Abstract
Objective
To examine the effect of clinical use on both force retention and the deactivation of closed-coil nickel-titanium (NiTi) springs in a 16-week trial.
Methods
The force-activation curves for NiTi springs were determined before and after clinical use. The rate of tooth movement and maximum force (MF), hysteresis between activation and deactivation, and mean force of the deactivation plateau (MDF) were examined and correlated as a function of 4, 8, 12 and 16 weeks of clinical use. To recover the force properties, the springs were heat treated at 100°C and the results were compared with the preceding data.
Results
A total of 36 springs were analysed. The MF loss after use was 60, 74, 55, and 48 g for the 4-, 8-, 12- and 16-week springs, respectively. Heat treating had little effect on the MF. Clinical use lowered hysteresis by a mean of 180 g*mm compared with the pre-clinical use data, and heat treating increased the hysteresis by a mean of 59 g*mm above the post clinic testing data. The MDF was nominally 100 g less than the MF. Teeth moved approximately 1 mm/month, independent of the force loss.
Conclusions
The loss of MF and the lowering of the MDF was not time dependent. Heat treating can partially recover the mechanical properties of the used springs.
Introduction
In contemporary orthodontics, light, continuous and constant forces are expected to obtain the most efficient orthodontic tooth movement.1–4 Numerous materials (elastomeric chains, closing loops and coil springs) have been used as light force delivery systems to close space between teeth after premolar extraction.2,5–12 Although many of these orthodontic materials can initially deliver light forces, clinical studies reported that most of these materials exhibit force loss over time.6–9,11 Since the mid-1980s, the use of nickel-titanium (NiTi) for orthodontic springs has been studied due to its promise to deliver low, constant forces. 13
The thermal transitions of pseudoelastic NiTi materials are relatively well understood.14–17 With heating, these materials begin transforming to the austenitic phase (AS) from the martensitic crystalline phase until near complete austenitic conversion occurs (AF) (Figure 1a). On cooling, the reverse process occurs as the material returns to the martensitic phase (MS and MF, respectively). Concurrent with the crystalline change, the moduli and thus the stiffness increase with the transformation from the martensitic to austenitic crystal phases, and these same properties decrease on cooling. 17

Heating and cooling thermogram of a nickel-titanium spring indicating the locations of the starts and finishes of the phase transitions from the martensitic form to the austenitic form on heating (AS and AF, respectively) and the transition from austenitic to martensitic on cooling (MS and MF , respectively) (a). Typical pseudoelastic curve for nickel-titanium alloys indicating the transition from the austenitic crystalline form to the martensitic form on activation and back to the austenitic form on deactivation. Hysteresis is the difference between the activation and deactivation data. Also indicated is the constant linear region (CLR) of the deactivation curve and the mean of the deactivation force in this linear region (MDF) (b).
The stress induced hysteresis of NiTi is more complicated and not as well understood.14,15,18–20 In vitro studies have examined the force levels of NiTi springs.6–9,11–13 In typical force-displacement plots, on activation the force initially rises quickly followed by a more gradual rise until a maximum force (MF) is reached at the end of activation (Figure 1b). If the release of the strain begins within the plateau region of activation, there is a mix of martensitic and austenitic phases, and the material generally returns completely to the austenitic phase. 14 If strain continues beyond the plateau region, elastic distortion of the martensitic phase begins and the residual pockets of austenitic phase are converted to martensite. Above a certain strain, the martensitic lattices start to slip and permanent deformation starts. If the stress continues to increase, failure occurs. Depending on where the stress is removed there may be regions within the material that cannot fully transform to the austenitic form. 19 On deactivation, NiTi springs have an initial non-linear force loss followed by a relatively constant linear region (CLR) of force during the martensitic to austenitic transformation (Figure 1b).7,8,12,13 This CLR can provide a relatively constant force for tooth movement. Unfortunately, the force loss of CLR has been found to increase over time, similar to stress relaxation in elastomers.6–9,11 The stress necessary for activation is rarely the same as the stress of deactivation. This stress difference is mechanical hysteresis (Figure 1b).
