Abstract
To be effective, healthcare technologies should be attuned to particular contexts of use. This article examines how such attuning is articulated in global innovation practices for tuberculosis and HIV diagnostics, and to what effect. It examines the development of point-of-care (POC) diagnostics – promised to be designed for users outside laboratories or in resource constrained settings – to study what developers and implementers do to align diagnostic technologies to the POC. Fieldwork among global health actors involved in diagnostic development, including manufacturers, donors, industry consultants, international organizations, policymakers, regulators and researchers, is combined with fieldwork among users of diagnostics in India, including decision-makers, NGOs, program officers, laboratory technicians and nurses. The article adds to STS’s theory of alignment and user interaction, where the setting and user to which developers and implementers of global health diagnostics align are multiple, varied, emerging and keep shifting. The characteristics of a local user setting include multiple engaged and imagined user settings, but also the settings of developers, of global intermediaries, competitors and diseases. As such, alignment is happening across multiple dimensions and scales and has an important temporal dimension. The results reveal how alignment happens to some extent in the dark, characterized by uncertainty about the elements that should align. Standardizing elements, politics and scarce resources cause frictions in the temporalities of aligning and over what constitutes a well-aligned diagnostic.
In this article, I take the case of the development of point-of-care (POC) diagnostics – promised to be designed for users outside laboratories or in resource constrained settings – to examine what developers and implementers do to attune or align diagnostic technologies to the point of care, and to reflect on STS’s theory around alignment and user interaction. Global health experts consider early diagnosis and correct treatment crucial for avoiding the emergence of drug resistance and more advanced stages of illness. POC diagnostics for infectious diseases such as tuberculosis (TB), HIV and malaria promise to bring laboratory-based precision to homes, communities, clinics and hospital wards where accessing a lab test is difficult or impossible, where it takes long or is too expensive. As such, these devices evoke promises similar to other, in Redfield’s words, ‘small-scale gadgets of humanitarian design’ such as the LifeStraw (Redfield, 2016), Plumpy’nut® (Scott-Smith, 2018) or the Zimbabwe bush pump (de Laet and Mol, 2000). While precise technologies differ, POC diagnostics include, for instance, rapid antibody tests based on lateral flow technology (such as pregnancy tests) as well as miniaturized molecular diagnostics that incorporate polymerase chain reactions (PCR) into devices the size of a small printer. Contrary to laboratory-based testing, where a few central labs are equipped with a handful of big devices running many samples simultaneously, POC testing usually means many small devices placed in many more testing sites. Promoters and other actors hope that this will allow for rapidly available results at low cost, as well as lower maintenance and user skills in settings without timely access to laboratory-based testing. If healthcare providers can diagnose a patient on the spot and initiate treatment, they can reduce misdiagnosis, mistreatment, delays, drug resistance and mortality, as well as freeing up time in overloaded, ill-equipped and under-resourced laboratories. The POC technology is presented as a way to overcome challenges that do not allow former technologies (laboratory-based testing) to function fully, by being more attuned to local contexts of use.
I examine how attuning to local contexts is articulated in POC test innovation practices. How do innovators of POC diagnostic and delivery programs for TB and HIV incorporate local settings into the innovation process? How do they know the POC and users? How and what forms of expertise and knowledge are made accessible? And what are implications of these innovation practices for meeting promises of what is considered a good POC test? The majority of ethnographic studies on POC diagnostics have focused on how such technologies are used and to what effect (Beisel et al., 2016; Engel et al., 2017; Hutchinson et al., 2015; McDowell et al., 2018; Umlauf and Park, 2018) but not on how these diagnostics have been attuned to local contexts, how they were imagined, designed and developed, and how these processes interact with their use. While the particular challenges of developers in innovating diagnostics for global health have been explored (Engel et al., 2016; Palamountain et al., 2012) and others have examined end-user needs (Rompalo et al., 2013), I examine the innovation process of POC diagnostics, and in particular the efforts to attune to local contexts and align multiple actors, settings and development steps.
Alignment work in constructing doable global health technologies
An emerging literature on the sociology of diagnosis (Brown et al., 2011; Jutel and Nettleton, 2011; Schubert, 2011) suggests that practices of diagnosing bind biological, technological, social and political aspects with lived experience. Such a composite activity can be contested, framed and enacted differently (Jutel, 2009). Similarly, taking a technology-in-practice perspective highlights that the result of medical practice is always a combination of very different elements: bodies, samples, professionals, research designs, patients, hospital or clinic organization, equipment, materials, reagents, conversations, questions, etc. (Timmermans and Berg, 2003). Using and implementing such technical artifacts involves constructing and manipulating them in daily practices (Crabu, 2014). Combined, these two perspectives highlight that aligning diagnostics to the POC involves continuously attuning to multiple social and technoscientific elements and the components that make up diagnosis.
Alignment, as I conceptualize it here, differs from the translation of knowledge from bench to bedside, as evoked in translational medicine, or the translation of knowledge from the Global North to the Global South. Sociologists and STS scholars studying translational medicine emphasize that translation is not limited to a one- or two-directional relationship between bedside and bench (Crabu, 2016; Lewis et al., 2014; Paul et al., 2008); it can be better conceptualized as movement of knowledge across different institutional, disciplinary, spatial, geographical or conceptual domains (Rajan and Leonelli, 2013). Mostly based on ethnographies of science and technology in the Global South, the STS literature on global health examines how science and technology travel or translate between the Global North and South (Crane, 2013; de Laet and Mol, 2000; Engel, 2012; Geissler and Molyneux, 2011; Hadolt et al., 2011; Montgomery, 2012; Mueller-Rockstroh, 2011; Pollock, 2014; Prasad, 2014). Through concepts such as fluidity (de Laet and Mol, 2000; Redfield, 2016), sticky technologies (Scott-Smith, 2018) and immutable mobiles (Latour, 1990), scholars have explored the characteristics of technologies that make them travel well. They may be fluid and effortlessly adaptable to very different contexts, sticky and not easily adaptable, or immutable, designed to retain shape during transport. The case of aligning to the POC highlights different settings or domains and some mobility between these. But rather than focusing on knowledge traveling from an origin to a destination, the case prompts us to ask how to conceptualize targeted efforts of making technology fit across different worlds and multiple social and technoscientific elements.
Scott-Smith (2018) argues that fluidity is a matter of degree and that most humanitarian technologies are characterized by stickiness, where change occurs at different levels and speeds, and with varying levels of friction between the immutable and the mobile (allowing only for a certain amount of adaptation). Developing such sticky technologies is exactly where the problem of aligning diagnostics to the POC lies and frictions emerge. How can actors strike the right balance between fluidity and immutability, especially if, as we will see, not all POCs, users and elements can be known? Suchman (2000: 322) writes that organizing alignment when building a bridge is simultaneously and ‘inevitably hybrid, practical, political technical, contested and negotiated and situationally specific’. Importantly, alignment to the POC not only happens across different actor communities, such as public and industry research communities (Vedel and Irwin, 2017), but across multiple settings and elements involved in innovating medical technologies and throughout the entire process.
