Abstract
“Pix” is one of the most common skin defects that reduce the quality of crocodilian leather. The name is derived from their resemblance to pit marks made by an ice pick. Histologically, each “pix” is associated with a focal dermal accumulation of immune cells, specifically lymphocytes and histiocytes. Consequently, these defects have been termed lymphohistiocytic proliferative cutaneous lesions (LPCLs). In farmed American alligators (Alligator mississippiensis), LPCLs have been associated with seropositivity against West Nile virus (WNV) and the presence of viral genome in the skin. Despite this association, the nature and pathogenesis of LPCLs remain unclear. Using immunohistochemistry and in situ hybridization, we unravel the microanatomy of LPCLs of alligators and localize WNV genome within the lesions. Our results show that LPCL lesions consist of de novo follicular aggregates of lymphocytes segregated into B- and T-cell zones, like tertiary lymphatic follicles of mammals and birds. Furthermore, the presence of WNV genome was highlighted by in situ hybridization in the macrophages of LPCLs, gut-associated lymphoid tissues, and the spleen. Our results suggest that LPCLs may form in American alligators’ skin as part of a generalized lymphofollicular proliferation, likely as an immune response against WNV infection.
Keywords
The focus of crocodile farming is the production of leather for the luxury leather market. The quality standards of the market are stringent, and every imperfection can considerably reduce the value of a skin. 4 For this, the financial sustainability of the crocodilian farming industry relies on the production of first-grade, flawless skins. 19
During the 2001 and 2002 West Nile virus (WNV) outbreaks, American alligator (Alligator mississippiensis) farms experienced severe losses not only due to significant mortalities14,21 but also because of a concurrent increase in skin defects that decreased the value of the hides. In fact, surviving alligators developed a cutaneous syndrome characterized by lymphohistiocytic proliferative cutaneous lesions (LPCLs). This syndrome is characterized histologically by multifocal dermal mononuclear cell infiltrates. Tanned hides with LPCLs show characteristics of 1 to 2 mm diameter, round pitted defects known as “pix” that have uneven dye uptake. These pix lesions appear as translucent foci when viewed on the light-table during skin grading.13,25
Nevarez et al 25 were the first to show an association between LPCLs and WNV exposure by measuring anti-WNV antibody titers in alligator serum. Furthermore, using reverse transcriptase-polymerase chain reaction (PCR), the same group detected WNV genetic material in LPCLs. 26 A similar association between the Australian subtype of WNV, Kunjin virus, and LPCL-like cutaneous lesions was recently found in Australian saltwater crocodiles (Crocodilus porosus). 13 Neither the group from Louisiana 26 or the Australian groups11,13 were able to isolate the virus from the cutaneous lesions or detect WNV virions in the skin by immunohistochemistry (IHC) or electron microscopy. After 2 decades from the first description, the pathogenesis of LPCLs remains elusive.
In this study, we investigated the pathogenesis of LPCLs in WNV-positive alligators. Considering their morphology, we hypothesized that LPCLs were lymphoid follicles formed in the dermis as a result of immune stimulation due to WNV infection. To investigate this hypothesis, we characterized the immune cell population forming WNV-associated LPCLs cutaneous lesions of American alligators using IHC and compared this to the structure of alligator gut-associated lymphoid tissue (GALT). Alligator splenic tissue was used to validate and optimize the IHC antibodies. In addition, we used in situ hybridization (ISH) to visualize WNV genome in the LPCLs cutaneous lesions, GALT, and spleen.
Material and Methods
Ethic Statement
The alligator samples from WNV-infected animals included here are formalin-fixed paraffin-embedded tissues that were collected in 2006 for a study that was approved by the Louisiana State University Institutional Animal Care and Use Committee. The control samples from WNV-negative alligators were collected in 2020 for a study that was reviewed by the Animals Research Ethics Committee at University College Dublin and was deemed full ethics review exempt (AREC-E-22-20-KELLY) as it involves samples collected exclusively during normal husbandry or veterinary clinical procedures. No procedures were carried out on live animals, and no animal was euthanized for the sole purpose of this study.
Animals
All alligators included in this study were farmed. Alligators with LPCLs were from a ranching farm in Louisiana that had a previous history of WNV and LPCLs, with the most recent outbreak in 2005. All alligators with LPCLs tested positive for WNV antibodies and WNV RNA were amplified from the LPCLs using PCR. 26 The LPCL-free animals were from a private ranching farm in Texas, where WNV and LPCLs had never been detected and thus served as controls. Alligators were 6 to 9 months of age, were of mixed gender, and were housed in rectangular buildings in groups of 20 to 80 depending on their size. The houses were insulated and divided into pools and feeding and basking decks. Pool temperatures were kept at temperatures between 29 and 32°C. The diet consisted of commercially available complete crocodilian pelleted feed purchased from a single manufacturer. The animals farmed in Louisiana were fed occasionally with whole ground chicken mixed with the commercial diet.
Sample Collection
Samples of skin, heart, lung, liver, kidney, intestine, and spleen were collected between March and June of 2006 from 10 alligators with WNV-associated LPCL lesions, which were farmed in a single private ranching facility in Louisiana. This farm had a previous history of WNV. 26 Animals with LPCL lesions were selected at random from a single building that contained all LPCL-positive alligators in the farm.