In the pseudoelastic region, the hysteresis represents the resistance to motion caused by the internal friction between the phase boundaries and the mechanical resistance to the crystalline shape changes. 19 While under stress, thermal cycling above and below the austenitic and martensitic thermal transformation temperatures initially caused a drop in the transition temperatures after ∼50 cycles and then rose with ∼100 cycles. 20 This cycling increased the dislocation density and pseudoelastic cycling significantly reduced the transformation hysteresis.
A 28-day study comparing the force loss of elastomeric chains and NiTi closed-coil springs found that the elastomeric chains lost a mean of 37% while the NiTi springs lost a mean of 23%. 9 An earlier study reported force loss of 8% and 20% for two types of springs. 11 In a recent 12-week clinical trial, there was an initial loss of approximately 12% force after 4 weeks, a further loss of 7% over the next 4 weeks, and stabilization for the last 4 weeks. 7 A clinical investigation reported that NiTi springs initially lost CLR force over 6 weeks and then the force plateaued with a mean overall loss of 48%. 6 In another study, the slope of the CLR was significantly higher after clinical use, but the mean force of the CLR decreased. 8
This current study examined the effect of clinical use on force loss and the plateau region of deactivation of closed-coil NiTi springs during a 16-week clinical trial. The study hypothesized that the lost CLR force was not time-dependent and could be recovered by heat treatment.
Patients and methods
Patient population
This prospective study enrolled consecutive patients in the Department of Orthodontics Clinic of the Affiliated Hospital of Jining Medical College, Jining, Shandong Province, China who met the following inclusion criteria between September 2015 and December of 2016: (i) required space-closure treatment in the maxillary dental arch; (ii) were 12–18 years old at the start of treatment; (iii) were in the permanent dentition stage; and (iv) consented to participate in the study. Note that the age range of the patient population (12–18 years) was chosen to minimize biological variations while covering 80% of the orthodontic patients. All clinical procedures were approved by the Ethics Committee of the Affiliated Hospital of Jining Medical University (no. M-990320-011; 8 June 2015). All patients provided written informed consent prior to study participation.
Initial force level testing (INIT)
Prior to clinical use, the initial force level (INIT) of each spring was determined using dynamic mechanical analysis (DMA). DMA measures the mechanical properties of materials as a function of time, controlled force, temperature and frequency. The temperature was maintained at 37°C ± 0.1°C. Each spring was suspended on the DMA equipment (Model 2980; TA Instruments, New Castle, DE, USA) with 0.032” stainless steel hooks for 2 min at the test temperature (Figure 2a) and were stretched at a force ramp of 0.5 N per min up to 2.942 N (300 g). The force ramp was reversed to zero and the activation length at maximum force was recorded. The manufacturer of the springs (Dentsply GAC International, Islandia, NY, USA) suggested that the stretch range was ‘up to 15 mm activation’. In a previous study, many of the springs reached the full 12 mm activation range proposed by the manufacturer when stretched to 300 g. 7 These tests were run with a single frequency of 0.1 Hz, which typically yields results equal to traditional mechanical testing.

Close-up photograph of the dynamic mechanical analysis tension clamps, support wires, and the test spring (a); and close-up photograph of the apparatus used to test the springs after clinical use in tension (b).