Fujimura describes scientists articulating alignment across three levels of work organization –experiment, laboratory and relevant (external) social worlds – in order to construct doable research problems (Fujimura, 1987). Articulating alignment means actively manipulating the various elements in a research situation through planning, organizing, coordinating, monitoring, evaluating, adjusting or integrating activities to ensure that, for example, ingredients, machines and components for an experiment are available. Abundant resources, clearly defined roles, standardization, modular and packaged tasks can support doability, as they reduce uncertainty and allow planning (Fujimura, 1987). The elements that scientists manipulate to articulate alignment include all actors, materials, technologies, skills and work organizations, but also regulatory groups, guidelines, theories, models and other representational entities in that situation. Elements are in the situation itself, even if they are abstracted (Fujimura, 1996). This helps in the conceptualization of the composite nature of diagnosing (composed of all the different elements in the situation in which diagnosing is taking place). The construction of the right tool for the job involves – aside from achieving doability of problems – tinkering, craftwork and ad hoc arrangements inherent to scientific practice, as well as standardizing or stabilizing the elements in the situation (Clarke and Fujimura, 1992).
As will become clear, the developers and later implementers of POC diagnostics oversee this alignment work, but they are also bound by their structural positions (Castel and Friedberg, 2010) within different innovation settings or worlds (Timmermans and Shostak, 2016) operating in a context of regulations, political interests as well as limited resources. Scholars using the notion of ‘research in the wild’ highlight trajectories of technological innovations localized outside conventional R&D settings or innovation environments (Crabu and Magaudda, 2018; Dittrich et al., 2002). The design activity is distributed to new actors and places where use, design in use, maintenance and development intertwine. With it, the object of design changes and includes the infrastructure of service provision (Dittrich et al., 2002). In the process of innovating, alignment between technical work necessary to build such infrastructure and political and cultural frameworks of those innovating is taking place (Crabu and Magaudda, 2018). Diagnostic innovation, especially molecular diagnostics, is increasingly driven by diagnostic companies and not – as it had before – by professional sub-disciplines or non-profit organizations. While this has actually strengthened the role of public bodies in regulation and adoption of new technologies (Hogarth et al., 2012), the practices of evidence-based medicine and health technology assessment that regulate current adoption decisions are not immune to political interests and market lobbies (Blume, 2013; Hogarth et al., 2012).
Analysing alignment therefore requires examining structural actor positions, resources, regulations and politics. It requires examining how the multiple social and technoscientific elements in the situation are continuously articulated, manipulated, tinkered with and stabilized, in co-constructing the ‘job’, the ‘tool’ and ‘rightness’.
The alignment literature seems to suggest that the elements that are being aligned are known or can be known. I show this is not necessarily the case. Instead, aligning is sometimes characterized by uncertainty about what all the different elements are and look like and happens to some extent in the dark. In addition, the case of POC diagnostics highlights the temporal dimension of alignment work. In an expansion of Fujimura’s framework, Penders and colleagues highlight how doability in large-scale research collaborations, such as nutrigenomics, is constructed in multiple labs in a network of local doabilities (Penders et al., 2009). Instead of modularizing the overall research problem, the doability of POC diagnostics is constructed by developers and later implementers in a phased manner across multiple settings, elements, actors and across development steps. This temporal dimension of alignment work, continuously ongoing and across development steps, means that alignment is only ever stabilized temporarily and one cannot easily draw a line to pinpoint a well-aligned global health diagnostic.
To understand how users matter in these alignment processes, I will bring STS’s alignment literature into conversation with the field’s work on users. STS scholars have pointed to the contingent and interactive nature of innovation processes and to the mutually constitutive relations between design and use (for a review of user studies in STS, see Hyysalo et al., 2016).The intensity of user engagement has increased since the early user studies in the 1990s. This now also involves engaging with patients as either expert patients-activists or consumers, which is itself the result of decades of social and political change (Blume, 2013; Callon and Rabeharisoa, 2003, 2008; Epstein, 1996). However, the degree of institutionalization of patient organizations and user participation in the governance of medical research is uneven across different countries (Akrich et al., 2008) and does not automatically lead to good technologies for human development (Hyysalo et al., 2016). Hyysalo and Johnson emphasize that the user ‘is a more complex entity than just people out there’ (Hyysalo and Johnson, 2015: 73), but is instead a relational term that describes the relation some people have to an object, service, technology, etc. This relational understanding of users is valuable to illustrate the shifting relationships multiple users have with POC diagnostics.
Anticipating users and usage during design stages is different from the engagements of users and designers during appropriation and adaptation of technologies (Hyysalo and Johnson, 2015). Others have similarly highlighted how imagined users, inscribed into devices and machines, prescribe human-machine interactions (Akrich, 1992) and how real users shape technology (e.g. Oudshoorn and Pinch, 2003) by attaching different meanings (Bijker, 1995) or literally changing the technology (de Laet and Mol, 2000). Hyysalo and Johnson (2015) analytically distinguish between an engaged user and user representations and study how the latter are shaped, maintained and translated into material characteristics of technologies. This also entails that the field is not only out there but also inside design organizations (Hyysalo and Johnson, 2015). My analysis highlights how users of POC diagnostics are dispersed, multiple, varied and shifting and can never be completely known, contributing to the sense of aligning in the dark. Making user representations and their sources visible – including those by dominant actors, those hidden and taken for granted – offers a way to mediate between these representations and thereby deal with the politics of POC diagnostic innovations. It also helps to study how actors manoeuvre users that remain unknown.
Overall, user-technology interactions require work to show effect (Loon et al., 2014). Alignment theory offers a way of conceptualizing how attuning to local context and use, and therefore user-technology interactions, are organized. At the same time, the STS literature on users, with its attention to the relational aspects of users, to engaged and represented users, helps to conceptualize the alignment to users in alignment theory. It also highlights the interactive and bidirectional nature of aligning (co-production between design and use).
Methods
The analysis draws on fieldwork among global health actors involved in diagnostic development for tuberculosis and HIV. These include diagnostic developers, donors, members of civil society, industry consultants, international organizations, policy makers, regulators and researchers. I draw on 52 semi-structured interviews in Europe, North America and via Skype or telephone with participants based in Asia, Africa, Europe and North America, and combine them with fieldwork at workshops and companies, conferences and document research. A second batch of material is based on fieldwork in Bangalore, India, including 15 interviews with diagnostic developers, decision-makers, NGO program officers, scientists, TB and HIV program officers, laboratory managers, technicians and nurses using TB and HIV diagnostics as well as visits to companies, clinics and labs. I focused on TB and HIV because both areas have seen recent developments of POC diagnostics, with several products in the pipeline. The TB pipeline contains many diagnostics still in early stages of development, while there are more experiences with implementing POC diagnostics in HIV. In this article I draw on empirical material from across the different development stages and diseases to highlight specific points about alignment.
The interviews covered, among other topics, the participants’ involvement with diagnostic development, steps of development and challenges, involved actors, evaluation and regulatory practices, participants’ understanding of the POC, and practices for making the diagnostic work at POC. In this article I anonymize the companies and technologies except when referring to those situations where technologies are in use.
The analysis and data collection overlapped. Halfway into the data collection, a global fieldwork report was compiled, highlighting findings from the notes taken during the interviews. This report was updated as interviewing continued and while cross-editing transcripts. After completing the data collection, a second fieldwork report based on the India material was compiled in the same manner. The fieldwork reports allowed for an overview of different alignment practices and dimensions of alignment work. A coding scheme was developed based on the topic list, research question, notes and fieldwork reports and codes that emerged from reading the material. Once coding was completed, this analysis was used as a starting point and further deepened by going back and forth between the coded material and these initial practices and dimensions, looking for examples, reordering, sorting and summarizing (Rubin and Rubin, 2005), writing memos on selected codes and themes and reviewing theoretical approaches to make sense of the data.