Samples of skin, heart, lung, liver, kidney, intestine, and spleen were collected in 2020 from 6 alligators reared in a single, LPCL-free ranching farm in Texas that had no previous history of WNV. 29 The LPCL-free samples from Texas were used as a control. Animals from the ranching farm in Texas were selected randomly at slaughter.
Histology
Following fixation in 10% neutral-buffered formalin, tissues were processed, embedded in paraffin, sectioned at 4 µm, and stained with hematoxylin and eosin. Histological sections were assessed for the presence of histopathological changes by pathologists P.A.K. (Diplomate ECVP) and I.M.P. (Diplomate ACVP).
Immunohistochemistry
For validation of IHC assay specificity in American alligator tissues, we designed positive and negative tissue controls for each antibody used. 37 As the distribution of B and T lymphocytes in the spleen is well defined, this tissue was elected as the control for antibodies against immune cells in these species. Three samples of spleen from American alligators with WNV-associated LPCLs were used for this purpose. B lymphocyte cells were assessed by IHC using Abcam antihuman PAX5 rabbit monoclonal antibodies (EPR3730-2) with antibody dilution of 1:200. T lymphocytes were assessed using Dako antihuman CD3 rabbit polyclonal antibodies (A0452) diluted 1:100. Antigen-presenting cells (APCs) were targeted with MHC-II Abcam monoclonal mouse antihuman HLA-DR antibodies (antigen alpha chain, Clone TAL IB5) diluted 1:200. Macrophages were targeted with WAKO antihuman IBA1 rabbit polyclonal antibodies (WAKO 019-19741) with a dilution of 1:1500. For all antibodies, the retrieval method was heat-induced epitope retrieval in sodium citrate buffer at pH6 (Sigma Aldrich). As a negative control, 1 reaction with rabbit or mouse IgG antibody serum was included per each antibody tested.
After validation, 4 samples of colon with GALT lymphoid hyperplasia and 8 samples of alligator skin (including 4 with LPCLs and 4 LPCL-free controls) were also immunolabeled with anti-PAX5, anti-CD3, anti-MHC-II, and anti-IBA1. Skin samples were bleached prior to the IHC reaction to eliminate melanin pigment, and tissues were protected during heat-induced epitope retrieval with an overlying slide as described by Vinod et al. 35 The detection method was horseradish peroxidase with 3,3’-diaminobenzidine as the chromogen for all antigens.
In Situ Hybridization
Nine samples, including 3 skin samples, 3 samples of large intestine, and 3 samples of spleen from animals with WNV-associated LPCLs were examined using ISH 36 for the presence of WNV genome. In addition, 2 samples of skin, 2 samples of colon, and 1 sample of spleen from LPCL-free control alligators were included as negative controls. To target WNV NY99P2 genome (GenBank#HQ_596519.1), we used RNAscope Probe—V-WNV-pp (catalog# 475091). RNAscope Negative Control Probe—dapB (catalog# 310043) for Bacillus subtilis dihydrodipicolinate reductase (dapB) gene, and RNAscope Positive Control Probe—Hs-UBC (catalog# 310041) for ubiquitin C were used as negative and positive controls, respectively. Colorimetric ISH was performed manually on 4 μm sections of formalin-fixed paraffin-embedded tissues on Superfrost Plus slides (Fisher Scientific, Pittsburgh, Pennsylvania) using the RNAscope 2.5 Red assay kit (cat. 322360, Advanced Cell Diagnostics, Hayward, California) as per the manufacturers’ recommendations. Deparaffinized and dehydrated tissue sections were boiled in a target retrieval buffer maintained at 100°C using a hot plate for 30 minutes. Slides were then treated with protease plus reagent for 30 minutes at 40°C. Slides were digitized using Motic EasyScan Infinity 100 NFC and an 80× objective with brightfield illumination.