Clinical study
The NiTi springs used in this study were reported to deliver 150 g of force (Sentalloy; Dentsply GAC International, Bohemia, NY, USA). The springs were nominally 9 mm long (eyelet to eyelet) and had 3 mm of active spring between two 3-mm eyelets. For the clinical study, the springs were attached to hooks that were cast onto archwires in the middle of the lateral incisor and canine to molar hooks or were placed between canine and molar hooks in some long interdental space cases (Figure 3). All the springs were stretched in the range of 3 to 15 mm. The brackets were MBT prescription (0.022 × 0.028 slot) and the archwires were 0.019 × 0.025 stainless steel. The springs were checked every 4 weeks during scheduled recalls. Nominally, 10 springs each were collected at 4, 8, 12 and 16 weeks. The sample size was determined using pilot data from a previous study. 7 Based on the pilot data, a minimum of nine samples (springs) for each time-point and 36 samples in total for four time-points were required to detect a maximum force loss of 30% with 90% power. Ten samples per group were expected to detect the difference. Following retrieval from the clinic, each spring was examined and photographed using a stereomicroscope (Model SMZ18; Nikon Instruments, Melville, NY, USA). Based on the clinical procedure as approved by the Ethics Committee, the operator replaced each coil with a new coil for each patient once the testing sample was removed.

Example of the coil spring attachment to teeth during clinical treatment.
Post-clinical force testing (PCT)
After clinical use, displacement control was used to test each spring in tension (PCT) (Model 4411; Instron, Norwood, MA, USA). While submersed in a double-chambered device at 37°C, the springs were pre-equilibrated for a minimum of 7 min prior to testing between two 0.032” stainless steel hooks (Figure 2b). The temperature in the inner bath was maintained by pumping water from a temperature-controlled water bath (HAAKE™; Thermo Electron, Karlsruhe, Germany) through the outer bath. The controller thermocouple was located in the inner bath. The springs were stretched at 5 mm/min to the maximum activation length determined by the DMA pretesting, and the load was recorded as a function of the activation length. When the maximum activation length was reached, the crosshead was reversed, and the spring was allowed to relax at the same rate.
Post-heating testing (HT100)
To examine the effect of heating, the clinically used springs were heated in air at 100°C for 30 min (Model F6038CM; Barnstead International, Dubuque, IA, USA) and retested on the mechanical tester as indicated above (HT100).
Hysteresis