Charting the field
For decades, TB practitioners have relied on sputum smear microscopy (which is often inaccurate), chest X-rays and clinical judgement to diagnose TB. In many places, they have to wait months for a laboratory diagnosis of drug resistance to any of the standard TB drugs. In recent years, a few new diagnostics have been introduced. Most notably for the purpose of this article is the Xpert MTB/RIF by Cepheid, a molecular test of TB and resistance to rifampicin (one of the main anti-TB drugs) that provides results in 90 minutes. The test involves a platform with, depending on the model, four cartridges connected to a computer and a printer. It was heralded as a new ‘while you wait test’ to be used outside conventional laboratories, revolutionizing TB control (World Health Organization, 2010). The World Health Organization now recommends its use as the initial diagnostic test for TB, acknowledging resource implications due to the higher cost of the assay (World Health Oganization, 2016). Challenges remain – as we will see below – with access, implementation and utilization. Following these developments, other test developers are working towards molecular-based TB diagnostics for the POC. Many of these developers are smaller start-ups with a technical background or a technology that they are trying to adapt to TB. Few are established diagnostic companies with an existing portfolio of rapid tests. Most are based in the US or Europe, with some exceptions in India and China. While the pipeline looked healthy and filled with new products a few years ago, a recent market analysis attests to a contracting pipeline, with some products being delayed due to technical development challenges and others underperforming in independent trials. So far, no diagnostic has been evaluated as robust and accurate enough to work in microscopy centres, where many TB patients become present to the health system (UNITAID, 2017).
In the field of HIV there is a long history of testing at POC, with disposable rapid tests and many different companies producing these tests. Most kits use lateral flow technique, which can be done using a finger-prick blood sample and takes up to twenty minutes to perform. Instrument-based testing for HIV at POC is much less common. Several companies have developed instrument based POC devices to do CD4 tests, counting the number of a type of immune cells to monitor a person’s immune system- important for those living with HIV. A CD4 test that is fairly widely implemented in resource-limited settings is the PIMA CD4 by Alere (Medicins sans Frontières, 2017). The test involves a small platform with an integrated printer and a single cartridge. It establishes CD4 counts in twenty minutes with a finger-prick blood sample. Since 2015, the World Health Organization has recommended HIV treatment for all, regardless of CD4 count, and instead monitoring by viral load (World Health Oganization, 2015). Viral load tests count the number of virus copies in the blood, are used to monitor treatment and detect possible infections. However, laboratory-based viral load tests are expensive and not available everywhere. Several near-POC diagnostics for viral load testing are in development or on the market, some of them using the Xpert platform (Medicins sans Frontières, 2017). The policy change had a profound effect on the market for POC CD4 devices with some companies stopping their product developments or exiting the market altogether.
Below, I analyse examples from across these different developments and discuss: (1) the challenges developers face to get to know the POC and user, and how that generates a sense of aligning in the dark, (2) how developing and manufacturing POC diagnostics means aligning across multiple global and local settings and across time and (3) how alignment is multiplied with consequences for access and utilization.
Getting to know the POC and user
The variability of the global health field multiplies POCs and users
Because of the inherent variability of global health, it is not easy to understand POCs, settings, health markets, users and their practices. The users of POC diagnostics involve more actors including patients, clinic staff, lab managers, ministries of health, NGOs, regulators and funders. These users are engaged in different aspects of use: supplying a bodily sample and receiving a result, ordering a test and acting on its result, conducting the testing procedure, maintaining the machine, evaluating safety and quality, overseeing the technology’s deployment, utilization and reporting, and purchasing devices and supplies. The variability of global health multiplies kinds of users and their settings. This means that for companies, it is often unclear who the users and customers are and how to appeal to them – unlike in a market such as the US, it is not individual hospitals that buy these technologies but mostly governments, donors and NGOs. And some of them are unexperienced customers, overwhelmed if they have to deal with hundreds of diagnostic POC machines in understaffed facilities (industry consultant 2).
According to several of my interviewees, knowing the POC requires understanding multiple social and technoscientific elements across multiple settings and local and global scales. This includes understanding what is feasible in terms of operational design and the clinical use, how work is organized at the clinic (test developer 11), understanding the markets and business case from the patient and the health system’s perspective, and understanding trends in policy, the WHO and global guidance (programme officer CHAI 1, scientist KNCV 3). Data to support such an understanding of the POC is difficult to access or generate (programme officer CHAI 2, industry consultant 2). Furthermore, many influencers might affect this data, including, for instance, institutional structures, procurement mechanisms, epidemiological context, clinician behaviour, workflow changes, and changing global health arena and guidance (industry consultant 2, programme officer CHAI 1).
According to one donor, no company big or small has the competence and all components needed to develop a successful, molecular POC test for infectious diseases for global health. Collaboration, multi-institutional projects and intermediaries are always needed: NGOs like FIND and PATH and CHAI are really helpful, because they can introduce the grantee to the stakeholders in those countries. And that is why I think it’s another asset to have a multi-institution kind of project, because no single institution really understands all that [relevant actors in countries]. (programme officer donor 1)
Many of these intermediaries are employed as consultants by international organizations, donors and big NGOs with a focus on technologies for global health like UNITAID, Clinton Health Access Initiative (CHAI), Foundation for Innovative New Diagnostics (FIND), Medicines sans Frontières (MSF), the Bill and Melinda Gates Foundation, the Stop TB partnership and working groups of the WHO. Others work as individual industry consultants. These global intermediaries support developers by organizing access to users and samples, by publishing market landscape reports and Q&As, doing cost-effectiveness modelling, and by organizing workshops and meetings. They also publish so-called target product profiles, essentially a user representation, specifying the minimal and optimum requirements a new test should meet for a specific purpose (for TB see World Health Oganization, 2014). They map out national TB or HIV programs, market needs and epidemiological profiles and regulatory requirements of different countries (programme officers WHO 1, 3). And they connect developers with key opinion leaders or distributors in countries (programme officer CHAI 3). As such, global intermediaries are important mediators, translators and gatekeepers at the same time.
Knowing the POC involves handling the variability of the global health field in terms of data, policy and actors and knowing global intermediaries to gain access to users, materials and data. The users of POC diagnostics are not a single easily identifiable group. They are multiple, dispersed and often represented by others and as such they are hard to access. This generates a sense of aligning in the dark.
Shifting users and difficult to access POCs
The users of POC testing are not easily contacted and good user feedback takes work to generate. According to an industry consultant, it is difficult for companies to access the POC, get permission to visit public clinics and public health programmes, and use time of clinic staff to try out a prototype. It often requires intermediaries and willingness to continually engage and establish relationships with users (industry consultant 2). According to a developer of HIV POC tests in a company with an established POC market segment, the challenge to knowing the POC is to get good constructive feedback from a user who is able and ready to anticipate and see through the design hurdles. For that, a user needs to take time, share an interest in the design of the test, and think alongside the developer. This particular developer dealt with the complexity of engaging users in a personal, organically growing and initially very localized way.
For example, the developers of one HIV CD4 diagnostic initially entered an African country using only a personal contact in only one lab, and then improvising and testing ideas with what they found there. Interest, contacts and relationships emerged that allowed the developers to receive feedback without letting the device leave their hands. They slowly engaged more people in the country, conducted joint studies, collaborations, and created interest among policymakers. These relationships not only helped design the test and make it fit better, but also with organizing evaluation trials in these sites and preparing the grounds for selling devices (test developer 6). According to the developers, they are continuously updating the product, setting up servicing and training infrastructure, servicing the POC, and engaging with ministries of health and users (test developer 8, 9).