Results
Histology of Lymphohistiocytic Proliferative Cutaneous Lesions and Gut-Associated Lymphoid Tissue
All WNV-positive animals (10/10, 100%) had skin lesions compatible with LPCLs, whereas LPCL-free control animals had normal skin (6/6, 100%). As previously described,24 –26 LPCLs in American alligators were characterized by large, discrete, well-defined unencapsulated dermal nodular aggregates measuring 1 to 2 mm in diameter and composed of dense sheets of round cells (Fig. 1a). The overlying epidermis was occasionally compressed by the mass effect of the LPCL. This compression happened without disruption of the basal membrane, inflammatory infiltration, or degeneration of the epidermal layers (Fig. 1a). The main cell type forming the lesions was lymphocytes, which measured approximately 15 µm, had variably distinct cell borders, high nuclear to cytoplasmic ratio, an oval nucleus with finely stippled chromatin, and scant homogeneous eosinophilic cytoplasm. Scattered throughout were low numbers of large macrophages (50-80 µm in diameter), with clear cytoplasm laden with a variable amount of basophilic to amphophilic granular cellular debris (Fig. 1b). These cells were interpreted as tingible body macrophages and conferred a characteristic “starry night appearance” to the lymphoid nodule. 37 Mitotic figures were present within the lymphoid cell population (Fig. 1b). Heterophils characterized by numerous large, bright, eosinophilic cytoplasmic granules, and a single oval, eccentric, hyperchromatic nucleus were occasionally present. The LPCLs were consistently adjacent to nerves and venules of the superficial and mid-dermal plexus (Fig. 1c). Lymphoid follicles of GALT were found in the lamina propria and submucosa of the alligators’ large intestine (Figs. 1d, 2a). Like LPCLs, GALT lymphoid follicles were composed of a nodular aggregate of lymphocytes, which were often mitotically active, and scattered tingible body macrophages. In WNV-positive alligators, colonic lymphoid follicles were 5.8 times more frequent compared to LPCL-free animals (1 lymphoid follicles every 1.5 mm of colon measured at the level of the muscularis mucosae compared with 1 lymphoid follicle every 8.8 mm in LPCL-free controls). The GALT follicles formed single nodules (Fig. 2b) or clusters within diffuse interfollicular lymphoid tissue (Fig. 2c). Clusters of follicles, reaching a size of 2.5 mm in diameter, were only seen in WNV-positive colons.
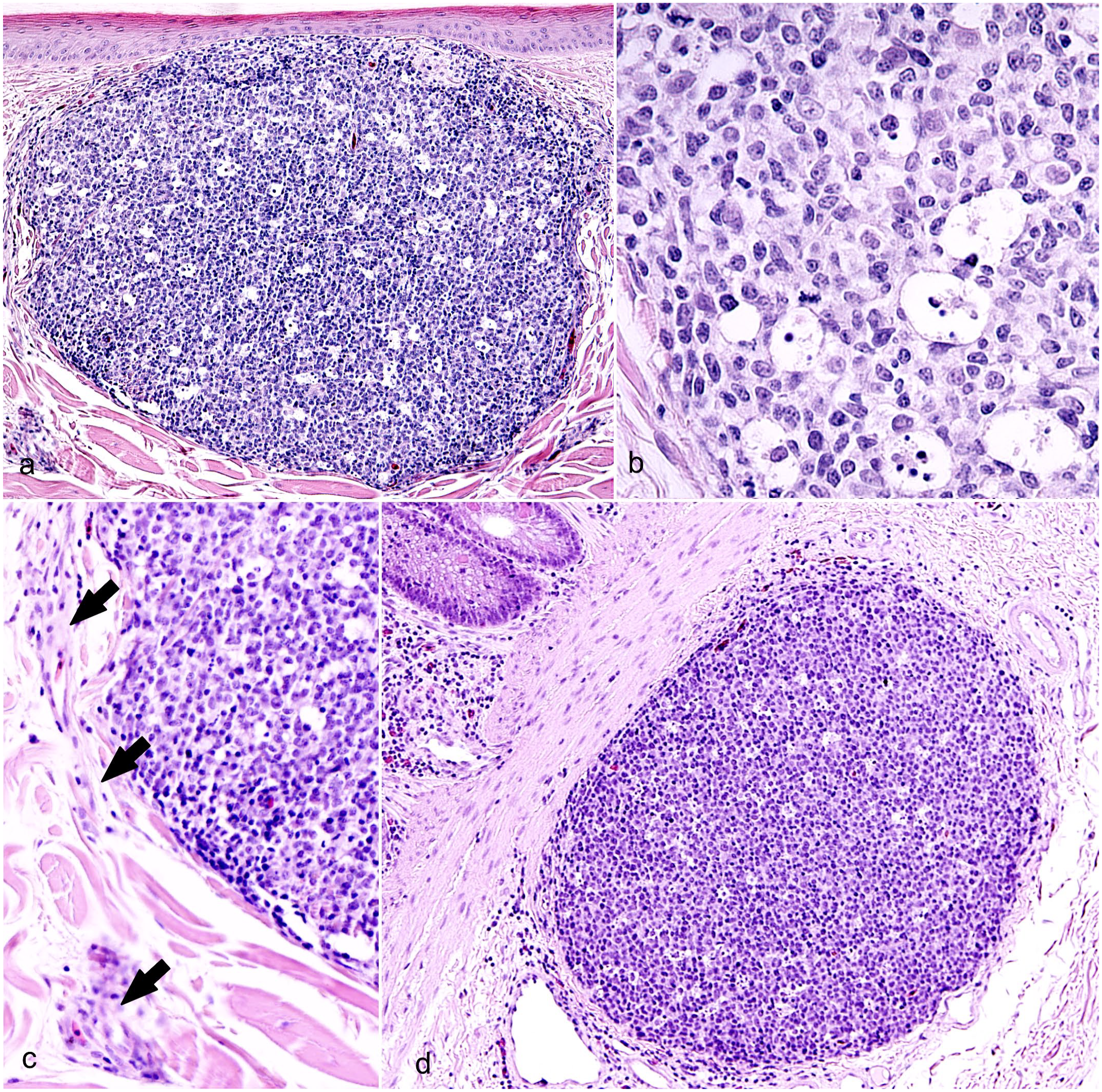
Lymphohistiocytic proliferative cutaneous lesions (LPCLs), American alligator. Hematoxylin and eosin. (a–c) Skin from the ventral abdomen. (a) LPCL presenting as a nodular, well-defined unencapsulated mass of mononuclear cells that focally replaces the collagen in the superficial dermis. The mass is compressing the overlying epidermis. (b) LPCL at higher magnification. Dense sheets of lymphocytes compose the mass. Mitotic figures are seen occasionally. Scattered throughout the lymphoid population are large macrophages with abundant cytoplasm laden with apoptotic debris (tingible body macrophages). (c) LPCLs are associated with venules and nerve bundles of the superficial dermal plexus (arrows). (d) Colon. The colonic lymphoid follicle shows similar morphology to LPCLs.
