Abstract
Obstructive sleep apnea (OSA), the most prevalent form of sleep-related breathing disorder (SRBD), is associated with cardiovascular, neurocognitive, and metabolic complications. Evidence suggests that these comorbidities are also risk factors for enhanced severity in COVID-19 patients. Hence, initial diagnosis or screening of OSA-risk is a major requirement of current times, which can be fulfilled by a noncommercial, easily accessible mobile application for self-assessment of OSA-risk. The current article mentions a prototype of an “OSA-Risk Assessment Tool,” a mobile application developed after prior testing of needs analysis and comprising various interfaces for OSA-risk assessment in all age groups, and further refined for user applicability through a cognitive, pluralistic walkthrough and heuristic evaluation by the authors and four volunteers. It has huge scope of application in orthodontic clinics, primary healthcare centers in middle and low-income strata of developing countries, and multiple educational and licensing institutions for the larger benefit of the society.
Background
Obstructive sleep apnea (OSA) is the most prevalent form of sleep-related breathing disorder (SRBD), which presents with prominent symptoms of snoring, pauses in breathing, and excessive daytime sleepiness. A worldwide estimate of 936 million adults (both men and women), aged 30 to 69 years have mild-to-moderate OSA, with a reported prevalence of 4-9%. High prevalence along with its significant association with cardiovascular, neuro-cognitive, and metabolic complications, has made OSA a major health concern. 1 Since these comorbidities are also risk factors for enhanced severity in COVID-19 patients, initial diagnosis or screening of OSA-risk is a major requirement of current times. 2 Additionally, a recently published meta-analysis reported a pooled prevalence of depression and anxiety in 35.0% and 32.0% OSA patients respectively. 3 These symptoms are also on the rise in COVID-19 patients as well as health care workers, hence making them more prone to OSA. 3 The dysregulation of the renin–angiotensin system (RAS) is believed to be commonly associated with OSA, as well as hypertension (HT), a comorbidity significantly associated with OSA. 4
There are high chances of OSA being underreported, as the hypopnea events occur in sleep. Its diagnosis in the current COVID-19 times additionally poses a greater challenge due to the requirement of sleep studies based on polysomnography (PSG), which is not currently preferable in a hospital or home setup. Hence, early screening by means of convenient self-diagnosis or minimal hospital interventions through customized mobile applications is the need of the hour to prevent further complications or comorbidities.
Study Goals
The goal of the current study is to assess the possibility of self-assessment of OSA-risk by means of a customized mobile-based application, “OSA-Risk Assessment Tool,” in people of all age groups.
Methods
The prototype development took place in multiple stages:
A preliminary project related to the assessment of craniofacial morphology related to adolescents at risk of OSA was undertaken in the department of orthodontics in a central-government dental school in Delhi.
5
It showed a high prevalence of OSA-risk (14%) in orthodontic patients who were completely unaware of any symptoms of sleep-disordered breathing. Hence, it highlighted lack of awareness related to OSA in orthodontic patients and can easily be extrapolated further for the general population.
The development of a customized mobile application, “OSA-Risk Assessment Tool,” is planned, with the application comprising the following interfaces: A disclaimer stating that the application is completely non-commercial and free to use: It would avoid any kind of discrepancy in terms of medical advice and data privacy. The disclaimer would also specify that the application is for pre-screening OSA-risk and is not a confirmatory diagnosis for OSA. Only after agreeing with the terms and conditions would prospective patients be allowed to proceed with screening. Collection of details of patients: Two types of patient details would be collected: (a) demographic details—name, age, socioeconomic status; and (b) anthropometric details—blood pressure, body weight, height, neck circumference, waist circumference (patients would be assisted in the measurement of neck and waist circumference through relevant explanations). A preliminary questionnaire including questions on sleep characteristics and medical history: If the number of responses in this questionnaire >2, the patient would proceed to the next interface of standardized questionnaires. If the number of responses <2, the patient would be notified of no OSA-risk and would not proceed to next interface. Standardized questionnaires: The questionnaire would be automated with respect to the age filled in the second interface. It would perform the function of textual analysis. If age is 2 to 18 years, the pediatric-sleep questionnaire (PSQ) would open. If age >18 years, the STOP-BANG questionnaire would open. The questionnaires employed would be:
Pediatric-sleep questionnaire
6
: This includes 22-point SRBD scale comprising of three prominent symptom complexes: snoring, excessive daytime sleepiness, and inattentive/hyperactive behavior. The sensitivity and specificity of PSQ are 0.85 and 0.87, respectively. STOP-BANG questionnaire
7
: It includes a subjective 4-item questionnaire (STOP) and a 4-item portion informed by demographics and anthropometric measurements (BANG). It has the highest methodological validity, reasonable accuracy, and ease of use. Sensitivity = 1.00 (0.91, 1.00), specificity = 0.37 (0.29, 0.46), and negative predictive value (NPV) = 1.00 (0.93, 1.00). Patient report regarding OSA-risk status: Results would be depicted as “OSA-risk”/“No risk” in PSQ and “High risk”/“Intermediary risk”/“Low risk” in the STOP-BANG questionnaire. Result translators for the patients: This additional interface would interpret the results obtained for OSA-risk in both the questionnaires, to guide the patients regarding further protocol and management (Figure 1; Table 1). Additional features: Doctor’s login: This interface would help doctors track the history of patients and patient responses, only accessible to associated doctors. Informational interface: This would explain the working of the application and enlighten patients more about OSA and related risk factors and complications. About Us: Information about the team of doctors and developers would also be incorporated.