To improve communication about errors and problems with users at the POC, this group of developers adjusted training schemes, communication channels and even the problem-solving software on the technology itself. If users log a complaint and email the machine’s archive, the developers can analyse the error pattern and, if necessary, send someone from the in-country or supplier team to solve the problem and retrain. The developers learned that users’ tolerance for errors and inclination to log a complaint differs from country to country. Users also shift at more micro levels: Frequent clinic staff turn-over and shared responsibility for the same technology mean that users are less likely to have devoted attention to or develop close relationships with the device. What is more, developers rarely interact directly with people operating the device, but instead with supervisors or clinic managers. According to the developers, the more one moves towards the POC and away from the laboratory, the less understanding there is for the intricacies of the test (test developer 9). ‘[Y]ou are not dealing with a dedicated lab personnel, but nurses, doctors, etc. … They are dealing with patients, not only with tests’ (test developer 11).
Engaging shifting users takes ongoing alignment work, involves tinkering, ad-hoc arrangements and substantive investment. This work helps establish relationships and access to the POC, to generate data and not lose sight of design goals. The innovation process becomes a strategic tool to align to the POC and prepare for making a sale. This points to the temporal considerations of aligning.
Doing too little too late to know multiple POCs, users and markets
Diagnostic companies vary in their efforts, beyond interacting with global intermediaries, to understand the local context of use. Some undertake their own user requirement studies, send someone to visit different clinics, and test early prototypes in clinics and small labs. Many of my interviewees argue that most developers do not do enough to know the POC, or do it too late in the product development (test developer 15, programme officer CHAI 1, 2, consultant KNCV 4). The advice of global intermediaries is not always well heeded and sometimes comes too late: [S]everal people in WHO and FIND commented that one of their problems has been manufacturers and developers saying that this is our product and this is how it will work. And they would say to different people over the years: ‘Well, have you thought about this and have you thought about that?’ To which the developers’ response was often in more cases than not, ‘Well, we’re already on a track so we’ll have to market it as it is’. (test developer 8)
According to a donor, most diagnostics companies do not have sufficient understanding of the global health context for innovating infectious disease diagnostics for the POC – including the business model, clinician behaviour, uptake, forecast and the cost of servicing many more testing sites across different countries (programme officer donor 1). A programme officer at CHAI argues that often the starting point of developers is the technology, rather than an understanding of markets, settings and users. He argues that having a design concept based on a target product profile, even if it has been handed over by global intermediaries, is not enough. Instead, one needs to spend time in the settings directly in order to arrive at priorities within that profile (programme officer CHAI 1) and to understand the importance of advice on technical requirements and product specifications (industry consultant 2, test developer 11).
An industry consultant argues that some of the global intermediaries themselves do not enough to know the POC and have little experience with, for instance, clinic settings in Africa: There are a lot of people even in Global Health, even sitting in Seattle I think in the Gates Foundation, who have never been to Africa. I mean I find that really startling. They have never gone and travelled … to these settings in Ethiopia and Kenya and Malawi and Uganda etc. And really understand what happens. What is the patient flow? How does it really work in real life? (industry consultant 3)
Intermediaries’ advice, such as ‘keep your turnaround time at twenty minutes’, is not always founded on detailed knowledge of POC settings and practices (test developer 15). Doing not enough or too little to get to know the POC contributes to the sense of aligning in the dark.
The composite nature of diagnosing challenges alignment
One of the inherent challenges of innovating diagnostics is that diagnosing is made up of different composite elements that need to align together for the technology to work. Aligning a technology to such a composite activity is challenging, as a senior officer at FIND highlights: [O]ne of the things about diagnostics is different than drugs and vaccines, … is that there’s loads and loads of different technologies and different reagents and sort of putting them together requires all that heavy R&D process. Whereas with a drug there’s a core clinical material and then it’s composed somehow and formulated and then that’s the end. You just give it to the patient. Whereas the diagnostics has all of the uptake and training and what it causes to happen in the entire system. So going from bench to bedside requires a lot of planning about how to handle this work. (programme officer FIND 1)
Integrated molecular POC devices such as PIMA or Xpert MTB/RIF harbour within themselves entire PCR (polymerase chain reactions) mechanisms made possible through the complicated interplay of different components, fluids need to be pushed around, reagents dissolved and applied, photos taken, reactions analysed, etc. To align these components with each other requires much research effort, often across disparate disciplines and company departments (test developer 18). It mobilizes many different disciplines, either bringing new specialists into the company or coordinating and aligning work across different company departments representing different components of the machine: biology, chemistry, optics, engineering, plastics, regulatory affairs, marketing, the developers of the platform and those of the assays, etc. (test developer 18).
Once the diagnostic instrument is placed in a clinic, people need to order the test, conduct it, act on the result, maintain the machine, order the reagents, conduct quality controls, stock cartridges and fit the device into different workflows and user practices. Many POC devices have, for instance, turn-around times of under an hour, but run only one sample at a time. This has consequences for stability requirements of reagents and patient journeys and workflows at the clinic. It can mean that the next sample is prepared and put into the cartridge, but then is waiting on the bench for its turn in the machine (during which time reagents need to remain stable even at room temperature). This particular trade-off between short turn-around time and limited throughput means that patients wait for results, rather than making two short visits (officer WHO 3). Diagnosing is an activity composed of many different elements and users, which span the social world of developers, patients, lab technicians and clinicians. Developing a test for the POC thus requires aligning to these various elements both inside and outside the box.
Test developers know that many of these elements that ensure usage at POC are not under their control. And some elements defy control, adding to the sense of aligning in the dark. A test developer explains: There has to be good reason for people to use it [the diagnostic technology], and you have to get rid of all these things that are standing in the way between the technology that you have developed and the use of it. And most of these things are completely not under your control. Because they are influenced by all kinds of other things, budgetary constraints, policies. (test developer 9)
Developing and manufacturing POC diagnostics
Aligning across different sites and the politics of alignment
During the R&D phase, developers face difficult trade-off decisions on a regular basis. Should they shorten the turn-around time or increase throughput? Should they keep the cost of the test low, typically compromising on materials, or increase the accuracy of the test results? Should they lower detection limits (possibly missing some patients) to allow for a shorter turn-around time (losing fewer patients if waiting times are short)? Should they invest in the quality of the test results, or instead in the ruggedness of the device? There are no easy answers to these questions, since various elements are unknown. The answers also differ depending on whether the technology is to exclusively address markets in resource-constrained settings or also better resourced markets. And yet, developers need to make a choice.
[Y]ou have to make a trade-off on whether you are going to invest $500,000 in improving your sensitivity or 200,000 in doing something else. Those are the decisions where the rubber meets the road and where they don’t really have the answers from the ministry on the country settings and on what to target. (officer CHAI 2)
The decision might go against some of the user feedback developers receive. For example, a developer of a new TB diagnostic was advised by global intermediaries to keep using the existing sputum cups to collect the sample and not involve different cups that are more biosafe to spit into. The reason for this suggestion was that too many sputum cups had been purchased globally, and they were asked to respect existing protocols of public health programs. When the developer talked to end-users of TB diagnostics in one country, though, a different user representation emerged. Providers, nurses, and doctors wanted a closed, more biosafe sample collection system.
[W]hilst you might be able to envisage a completely new approach to processing a sputum sample, both WHO and FIND said that really you should try and stick to the current protocol where people are coughing into a plastic cup that’s not been engineered. … And it’s a balance, because when we talk to users in clinics, … with clinicians and district nurses and people who’re on the front line, off the record they were saying they really hate dealing with sputum in an open cup. (test developer 5)
The developer plans to offer the biosafe sputum cup in a second step, after having released their test assay for the conventional cup first.
