEDro score ≤8; internal validity score ≤5) was not found.
Conclusion:
Mirror therapy, motor imaginary, and virtual visual feedback reduce phantom limb pain; however, there is limited scientific evidence supporting their effectiveness. Future studies should include designs with more solid research methods, exploring short- and long-term benefits of these therapies.
Clinical relevance
This systematic review investigates the effectiveness of mirror therapy, motor imagery, and virtual visual feedback on phantom limb pain, summarizing the currently published trials and evaluating the research quality. Although these interventions have positive benefits in phantom limb pain, there is still a lack of evidence for supporting their effectiveness.
Background
Phantom limb pain (PLP) is a painful sensation perceived in a part of the body which no longer exists. 1 The therapeutic approach for this kind of pain after amputation is complex.2,3 The “phantom sensation” is a common consequence after limb amputations, consisting of additional sensations of numbness, tickling, or muscle cramp in a non-existent limb. The type, frequency, and stability of perception can vary. 4 PLP is reported in 50%–85% of amputee patients. Although the effect of cortical reorganization on PLP is still unclear, clinical interventions on the central neural mechanism could lead to promising strategies for symptomatic management of central pain sensitization. 5 Previous studies have shown that pain can be reduced by mirror therapy, motor imagery, sensory discrimination, or virtual visual/optical feedback.2,3,5
Visual feedback therapy is a fairly new paradigm that allows patients to visualize and monitor real-time changes in the parameters of activity of the injured area. 6 Patients visualize their missing limb either using mirrors,7,8 virtual systems, or videos of the previously recorded intact limb. At the same time, they are encouraged to synchronize their phantom limb with the movements observed.9,10 Some investigators such as Mercier and Sirigu 10 have suggested that the success of the treatment may depend on the degree of participation of the patient.
Mirror visual feedback, termed as mirror therapy, was proposed in the early 1990s as a treatment to relieve PLP in amputees. Its use is also increasing for treating other types of chronic pain conditions. 11 Mirror therapy involves the patients using a mirror to see the reflection of the uninjured side, resulting in an illusion of function in the missing area.11,12 The mirror is placed in a box, allowing amputees to see their anatomical limb in the visual space occupied by their phantom limb.7,13 This intervention induces vivid sensations of movement from the muscles and joints of the affected limb, reducing phantom limb sensation and gaining motor control, when the residual limb is paralyzed.7,13 The movement of the unaffected limb induces a feeling of movement in the phantom limb, reducing phantom pain in some cases.2,3 Ramachandran et al. 3 have shown how the feedback provided by mirror therapy can improve voluntary control and pain relief of PLP. In fact, this therapeutic approach has been described as the most promising method for treating phantom pain. 14 However, the mechanisms underlying mirror therapy effect are not yet clear. 15 Some theories claim that this therapy produces a correction of imbalance between the motor and sensory systems;7,16–18 other authors state that the effectiveness of the technique lies in producing an increased focus on the painful limb, improving the perception of the limb itself. 19 In this sense, Selles et al. 20 have classified treatment with a mirror as a simple distraction therapy.