The evaluation of the application’s usability was executed by three methods:
Cognitive walkthrough
8
: This was done by defining the tasks expected to be performed by the user and identifying problems faced by them in the functionality of the interfaces. Figure 1 (flowchart) depicts the sequence of tasks outlined for the user. The details of the interfaces have been explained in the section on the prototype development stage. The authors and four interns volunteered to test the user interfaces and outline the problems in usability. Pluralistic walkthrough
8
: Group meetings were organized with the authors, volunteer interns, and developers to discuss each interface and problems faced in navigating through the various interfaces. Heuristic evaluation
8
: For evaluating the extent to which the design principles of the mobile application conformed to the screen designs, the following aspects were appraised:
Visibility of system status: This involved assessment of the navigation through various interfaces. Specific feedback would be provided to the user related to leaving the application when the number of responses to the preliminary questionnaire was less than 2, and also while providing the final result of OSA-risk to the user. Match between the system and the real world: This involved assessing the level of understanding of the terminologies or phrases used in the application by the user and ensuring the provision of proper explanations for terms like OSA, abdominal circumference, and neck circumference, besides assessing the user-friendliness of the questionnaires. User control and freedom: The ease of the user in going back and forth with their responses, in case any change is intended, and also in doing an emergency exit of the application when required, was assessed. Consistency and standards: This involved assessing whether different words and situations used in the application holding the same meaning were clearly defined and comprehensible to the user. For example, “obstructive sleep apnea” and “sleep-disordered breathing,” whether they were clearly explained, and whether they were used in their short form or otherwise. Error prevention: Error-prone conditions whereby users would not be able to understand a few terminologies unless explanations were given, like abdominal circumference or nocturnal enuresis in children, were identified. Also, a final interface was added where the results of OSA-risk would be clearly flashing on the screen without the score of the questionnaire for the benefit of the users. Recognition rather than recall: The aspect of elimination of recall of information for users was ensured. Each interface would have exclusive questions with no repetition. Also, the age filled in the second interface would automatically open the relevant questionnaire in the fourth interface. Flexibility and efficiency of use: Whether the user is a novice or an expert, the system should be able to be used by anyone. Help and documentation: All responses filled by the volunteers were double-checked using a doctor’s login. Any discrepancy in responses was rectified.
The following flowchart is a cognitive walkthrough depicting the sequence of tasks outlined for the user, the different interfaces (I-V), derivation of results of questionnaires, and interpretation of the results.
Tasks Still Unaccomplished
Testing end-user usability through the recruitment of subjects of different age groups and experience.
