Abstract
Background
Orthodontic appliances such as wires and brackets received from manufacturers come unsterilized and may be contaminated with various microorganisms before being used in the mouth. In this study, we evaluated and identified the bacterial contamination on orthodontic appliances along with the disinfecting efficacy of ultraviolet (UV) light and various sterilization methods.
Methods
Different orthodontic appliances were obtained from manufacturers divided into 5 sterilization methods and a control group (control, UV, dry heat and steam autoclave, ethyl alcohol, and 2% glutaraldehyde). Microbiological and DNA sequencing was performed on the appliances to identify the contaminated bacteria.
Results
Bacterial contamination identified on the orthodontic appliances were Staphylococcus aureus, Staphylococcus epidermidis, Lactobacilli, Klebsiella pneumoniae, Bacillus licheniformis, and Bacillus cereus. UV sterilization method effectively prevented the bacterial growth when compared to the control (unsterilized) orthodontic appliances.
Conclusion
We concluded that the orthodontic appliances received from the manufacturer showed bacterial contamination. All of the tested sterilization methods including UV light were effective in eliminating the bacterial contamination on the orthodontic appliances. Since UV light does not cause change in material properties and is cost effective with relative ease of use, its use in clinical practice for the disinfection of orthodontic appliances is suggested before placement in the mouth.
Introduction
Orthodontic appliances have been associated with increased plaque accumulation and bacterial colonization on the teeth and gingiva, predisposing them to enamel decalcification and periodontal inflammation. 1 This increase in oral biofilm and a shift in the composition of oral microflora could be due to a reduction in tooth brushing efficacy, contamination of the orthodontic appliances during handling, or unhygienic manufacturing processes. Infection control is an important aspect of clinical dentistry and routine precautions are taken to prevent the transmissibility of microorganisms to patients and staff. Even though there have been major advances in orthodontics with the introduction of new materials and processes, adherence of microorganisms to these materials during orthodontic treatment remains a concern. Many studies have been conducted to study the bacterial adhesion on orthodontic appliances.2-5 These studies indicate the need for sterilization of orthodontic materials before placement into the oral cavity. Although dental instruments are sterilized before use, orthodontic materials like brackets, bands, archwires, elastomeric ligatures, and chains are used “as received” from the manufacturers for clinical use.
Recent studies have demonstrated the efficacy of different sterilization and disinfection methods including, steam, dry heat, ethylene oxide gas, and glutaraldehyde. Steam autoclave (moist heat) sterilization is the most widespread technique in dentistry that causes microbicidal activity by the irreversible denaturation of enzymes and structural proteins in microorganisms. It requires maintaining a high, constant temperature and pressure of 121°C at 15 pressure per square inch (psi) for 20 min. However, a major disadvantage is the corrosion that results due to the exposure to water over time. Dry heat sterilization is also another very popular sterilization technique that results in oxidative damage to bacterial cells as opposed to denaturation and coagulation of proteins in moist heat sterilization. However, this method can degrade the properties and plasticity of many orthodontic materials. Ethylene oxide gas is an excellent reagent for sterilization due to its high level of penetration at room temperature, and it is also noncorrosive. However, it is a very slow process, and the reagent is very toxic and explosive. Although it is not sporicidal, ethyl alcohol shows high effectiveness for the elimination of bacteria without degrading its mechanical properties. Glutaraldehyde is commonly used in cold sterilization of heat-sensitive materials and metal instruments where it has been shown to be highly efficient. Each technique has its advantages and disadvantages that make it the best option in different situations.6-10
There are contradictory reports on the effect of ultraviolet (UV) light on orthodontic appliances against contamination. Thus, in our study we evaluate the effectiveness of UV light in addition to other sterilization methods and demonstrate its possible use in clinics. It has been reported that UV light deactivates the DNA of bacteria, virus, and other pathogens. The lack of heat and chemicals in the UV light allows the orthodontic appliances to retain their properties, while the radiation of the light destroys the bacteria. In this study, we note that manufacturers commonly deliver orthodontic supplies to health-care professionals without being sterilized and the supplies are then immediately used on patients without any form of sterilization. Based on the previous studies, we hypothesize that UV sterilization is an effective way to sterilize orthodontic appliances. Here, we direct our focus to the efficacy of different sterilization technique on different orthodontic supplies and compare the differing methods, also providing a control group of orthodontic materials without exposure to any form of sterilization.11-13
Materials and Methods
This was an in vitro microbiological investigation. The most commonly used orthodontic supplies in patient care, that is, stainless steel and nickel titanium (NiTi) archwires, elastomeric power chains (e-chain), and ties were received from different vendors. Upon inspection, there was no packaging information provided regarding sterility from any of the companies. The packages were opened, and the contents were removed using a sterile technique. Samples were cut into appropriate sizes for testing. Archwires were cut into 1-cm length, elastomeric chains into 2 links, and the elastomeric ties individually. Samples of the 4 materials were divided into the following 6 disinfection/sterilization groups (control, UV, dry heat and steam autoclave, ethyl alcohol, and 2% glutaraldehyde; Table 1). Ethical approval from Institutional Review Boards (IRBs) was obtained.