Trade-offs and alignment work cut across multiple social and technoscientific elements in different settings: the setting of the developer, of (imagined) users (equipment, user steps, clinic workflows, maintenance, infrastructures), of global intermediaries (standards, agendas), of diseases/micro-organisms (in samples, public health programmes, guidelines) and of competitors (existing diagnostic equipment and suppliers). Figure 1 provides a schematic illustration of the situation faced in developing a POC diagnostic. The developer and later implementers are aligning the different elements of the innovation situation (the dots) across these different settings. In doing so, they might be unaware of some or many of these elements, yet they need to make a choice for aligning to certain elements while ignoring others (aligning in the dark). The example with the sputum cup also reveals the politics of aligning and how powerful actors can define how trade-offs are handled. Aligning with one setting or user can conflict with aligning with another: Whether to align to ‘diagnosing TB’ or ‘diagnosing TB in a biosafe way’ is situationally dependent, pointing to the layered meanings of diagnostic technologies.
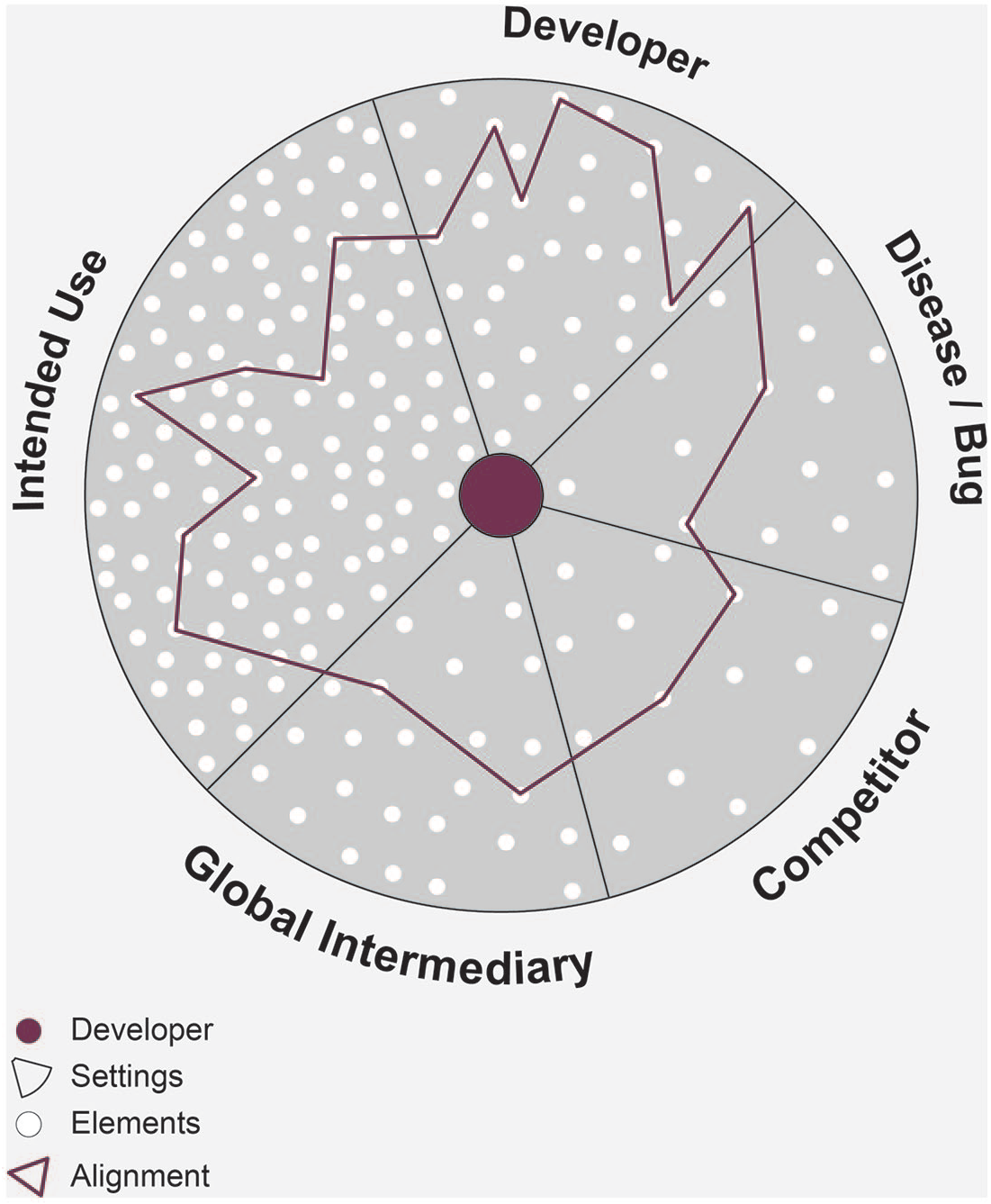
Aligning elements across different settings.
Aligning across time and development steps
According to my interviewees, alignment not only happens across different settings but also across time and development steps. To develop and manufacture POC technologies, business and funding models need to align with development and manufacturing phases, market entry with infrastructure development plans and procurement timelines.
The majority of all TB and HIV testing in public programs is funded by external donors, who then turn from R&D funders into customers (officer StopTB partnership 1). While funders are willing to fund R&D, they are usually not funding manufacturing and production lines or committing to pre-purchase agreements.
At the end of the day [developers need] to figure out … what manufacturing equipment they need to invest in. Is it a facility that does a million tests per year or is it 10 million? And they are not going to invest in the one that requires 10 million tests per year until they have a list of customers. (industry consultant 2)
For developers, it is nearly impossible to make investments in manufacturing plants without having pre-purchase or funding agreements with donors or ministries of health, or partnerships with a big corporations convinced that there will be sufficient returns on investment (test developer 10, 20).
Similarly, it matters how market entry, infrastructure development plans, and procurement timelines align to avoid delays: If ministries of health plan, budget and set up evaluation studies based on company projections of market entry, the diagnostic might not be ready, as companies tend to project optimistically. Thus, country-level decision makers delay planning evaluation studies and making deployment decisions until the technology is on the market (industry consultant 1). Most funding for TB and HIV control comes from external donors (officer StopTB Partnership 1), with funds for hardware and consumables separately earmarked and mostly spent in cycles of tenders. Companies with diagnostics that consist of a platform and cartridges – and not just a disposable rapid test – try to find open tenders that allow for buying hardware and consumables together, and fit into the timelines (test developer 9). Some countries have invested heavily in strengthening their transport and laboratory infrastructure (also in line with global guidelines of the past ten years) and might not be willing or able to make changes for a small POC device (test developer 9) about which they are not sure how guidelines will develop in the next few years. These infrastructural frictions (Paul et al., 2017) point to the temporal dimension of aligning.
Iterations, re-alignment and frictions in the temporalities of aligning
Friction in the temporalities of aligning also occur due to regulatory requirements. According to developers, it is common to go through design iterations during R&D stages. But it also shows that it matters how earlier alignment practices, for instance when getting to know the POC and establishing relationships, affect later stages of design iterations. Yet, there are also frictions in these temporalities of aligning. For the most part, design iterations have to stop in order to perform evaluation studies for regulatory approval by the FDA, EU or WHO, and to create comparable evidence across evaluation studies. On the basis of these studies, various actors assess quality and draft guidelines. From this so-called design lock onwards, the technology has to be a ‘finished’ product. Substantial design changes after this stabilizing element require costly and time-consuming re-evaluation studies to acquire regulatory approval, even though the variability of the global health situation continues to demand localized and flexible solutions.
