Abstract
Objectives
The objective of this study was to evaluate the ability of the bioactive glass-bonds to inhibit the demineralization surrounding the orthodontic brackets and evaluate the physical and mechanical properties in order to be accepted as an orthodontic bonding agent.
Materials and Methods
Preparation of mesoporous bioactive glass (MBG) used an improved sol-gel method. The samples were prepared with different surfactant to oil ratio with 12 groups and one control group with six samples each. The debonding characteristics were analyzed using an Instron Universal Testing Machine (UTM). After six months of pH cycling in vitro saliva solution to simulate the oral environment, the microstructure was analyzed using a field emission gun-based scanning electron microscope (SEM). The sample, which is viable in terms of physical and mechanical properties, is converted into a single paste form by addition of resins and compared with the control group Transbond XT adhesive.
Results
SEM analysis showed that the prepared MBG particles possess spherical morphology. Novel orthodontic bonding material (BG) provides adequate mechanical properties with bond strength of 7.2 MPa (p = .304) in order to be accepted as an orthodontic bonding agent since its bond strength both at the time of bonding (p = 1) and after six months (p = .325) in simulated salivary conditions is in par with conventional bonding resin (TBXT) with reduced demineralization potential.
Conclusion
This novel adhesive results in reduced demineralization surrounding orthodontic brackets and possesses optimal physical and mechanical properties to be accepted as an orthodontic adhesive.
Introduction
Decalcification of the enamel surface adjoining the bracket bonded area of the fixed orthodontic appliance is an important ubiquitous iatrogenic effect of orthodontic therapy.1, 2 Due to the challenges in maintaining oral hygiene with fixed orthodontic appliances, the bracket often acts as a reservoir for plaque buildup. The remineralization process can be impeded by the acidic environment of plaque surrounding orthodontic brackets, potentially leading to the development of white spot lesions (WSLs), which are typically indicative of enamel decalcification.3, 4 Fejerskov and Kidd 5 defined WSL as the “first sign of a caries lesion on enamel that can be detected with the naked eye.” Gorelick et al. 6 found that the incidence of at least one WSL was 50% in patients who had treatment with fixed orthodontic appliances compared with only 24% in untreated controls.
The field of orthodontic adhesives has seen several advancements aimed at creating materials with minimal enamel damage while still maintaining sufficient mechanical and physical properties. Over time, bonding materials have progressed from Glass Ionomer Cement (GIC) to the latest nanobioadhesives.7, 8
Bioactive glasses (BAGs) have found extensive application in hard tissue regeneration, including bone and dental restoration. When exposed to simulated body fluids (SBFs), BAGs exhibit biomimetic behavior, resulting in the formation of tooth-like hydroxyapatite that can even deposit on organic polymers. 9
The microemulsion phase diagram 10 illustrates the different phases that can be formed when oil, water, and surfactant are mixed together. These phases are influenced by factors such as the composition of the mixture, temperature, and pressure. The diagram helps to identify the boundaries between various phases, including oil-in-water, water-in-oil, bicontinuous, and microemulsion, while most nanoparticle synthesis is conducted in the single-phase region, a relatively unexplored area known as “bicontinuous” offers the opportunity to produce a network of uninterrupted BAGs. This network has the potential to impede crack propagation, thus preventing microfractures and increasing adhesive properties. Our approach will involve utilizing this process to prepare uninterrupted BAG networks. The morphology of the gel surface layer is a key component in determining the bioactive response. The inherent high porosity of bioactive glasses derived from the sol-gel process is thought to contribute to high bioactivity of this material. 11
To date, rarely any bracket bonding material has been able to address all three factors that can impact enamel demineralization around brackets: pH control, antimicrobial activity, and ion release. Therefore, the aim of this study was to develop an adhesive with optimal physical and mechanical properties, along with an added benefit of reduced demineralization.
Materials and Methods
Preparation of Mesoporous Bioactive Glass (MBG) Powder
A new sol-gel method 12 was used to synthesize bioactive glass by utilizing micro-emulsion droplets containing a soft template of EA-CTAB-water. In this method, 0.7 g of hexadecyltrimethyl ammonium bromide (CTAB) was dissolved in 33 ml of deionized water (DW) at 303 K with stirring until it was completely dissolved. Afterwards, 20 ml of ethyl acetate (EA), 7 ml of aqueous ammonia, 3.6 ml of tetraethyl orthosilicate (TEOS), 0.36 ml of triethyl phosphate (TEP), and 2.277 g of calcium nitrate tetrahydrate (CN) were added sequentially at 30-minute intervals while vigorously stirring the solution for four additional hours. The white precipitate produced was collected through centrifugation, washed three times with ethanol absolute (EtOH) and DW alternately, and dried at 333 K for 24 hours. Finally, the organics and nitrates were removed by calcination at 923 K under air pressure. To produce different surfactant oil ratios, the concentrations of CTAB and EA were varied, and 12 different variants were synthesized and grouped as indicated in Table 1. For example, 0.7 g of CTAB with 20 ml of EA having all other components standard falls under Group 1 with surfactant-to-oil ratio of 0.016514. The control group is composed of Transbond XT adhesives from 3M Unitek.