Different Sterilization Method Was Performed.
Effects of Different Sterilization Methods on Orthodontic Appliances
Orthodontic supplies were subjected to different sterilization and disinfection treatments as follows. Group 1: the appliances were not subjected to any sterilization method (control group); group 2: the appliances were immersed overnight in sterile Eppendorf tubes containing 2% glutaraldehyde; group 3: the appliances were immersed in sterile Eppendorf tubes containing 70% ethanol for 1 min; group 4 and 5: the orthodontic appliances were sterilized either by dry or wet cycle at 121°C at 15 psi pressure for 30 min using STERISR Life Sciences, AMSCO Lab 250; LV 250 Laboratory Steam Sterilizer, and group 6: the orthodontic appliances were UV sterilized (115V UV light) using NUAIRE NU-427 Biosafety Cabinet for 15 min. The samples were then turned over using sterile forceps and kept in the chamber for another 15 min (Table 1).
Light Microscopic Observation of Microbial Growth on the Orthodontic Materials
After sterilization, the orthodontic materials were placed on a Tryptic soy agar (TSA) plate and incubated at 37°C for 3 days. TSA is a universal growth medium (contains a pancreatic digest of casein, a peptic digest of soybean meal, yeast extract, sodium chloride, and agar) that supports the growth of a wide variety of both gram-positive and gram-negative bacteria. Subsequently, the microbial growth on the plates were photographed using DP12 Microscope Digital Camera System (Olympus).
Identification of Bacterial Contamination by Direct Polymerase Chain Reaction (PCR) Amplification of 16S rRNA Genes
Genomic DNA from different orthodontic microbial contamination was extracted using a QIAamp DNA Kits as described by the manufacturer (Qiagen, Inc). The PCR amplification of 16S rRNA genes was conducted using the 16S rRNA universal primers. The bacterial DNA samples were then performed PCR by using Techne TC-412 with the following conditions: initial denaturation at 95°C for 2 min, 30 cycles of 95°C for 30s, 55°C for 30s, and 72°C for 2 min, followed by a final extension at 72°C for 5 min. The PCR products were then purified using a QIAquick PCR Purification kit and quantified by a NanoDrop (Thermo Fischer Scientific). To identify the microbial contamination, DNA samples were then sequenced using ABI BigDye Terminator chemistry on an ABI 3130×l Genetic Analyzer (Molecular Resource Facility, NJMS). All bacterial identification was performed using National Center for Biotechnology Information-Basic Local Alignment Search Tool (NCBI BLAST) searches.
Results
Morphological Growth on an Agar Plate
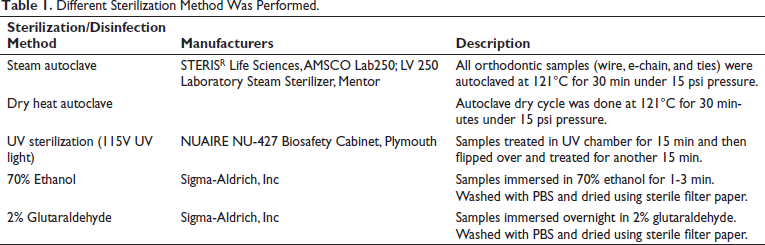
Bacterial contamination was seen on “as received” samples of the orthodontic appliances (archwires, e-chains, ties) when they were placed on TSA plates and incubated at 37°C for 3 days. All the appliances were contaminated with microbial growth that appeared white, cream, or yellowish color and circular in shape. We also observed a few circular and white colonies that had a glossy surface. The wire samples showed yellow and cream circular microbial growth and a few white-colored microbial growths, both with a smooth and glossy appearance. Two wire samples showed white colored irregular-shaped colonies with a dull appearance. The e-chain samples showed white colored circular and irregular colonies that appeared glossy and smooth on the surface. One e-chain link showed white and irregular microbial growth with a dull appearance. The tie samples showed a mix of all 3 colors (white, yellow, and cream) of microbial growth. The yellow and cream colored appeared to be circular and glossy. The white colored colonies showed circular as well as round growth, which appeared smooth and glossy. One of the white microbial growths around the tie was irregular in shape with a dull appearance (Figures 1-3).