One developer (test developer 10) of a CD4 test for HIV monitoring describes several iterations in product design, technical specifications, and expectations of how operators will interact with their device. These changes were made after receiving feedback on their prototypes from operators and their trainers at a local research institute, and from Ministries of Health, national-level NGOs and CHAI (a global intermediary), with whom they had established relationships since the early phases of design and getting to know the POC. They had not anticipated the calluses of Zimbabwean farmers that needed a different lancet to make a slice in the finger to get at the blood; this led to changes in the lancet and sample volume. Optimization in manufacturing meant that they had to weaken the rugged design of their device to allow replacements of the battery by service providers.
Innovation efforts aim at aligning composites across multiple settings, local and global scales, time and development steps. Figure 2 shows how alignment work is continuous and changes through development phases. It also illustrates iteration during the R&D phase when developers rely on feedback by users they have established a relationship with during earlier phases. Efforts to continuously align across time and development steps, infrastructural and regulatory practices reveal frictions in these temporal dimensions of aligning. And, as the example of the sputum cup shows, the different structural actor positions across settings can cause frictions between the diverse interests and politics at play in negotiating the alignment between sociomaterial and technological aspects of POC diagnostics. In doing so, the job and the rightness are situationally constructed and are shifting, shaped by competing user representations, interests and layered meanings of diagnostic technologies.
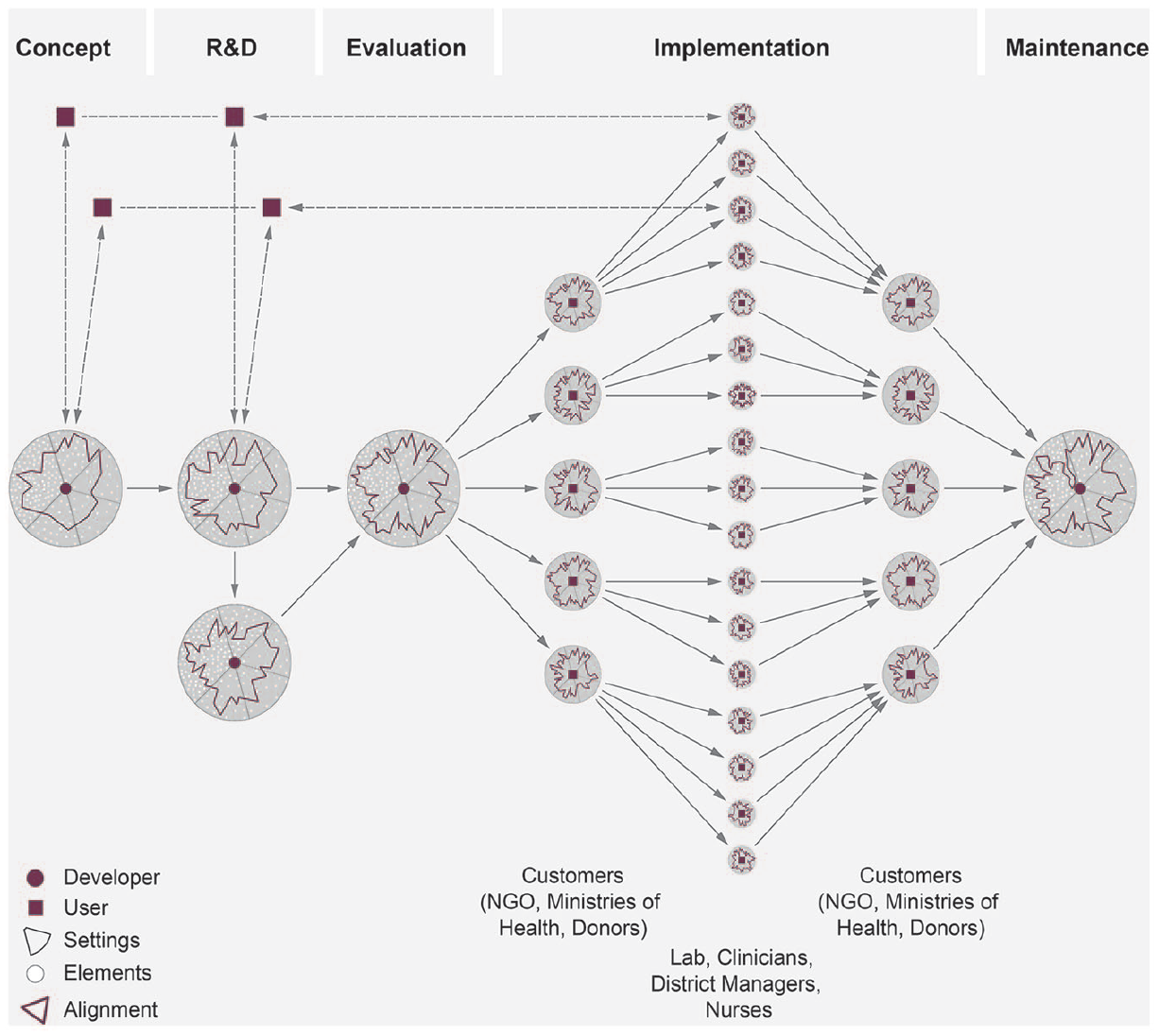
Alignment work throughout product development for POC diagnostics.
Implementing POC diagnostics
Multiplied alignment with implications for access and utilization of machines
Aligning does not end with design lock or market launch of POC devices, but is continuous throughout product development from initial stages to distribution, maintenance and servicing. The examples of the Xpert MTB/RIF for TB show that when implementing POC devices, alignment demands multiply across many different clinics and laboratories, at district and subdistrict levels. While in earlier stages it is mainly the developer who is responsible for the alignment work, during implementation, many heterogeneous actors are engaged in it, including a few larger customers such as NGOs, ministries of health and donor groups, as well as many different lab and clinic managers, nurses and clinicians (see Figure 2). The implications of earlier and ongoing alignment practices for access and utilization of diagnostic technology come to the forefront.
Xpert MTB/RIF fails if it is too dusty, too hot and if power fluctuates – all of which are frequent in India (implementer 3). In the first years of implementation, new filters were developed by the manufacturer to deal with the dust, and air-conditioning needed to be installed wherever the machine was placed. Its specific infrastructure requirements, but also its cost, human resources and servicing requirements mean Xpert cannot be placed in the primary health centres where most of the TB patients in India are. Either the patients need to travel to the machine or the sputum samples need to be transported. These are challenged by distance, transport facilities and non-referring medical officers. Limited funding and the high cost of the Xpert MTB/RIF cartridges and maintenance contracts furthermore mean that the test is restricted to certain patient groups at these sites (those failing treatment, pregnant women, children and HIV positive patients) and not available to everybody who needs it (implementer 5). For patients, this specific alignment means access is limited.
Limited access is compounded by underutilization. Where Xpert is placed, it is added onto existing workload of lab technicians. Yet the cost of cartridges and operational costs forbid a senior TB officer from re-aligning workload by decentralizing further than the district level and easing the workload on district level lab technicians (implementer 3). As a result, workflows are not necessarily adapted, delaying testing and undermining maximum utilization. In none of the settings I visited, Xperts were actively running samples at the moments of our visits, which all happened during office hours. It was also common that samples would be batched and run in two cycles per day, even if there were only a few samples to process. This delays testing (the device promises a turnaround time of 90 minutes) and patients would wait longer or were asked to come back the next day, risking that they might not (be able to) come back. As a result, the Xpert machines in those centres are underutilized (laboratory technician 4). Underutilization can further limit access, because it is difficult to reach these machines even if a patient is eligible. In settings where staff is overburdened and patients have scarce resources to spare on accessing healthcare, staff might not refer them for Xpert testing (scientist KNCV 2).