Surfactant-to-Oil Ratio in Various Groups.
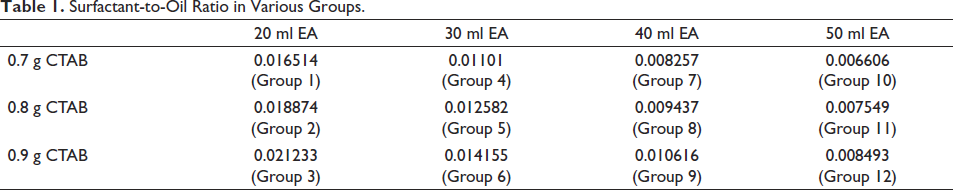
Sample Preparation
Based on the study conducted early, the values were substituted in G power software 3.1.9.7. The power of the study was 0.85 and α error was -0.5%. The sample size estimated was 6 per group and the effect size was 1.92. Since it is an in vitro pilot study, the minimum sample size of 6 was chosen. A total of 165 human first premolars extracted for orthodontic reasons, which are devoid of any pathology or defects on the buccal surface, were collected and stored in Chloramine-T trioxide (5%) at approximately 298 K. Of the collected teeth, nine teeth with any pre-existing WSL were excluded. The samples were randomly assigned to different groups. Each group was assigned with 12 teeth, first six being used for initial bond strength and the latter six for six months bond strength determination.
Preparation of BAG-bond
To prepare BAG bonds, the powder obtained from the sol-gel method discussed earlier was mixed with monomer resin, specifically the primer, that is, Transbond XT Primer from 3M Unitek, in a 4:1 powder-to-liquid ratio by weight, which is the ratio used in the control sample (Transbond XT adhesives). The standard bonding protocol was followed for both the groups.
pH Cycling
To simulate a six-month caries challenge, a pH cycling process was conducted in an artificial environment. Each sample was immersed in 40 ml of neutral pH saliva for 18 hours, followed by 6 hours in an acidic saliva solution. The cycle was repeated for six months, with fresh solutions prepared each week. 13 This pH cycling process exposes the teeth to continuous cycles of demineralization and remineralization, designed to mimic the dynamics of mineral loss and gain involved in caries formation.
Bond Strength
A UTM was used to measure the shear bond strength (SBS) where the bonded teeth were loaded with a crosshead speed of 0.5 mm/min. The force to debond was recorded in pounds. The SBS is calculated using the formula:
Shear bond strength (MPa) = Force (lbs) / bracket base area (9.61 mm2)
SBS was measured for all the groups both for the initial and six months caries challenged samples.
Adhesive Remnant Index
After testing with UTM, the brackets and respective teeth were observed for the Adhesive Remnant Index (ARI). The bracket base and the bonding surface were viewed under 10× magnification using a light stereomicroscope to determine the location of bond failure and derive at an ARI scoring. The scoring was done according to the modified ARI. 14
Scanning Electron Microscopic Analysis
The powder of all 13 samples was viewed under SEM to qualitatively assess the nanostructure of the samples. For control group, powder is obtained by heating the transbond adhesive in a furnace at 500ºC for 6 hours. All pH cycled samples were debonded and were stored in 0.5% sodium hypochlorite and copper for decontamination. The teeth were sectioned parallel to the long axis of the teeth in a buccolingual and mesiodistal direction. The teeth were cut using a water-cooled diamond wafering blade on a slow speed micromotor and serially polished through 4,000-grit silicon carbide polishing paper. Each specimen was sectioned into a 2 mm section to better fit the SEM stub. Each of the sections were washed with hydrogen peroxide and air dried to remove all debris collected during the sectioning and viewed under SEM to qualitatively check for the demineralization zones differentiated by density-dependent color (DDC) detection.