Using each data point and the trapezoid method to calculate area, 21 the magnitude of the hysteresis for each test (INIT, PCT and HT100) was determined by subtracting the area below the deactivation curve from the area below the activation curve.
Constant linear region
The evaluation of the CLR was simplified by using the deactivation data between 2 mm below the maximum activation and the last 2 mm of deactivation for all curves (area between the two vertical dashed lines of Figure 4). These segments were evaluated using two methods: (i) the mean of the data within this segment was calculated (mean deactivation force, MDF); and (ii) the linear regression was determined for this segment of the deactivation. Slopes and y-intercepts were obtained for each curve.
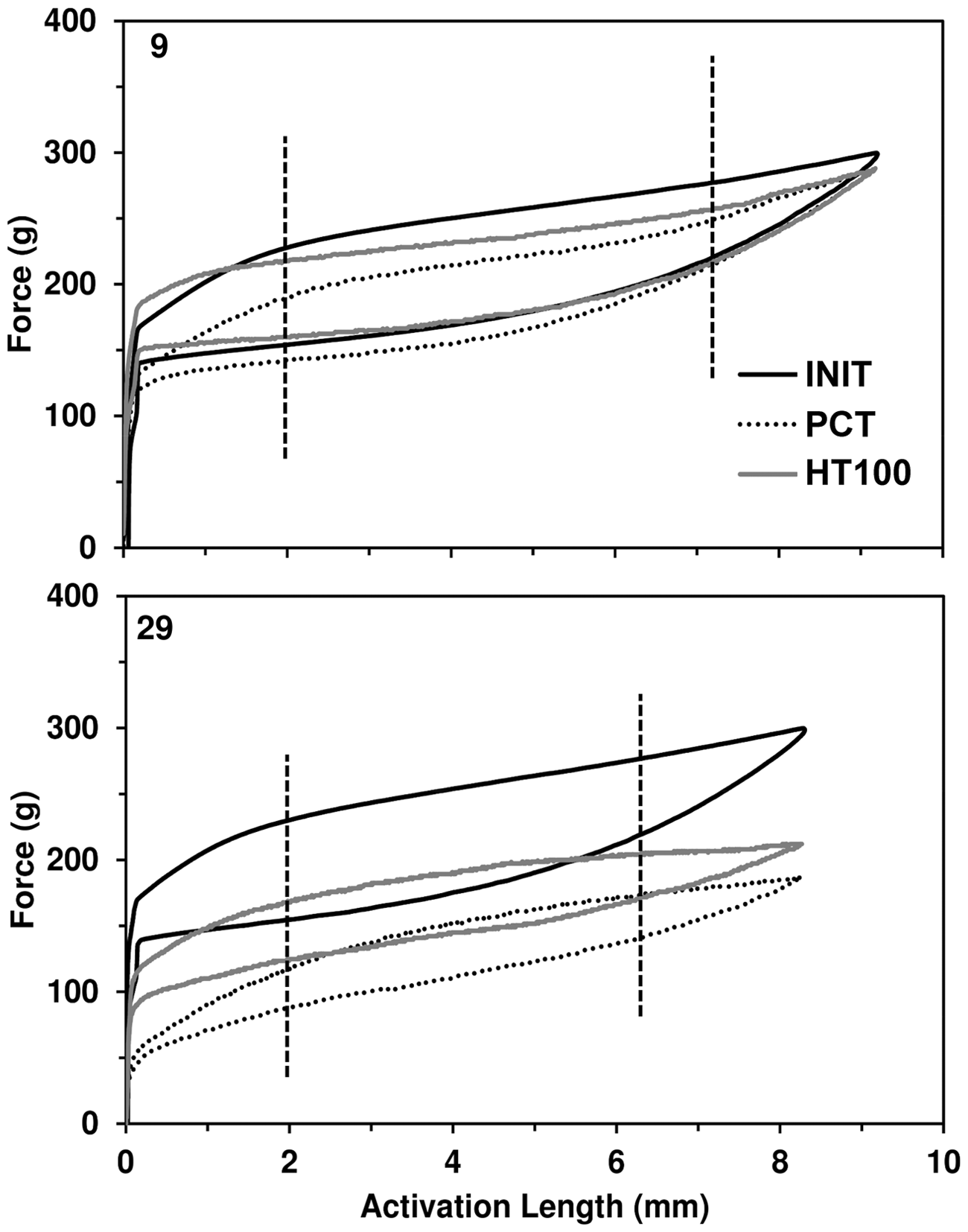
Representative force-activation curves for two 12-week springs, 9 and 29. Shown are the initial data (INIT), post-clinical data (PCT) and data after heat treating at 100 °C (HT100). The two vertical lines indicate the region used for the analysis of the constant linear region of the deactivation data.
Statistical analyses
Descriptive statistics, including mean and SD, were calculated for the maximum force and the space closure data using Microsoft Excel (Microsoft Corporation, Redmond, WA, USA). The space closure data were also examined using one-way analysis and plotted as box plots with the default quantiles using JMP software version 13.1 (SAS Institute, Cary, NC, USA). These data were additionally examined using linear regression to determine the P-value of the data using JMP software version 13.1 (SAS Institute). The significance of the CLR data was determined using linear regression analysis (Microsoft Excel).
The statistical significance of the relationships of the INIT, PCT and HT100 data was verified using two-way and one-way analysis of variance followed by Tukey–Kramer Honest Significant Difference test, post-analysis using JMP software version 13.1 (SAS Institute). A P-value < 0.001 was considered statistically significant.
Results
Microscopic examinations showed that only one spring was obviously damaged by clinical use (spring 30; Figure 5). This 12-week spring was removed from further analysis. Several springs were found to have eyelets that were not parallel (e.g. springs 9 and 29, Figure 5). Because this had been observed in these springs when they were received from the manufacturer, these springs were not removed from the study. One spring at the 8-week time-point and two springs at the 16-week time-point did not have any clinical data reported for space closure so these were eliminated from the study. As such, the data for 36 springs were analysed.

