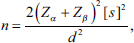Abstract
Objective
The research purpose was to inspect the impact of deproteinization of enamel through the use of 10% papain on the shear bond strength (SBS) of resin-modified glass ionomer cement (RMGIC) and single-step composite (Biofix), and compare it with the regularly used composite bonding material (Transbond XT).
Methodology
Extracted premolars (120) were equally separated into four groups. Group A (RMGIC + papain), Group B (Biofix + papain), Group C (Transbond XT + papain), and Group D (Control) (Transbond XT without papain). The SBS and adhesive remnant index (ARI) were studied and juxtaposed.
Results
There were statistically significant differences (P < .01) observed in mean SBS values among all groups. According to the study results, the mean SBS value of Group C (12.77 + 3.13 MPa) was statistically more than that of the other groups (P < .01). Although, differences in values among Groups A (8.55 + 3.61 MPa), B (7.44 + 2.34 MPa), and D (9.71 + 4.19 MPa) were not significantly different (P > .05). The study did not find any relevant difference in the mean ARI scores between all groups (P > .05).
Conclusion
Deproteinization with the use of 10% papain has significantly improved the SBS values of RMGIC and Biofix. Making these materials comparable to conventional composite.
Introduction
White spot lesions (WSLs) are a usual issue that patients often experience during orthodontic treatment, caused by plaque accumulation around brackets. The incidence and prevalence of WSLs among orthodontic patients are significantly more, 1 as reported by Boersma et al., who found that 97% of their study participants receiving fixed orthodontic treatment were affected by WSLs. 2
Resin-modified glass ionomer cements (RMGICs) have been shown to continuously release fluoride during orthodontic treatment, serving as a “fluoride pump” by absorbing fluoride from dentifrices and oral rinses and re-release it in areas that are most vulnerable to WSL.3, 4 Research studies have demonstrated that RMGIC results in less demineralization compared to a conventional composite during fixed orthodontic treatment.5–7 Biofix, a single-paste system, has been found to release fluoride after bonding, with the added advantage of reducing one step (primer application) during bonding. 8 However, despite all the favorable characteristics, both Biofix9, 10 and RMGIC11–13 have been found to have lower shear bond strengths (SBSs) as compared to conventional composites. Hence, any method that can improve the SBSs of these materials and make them comparable to conventional composites can provide an alternative bonding material with enhanced tooth surface protection.
Despite pumicing the teeth prior to acid-etching, organic matter in the form of pellicles can remain attached to the tooth surface. This material controls the mineral dissolution process, making it resistant to acid attack and thus preventing optimal etching. Subsequent inadequate etching can result in bond failure and WSLs formation around the bracket base. 14 To address this issue, deproteinization can be done prior to acid-etching to provide Type I and II etching patterns, which in turn increase resistance to orthodontic traction. 15 Justus et al. first proposed using 5.25% sodium hypochlorite to remove organic material. 16 Moreover, Pithon et al. propounded using 10% papain gel as a deproteinizing agent before acid-etching and confirmed that its use intensifies the bond strength of RMGICs.17, 18
Hence, the present study aimed to investigate whether 10% papain could enhance the SBS of RMGIC and single-step composite (Biofix) and to compare it with the conventional composite. Additionally, the study aimed to appraise and contrast the adhesive remnant index (ARI) of these materials. The selection of these materials was based on their ability to release fluoride, which is an added advantage over conventional composite resin.
Material and Methods
Preparation of Study Samples
For this study, 120 human premolars that have been extracted were utilized. As it is an in vitro study, the samples taken were extracted premolars; therefore, patient consent was not mandatory.
Inclusion Criteria
Teeth with intact buccal surfaces.
No known exposure to pre-treatment chemicals.
No cracks or fractures due to extraction forceps.
Free of caries and restorations.
Exclusion Criteria
Teeth with any developmental anomaly and hypoplastic/hyperplastic abnormalities were excluded.
The mean and standard deviation values were obtained by using the following sample size calculation formula:
Where Zα is the z-variate of alpha error (a constant with a value of 1.96), and Zβ is the z-variate of beta error (a constant with a value of 0.84). The calculated sample size, using this formula, was 28; however, to be on the safer side, we included 30 samples in each group.