Preparation of BAG-based Adhesive in Single Paste Form
The premier group was selected from the 12 groups based on the initial and six months bond strength, ARI, and SEM evaluation. The sample found more reliable after all the experimental testing was used to make the single paste form, which is easily dispensable. The hydrophilic glass fillers are hydrophobized through surface modification using an overhead stirrer, before using in an adhesive form. BisGMA (10–20%), TEGDMA (10–15%), BAG fillers (50–55%), and Glass/Silica fillers (5–15%) are weighed and physically blended with pestle till a homogenous paste is obtained. The paste is then tested for mechanical properties and packed in black syringes.
Statistical Tests
Statistical analysis was performed using statistical software (SPSS version 16, IBM). The level of significance was set to 0.05 for all statistical inferences. For intergroup comparison of parametric values, ANOVA was used. Further, Tukey’s post hoc test was used for the individual comparison between groups and with control group. For non-parametric values, Mann–Whitney test was used.
Result
The average initial and six months bond strength of all 13 groups with the p value derived out of ANOVA test is mentioned in Table 2. The p value of .00 indicates a significant difference in bond strength values among different groups. The comparison of each experimental group with the control group is done with a post hoc analysis (Table 3). The p value = 1 indicates equal values of bond strength of experimental and control group.
One-way Analysis of Variance (ANOVA) for Initial Bond Strength.
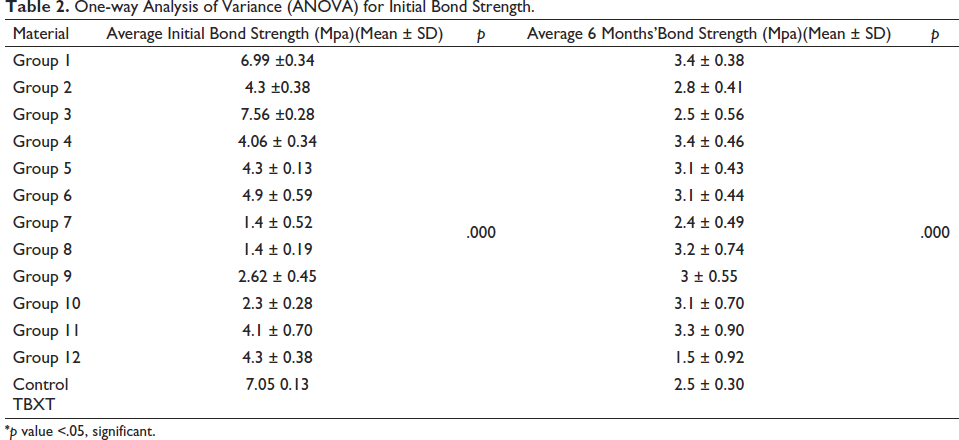
Post Hoc Analysis for Comparison of Initial and 6 Months’ Bond Strength of BAG and Control Group.
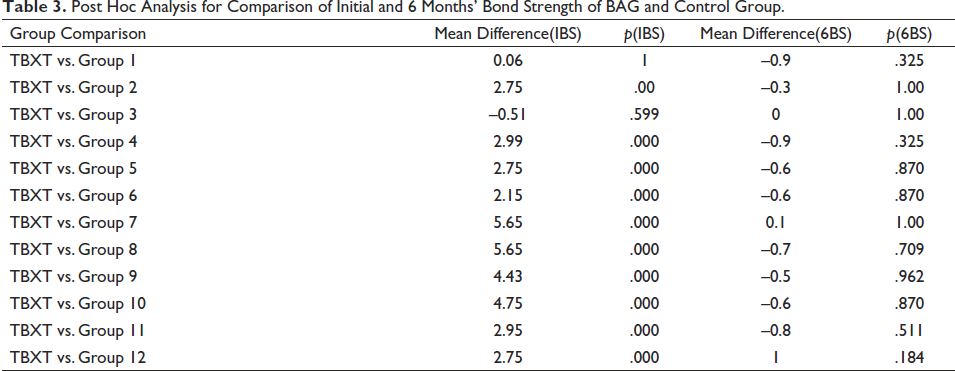
Characterization of Mesoporous Bioactive Glass Nanoparticles
SEM analysis shows that the prepared MBG particles of Group 1 possess spherical morphology with generally uniform size as shown in Figure 2, which shows a great mechanical advantage. And when viewed after the pH cycling for six months, it shows regular packed structure of remineralization with deposition of mineral content. No specific shape was made out in the SEM image of the control group and numerous areas of demineralization are present, which is indicated in Figure 3.
Graphical Representation of Initial and 6 Months’ Bond Strength of Group 1 (Left) and Control Group (Right).