Motor imagery is another technique used to reduce this phenomenon, involving a complex cognitive, sensory, and perceptual process. Motor actions are activated from central nervous system at the level of working memory. This produces a mental representation of movement without any physical body movement. The voluntary recreation of scenes or tasks, called “mental practice,” can improve the motor performance of the individual. 21 The study conducted by Maciver et al. 22 reported that imagining the movement and perceiving sensations in the lost limb can result in significant pain relief. Motor imagery seems to be less effective for treating chronic pain than mirror therapy.8,23
Comparing those modalities, the technique of the mirror box requires the patient to remain with the head facing the mirror in a relatively fixed position and the body in the medial sagittal plane with respect to the mirror. 24 The patient has to ignore the intact limb, focusing on the reflected image of the phantom limb. However, this method can create a rather restrictive and provisional illusion. For this reason, immersive virtual reality has appeared as a possible solution to this problem. The movements of the intact limb are transformed into virtual limb movements in the space occupied by the phantom limb, giving a similar illusion to that of the mirror box, but without the limits imposed by the reflection box. 24 In the virtual environment, the illusion is plausible, irrespective of the patient’s orientation. 25 However, lower limb movements are usually not symmetrical in activities of daily living, except in some activities such as swimming. The mirror creates a symmetrical illusion; however, virtual reality can create a more appropriate image, more similar to the real asymmetrical movements from daily living. 26
The phenomenon of PLP is often detrimental to the patient’s quality of life. 4 Virtual visual feedback and mirror therapy seem to be promising treatments for relieving PLP, alone or in combination with motor imagery. 10 However, to our knowledge, the research on effectiveness of these approaches has been shown to be variable in methodology and findings. Therefore, the aim of this study was to evaluate the efficacy of mirror therapy, motor imagery, and virtual visual feedback for treating PLP after amputation.
Methods
Study protocol
A systematic review following the principles of the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement was performed.
Information sources and search strategy
We conducted a literature search using PubMed, Scopus, CINAHL, MEDLINE, ProQuest, PEDro (Physiotherapy Evidence Database), EBSCOhost, and Cochrane Plus databases. The following search categories were used: amput*, phantom limb, motor imagery, mirror therapy, and pain. Terms were established based on initial searches to find keywords (MeSH terms) with PubMed. Several combinations of the following search equation were used depending on the database (except for PEDro database): (amput* OR stump* OR residual limb OR amputation phantom) AND (mirror therapy OR virtual visual feedback OR motor imagery OR proprioceptive memory OR treatment phantom OR mirror therapy phantom) AND (decrease pain OR reduction pain). The search strategy for PEDro was: amput* AND pain.
Study selection
For the selection of potentially relevant studies, first, titles and abstracts were reviewed. The inclusion criteria were (1) studies published in English or Spanish since April 2017; (2) studies focused on amputees or stumps with PLP; (3) studies using mirror therapy, motor imaginary, or virtual visual feedback; (4) experimental studies; (5) randomized controlled trials (RCTs); (6) using the adult population; and (7) accessible in full text. The following exclusion criteria were established: (1) studies focusing on pre-amputation treatments, (2) duplicates in different databases, (3) case report studies, and (4) studies with a PEDro total score <3.
Data extraction and quality assessment
Items were searched and extracted by one reviewer. The reviewer registered information relating to identification of pain outcome measurements, methods (characteristics of the intervention and the control group, technique, description of interventions, and obtained results). The PEDro scale was used to evaluate the methodological quality of the included studies, selecting only articles scoring over 3 points. 27
The internal validity of each item was assessed by calculating the internal validity score (IVS).28,29 To calculate the IVS, the criteria 2, 3, 5, 6, 7, 8, and 9 from the PEDro scale were selected, since they have been reported to be representative of internal validity.28,29 Methodological quality was scored and interpreted as follows: 28 (1) studies with an IVS of 6–7 were considered to have a high methodological quality, (2) IVS of 4–5 were considered of moderate methodological quality, and (3) 0–3 points represent limited methodological quality.
To reduce inter-examiner bias, the quality of the studies was evaluated by three independent reviewers. A consensus method was used to discuss and resolve discrepancies between the PEDro scale scores (included in the “Results” section).
Data synthesis
The search results showed that current literature regarding effectiveness of these therapies on PLP after amputation contained heterogeneous information, precluding the performance of statistical analysis. It was not possible to group extracted and analyzed data, and the overall effect size could not be calculated. For these reasons, qualitative methods, recommended by the Cochrane Back Review Group, were applied,30,31 using the following levels of evidence for the synthesis of the data: (1) strong evidence for studies with multiple high-quality RCTs; (2) moderate evidence, multiple low-quality RCTs and one high-quality RCT; (3) limited evidence, one low-quality RCT; (4) conflicting evidence, incoherent findings in multiple RCTs; (5) no evidence; and (6) no use of RCTs.30,31
Results
Study selection
The electronic search in the different databases produced 3214 items from a total of eight databases: 92 studies from PubMed, 155 from Scopus, 9 from CINAHL, 93 from MEDLINE, 2766 from ProQuest, 24 from PEDro, 14 from EBSCOhost, 21 from Cochrane Plus, and 20 from gray literature.