Lymphoid hyperplasia, colonic gut-associated lymphoid tissue (GALT), West Nile virus-infected American alligator. Hematoxylin and eosin. (a) Multiple lymphoid follicles are present in the submucosa. Note the diffuse increase in cellularity in the lamina propria. (b) Higher magnification of an isolated lymphoid follicle in the submucosa. The follicle is mostly composed of a central light zone and a marginal crescent-shaped dark zone. (c). A cluster of lymphoid follicles within diffuse interfollicular lymphoid tissue. The inset shows the interface between the light zone and the dark zone, which includes small, intensely basophilic round lymphocytes with scant cytoplasm, whereas the light zone includes large lymphocytes with a moderate amount of cytoplasm and lightly stained nuclei.
Occasionally, GALT lymphoid follicles displayed a central light zone and a peripheral, crescent-shaped dark zone. The dark zone included small, intensely basophilic round lymphocytes with scant cytoplasm and hyperchromatic nuclei, whereas the light zone was made of large lymphocytes with a moderate amount of cytoplasm and lightly staining nuclei with occasional prominent nucleoli (Fig. 2b, c).
Histology of Splenic Tissue of American Alligators
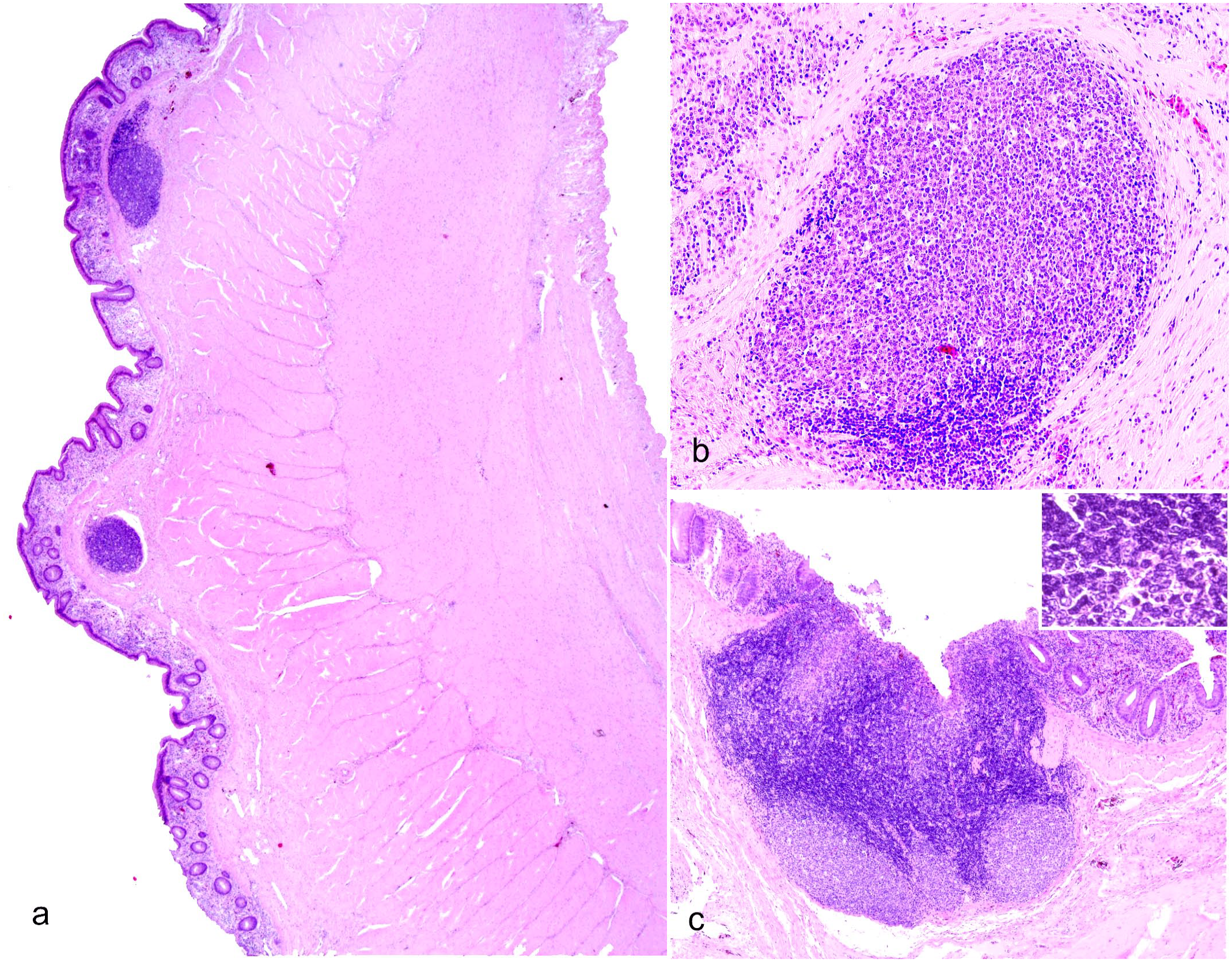
Owing to the characteristic organization of immune cells in the spleen, splenic tissue was used to validate antibodies in crocodilian tissue in this study. Before proceeding with IHC, due to the limited characterization available for the histology of crocodilian spleen, we detailed the normal histology of the organ using hematoxylin and eosin-stained sections and compared splenic morphology of WNV-positive alligators to LPCL-free control alligators. Results from histology indicated that the branches of crocodilian arteries raised from the splenic artery and then ramified into arterioles and penicilliform capillaries (Fig. 3a). Arterioles had a prominent muscular layer and were surrounded by a dense population of lymphocytes that formed the periarteriolar lymphoid sheath (PALS) (Fig. 3b). Penicilliform capillaries were surrounded by a sheath of ellipsoid cells, which were round to ovoidal and were in turn cuffed by lymphocytes, the periellipsoid lymphoid sheath (PELS) (Fig. 3c). The red pulp was clearly distinguished from the white pulp and consisted of venous sinuses. The cell population of the red pulp comprised mostly erythrocytes, and cords formed by macrophages, fewer lymphocytes, some plasma cells, and scattered granulocytic leukocytes (Fig. 3a). The presence of frequent brown pigment-laden macrophages (hemosiderophages) reflected the main function of the red pulp in the removal of senescent erythrocytes from the blood circulation (Fig. 3a). Occasionally, scattered nodules