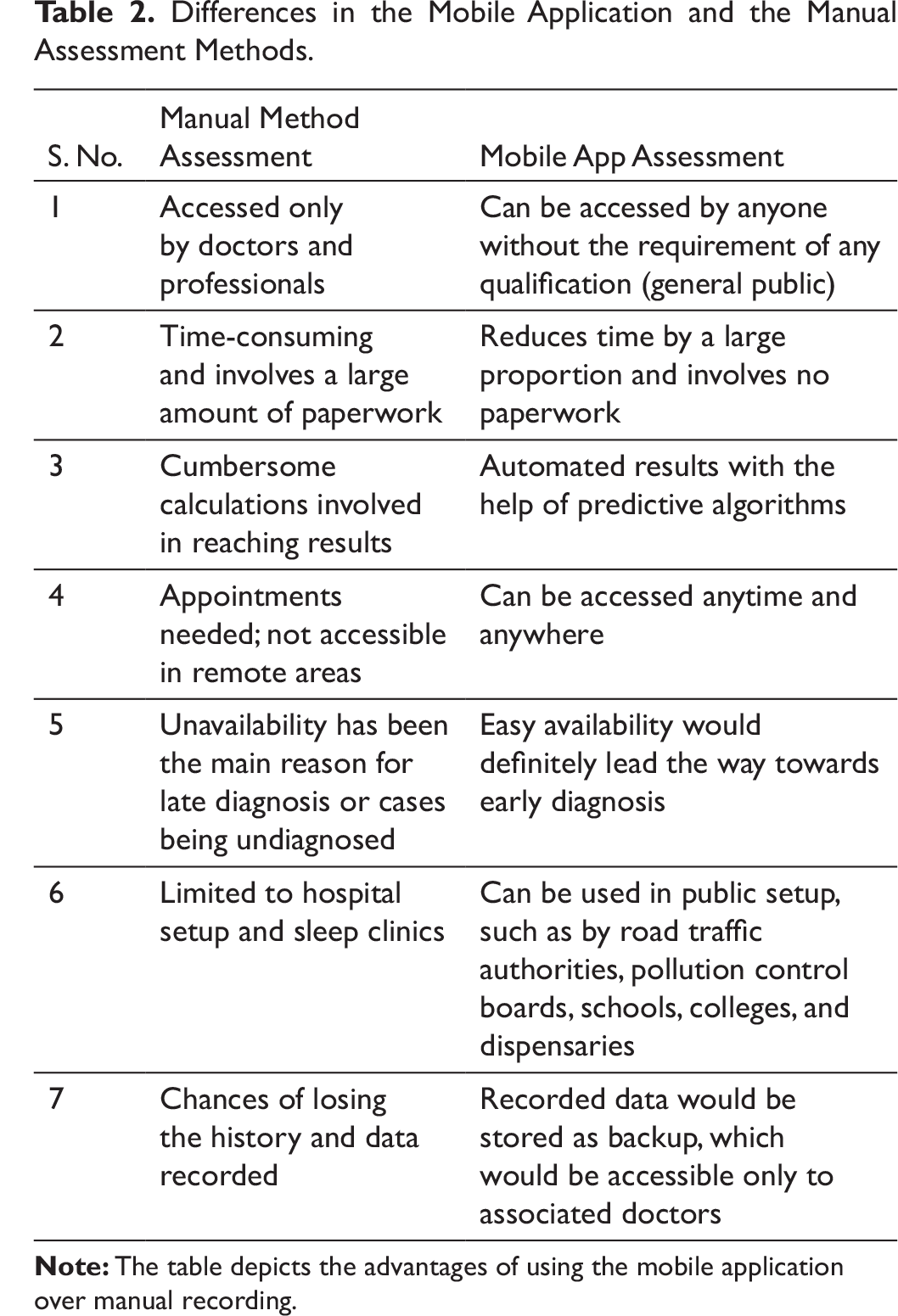
Time taken on average for OSA-risk assessment through the mobile application. Table 2 presents the advantages of mobile testing over manual testing of OSA-risk.
Objective assessment of user satisfaction.
Novelty
Currently, there are no non-commercial, easy-to-use mobile-based OSA-risk screening tools in use. Mobile applications have been tested previously for screening OSA, including “SleepAp” 9 and “Firefly App,” 10 to name a couple of them. However, these are commercial applications that may present cost limitations in a developing country like India. Additionally, they are not used for pre-screening OSA-risk as in our mobile application but use varied signals, as well as artificial intelligence (AI) algorithms, to assess OSA, comparable to ambulatory OSA screeners. For example, SleepAp uses actigraphy, position of the body, photoplethysmography, and audio for inputs in a vector machine classifier, as well as a standardized STOP-BANG questionnaire. 9 The “Firefly” app, 10 on the other hand, utilizes AI and advanced digital signal processing (DSP) for identifying stages of sleep, snoring, OSA patterns, and respiration rate. Additionally, the questionnaire used in “SleepAp” 9 has clinical validity only in adults, and hence pediatric OSA cannot be screened, while our mobile application would be applicable for all age groups. Moreover, our mobile application would be user-friendly and non-commercial and also use a preliminary questionnaire that can guide a user regarding whether he/she requires an OSA-risk assessment or not; hence, self-assessment is a possibility. However, the current application is not diagnostic for OSA, unlike the other mobile applications mentioned above.