Photomicrographs of three springs after clinical use. Spring 30 was damaged during clinical use and was removed from the data analysis.
For the springs in this current study, activations from 6.4 mm to 10.0 mm were required to obtain the initial DMA force of 300 g. The force-activation curves for each spring were obtained for INIT, PCT and HT100 data. As an example of the extremes observed, the curves for two 12-week springs are shown in Figure 4. In spring 9, there was only 12.6 g difference in the maximum force between the INIT and PCT data; and there were only minor differences after heat treatment (11.7 g). In contrast for spring 29, the maximum force after clinical use was 113.0 g lower and the difference was 87.7g for HT100. The three unloading curves for spring 9 were similar in appearance but the forces for PCT were slightly lower. When the CLR data were compared, more curvature was observed in the INIT data of spring 29 than in the data for PCT and HT100, and the force values for the PCT and HT100 CLR were lower than the INIT forces. Heat treating raised the forces within the CLR for both of these springs.
The mean maximum force loss and the percentage loss was calculated for the PCT and HT100 data as compared with the INIT data (Table 1). Note in this table and subsequent figures, INIT values are the results for the pre-clinical data and are shown alongside the respective post-clinical use time-points for comparison. For the PCT data, the mean maximum force loss ranged from a low of 48 g for the 16-week springs to a high of 74 g for the 8-week springs (16% to 25% loss). After heat treating, the mean loss ranged from 44 g to 59 g (15% to 20% loss). For both post-clinical use data sets, the lowest force losses were observed for the 16-week springs and the highest were observed for the 8-week springs. The maximum forces for PCT and HT100 were significantly lower than INIT for all weeks (P < 0.0001), but not significantly different from each other. The difference between the initial values and the two post-clinical use data values did not increase after the 4-week time-point.
Summary of maximum force data at activation of the springs as a function of the duration of clinical use.

Data presented as mean ± SD.
Data before clinical use; bdata after clinical use; cdata after heat treatment of the used springs at 100°C.
Mean ± SD for all springs.
Differences between the data before clinical use (INIT) and the data after clinical use (PCT and HT100).
Percentage differences between the mean data before clinical use (INIT) and the data after clinical use (PCT and HT100).
Space closure over the 16-week clinical trial progressed at an overall rate of 0.22 mm/week (P < 0.001; regression analysis across the 16 weeks; dotted line, Figure 6). The mean ± SD cumulative space closure at the individual 4-, 8-, 12- and 16-week time-points were 1.2 ± 0.3 mm, 2.2 ± 0.7 mm, 2.9 ± 0.4 mm and 4.0 ± 0.5 mm, respectively. The mean ± SD closures rates were 0.30 ± 0.08, 0.28 ± 0.08, 0.24 ± 0.04 and 0.25 ± 0.03 mm/week for the four clinical time-points, respectively (1.1 mm/month).

Box-whisker plots of the space closure as a function of the duration of clinical use using standard box plot quantiles. The horizontal line represents the median of the data. The box delineates the 25th and 75th percentiles. Where present, the horizontal lines connected to the whiskers represent data in the outer quartiles. The dotted line is the linear regression of the data (circles, Y = 0.22X + 0.34; P < 0.001).
The areas of the hysteresis curves were statistically independent of the clinical time-point (Figure 7a). The mean hysteresis area for the PCT was 180 g*mm lower than the INIT data. Heat treating increased the area by 59 g*mm above the PCT data, but these areas were still substantially lower than the INIT data (P < 0.001 for all comparisons). The HT100 data were significantly different from the PCT data at all time-points (P < 0.001 for all comparisons), but the magnitude of the differences did not increase with use.