composed of large lymphocytes were identified close to the arterioles, at the interface with the PELS (Fig. 3d). These nodules were partly surrounded by a thin, 1-cell thick layer of fibroblasts (capsule), as seen in avian germinal centers, and contained scattered tingible body macrophages (Fig. 3d). The lymphocytic population displayed frequent mitotic figures, indicating proliferation. These nodular lymphoid structures were identified in 8 of 10 spleen sections from WNV-positive alligators and only in 1 of 6 sections of LPCL-free control spleens. Splenic lymphoid nodules of WNV-positive alligators were also larger, with an average size of 400 µm in diameter, and displayed up to 10 mitoses per nodule. The single splenic lymphoid nodule found in LPCL-free control alligators measured 200 µm and had low mitotic activity, with 0 to 1 mitotic figure identified per nodule.
Spleen, West Nile virus-infected American alligators. Hematoxylin and eosin. (a) The crocodilian spleen is segregated into red pulp and white pulp. The white pulp contains arteries that are sheathed primarily by small lymphocytes called periarteriolar lymphoid sheaths (PALS, black arrows). At the periphery of the PALS, the splenic arterioles further branch into the penicillary capillaries and terminate into the splenic cords. Penicillary capillaries are surrounded by layers of cuboidal reticular cells (ellipsoid or Schweigger Seidel sheath), which are in turn surrounded by periellipsoid lymphoid sheath (PELS, white arrows). Red pulp is formed from venous sinuses and anastomosing cords of reticular cells, macrophages, lymphocytes, and red blood cells. (b) Higher magnification of PALS with arterioles surrounded by numerous (up to 15) layers of small lymphocytes. (c) Closer view of PELS, which are up to 6 cells thick, surrounding the inner sheath of ellipsoid. (d) Follicular structures made of large proliferating lymphocytes form occasionally at the interface of PALS and PELS. Tingible body macrophages are sprinkled within the lymphocytes. The follicles are well defined and partly surrounded by 1 layer of fibroblasts and thin collagen.
Lymphofollicular Proliferation in the Heart, Lung, Kidney, and Liver of American Alligators
No ectopic follicles were evident in the heart or liver in the tissues examined. One ectopic lymphoid follicle was evident in the kidney of one of the WNV-positive animals. No difference was seen in the number or size of lymphoid follicles of bronchial-associated lymphoid tissue between animals with and without LPCLs. Two alligators, 1 with WNV-associated LPCLs and 1 LPCL-free, had granulomatous pneumonia. One with WNV-associated LPCL alligator had heterophilic pneumonia. Pneumonia was not accompanied by lymphofollicular proliferation in any of these cases.