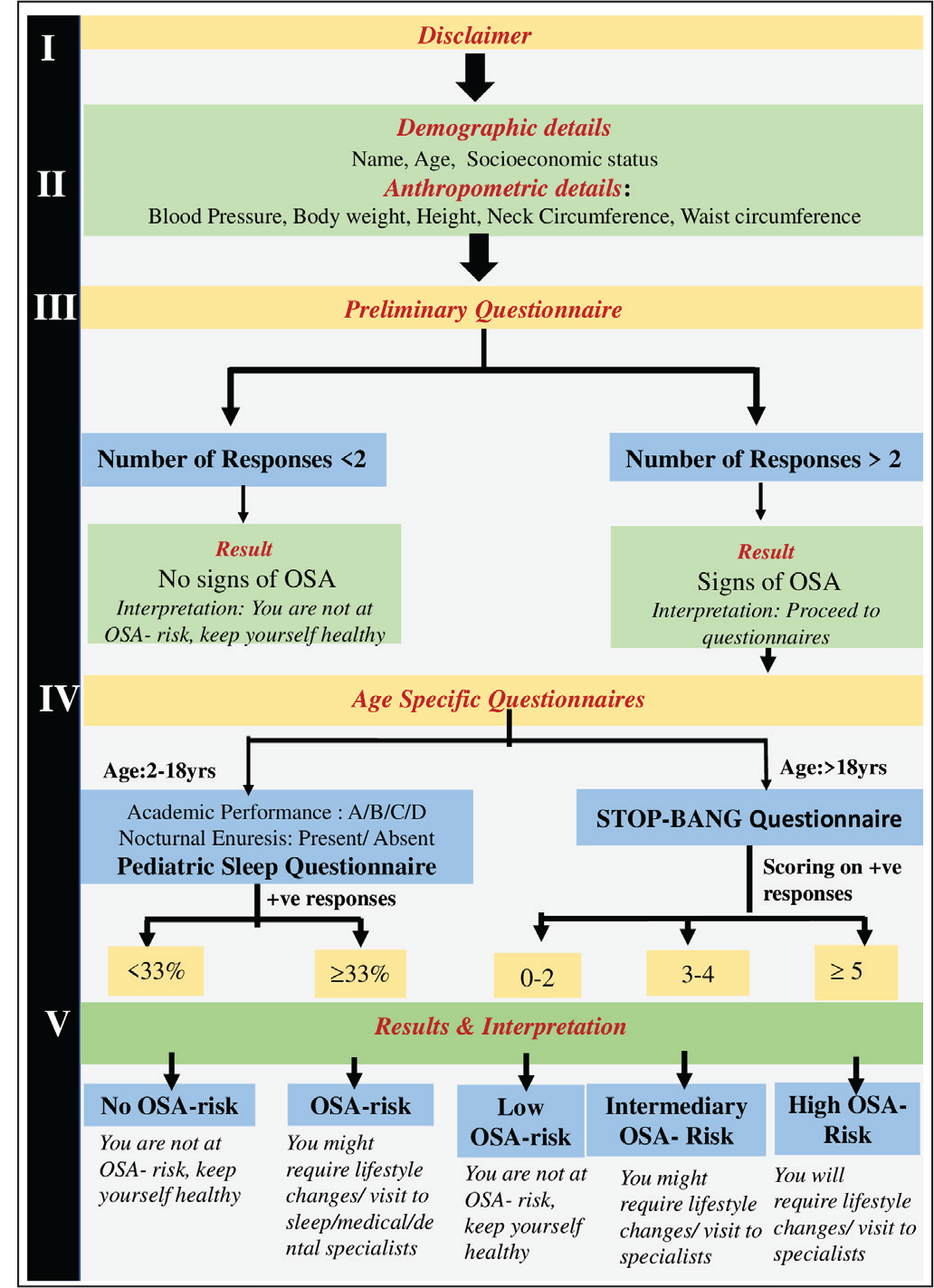
Interpretation of Results/Implications for Patients in Interface 5.