Analysis of the pseudoelastic curves as a function of the duration of clinical use: (a) hysteresis area; (b) mean force of the deactivation plateau (MDF) of the constant linear region (CLR) data between 2 mm less than the maximum force and 2 mm strain (see Figure 4); (c) mean value of the slopes from the linear regression of the CLR data; (d) mean value of the y-intercepts from the linear regression of the CLR data. INIT, initial data; PCT, post-clinical data; HT100, data after heat treating at 100°C. Data presented as mean ± SD.
Even though there were inherent errors in making the identical 2 mm data cuts to examine the deactivation data, the regression lines of all of the CLR were statistically significant (P < 0.001 for all comparisons). Just as was the case with the hysteresis data, the mean values of MDF were independent of the clinical-time point (Figure 7b), but the differences between the three test results (i.e. INIT, PCT and HT100) were significant (P < 0.001 for all comparisons). The mean ± SD PCT data (148.14 ± 19.9 g) were always lower than the mean ± SD INIT data (189.9 ± 4.9 g); and heat treating always raised the value of the mean ± SD MDF (164.0 ± 16.1g). These differences were significant (P < 0.001 for all comparisons); however as above, the difference did not increase with clinical use. For all data sets, the slopes of the plateau region significantly decreased after clinical use (P < 0.001 for all comparisons) (Figure 7c). Heat treating the used springs further reduced the slopes, but by a lesser amount. The mean of the slopes for the four clinical time-points were 11.3 g/mm and 9.7 g/mm for the PCT and HT100 data, respectively. Heat treating significantly lowered the slopes (P < 0.013 for all comparisons). Other than a slight increase at week 12, the differences with use remained relatively constant. The y-intercepts of the PCT data (Figure 7d) dropped 18 g following clinical use (P < 0.001), and there was an increase between PCT and HT100 (23 g). The HT100 intercepts were slightly higher than the value for INIT, although the differences were not significant.
Discussion
In this current study, the mean space closure rate (1.1 mm/month) agrees with other values reported in the literature.5–7,22 Regardless of the decreases observed in other properties in this current study, the closure rate was consistent across the 16-week clinical trial. So the observed force losses did not affect the closure rate. The data suggested that the force decay of NiTi coils during clinical use did not drop below the stimulation threshold for tooth movement. Generally, high standard deviations were observed (Figure 6), with much of this deviation probably due to the non-uniform force characteristics of individual springs and biological differences between patients. For example, spring 9 required 1 mm more activation than spring 29 to reach the same initial force (Figure 4). The inconsistency in activation length to obtain a predictable force (range of 3.6 mm in the DMA tests) continues to be an issue for the clinician who needs to accurately predict the applied force with a specific extension. The specific cause of this deviation is not known, but if eliminated, would greatly improve the clinical application of NiTi springs.
In a previous paper using the same springs as this current study, the thermal martensite to austenite transformation was shown to finish (AF) (Figure 1a) at around 44°C, though the full conversion did not occur until even higher temperatures. 7 Ideally, to have the optimum pseudoelastic plateau, the temperature for activation should occur when the NiTi has thermally transformed completely to the austenitic phase (AF).14,15,18 At 37°C, these springs were activated at a temperature below the AF, which increased the complexity of the crystalline phases and possibly contributed to the force losses.