After reading the title, abstract, and full text, 24 (0.75%) studies met the criteria. The studies with a methodology of case study and those with a PEDro score <3 were eliminated (k = 10),31–33 resulting in a total of 12 studies. Figure 1 shows the flowchart of the process of selection and exclusion of studies.
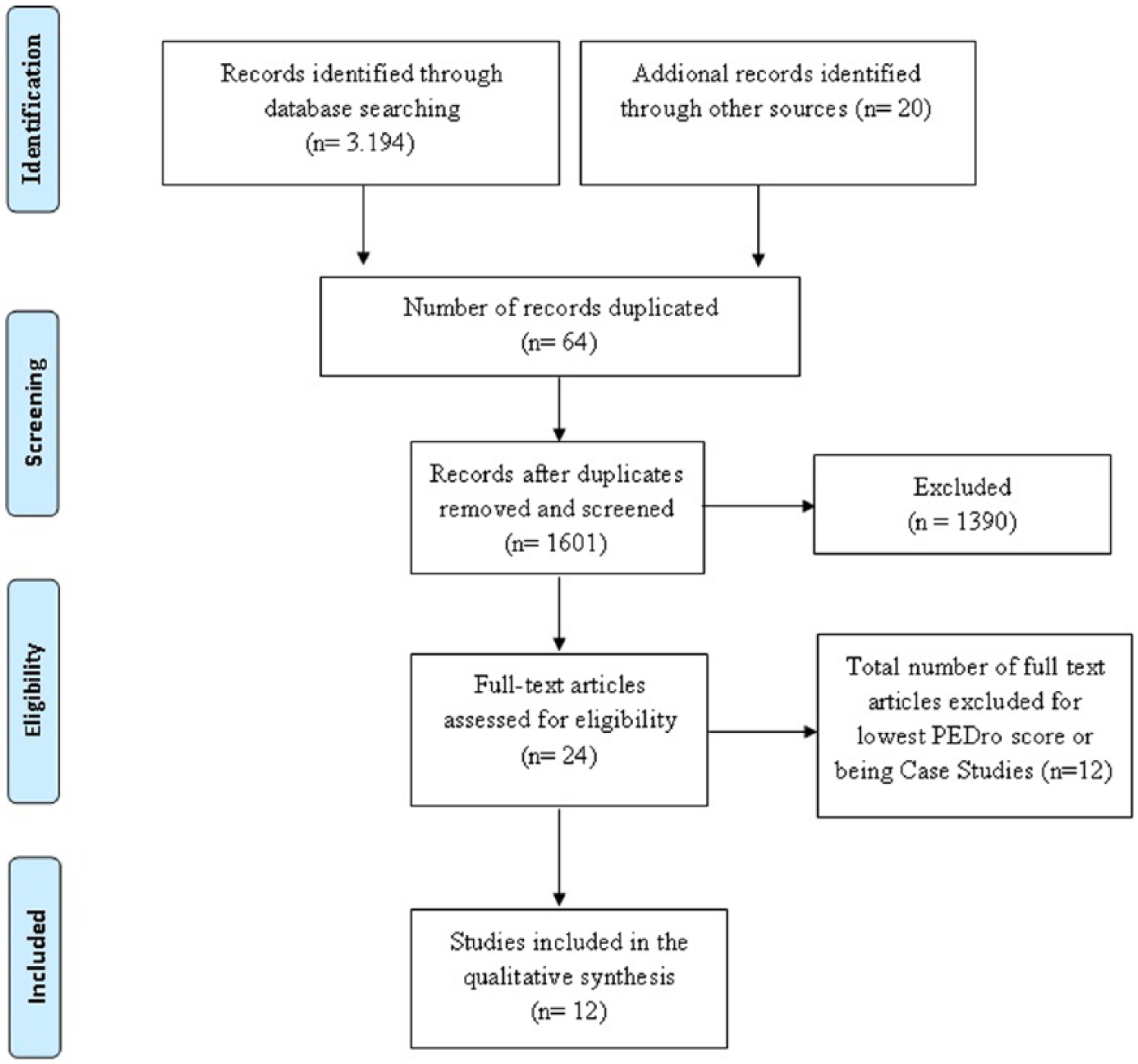
Flowchart according to the PRISMA system. Selection process.