The current alignment of cost, human resources, location and infrastructure requirements of the POC and Xpert MTB/RIF compromises utilization patterns of devices and access for some patients, while generating a better diagnosis for those who manage to access the test. Further re-alignment, to ensure linkages to care and policy change, to make it the right tool for the job of diagnosing TB at these particular points of care is challenged by resource constraints of patients or the health system. Continuous aligning involves ad-hoc arrangements and, depending on alignment practices prior to design lock, requires extra commitment by developers and implementing users, to avoid rendering the diagnostic test useless at the POC.
After a positive Xpert MTB/RIF result for rifampicin resistance, treatment for multi-drug resistant TB needs to be initiated, adverse responses to medication need to be monitored for at least a week, and patients need to be counselled to cope with the very harsh treatment. Not every institution in India where the Xpert result is reported can provide these services, and hospitals that can are not always conveniently located for patients. A district TB officer managed to arrange a deal with the nearby medical college: [W]e had a dialogue with the Medical College Director, we requested them [to care for MDR-TB patients diagnosed at our site], because it needs Isolation Ward, staff to engage them, without giving funds other institutions may not accept. So we were able to convince the Director and started this. For the treatment part and other medicines we’re paying for the institution. The rest will be free of cost. (Implementer 5)
It requires extra commitment to arrange for treatment initiation at convenient locations for patients diagnosed at the program manager’s Xpert site and overcome hurdles with hospitals who might request payment for this extra burden. Without those extra efforts, the loss to follow up of patients who are unable to initiate treatment after diagnosis would have increased and the test rendered useless in this particular situation.
As we have seen, ongoing alignment practices have consequences for access and utilization of the diagnostic technology. Aligning also means fitting the diagnostic technology across laboratory and wider diagnostic infrastructures, including the temporal dimensions of sample transport, follow-up visits, diagnostic algorithms, clinic workflows, supply and repair services. Aligning involves positioning the technology towards competing diagnostics, to arrange for linkages to care and policy changes in such a way that tests function at a particular POC and there is coordination among different actors and sites involved in testing. These are dynamic processes that take years and are never really completed. Material and human resource constraints at the POC can hamper (re-) alignment efforts, especially for POC testing technologies that – after design lock – need extra commitment and entrepreneurship to be made to work. Aligning is not just a personal skill of cognitive bricolage (Castel and Friedberg, 2010) but also explained by the actor’s structural positioning and the dimensions of contexts.
Discussion
Aligning diagnostics to the POC includes aligning to multiple settings, actors, elements and scales at different moments in time. Shifting and multiple users, the composite nature of diagnosing, the variability of global health, low profit margins and the poverty-related nature of TB and HIV complicate aligning to any context of use. It is hard to create or identify the right tool for the job, especially if rightness and the job are shifting. Some amount of aligning in the dark is inevitable.
I put forward three main points: First, the alignment situation multiples and includes more elements, heterogeneous actors, settings and temporalities than the current alignment literature recognizes. Second, the shifting users, settings and composite nature of diagnosing at the POC mean there is a certain amount of aligning in the dark, characterized by tinkering and directed efforts of attuning to imagined and partly unknown users and elements. Third, standardizing elements, politics and scarce resources cause frictions in the temporalities of aligning and over what constitutes a well-aligned test. In the following, I discuss these three points in more detail, along with the contribution to studies of users and alignment.
Continuously aligning across multiple settings, elements and time
The social science literature has criticized simplified ideas of technology diffusion and transfer underlying much technology design for global health (Engel, 2012; Hadolt et al., 2011; Pfotenhauer and Jasanoff, 2017; Prasad, 2014). Many have argued – in different ways – that the local context of use and the additional work that is required to enact technologies should be taken into account more and earlier during technology design. Existing literature on medical technology development often differentiates between top-down and bottom-up development processes. POC testing promises to solve global problems with solutions attuned to local context of use from the start. As I have shown, when innovating POC diagnostics for TB and HIV, the lab consciously anticipates the field. Moreover, the field talks back in various ways, through various sources, user representations and at moments in time, and sometimes through global intermediaries. I propose that the concept of alignment offers a fruitful way of refining our understandings of these issues.
Aligning diagnostic technologies to the POC involves aligning with multiple engaged and imagined user settings, some of which compete with each other. In so doing, the innovation situation expands and includes aligning to the setting of the developer, of global intermediaries, competitors and diseases or microorganisms and a myriad of diverse elements associated with these settings (see Figure 1). Aligning to the POC also includes activities such as strategizing, getting to know different settings to which one believes alignment is necessary, continuously engaging various users and building relationships over time, handling competing demands and user representations, making trade-off decisions, adding steps within devices to control user engagement, handling infrastructural and regulatory requirements, overcoming temporal frictions, coordinating planning steps over time, coordinating staff and policy actors, deployment of instruments, sample transport, care availabilities, supply and repair services. Different actors are involved with this alignment work at different steps.
The alignment literature has highlighted that alignment happens within social worlds. In her classic study of alignment, Fujimura’s (1996) scientists need to convince management and their funders and align with research problems in the scientific worlds related to cancer genetics, but they do not aim to fit their products to a variety of users in different settings, position themselves towards direct competitors, work with the specific control culture of disease programs and their public health histories, handle a variety of global health intermediaries and agendas, and hand over the alignment work to multiple and heterogeneous implementers (see Figure 2). The case of POC diagnostics happens in a constant back and forth between global and local practices and concerns and thus is always global and local at the same time. Instead of the bidirectional co-production between design and use that the literature on users suggests, aligning is multi-directional and multi-scale. As translation between laboratory and bench is better understood as a multi-directional and multi-modal process where objects, materials, knowledge, phenomena and practices are circulating between different sites (Crabu, 2016; Lewis et al., 2014), aligning to ‘local contexts of use’ is ongoing between many different settings, global and local scales and elements in the situation.
In aligning between the different elements across these settings, new POC diagnostics are being shaped and organizations of work are constituted. For instance, global intermediaries can suggest aligning to a different, competing user representation, or evaluation work at the laboratory can be changed to account for regulatory requirements. Kelly and Lezaun highlight how different sites and topologies – in their case the breeding ground for mosquitoes – matter for what is considered a doable problem for malaria control in Tanzania (Kelly and Lezaun, 2013). In the case of POC diagnostics it is not so much the local alignment in one specific setting but the alignment across different settings and across time that matters. This is also different from Penders and colleagues’ local doability, plugged back into the overall global doability of the research collaboration (Penders et al., 2009). The innovation problem of global health diagnostics is not so much modularized but phased, and it is the developer (and later the implementing program officer or lab manager) who aims to align what is locally doable across a range of other settings at different moments in time (including projecting out into the future). Viewing this as a collective trajectory over time (Timmermans, 1999) shows that aligning is emergent and open-ended, but also that there is a temporal dimension to it. Alignment work is ongoing throughout and across the development of a diagnostic technology, from initial idea until servicing, maintaining and operating the technology. It seems to never be completely done. Aligning across time and between different development steps can avoid delays and wasting resources, such as if market entry aligns with infrastructure development plans, or if practices when getting to know the POC align with later stages of design iterations. The temporal nature of the efforts points to extra uncertainties and frictions, and to the consequent sense that aligning takes place in the dark.
Aligning in the dark across shifting users and settings, and the composite nature of testing
Most alignment literature assumes that what is to be aligned is known. Yet managing uncertainty is regarded as an inherent and necessary characteristic of medical work, scientific work and knowledge production (Fox, 1980; Moreira et al., 2009; Pickersgill, 2011; Star, 1985). How is alignment done when those who are attempting to align cannot know everything that they need to know? In the case of POC diagnostics, some sites and elements will always be underrepresented, because they, in the variability of global health, can never be known completely – if certain sites are underrepresented in the innovation practices, then they are not part of the local alignment at that site – resulting in what Penders (2010) calls incomplete alignment. Further complicating the situation is the shifting nature of users, settings and tests. What is more, like research in the wild, where design activity is distributed to new actors and places (Crabu and Magaudda, 2018; Dittrich et al., 2002), the alignment situation in the case of POC diagnostics is multiplied and dispersed among multiple heterogeneous actors acting across local and global scales.