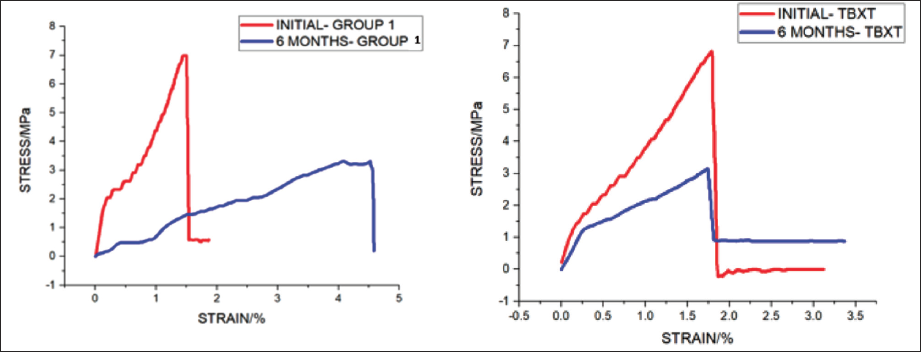
SEM Image of Powders of Different Groups: Left - Group 1; Right - Control (TBXT).

SEM Image of Tooth Bonded with Left - Group 1 and Right - Control Group.

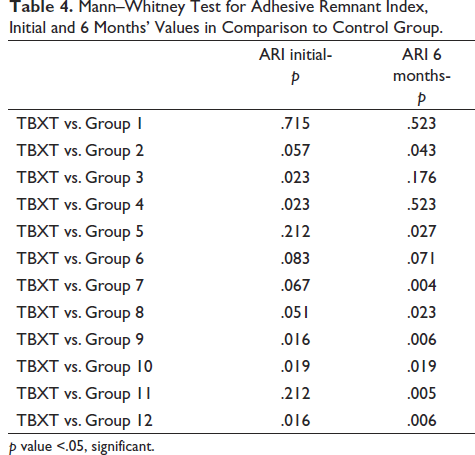
Adhesive Remnant Index
Mann–Whitney test values indicate that Group 1 shows ARI similar to that of the control group.
Single Paste System Characters
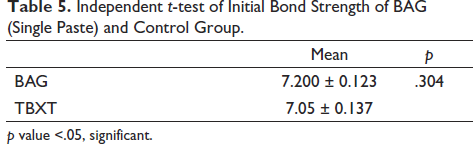
The independent t-test indicates that the mean value of bond strength of the BAG group is higher than the control group, that is, TBXT but is statistically insignificant.
Discussion
Various methods have been developed to reduce WSLs around the bracket, such as the use of fluorides, GIC, and CPP-ACP. While these agents have shown some success in reducing lesion depth and volume, they do not eliminate WSLs entirely.7, 8 However, a promising new option is the use of bioactive glass modified adhesives as a novel orthodontic bonding agent. These adhesives contain bioactive glass, which can release calcium and phosphate ions and rapidly dissolve to maintain a high ionic concentration. This material has properties such as antibacterial action, 15 pH control, 16 and the ability to maintain enamel’s mechanical properties while inhibiting demineralization and promoting remineralization. 17 The discovery of such material could be crucial in preventing the complications of orthodontic treatment caused by enamel damage after bracket bonding procedures. From all the tests and statistics above, it is established that Group 1 having a surfactant-to-oil ratio of 0.016514 was found to be better than the other compositions, in terms of both physical and mechanical properties. So, the Group 1 composition was used for the conversion into a single paste system.
Bond Strength
The new bioactive glass (BAG) adhesive has demonstrated an optimal bond strength of 6–8 MPa, 18 which falls within the safe range for bracket bonding, debonding, and adhesive removal without causing enamel damage. The present study compared the bond strength of the experimental resin, which contains bioactive glasses, with the gold standard resin (Transbond XT) and found that there was no statistically significant difference between the two (p = .304). However, the bond strength of the experimental resin was within the optimal range and even superior to the control group. Group 1, which had the experimental resin, showed optimal properties in terms of initial bond strength (6.99 ± 0.34 MPa) and six-month bond strength (3.4 ± 0.38 MPa) compared with the control group (Table 2). Moreover, when the BAG adhesive was formulated into a single paste, it maintained its physical and mechanical properties and had a bond strength of 7.2 MPa (Table 5), which was higher than that of the control group (TBXT). These findings indicate that the BAG adhesive is a promising alternative to the gold standard resin for orthodontic treatment.
Mann–Whitney Test for Adhesive Remnant Index, Initial and 6 Months’ Values in Comparison to Control Group.