Study characteristics
Table 1 summarizes the main information of the selected studies. The studies included in this review used 13 different measures of pain: Numerical Rating Scale (NRS),22,36 Nuclear Magnetic Resonance Imaging (NMRI), 22 PLP Questionnaire, 22 Visual Analog Scale (VAS),8,26,33,34,37,40 McGill Pain Questionnaire,26,34,35,41 West Haven–Yale Multidimensional PLP Inventory,33,38 Functional Magnetic Resonance Imaging (fMRI), 33 Universal Pain Score (UPS), 40 Brief Pain Inventory (BPI), 39 Patient Information Form, 42 Mirror Therapy Practice Follow-Up Booklet, 42 Pain Rating Index, 41 and Weighted Pain Distribution Scale. 41
Description of selected studies.
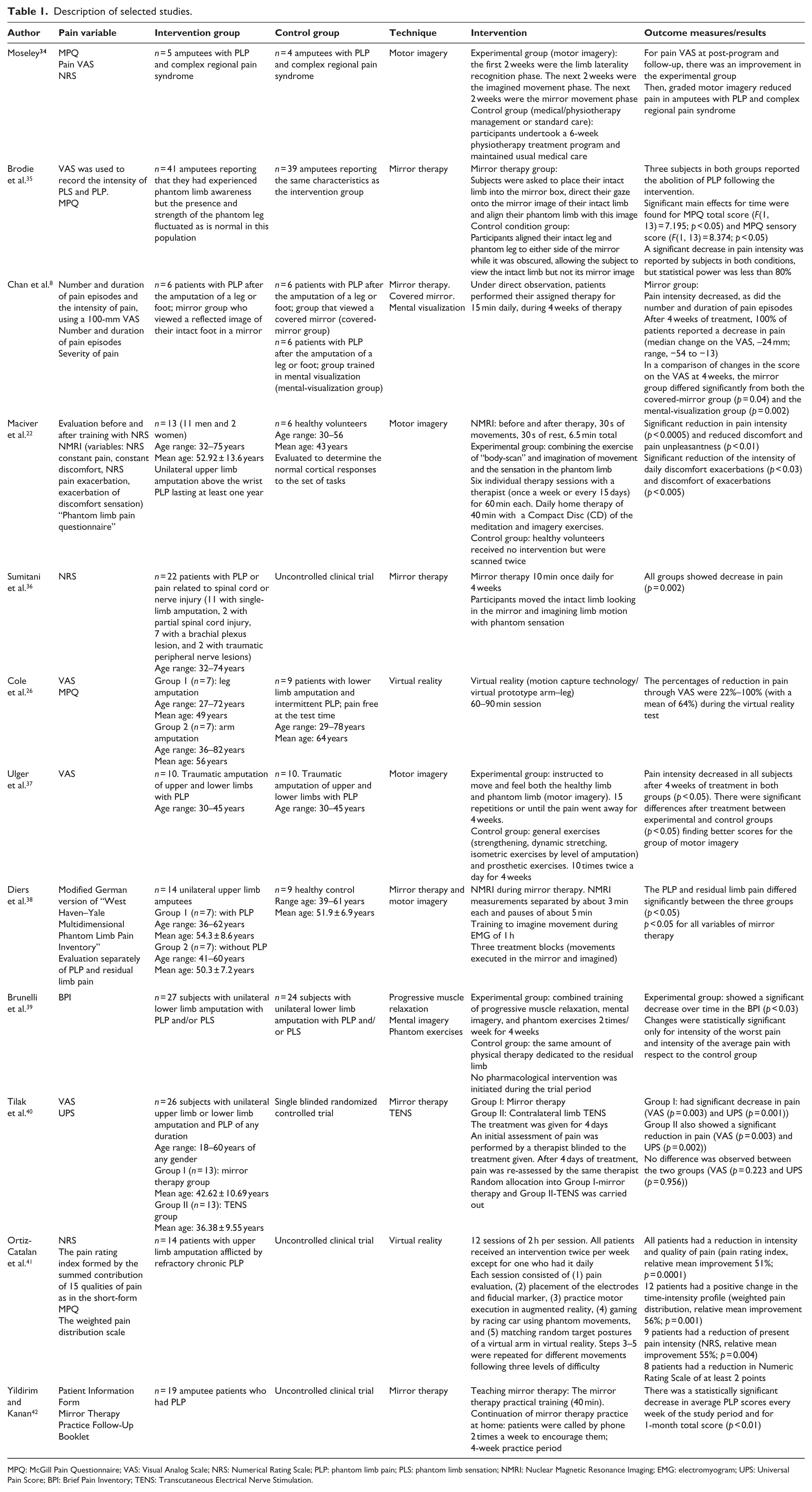
MPQ: McGill Pain Questionnaire; VAS: Visual Analog Scale; NRS: Numerical Rating Scale; PLP: phantom limb pain; PLS: phantom limb sensation; NMRI: Nuclear Magnetic Resonance Imaging; EMG: electromyogram; UPS: Universal Pain Score; BPI: Brief Pain Inventory; TENS: Transcutaneous Electrical Nerve Stimulation.
The largest sample of amputees in the experimental group was 41 patients, 35 and the lowest, 5 patients. 34 Four papers presented a sample of upper limb amputees,22,33,38,41 three studies used lower limb amputees,8,35,39 and six used a mixed sample of upper and lower limb.26,34,36,37,40,42