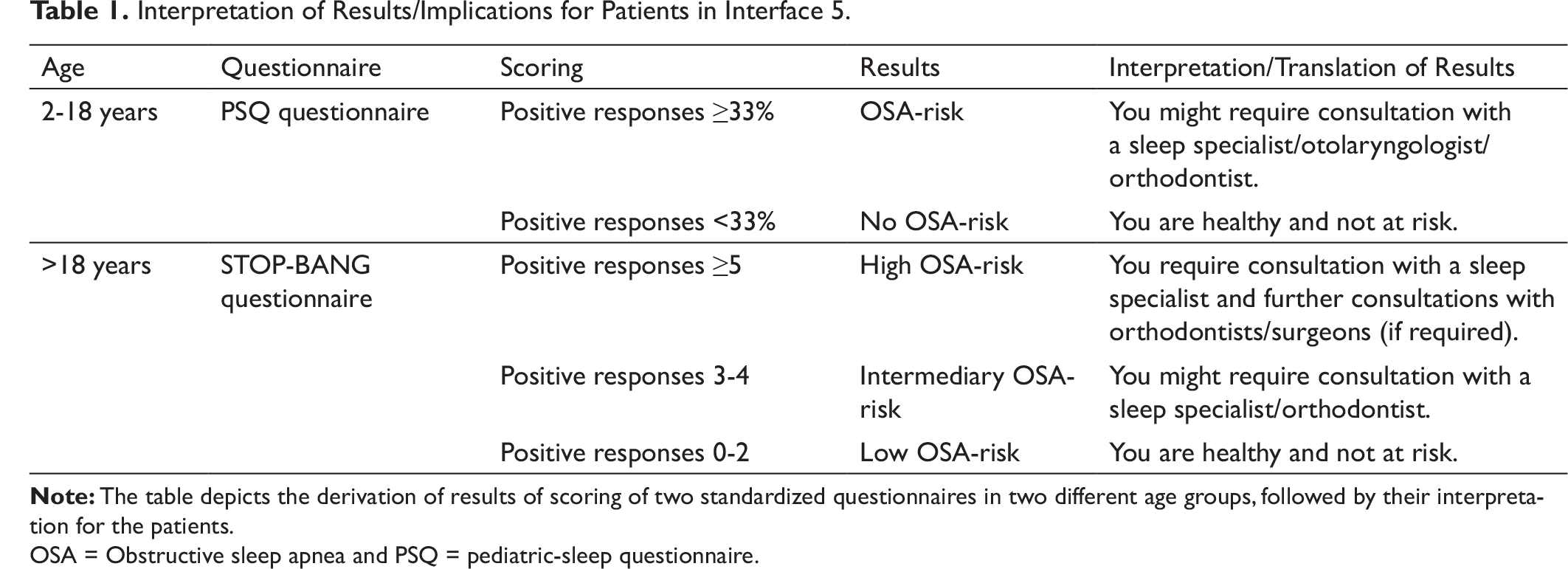
OSA = Obstructive sleep apnea and PSQ = pediatric-sleep questionnaire.
Differences in the Mobile Application and the Manual Assessment Methods.
Scope of the Application
Solution for OSA-risk screening in COVID-19 times, with a clinician monitoring/self-assessment option;
Non-commercial, cost-effective application available to all strata in low-income countries for automated OSA-risk screening;
Awareness through patient education interfaces for symptoms/implications of OSA;
Lowering of the curve of undiagnosed OSA-risk cases;
Scope for prescreening in orthodontic clinics, primary healthcare centers, and hospitals;
Automated initial assessment in schools, educational institutions, and driving license authorities; and
Research avenues:
Data stored by users can be used to establish significant correlations of socioeconomic, demographic, anthropometric, medical, and behavioral variables with OSA-risk. Further oral microbiological assessments can be conducted to outline differences between OSA-risk and non-risk groups, in dental and orthodontic clinics. People from different educational backgrounds or belonging to different age groups or ethnicities can be tested for universal applicability.
Conclusions
The prototype of a customized mobile application, “OSA-Risk Assessment Tool,” has been developed after a potent needs analysis of various interfaces duly tested for usability through a cognitive walkthrough, a pluralistic walkthrough, and heuristic evaluation. This application still needs to tested for end-user applicability but promises to have huge scope in the self-assessment and initial identification of patients at OSA-risk, especially in COVID-19 times.
Footnotes
Contribution of Authors
Declaration of Conflicting Interests
Funding
This research was self- funded. Hence, no funding agency to disclose.
Statement of Informed Consent and Ethical Approval
Permissions for the mentioned questionnaires would be acquired and security protocols for the mobile application would be followed as per regional/country rules. Ethical standards and proper credits would be given to the respective authors/creators.