Determination of the bacterial contamination of tie. The tie was sterilized with different methods as described in the materials and methods and control was placed on the TSA agar plate and incubated at 37°C for 3 days. The tie was then photographed using stereo microscope. (a-c) Representative e-chain of different sterilization method; no bacterial growth was seen; (d-i) The nonsterilized tie showed bacterial contamination.
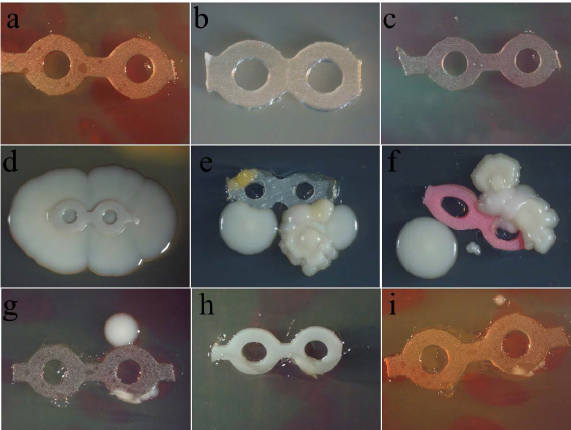
Determination of the bacterial contamination of e-chain. The e-chain was sterilized with different methods as described in the materials and methods and control was placed on the TSA agar plate and incubated at 37°C for 3 days. The e-chain was then photographed using stereo microscope. (a-c) Representative e-chain of different sterilization method; no bacterial growth was seen. (d-i) The nonsterilized e-chain showed bacterial contamination.
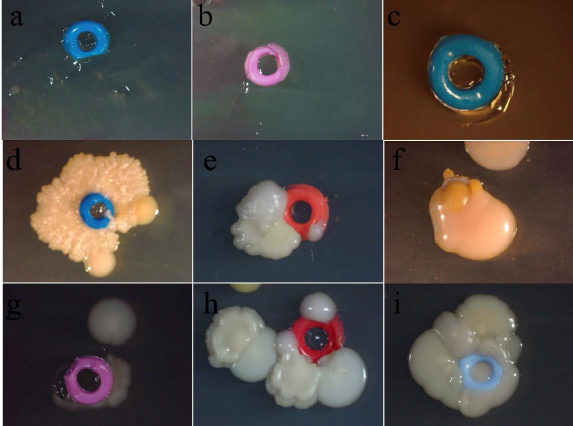
Determination of the bacterial contamination of NiTi wire. The NiTi wire was sterilized with different methods as described in the materials and methods and control was placed on the TSA agar plate and incubated at 37°C for 3 days. The NiTi wire was then photographed using stereo microscope. (a and b) Representative e-chain of different sterilization method; no bacterial growth was seen. (c-f) The nonsterilized e-chain showed bacterial contamination.