Validation of Antibodies for Immunohistochemistry
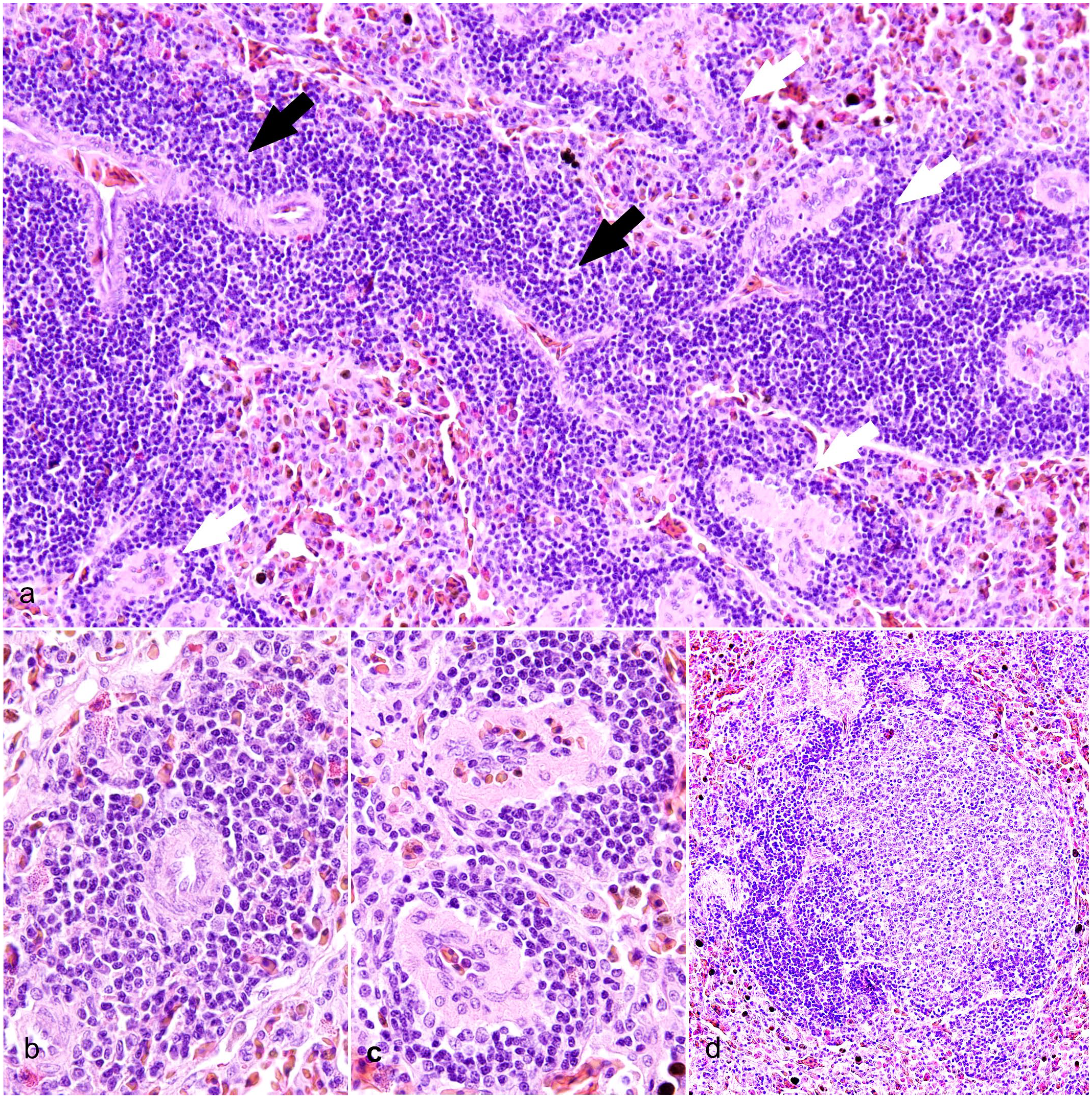
As expected, immunohistochemical labeling showed PAX5+ B lymphocytes segregated in PELS and in nodular lymphoid structures, similar to lymphoid follicles found at the interface between arterioles and PELS (Fig. 4a). PAX5 labeling was nuclear. CD3 labeling was membranous and highlighted T lymphocytes organized in cuffs around arterioles (Fig. 4b), similarly to avian and mammalian PALS. CD3+ T lymphocytes also surrounded splenic lymphoid nodular structures composed of B-cells and, in low numbers, were scattered within lymphoid nodules (Fig. 4b, inset). IBA1 positive cells were the most frequent mononuclear cells in the red pulp (Fig. 4c). A network of MHC-II+ APCs was embedded within crocodilian splenic B- and T-cell zones (Fig. 4d).
Immune cell organization of periarteriolar lymphoid sheaths (PALS) and periellipsoid lymphoid sheath (PELS). Spleen, West Nile virus-infected American alligators. Immunohistochemistry (IHC). (a) PELS are strongly PAX5-positive, as expected for B lymphocytes. The insets show PELS at a higher magnification (upper inset) and proliferating B-cell lymphoid nodules at the interface of PELS and PALS (lower inset). IHC for PAX5. (b) PALS are CD3-positive, as expected for T lymphocytes. The upper inset shows PALS at a higher magnification. In the lower inset, CD3 immunolabeled cells surround proliferating lymphoid nodules and, in low numbers, they are also scattered within. IHC for CD3. (c) IBA1-positive cells are concentrated mainly in the red pulp and, to a lesser extent, into PELS. These cells are likely macrophages. The inset shows the red pulp at a higher magnification. IHC for IBA1. (d) MHC-II immunolabeled cells are scattered within the PELS and PALS. The inset shows a higher magnification of PALS in the upper side of the photo and PELS in the lower side. IHC for MHC-II.