Study quality
Of the 12 studies included in this review, a PEDro score between 8 and 7 was achieved by only 2;34,40 4 studies gave a score of 6/108,35,39 or 5/10, 22 and 3 studies gave a score of 4/10.37,38,42 Finally, 6 studies showed a very low PEDro score (≤3).26,31–33,36,41 Regarding IVS, all selected studies had a limited to moderate methodological quality.8,22,26,36–42 Table 2 presents the methodological quality of the studies analyzed.
Methodological quality of the studies using the PEDro scale.
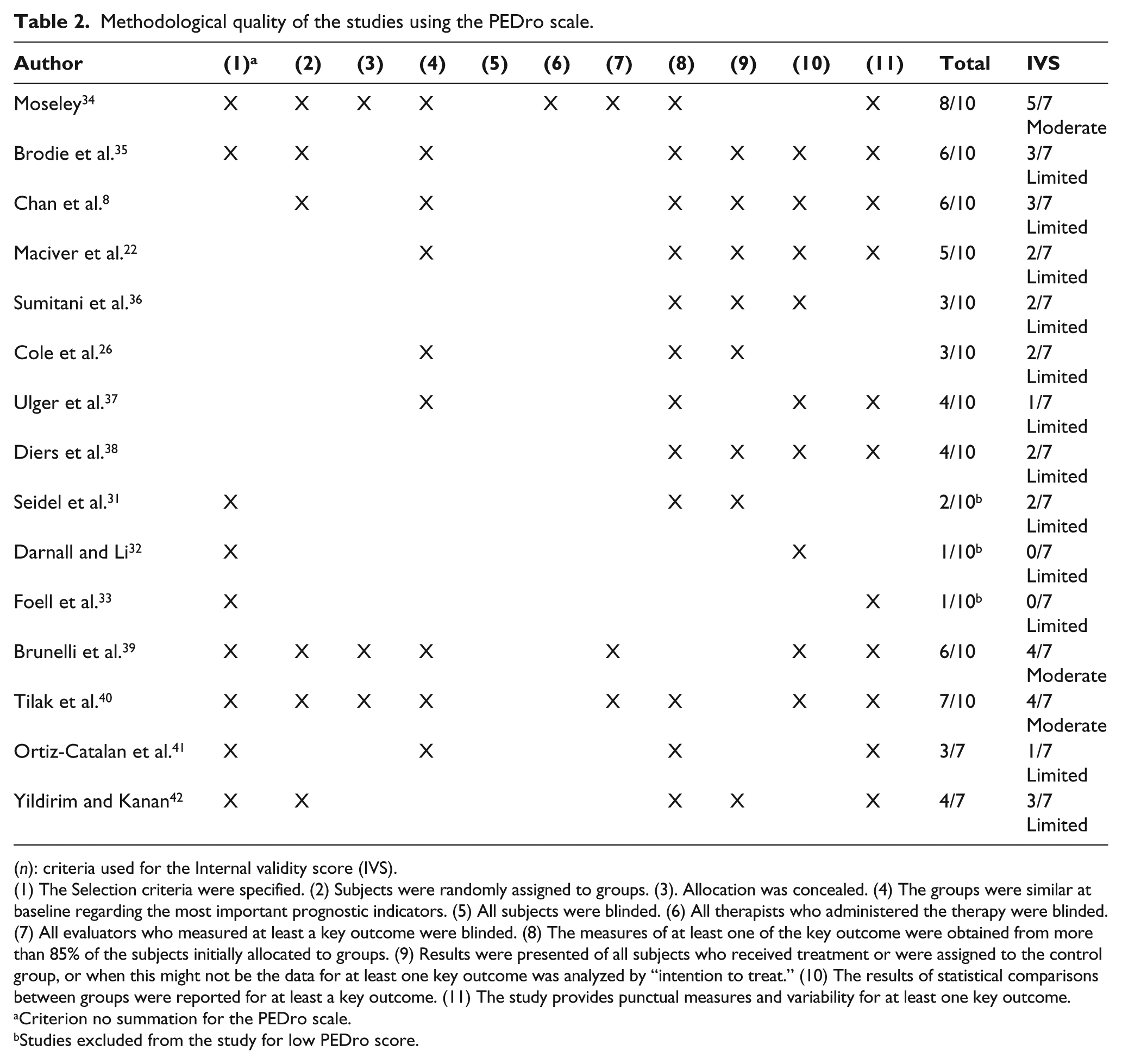
(n): criteria used for the Internal validity score (IVS).
(1) The Selection criteria were specified. (2) Subjects were randomly assigned to groups. (3). Allocation was concealed. (4) The groups were similar at baseline regarding the most important prognostic indicators. (5) All subjects were blinded. (6) All therapists who administered the therapy were blinded. (7) All evaluators who measured at least a key outcome were blinded. (8) The measures of at least one of the key outcome were obtained from more than 85% of the subjects initially allocated to groups. (9) Results were presented of all subjects who received treatment or were assigned to the control group, or when this might not be the data for at least one key outcome was analyzed by “intention to treat.” (10) The results of statistical comparisons between groups were reported for at least a key outcome. (11) The study provides punctual measures and variability for at least one key outcome.
Criterion no summation for the PEDro scale.
Studies excluded from the study for low PEDro score.
According to the classification of the Cochrane Back Review Group, 30 there was limited evidence for mirror/virtual therapy and motor imagery because there were no high-quality RCTs.
Descriptions of mirror, motor imagery, and virtual visual treatment
The studies analyzed showed different characteristics in terms of the progress of treatment sessions. They varied with respect to prior training, duration of treatment and session, and the type of technique performed. In addition, some authors took decisions about medication to which the participants were subjected during the treatment period.