Effect of Different Sterilization Methods on Orthodontic Supplies
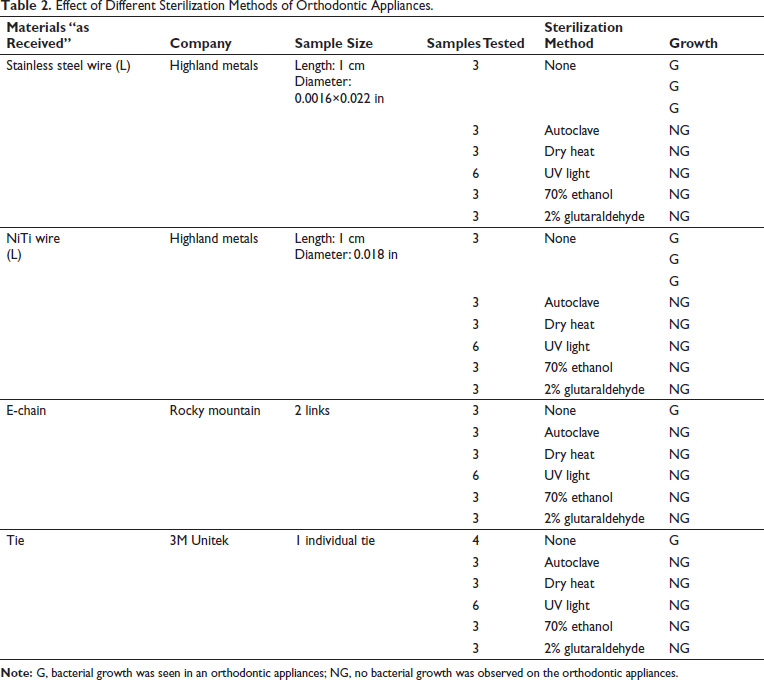
In this study, we used the “as received” orthodontic samples as the control group. Before treating with the different sterilization and disinfection methods, the “as received” samples showed microbial contamination. Out of the 6 samples of 2 types of control wires tested (Stainless Steel and NiTi), 4 samples showed microbial growth. The “as received” samples of the 3 e-chains (2 links each) and 3 ties (individual) all showed microbial contamination. The evidence of bacterial contamination was 100% in e-chains and ties. In this study, all the orthodontic samples were subjected to different sterilization and disinfection methods (steam autoclave, dry heat autoclave, UV light, 70% ethanol and 2% glutaraldehyde). All of the different treatments effectively prevented the microbial growth. In this study, UV light treatment of the orthodontic appliances was as effective as the other sterilization and disinfection methods and demonstrated complete elimination of the microbial contamination (Table 2 and Figures 1-3).
Effect of Different Sterilization Methods of Orthodontic Appliances.
Identification of Bacterial Contamination
To identify the microbial contamination, genomic DNA was isolated from different orthodontic microbial contamination and analyzed using DNA sequencing using a specific primer for identifying the bacteria. Following sequencing, the data was processed for analysis. All bacterial identification was performed using NCBI BLAST searches showed that the “as received” samples were identified Staphylococcus aureus (S. aureus), Staphylococcus epidermidis (S. epidermidis), Klebsiella pneumoniae (K. pneumoniae), Bacillus licheniformis (B. licheniformis), and Bacillus cereus (B. cereus).
Discussion
Contamination is a common issue that could possibly be a contributing factor to an imbalanced oral microbiota leading to the aforementioned orthodontic concerns and complications. In this study, we observed that the manufacturers usually delivered the orthodontic supplies to health-care professionals without being sterilized and the supplies are then immediately used on patients without any form of sterilization. We tested the orthodontic samples by subjecting them to different sterilization and disinfection methods namely autoclave, dry heat, UV light, 70% ethanol, and 2% glutaraldehyde. All treatment methods were effective in complete elimination of the microbial growth and colonization. In this study, UV light treatment of the orthodontic appliances was as effective as the other sterilization and disinfection methods and demonstrated the complete elimination of the microbial contamination.
Several studies have shown that the orthodontic materials were cross-contaminated, and our study also demonstrated that the “as received” samples showed cross-contaminated with S. aureus, S. epidermidis, Lactobacilli, K. pneumoniae, B. licheniformis, and B. cereus.2-5 Barker et al 2 conducted a pilot molecular study to evaluate microbial contamination of a wide variety of orthodontic supplies as received from manufacturers and after exposure to clinical environment. The results of the study showed low levels of bacterial contamination on both “as received” archwires, bands, and impression trays and those exposed to the environment. They considered this contamination to be low and insignificant considering the potential for aerosol and operator contamination. The identified bacterial species were S. epidermis, Kocuria, Maraxella, and Micrococcus species. Rastogi 14 conducted an invitro study to assess the sterility of packed orthodontic materials “as received” from the manufacturer and “bench top exposed.” Out of the materials tested (elastomeric chains, molar bands, buccal tubes, and lingual sheaths), the buccal tube and molar bands showed the highest levels of contamination. Bench top exposed materials showed more contamination than as received and the bacterial species identified were Klebsiella, Streptococci, Citrobacter, and Escherichia coli. 14 Another study also found some of the as received stainless steel and Nitinol archwires from several companies to be contaminated. S. epidermidis was found on stainless steel wires but not on Nitinol wires. S. aureus was found on both types of wires. Stainless steel wires were more contaminated compared to Nitinol wires. There was a difference in the level of contamination amongst the 4 companies with some showing none. 6 Harikrishnan et al 5 found less microbial adhesion and growth on stainless steel wires compared to Teflon-coated wires and elastic ligatures. Saliva accelerated microbial growth and adhesion.