Immunohistochemistry of Skin and Gut-Associated Lymphoid Tissue
IHC results revealed strong nuclear anti-PAX5+ immunolabeling of GALT lymphoid follicles, which was compatible with B lymphocytes in both dark and light zones (Fig. 5a). T lymphocytes formed dense sheets of cells within interfollicular areas and were scattered in low numbers among B-cells (Fig. 5b). CD3+ T lymphocytes were also often seen infiltrating the intestinal epithelium (Fig. 5b). IBA1+ macrophages were mixed within B- and T-cell populations within follicles (Fig. 5c). MHC-II+ APCs were embedded within crocodile B- and T-cell zones (Fig. 5d).
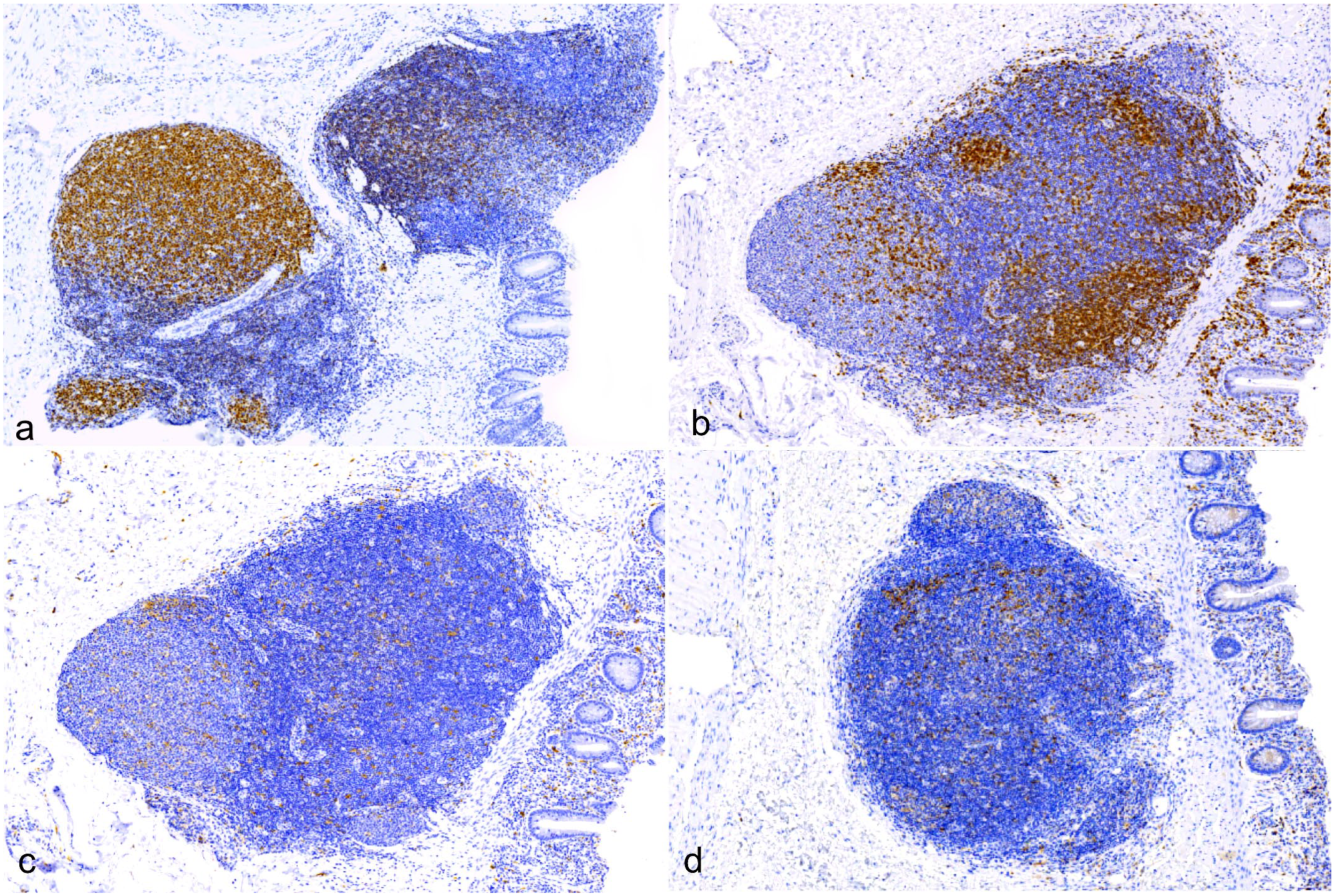
Immune cell organization in the colonic gut-associated lymphoid tissue (GALT), West Nile virus-infected American alligator. Immunohistochemistry (IHC). (a) PAX5+ immunolabeling reveals strong nuclear immunoreactivity of GALT lymphoid follicles, compatible with B lymphocytes in both dark and light zones. IHC for PAX5. (b) T lymphocytes form dense sheets of cells within interfollicular areas and are scattered in low numbers among B-cells. CD3+ T lymphocytes are also often seen infiltrating the lamina propria. IHC for CD3. (c). IBA1+ macrophages are mixed within B- and T-cell populations within follicles. Some areas of increased IBA1+ cell density are seen in the perifollicular areas. Rare, scattered IBA1+ cells also infiltrate the lamina propria. IHC for IBA1. (d) MHC-II+ antigen-presenting cells are embedded within crocodile B- and T-cell zones. In the lamina propria, low numbers of MHC-II+ cells are also present. IHC for MHC-II.