Participants from seven studies were trained before the sessions to feel their phantom limb movements or imagine them.22,34,35,37,38,41,42 Of the 12 studies included, the training period consisted of a minimum of 4 weeks in total, except for 2 based on a single session26,35 and another study where the training period was unspecified. 40 The method sessions followed during treatment varied from one study to another. Most of them were based on real-time working,8,22,26,34,36,38,41,42 while only two studies used repetitive exercises.35,37 Three studies also conducted treatment sessions at home, after previous training by the therapist.22,38,42
Regarding pain medication, four studies withdrew pain medication during the treatment period,22,34,37,38 and three studies did not change medication or possible standard physiotherapy techniques,26,38,41 and the other studies did not specify this medical aspect.
Discussion
Summary of main results
This review evaluated the effectiveness of mirror therapy, motor imagery, and virtual visual feedback in amputees with PLP. All studies examined showed an effective improvement in perceived pain; however, according to the criteria specified by the Back Review Group, 30 there was limited evidence for the use of these therapies in reducing PLP.
This lack of evidence can be explained because previous research used pilot studies or case study designs where the treatment protocols were not adapted to clinical practice. Moreover, the overall number of studies on this topic is low. 11 In our study, the number of studies selected was only 12 due to different causes. First, subjects who had phantom sensation without phantom pain were not included in this review; second, bilateral amputees were not included because the existence of an intact limb is essential for the treatment process and for achieving the effect of these therapies; third, a study including different therapeutic interventions in combination with the therapies targeted in this study was also excluded. 37 The internal validity of studies was also limited and the methodological quality was low to medium, as a consequence of the study designs: lack of randomization and blinding, small or uncontrolled groups.
Populations
As McCabe 11 suggested, studies used heterogeneous populations. In this review, some studies did not distinguish between upper or lower limb amputation,26,34,36,37,40,42 cause of amputation,22,26,36,38 or sex of subjects.22,26,37,38 Therefore, in some cases, groups were not balanced by gender. The lack of similarity across studies in subjects and methodologies could lead to significant differences in the size effect for post-amputation PLP. Previous studies showed that the probability of presenting PLP seems to be lower in men than in women and reducing over time in lower limb amputees compared to upper limb amputees. 43 Cole et al. 26 found that people with upper limb amputation felt more intense and constant pain than those with lower limb amputation.
The age range of subjects included in studies was wide, ranging from 19 to 82 years. A research group from the World Health Organization stated that neuronal plasticity is age-related, resulting in distortion and loss of sensation and perception in later life. The molecular phenomena of neuronal aging induce changes in psychomotor performance, increasing reaction time and decreasing speed and accuracy of movements and activities. 44 Therefore, the disparity between the ages of different groups could also help to explain the discrepancies of the results.
The post-surgical period should be recorded and considered since early rehabilitation has been recognized as a positive prognostic factor. 45 However, to the best of our knowledge, no studies reported the time since the amputation was performed and the best time to apply these types of therapies.
Interventions and outcomes in PLP
Heterogeneity has been observed in the type and intensity of PLP within and among studies, especially before treatment. However, all studies showed a significant reduction of PLP. Four of the studies reported that the reduction in pain could be explained by the treatment benefits at the motor and cortical sensory level.22,37,38,41 Maciver et al. 22 claimed that the main sensory and motor structures involved in those therapies were the right insula, posterior cingulate cortex, and the premotor cortex. Diers et al. 38 reported that the attempt to restore the activation of the cortical representation of the amputated limb can potentiate brain plasticity of those areas and consequently to PLP reduction. Chan et al. 8 suggested that pain relief in mirror/virtual therapy can be also explained by the activation of mirror neurons in the contralateral hemisphere of the brain to the amputated limb. This happens when a person performs an action or even observes another person executing a movement, reducing in turn the activity of systems that perceive protopathic pain. However, Ulger et al. 37 demonstrated that the phantom limb exercises modified muscle tension. They also showed that the position of the residual limb can affect the intensity of PLP. For this reason, exercises should be bilateral, decreasing muscle tension and relaxing the residual part of the amputated limbs. 37
In those therapies, primary motor cortex reactivation by visuomotor training seems to play a major role in reducing PLP. 9 However, Moseley 34 claimed that imagined movements can activate brain areas, premotor cortex, and primary motor cortex, influencing hand laterality recognition, imagined movements, and type of mirror movements in pain reduction. In contrast, Brodie et al. 35 reported that repeated movements of the contralateral limb reduced PLP by decreasing cortical activity in the somatosensory cortex and the anterior cingulate cortex.