Vivek et al 15 found all brackets obtained from 4 different manufacturers (American Orthodontics, 3M Unitek, Ortho Organizers, and China Dental Orthodontics) had microbial contamination. The bacteria identified were S. aureus, S. epidermidis, Lactobacilli, K. pneumoniae, B. licheniformis, and B. cereus. On treating with 0.01% Chlorhexidine, 3M Unitek brackets were completely disinfected, whereas the brackets from the other manufacturers showed disinfection on treating with 2% chlorhexidine. The study suggested the use of 2% chlorhexidine for disinfection before placement of brackets in the oral cavity. Gerzson et al 3 also reported biological contamination of some of the brackets in the original packages that were evaluated in their study. S. aureus was confirmed in full case metal brackets (Morelli) and S. epidermidis in ceramic replacement brackets (Abzil-3M Unitek) supplied by 2 of the 4 manufacturers. The authors suggested that manufacturers needed to improve quality control of contamination in packages of brackets. Brusca et al 16 found that microbial adherence (Streptococcus mutans and Candida sp.,) to brackets varied according to the material type. Composite brackets showed the most microbial adherence followed by ceramic brackets and lastly metallic brackets. They suggested that the porosity and roughness of the composite and ceramic bracket surface provided a favorable ecological niche resulting in greater adherence of the microorganisms. Purmal et al 17 conducted an in vitro study to test the sterility of orthodontic buccal tubes as received from the manufacturer. The results demonstrated the contamination of bands from all 4 manufacturers with opportunistic pathogens namely Micrococcus luteus, Staphylococcus haemolyticus and Acinetobacter calcoaceticus. Irfan et al 18 conducted a study to assess bacterial contamination of orthodontic bands that had been precleaned and then steam autoclaved. The results showed that all bands that were not precleaned and sterilized had bacterial growth. Whereas both manual scrubbing and emersion in an enzymatic solution for 10 min were more effective. However, 5% of those treated only with the enzyme solution had presence of Staphylococcus nonaureus bacterial species. It has been reported that UV light works by destroying nucleic acids, thus damaging the DNA of the bacteria. The lack of heat and chemicals in the UV light keeps the properties of the orthodontic appliances intact whilst the radiation of the light simply kills the bacteria. 19 Brindha et al 20 conducted a study to determine if the 4 different types of sterilization methods (autoclave, hot air oven, glutaraldehyde, and UV light disinfection) changed the tensile strength (TS) or the surface topography (ST) of 3 different types of orthodontic wires (stainless steel, TMA alloy, cobalt-chromium). For UV light, TS of stainless-steel wire was not affected, TS of TMA was increased compared to the control but still less than stainless steel, and the TS value for CoCr was inconclusive. UV light disinfection was shown to have the least effect on the ST of all the wire types when compared to the other 3 sterilization procedures. Based on our study, we recommended that the orthodontic materials should be UV sterilized before putting them into patient’s mouth.
Conclusion
We concluded that orthodontic brackets received from manufacturers showed high bacterial contamination. All of the methods, UV, dry heat, steam autoclave, ethyl alcohol, and 2% glutaraldehyde were effective in eliminating bacterial contamination. Since UV light does not cause change in material properties and is cost effective with relative ease of use, suggested that UV sterilization would be a good choice in clinical practice for the disinfection of orthodontic appliances prior to placement in the oral cavity.
Footnotes
Authors Contribution
KV and AA: Critical review; KV, KC, AA, and DS: Experimental design; KV, KC, PA, DS, and DA: Literature search; and KV, KC, PA, DA, AA, and DS: manuscript writing.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Statement of Informed Consent and Ethical Approval
Necessary ethical clearances and informed consent was received and obtained respectively before initiating the study from all participants.