Independent t-test of Initial Bond Strength of BAG (Single Paste) and Control Group.
Adhesive Remnant Index
Bond failures during orthodontic treatment can occur as tooth/adhesive and bracket/adhesive failures (adhesive failure) or within the adhesive itself (cohesive failure). Adhesive fracture can result from inadequate primer penetration due to reduced enamel demineralization, 19 making it easier to remove any remaining material from the enamel surface. To minimize enamel damage during debonding, it has been suggested that a weaker adhesive with a lower bond strength value may be preferable to increase bracket failure at the resin-enamel interface. 20 It is also desirable to have an equal amount of adhesive remaining on the tooth surface and bracket base to decrease the potential for enamel damage during debond procedures as suggested by Greenlaw. 21 The current study found that BAG Group 1, which had bond strength values and ARI scores similar to or greater than the control group, is a viable alternative to the gold standard resin for orthodontic treatment with a minimal risk of enamel damage during debonding procedures.
Remineralization Potential
The key feature of an orthodontic adhesive is its ability to promote remineralization in the circum-bracket area and prevent the formation of enamel WSLs. WSLs are a sign of early caries and are caused by an imbalance between demineralization and remineralization processes.22–24 The present study shows that BAG-containing adhesives have a higher remineralization potential compared with the control group on observation through a density-dependent color-SEM (DDC-SEM) estimation (Figure 3), which reveals more dark areas of gray scale as compared to the control group, which is indicative of demineralization.
Other studies have also demonstrateonds in maintaining enamel surface hardness and preventing WSLs. Manfred 25 found that teeth bonded with BAG had less reduction in enamel microhardness at depths of 25 and 50 nm compared with traditional bonding agents, indicating reduced superficial enamel softening surrounding orthodontic brackets. Brown et al. 26 also identified three approaches by which BAG promotes remineralization: raising the pH of cariogenic environments, providing an ion source for precipitation of Ca-P onto the tooth surface, and decreasing the resources for bacterial metabolism. The transient increase in pH of saliva when BAG is exposed to saliva is due to the release of ions, which leads to remineralization, which may be the possible explanation for reduced demineralization in the experimental group. This result is consistent with the study by Delgan et al., 27 which states that the transient increase in pH of the saliva when BAG is exposed to saliva is due to the release of ions, which is responsible for the remineralization effect. Wanitwisutchai et al. 28 also stated from their experiment that the orthodontic adhesives containing BAGs demonstrated a significant buffering capacity but did not show significant antibacterial properties against S. mutans and S. sanguinis.
Based on the results of the present study and previous research, the novel BAG of Group 1, with a surfactant-to-oil ratio of 0.016514 (Table 1), is considered optimal for preparing a sample with regular bicontinuous structure, as well as optimal physical and mechanical properties and remineralization potential. This finding suggests that BAG-bonds have the potential to be an effective orthodontic adhesive for preventing WSLs and maintaining enamel surface hardness.
Conclusion
The bioactive glass based adhesives can reduce superficial surface demineralization of enamel when exposed to an in vitro caries challenge test.
This novel orthodontic bonding resin provides adequate mechanical and physical properties to be used as an orthodontic adhesive.
The BAG-based adhesive was found to be best in its both physical and mechanical qualities, and demineralization effect when the surfactant-to-oil ratio was maintained at 0.016514 in the bicontinuous phase. This bicontinuous phase adhesive comprises a cross-linked matrix comprising hydrolyzed alkoxides of SiO2, CaO, and P2O5.
BAG-based resin was found to have a score of ≥2 (>25% adhesive left on the bracket), that is, 83.4% when compared to conventional adhesive with a frequency of 66.7%.
The upscaled bioactive glass adhesive, formed into one paste form by the addition of resin and other fillers to the BAG-based powder, is easy to dispense and can be used clinically, which is advantageous.
Limitation
The study was conducted in vivo with simulated oral environment, but according to Pickett et al.,[29] in vivo bonding systems are exposed to numerous intraoral factors, including saliva, acid, masticatory forces, variable patient abuse, and orthodontic mechanotherapy during the time period of comprehensive orthodontic treatment, which could not be simulated.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical Approval
The ethical clearance was approved by Banaras Hindu University, Institute of Medical Sciences. The IEC No. 2021/IEC/2707.
Funding
The research is funded by Indian Council of Medical Research (ICMR). Referece No.3/2/Dec-2021/PG-Thesis-HRD(46D), dated 18/04/2022.
Informed Consent
Since it is an in vitro study, patient consent was not taken.