Methodology
To keep the extracted teeth viable, they were placed in a normal saline solution and kept at room temperature. To facilitate identification, notches were made on the roots, and the teeth were fixed in custom-made cylindrical acrylic blocks, and they were positioned parallel to the surface of the blocks (Figure 1). After polishing with a rubber cup and polishing paste, they were split into four groups (n = 30), and the blocks were color-coded to facilitate easy identification of the groups.
Bonded Specimens of All Four Groups with Color Coding (Group A: White, Group B: Yellow, Group C: Red, and Group D: Blue).

Group A (RMGIC + Papain)
Deproteinization was done with 10% papain for 60 s, and after that, it was rinsed thoroughly.
Air-dried with compressed air (oil-free).
Acid-etching is done with 10% polyacrylic acid for 30 s, followed by rinsing and air-drying.
Bracket bonding is done with RMGIC (Fuji Ortho LC, GC Corporation, Tokyo, Japan), followed by photopolymerization.
Group B (Single-step Composite + Papain)
The enamel surfaces were deproteinized using a 10% papain solution for 60 s, following rinsing and air-drying.
Enamel etching is done with 37% phosphoric acid (3M ESPE Scotchbond etching gel, St. Paul, MN) for 30 s, and again washed, and then air-dried.
Bonding with single-step composite (Biofix, Biodinamica, Brazil).
Followed by photopolymerization.
The application of primer was not required.
Group C (Conventional Composite + Papain)
The enamel surfaces were deproteinized in a similar manner to Group B.
Etching with 37% phosphoric acid is done for 30 s.
After applying the primer, orthodontic brackets were bonded using Transbond XT (3M Unitek, Monrovia, California, USA) and cured using photopolymerization.
Group D (Conventional Composite Without Papain; Control Group)
The enamel was etched for 30 s using 37% H3PO4, followed by rinsing and air-drying.
After primer application, brackets were bonded using Transbond XT and cured through photopolymerization.
Bonding of Brackets
The procedure of bonding was followed as per the company’s instructions. Bondable metal premolar brackets with 0.022” slots, McLaughlin–Bennett–Trevisi (MBT) (American Orthodontics, USA) were used. The surface area of the bracket base was determined to be 8.686 mm2, based on the company specification. The brackets were positioned on the teeth, and any surplus bonding material was removed. A light-emitting diode curing unit (woodpecker LED.D) of an intensity of 850 mV/cm2 and a wavelength of 420-480 nm is used for curing for a total of 40 s (10 s on each side). A metal jig was prepared to ensure that the curing of every sample was done while maintaining a constant distance from the tip of the curing light (Figure 2).
A Metal Jig was Used for the Curing of Every Sample Ensuring a Constant Distance from the Curing Light.
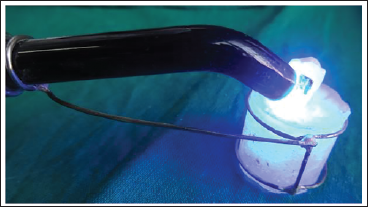
Application of Shear Pull Force with Universal Testing Machine.

Assessment of SBS
Samples underwent SBS testing after being preserved in distilled water for 24 h. A universal testing machine (Instron Machine, model number: 3345R3093, USA) (Figure 3) at a crosshead speed of 0.5 mm/min is used for SBS testing. To stabilize teeth during debonding, a custom-made stand was employed. A looped stainless-steel wire (0.021” × 0.025”) jig was placed beneath the bracket to apply a shear pull force; SBS was noted at the point of debonding. The debonding forces were recorded in Newtons and subsequently converted to megapascals (MPa) using the following formula:
ARI
The total adhesive resin left on the enamel surface was evaluated using a stereomicroscope having 16× magnification. The samples were categorized based on their ARI scores, which measured from 0 to 3. According to Artun and Bergland, 19 0 implies that no composite was left on the enamel, while a score of 1 meant that less than 50% of the composite remained. If greater than 50% of the composite is left over, then the score given was 2, and if all of the composite persists on the tooth surface, the score was 3. To ensure unbiased results, a sole investigator conducted all the measurements and data analysis without any prior knowledge of the groups.
Ethical Approval and Waste Disposal
The ethics committee of Nair Dental College and Hospital approved the study, and after completion of the study, waste was appropriately disposed of in yellow-colored non-chlorinated polythene bags for incineration, according to the guidelines of the Indian Council of Medical Research.