Other authors, studying cortical reorganization after using virtual systems or visualization of images, have hypothesized that pain relief is due to the mental effort of imagining the movement itself.2,26 Ramachandran et al. 2 reported that PLP may be reduced by a strategically placed mirror, giving the illusion that the amputated part has been returned and can be moved on purpose, producing a voluntary activation of visual motor imagery of the phantom limb. 36 In addition, the illusion or imaginary movement can be perceived by the patient as not painful, making the therapy pleasant and entertaining. Virtual reality may also decrease situational stress in the clinical setting, especially in those patients with kinesiophobia, preventing exacerbation of PLP during treatment. 41 Therefore, the reduction of pain depends on the quality of the illusion created and this should be considered in planning the treatment in the clinical setting.
Implications for future research and clinical practice
There are some implications for these techniques in clinical practice and research. Although there is evidence that programs incorporating mirror therapy, motor imagery, and virtual reality may be helpful for patients with PLP, the current evidence for the efficacy of these interventions is inconclusive. Therefore, this research topic needs more research and practical models of interpretation. 46 The new treatment protocols should specify the frequency, duration, sequence, difficulty, and progression during sessions. Even the importance of the duration of post-surgical period needs to be noted, to enable drawing conclusions about the most effective recovery phase for the technique. Since research in these innovative therapies is increasing in recent years, we would strongly recommend conducting studies with higher quality, with a homogeneous sample, adequately monitored, and with RCT design.11,25,47
The results of the studies included in this review highlight that mirror therapy could help in relieving pain under conditions not considered until now, such as dystonias. 46 Visual information of the movements provided via virtual applications can be used before proceeding to the movements themselves, when patients suffer PLP and kinesiophobia. Although the brain mechanisms involved in mirror/virtual and imagery techniques are similar, motor imagery requires higher cognitive resources and this strategy seems to be less motivating for patients. Mirror/virtual therapy may be especially suitable for those patients who find motor imagery difficult, 46 taking into account that its effectiveness depends on the quality of illusion created.
Motor imagery is also usually used for non-amputees with chronic pain. The challenging nature of chronic pain should inspire scientists and clinicians to investigate the physiological and psychological elements involved in this clinical manifestation. This could facilitate and enhance the creation of new treatments. 47 Research should focus on the influence of sequential activation of premotor and motor cortical networks and the effectiveness of attention resources as a treatment for the affected limb. 19
Strengths and limitations of this review
The main strength is that this review focuses on the effectiveness of relevant and new intervention techniques on amputees with PLP. This study is a meta-analysis of the trials published from a large number of scientific databases. It also evaluated the quality of each primary study included in the review, using the PEDro scale. 27
However, this review has several limitations. First, the studies were heterogeneous in subjects and methodologies used. Second, there were difficulties in obtaining several articles in a full-text format. Third, the inter-observer reliability of the search could not be calculated since the search process was performed by a single investigator.
Conclusion
In conclusion, all the studies reported a significant decrease in pain, regardless of the technique used. However, the evidence for the effectiveness of these techniques for PLP reduction in amputees was classified as limited. Future research with higher methodological quality studies are needed to demonstrate effectiveness in reducing PLP.
Footnotes
Author contribution
MEAF and LHC contributed to the study conception and design. JMPM and RMTH extracted data from the trials and evaluated the inclusion and exclusion criteria. AMCS, MAQZ, and LHC assessed the methodological quality of the trials. MEAF, CMG, RMTH, and JMPM contributed to the interpretation of the data and drafted the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