Though these NiTi springs did not encounter controlled thermal cycling, 20 the springs did undergo temperature cycling due to the daily intake of food and drink. Surface temperatures during a typical 30 min meal have been reported to cycle down to 23°C with a cold drink, have a slight increase to 25°C with a salad, cycle between 30°C and 43°C while eating the main course, go down to 4°C with ice cream, and go up to 43°C with coffee. 23 This range of temperatures are below and above the austenitic–martensitic start/finish thermal transition temperatures (Figure 1a). Others have reported the effect of temperature on NiTi springs and wires in the pseudoelastic region.24–29 The rise in temperature above AF overrides the stress-induced crystal transformation as the material partially converts to the austenitic form. This increase in the percentage of austenite would raise the modulus of the wires, 17 and thus increase the forces. In bending and torsion, previous research reported that the effects of short-term heating on superelastic wires under activation of deactivation were transient, but after short-term cooling the recovery was slower.26–29 The long-term effect of repeated cooling and heating cycles of springs in vivo is not known. If the results of controlled thermal cycling 20 is extrapolated to oral cycling, the dislocation density in the crystal lattices would increase, which would resist the transformation to the austenitic crystalline form. Thus, the amount of untransformed martensite would keep the modulus lower and yield lower forces. Lower forces are desirable, but the non-predictability of these changes complicate clinical treatment plans.
The drop in hysteresis following clinical use (Figure 7a) would suggest either a stress- or thermal-induced change has occurred in the crystalline lattice to lower either the internal frictional resistance between phase boundaries and/or lowered the mechanical resistance to the crystalline shape change. 19 For this to occur, either the AF temperature has shifted to a higher temperature or the martensite lattice has been trapped when the stress was removed. Post-clinical use heating at 100°C was sufficient to partially alleviate the change to begin returning the alloy to the preclinical configuration.
The procedure used to determine the MDF was comparable with the technique proposed by others8,15 and was based on a procedure originally used to analyse the bending data of superelastic archwires. 30 Only after clinical use did the MDF for the springs equal the manufacturer’s declared value. The mean difference between the maximum forces and the MDF were around 100 g (Table 1 and Figure 7b). Based on the range of activation used in the clinical trial some springs exerted much higher forces than expected. After use and/or heat treatment, the drop in the slope indicates the deactivation force plateau became more constant.
Because the length of clinical use did not significantly affect any of the calculations after the 4-week time-point (maximum force, hysteresis, MDF, y-intercept and slope), the physical causes for the force changes must occur within the first 4 weeks of use and must be non-reversible after this 4-week period. Within these 4 weeks, the spring would be at the highest stress due to placement and the longest elongation. The primary cause of the force losses may be in part due to the initial placement of the spring. A previous study demonstrated a decrease in MDF force with increasing activation. 31 With an initial spring length of 3 mm, the range of activation in this current study of 3 mm to 15 mm represents activations of 100% to 500%. Even though no obvious permanent deformation (except spring 30) was seen in the springs or the curves, any internal deformation and crystal lattice changes would affect the realignment of the lattice. 19
While the heat treatment at 100°C was not sufficient to completely return the material to the initial mechanical properties, some recovery was found in the maximum force, hysteresis, MDF and y-intercept in the current study. The recovery of the y-intercepts to pre-use levels might indicate the material was able to return to the original austenitic phase concentration at 0% strain. The understanding of the pseudoelasticity of NiTi springs is further complicated because when coil springs are stretched, in addition to tension and compression, the wire is primarily in torsion. The effects of torsion on the pseudoelasticity of NiTi has not yet been thoroughly reported in the literature.
Even though NiTi springs have the capability to produce low forces on deactivation, the magnitude of the forces is currently not predictable, which complicates their clinical use. Further studies are needed to examine the effect of the thermal cycling under stress on AF and to examine if there are any permanent modulus changes due to these internal changes.
In conclusion, this current study demonstrated that the loss of maximum force and the lowering of the forces in the deactivation plateau were not time-dependent in NiTi closed-coil springs. Care must be taken not to over strain springs at placement within the mouth. The thermal cycling from daily food and drink may contribute to the force losses observed with NiTi closed-coil springs. Heat treatment can help return the springs to their initial states, but the current conditions were not totally sufficient. The clinical rate of tooth movement was confirmed to be approximately 1 mm/month using NiTi closed-coil springs and was consistent over the 16-week clinical trial.
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Footnotes
Funding
This research was supported, in part, by a grant from the US National Institutes of Health/The National Institute of Dental and Craniofacial Research (no. R01DE022816).