Statistical Analysis
The collected data from the research study underwent statistical analysis using IBM’S Statistical Package for the Social Sciences (SPSS v. 21.0). To collate the mean flexural strength, one-way analysis of variance (ANOVA) was conducted since the data showed a normal distribution according to the Kolmogorov–Smirnov test (P > .05). Pairwise collations were then performed using Tukey’s post hoc test. ARI scores in all four groups were compared using the chi-square test, and the medians were compared using the Kruskal–Wallis ANOVA test. A P value less than .05 was considered statistically significant for all analyses, while maintaining an α error of 5% and a β error of 20%, which provided the study with a statistical power of 80%.
Results
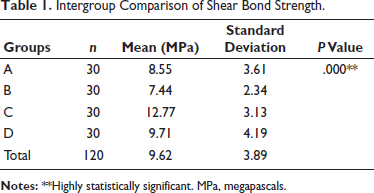
The analysis showed a statistically significant difference in the mean SBS values between all four groups (P < .01). The highest value of 12.77 ± 3.13 MPa was observed in Group C (Transbond XT + papain), followed by 9.71 ± 4.19 MPa in Group D (Transbond XT without papain), 8.55 ± 3.61 MPa in Group A (RMGIC + papain), and 7.44 ± 2.34 MPa in Group B (Biofix + papain) (Table 1).
Intergroup Comparison of Shear Bond Strength.
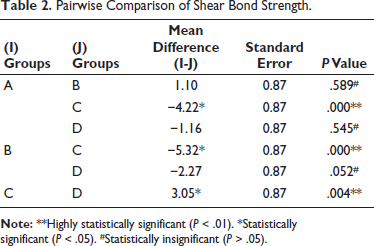
In addition, the analysis revealed that the mean SBS value in group C was significantly greater than the other three groups (P < .01); however, there were no significant differences in the mean SBS values among Groups A and B, Groups A and D, and Groups B and D (P > .05). This suggests that Groups A, B, and D were comparable, as shown in Table 2.
Pairwise Comparison of Shear Bond Strength.
The comparison of mean ARI scores across all groups showed no statistically significant difference (P > .05), as presented in Table 3. The mean scores across the four groups also did not differ significantly (P > .05), as stipulated in Table 4. Additionally, pairwise comparisons using the Mann–Whitney test revealed that there was no relevant difference between any of the pairs, as presented in Table 5.
Comparison of Means of Adhesive Remnant Index (ARI) Scores Across the Groups.
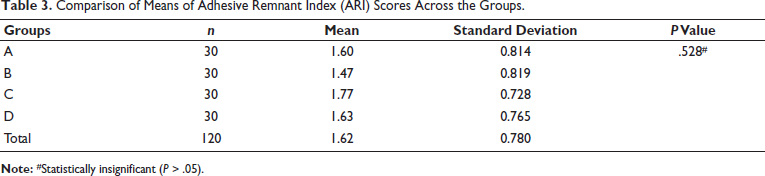
Adhesive Remnant Index (ARI) Scores Among the All Four Groups.
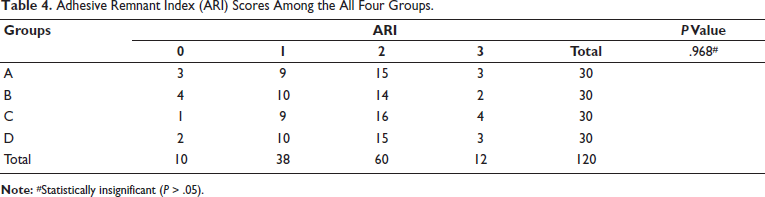
Pairwise Comparison of Adhesive Remnant Index (ARI) Scores.
Discussion
In orthodontic treatment, achieving clinically acceptable bond strength at the interface between enamel, adhesive, and brackets is crucial. Reynolds 20 has established a criterion for a clinically acceptable bracket adhesive, which states that it should possess at least 5.9 MPa of tensile bond strength, known as the Reynolds number. However, orthodontists tend to focus more on SBS than tensile bond strength, as bracket failure mostly results from shear forces, somewhat more than tensile forces. Despite this, the Reynolds number remains a commonly accepted parameter in orthodontics.
