Abstract
Introduction
Among fixed orthodontic appliances, Forsus fatigue-resistant device (FRD) is a popular choice for treating Class II malocclusion in permanent dentition due to its patient-friendly design. FRD utilizes nickel titanium coil springs to deliver precise and adjustable forces for clinical applications.
Material and method
A 16 gauge stainless steel wire is used for the fabrication of push rods.
Results
The fabricated push rods work with efficacy in advancing the mandible in forward position.
Conclusion
In cases of push rod size mismatch our innovative design overcomes these limitations, ensuring optimal fit and performance.
Introduction
Class II malocclusion is among the most frequent problems encountered in orthodontics. The most common feature of Class II malocclusion is mandibular retrognathism rather than maxillary prognathism, according to McNamara. 1 Functional orthopedic appliances are generally used to treat Class II malocclusions occurring due to mandibular retrusion.2, 3 The appliance selection usually involves removable or fixed functional appliances depending upon the existing anteroposterior discrepancy, cooperation, and residual growth of patients. Noncompliant Class II patients with mandibular retrognathia during growth spurts can be treated with a fixed functional appliance.4–6
The Forsus fatigue-resistant device (FRD) (Forsus: 3M Unitek, Monrovia, CA, USA) is a three-piece (L-pin module) or two-piece (EZ module), semi-rigid telescoping system incorporating a stainless steel rod that can be easily adjusted at the chairside, is compatible with fixed appliances, and is easy to place in the patient’s mouth.
Forsus FRD is attached to the maxillary first molar and mandibular archwire distal to the canine bracket. Different patients have different distances between the distal of the maxillary first molar and the distal of the mandibular canine. The primary size range of FRD includes six sizes, but we apply an innovative approach to address sizing issues.
Appliance Design and Fabrication
The initial measurement was recorded incorrectly. It might be because the patient did not bite into the proper position at the time of bite registration. Thus, a discrepancy of 10 mm was noted. We thought of getting some changes done to the push rod so that we could use the same appliance.
The Forsus FRD requires a rigid stainless steel push rod for the correction of mandibular retrognathia. Since the push rod must withstand masticatory forces during use, the wire needs sufficient rigidity. The wire used in our innovation is a 16-gauge, 0.052-inch, or 1.29 mm diameter thick stainless steel wire. The length of the wire can vary according to the size required by the clinician. The wire bending for the push rods on the left and right sides can be done with the universal plier, which is easily available in an orthodontic setup. After the wire bending is complete, use the provided template to mark 22 mm from the free end, where the stop should be soldered. Thus, push rods of different lengths can be fabricated, which are more economical than replacing the entire appliance (Figures 1 to 6).
Forsus Fatigue-resistant Device (FRD)—Push Rod.

16-gauge Stainless Steel Wire.

Fabrication of Push Rod with 16-gauge Stainless Steel Wire.

Soldering of Stop to the Push Rod.

Adjusting the Soldered Stop According to the Measuring Scale.

Comparison of Forsus Push Rod and Fabricated Push Rod.

Intraoral images with fabricated push rod (Figures 7 to 9).
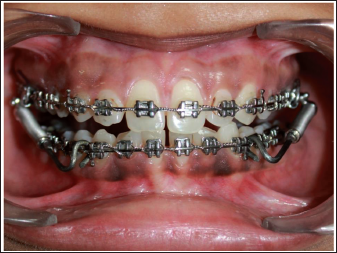
Frontal Intraoral Image with Fabricated Push Rod.
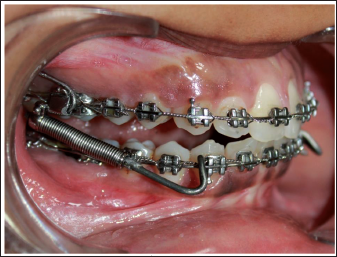
Right Lateral Intraoral Image with Fabricated Push Rod.
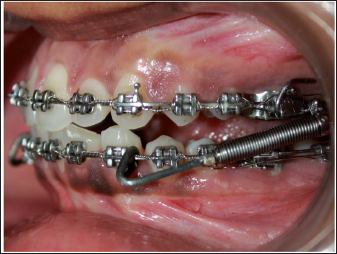
Left Lateral Intraoral Image with Fabricated Push Rod.
Advantages
Easily adjustable push rod size of the appliance.
Minimal armamentarium (16-gauge stainless steel wire, silver solder) needed for fabrication.
It is economical.
Footnotes
Declaration of Conflicting Interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent and Ethical Approval
Necessary ethical clearance and informed consent were received and obtained, respectively, before initiating the study from all participants.