The aligning of POC diagnostics takes place in the dark, because those aligning cannot know everything they need to know and some of these elements they aim to align defy control. Aligning in the dark is one of the challenges that may be faced by research in the wild involving new actors, objects and settings. In the case of POC diagnostics, the multiplied alignment situations and the temporal dimensions of alignment deepen the darkness and the awareness of it.
Variability, multitude and uncertainty are managed through the various modes of alignment work I have outlined. Here I focus on one important contributor to alignment. User representations emerge from the local settings of intended use (sourced through various modes of requirement gathering) and the local settings of development (sourced through professional experience, available technologies and business concepts), but also from the settings of global intermediaries (sourced through standards, policy and intermediaries), of competitors (sourced through earlier technologies and regulatory standards) and of the disease (sourced through local epidemiology, technical specifications and scientific knowledge).
This means that not all engaged users are hands-on all the time and not all voices are equally heard in representing their user needs. The ‘POC’ is at times silenced, implicated, partialized, or partially represented by global intermediaries, and takes work to involve. Much as the trial community in drug development is not a static and singular group of people, but is better conceptualized as dynamic and transient publics enacted in concert with the trial (Montgomery and Pool, 2017). The POC, including the potential end-users, are multiple and emergent and not a singular and static setting or stakeholder a diagnostic developer aims to address. This adds to STS literature on user innovations, in which users are typically clearly identifiable and – to some extent – organized groups with whom innovators interact, as in the studies on patient organizations who engage in their own bottom up research (Callon and Rabeharisoa, 2003). Montgomery has emphasized that, contrary to the user/designer dichotomy inherent in scripting or descripting theories (Akrich, 1992), technology design for global health is a political and normative process involving a range of actors in many-to-many relationships (Montgomery, 2012). Analyzing POC diagnostics innovation as alignment shows that not only are multiple heterogeneous actors involved, they are also shifting and operate in different settings and social worlds that cannot be completely known, complicating the normative and political processes.
Constituting rightness, frictions and scarcity
One cannot easily draw a line to identify a well-aligned global health diagnostic. In Fujimura’s (1987) case study, scientists decide what is doable and tinker to align different elements until they have as much overlap as possible. For global health innovation situations, being doable is stabilized only temporarily and, as Clarke and Casper illustrate with the Pap smear (1998), depends on the standpoint because it entails constructing rightness through different tinkering strategies. The job of innovating POC diagnostics and the rightness of them are situationally constructed and are shifting. Alignment work is shaped by competing user representations and layered meanings of diagnostic technologies. The different structural actor positions across multiplied alignment situations have important consequences for how the diverse interests, politics, regulatory practices and resource constraints at play in alignment are negotiated.
A POC diagnostic can be considered good enough if it does important work for those in power (Casper and Clarke, 1998); given the composite nature of diagnosing and the variability of global health, those in power vary from situation to situation, and include those who evaluate, fund, purchase, order and utilize diagnostic technology. Additionally, many more elements are being aligned within and outside the diagnostic device in a context of politics, regulation and limited resources, adding to the multiplicity and complexity and the potential for frictions and trade-offs in constituting rightness.
Aligning with one element (or user) can conflict with aligning with another. In the politics of alignment, some tools are regarded as better aligned than others. Funders, and what I termed global intermediaries, can influence those co-construction processes to achieve the science and technologies they want (Taylor, 1992). There have been many calls for more participatory decision-making on directions of global health technology development and the values it serves (Epstein, 1995; Leach and Fairhead, 2007). In drug development, self-experimental drug consumption by end-users presents a new source of innovation and business model for pharmaceutical companies (Cooper, 2012). STS work on users has pointed out that user involvement and bottom-up technology development is not uncontested or necessarily more democratic (Hyysalo et al., 2016). Similarly, community engagement, widely regarded as an essential feature of good global health research conduct, is at times instrumentalized to improve study participation and operation (Geissler and Molyneux, 2011; Petryna, 2009; Reynolds and Sariola, 2018). Scarce resources, narrow profit margins and poverty-related diseases make for different dynamics of user representation and participation in global health research, especially in the absence of alternatives for accessing care (Crane, 2010; Kingori, 2015). What is more, the alignment dynamics analysed here defy traditional distinctions between top-down and bottom-up innovation processes prevalent in existing studies of medical innovation and users. Users of POC diagnostics are neither organized nor a clear group of stakeholders that act from the bottom-up, but dispersed, multiple, varied and shifting and alignment situations are multiplied. Alignment is multi-scale and multi-dimensional.
Whether a technology is well aligned depends on the tinkering strategies that continuously need to maintain rightness. Standardizing elements, politics and scarce resources cause frictions with the temporal dimensions of aligning and between the immutable and the fluid of sticky technologies (Scott-Smith, 2018). According to Fujimura (1996), standardizing elements increase doability, because they reduce uncertainty and the necessity for ad-hoc arrangements. When these standardizing elements travel, alignment can be achieved partly because the standards are not as rigid as they seem. Tinkering with standards to integrate them in each situation is part of articulating alignment (Fujimura, 1996). Notably, standardizing is easier for tasks at experimental level and harder for tasks at laboratory or social worlds level, because there is more variation between sites (Fujimura, 1987). Yet the ability to align standards is not necessarily problematized. My results show that aligning is not just a personal skill of cognitive bricolage (Castel and Friedberg, 2010) but is also explained by the actor’s structural positioning in particular contexts (Timmermans and Shostak, 2016). One of the standardizing elements in innovating POC diagnostics is the moment of so-called design lock. The alignment inside the box has to stop and devices are black-boxed. But in this case, the tinkering, ad-hoc arrangements, and re-alignments necessary to align these ‘finished’ products to the POC require extra effort and commitment by implementers to enact, make functional or tolerate the technology. In the case of Xpert MTB/RIF, alignment to essential components of POC, such as linkages to care, connectivity, repair or training, happened only in the years after market launch (Albert et al., 2016) and required commitment and entrepreneurship. When innovating POC diagnostics, the ability to tinker and continuously (re-)align heavily relies on available resources and enabling circumstances. Tinkering and improvisation should not be romanticized, though, because there are some impossible circumstances (Rieder, 2017).
No one actor alone can foresee and account for alignment work beforehand, or involve all possible users into design processes. Much aligning of diagnostics to the POC happens in the dark. Rather, the insights of shifting users and varied user representations and their sources allow for stratifying input and feedback across different actor groups, users, sources and moments in time, and for brokering between different ideas of what constitutes a POC diagnostic, a user (-setting) and a well-aligned test: between different ideas of the right tool for the job with the right user in the right place.
Footnotes
Acknowledgements
I am grateful to all the participants for granting me their insights and valuable time, and for Vijayashree Yellappa and the Institute of Public Health in Bangalore India for supporting me with part of the data collection. I am indebted to Anja Krumeich, Klasien Horstman, Harro van Lente, and Petra Wolffs for their support throughout this project and their comments on earlier versions of this manuscript. I would also like to thank Bart Penders and members of the HEAD discussion group at the Department of Health, Ethics and Society for their constructive feedback.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a VENI grant from the Dutch Science Foundation NWO (Grant number 16.158.004).