O’Reilly and Featherstone 21 observed demineralization around the orthodontic bands and brackets due to increased plaque retention, difficulty in removing plaque, and bacterial adhesion to composite resin. The most important factors to be considered for the success of fluoride-releasing bonding material are acceptable bond strength and sustained fluoride release. Fluoride-releasing resin materials, previously used as bonding agents, failed to reveal potential long-term fluoride release, although exhibiting an adequate bond strength.22, 23 Glass ionomer cement (GIC) has the advantage of fluoride release, which makes it a promising material. While its ability for successful clinical bonding is doubtful.11–13 However, the newly introduced single-step orthodontic bonding material (Biofix) has been shown to have better fluoride release as compared to conventional composites, but it has lower SBS as compared to conventional composites.8–10 Hence, any method that would improve the bond strength of RMGIC and Biofix and make them comparable to composite resin will be beneficial for orthodontic bonding.
The acquired pellicle on the enamel is considered one of the primary factors that impact bond strength. This membrane (biofilm) contains proteins, glycoproteins, enzymes, and mucins or their by-products and is free of bacterial colonization. Justus et al. 16 demonstrated that removing this organic material before acid-etching can create Type I and II etch patterns. Amongst the various enamel-deproteinizing agents, papain has shown excellent results.17, 18, 24 Papain, an enzyme extracted from papaya, acts as an alkaloid and exhibits proteolytic, antibacterial, and anti-inflammatory properties, and it selectively targets the organic component without causing harm to the surrounding inorganic tissue. 25
The present study is performed on extracted teeth, making the need for a storage medium mandatory. Several researchers have provided evidence that long-term storage of specimens prior to bonding in normal saline or distilled water had no significant effect on SBS.26, 27 Thus, specimens were preserved in normal saline. Moreover, they were stored for 24 h in distilled water after bonding as well. The literature showed that RMGIC initially has less bond strength than composite adhesives; later on, it increases more than 20-fold within 24 h, unlike the two-fold increase in the case of composite adhesives. 28
The research findings suggest that deproteinizing the enamel surface using a 10% papain leads to an increase in SBS of orthodontic bonding materials. Group C showed the highest SBS in this study, and Group B showed the lowest. A statistically significant difference in the mean SBS values was seen between Group C and all the other three groups. However, the mean SBS values were not significantly different among Groups A, B, and D.
No relevant distinction was found in the values of Group A and Group B, indicating that the mean SBS values of both groups are comparable. Although both materials have the added advantage of fluoride release, there is no study in the literature that compares their SBSs.
A notable difference was found in the values of Group A (RMGIC + papain) and Group C (Transbond XT + papain), with the latter showing the highest mean SBS value. The results were in consonance with the study done by Pithon et al. 17 When the mean SBS values of Group A (RMGIC + papain) were compared with Group D (Transbond XT without papain), there was no remarkable difference in the values. Literature suggests that conventional GIC has less SBS as compared with composite resin.11–13 Hence, this proves that papain is an effective way of increasing the SBS of RMGIC.
A remarkable distinction was found in the values of Group B (Biofix + papain) and Group C (Transbond XT + papain), and no relevant distinction was found in Group B (Biofix + papain) and Group D (conventional composite without papain). Studies done previously show that the SBS of Biofix was less as compared to conventional composite.9, 10 Thus, it has now been established that enamel deproteinization before the bonding procedure increases the SBS of the single-step composite, making it comparable to the conventional composite.
A remarkable distinction was found in the values of Groups C and D (Transbond XT without papain), with the values of Group C being higher than Group D.
Moreover, the ARI score did not display any significant differences among the groups. The majority of test specimens had scored 2, demonstrating cohesive failure, with the adhesive substance mostly staying on the enamel after the bracket was debonded. The bonding performance of these materials was improved by the deproteinization treatment, which led to a higher ARI score. By leaving excessive adhesive during debonding, damage to the enamel is mitigated, as the fractured link moves towards the bracket-adhesive interface, thus providing better safeguarding for the enamel.
The results suggest that deproteinizing the enamel surface with a 10% papain solution leads to an increase in SBS for bonding materials. Therefore, it can be concluded that this method may be beneficial for improving the success of orthodontic treatment.
Conclusion
The study’s result allows for the following conclusion to be drawn:
Using 10% papain for enamel deproteinization results in higher SBS of orthodontic bonding materials. RMGIC and Biofix can be good alternatives to conventional orthodontic composites, especially in cases of high caries risk patients. ARI scores among the groups demonstrated a cohesive failure, preventing enamel microfractures.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval was obtained from the Institutional Ethics Committee of Nair Hospital Dental College and Hospital.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Not applicable.