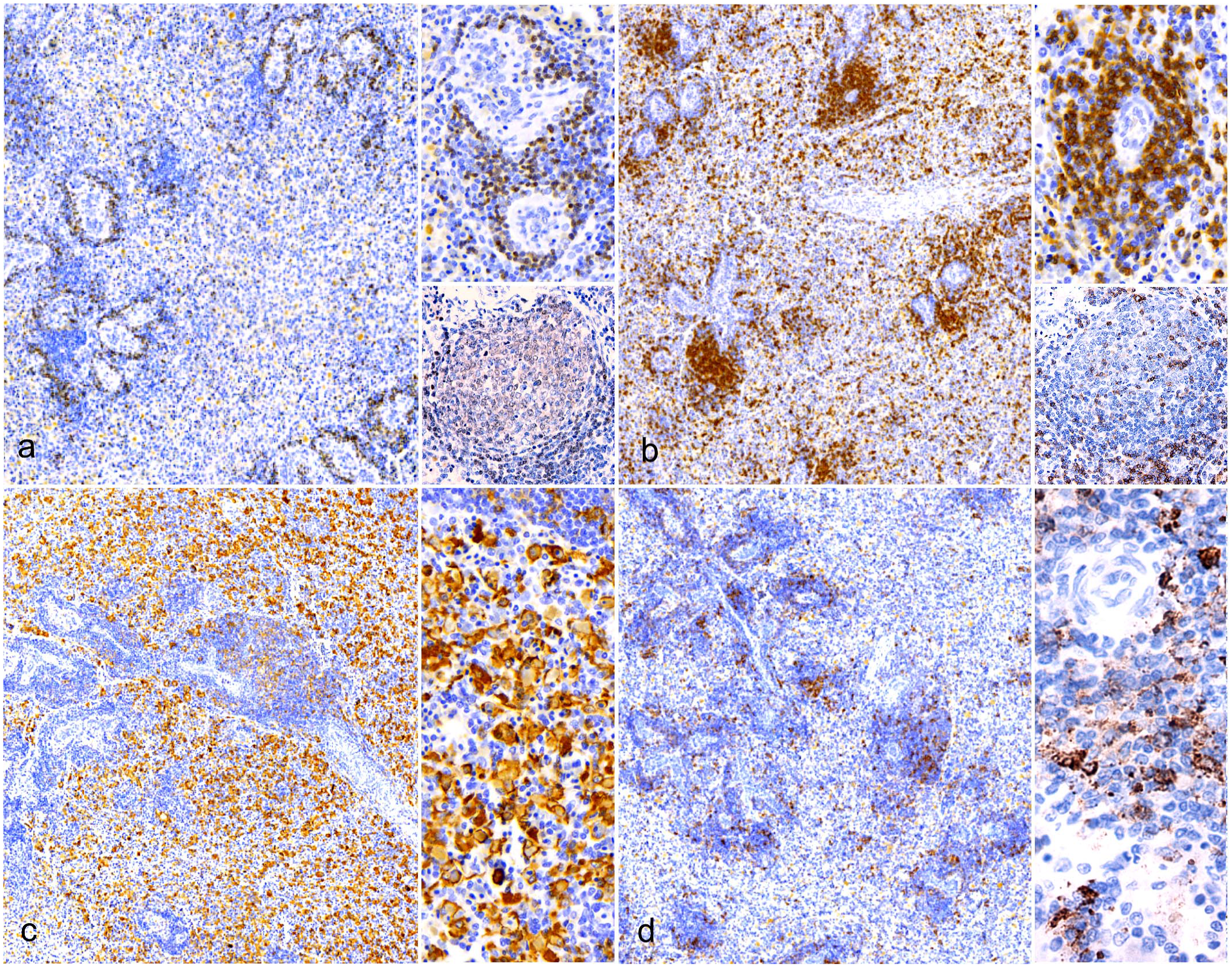
Like GALT lymphoid follicles, LPCLs were composed of an irregularly round to oval follicular aggregates of small, densely packed PAX5+ B lymphocytes (Fig. 6a). CD3+ T lymphocytes were segregated at the periphery, forming a partial rim to the B-cell follicle that was consistently associated with the dermal vasculature (Fig. 6b). IBA1+ histiocytes were scattered throughout the lymphatic population, forming a delicate web (Fig. 6c). As expected, tingible body macrophages were IBA1+ (Fig. 6c). MHC-II+ APCs formed a delicate web within the lesion and, in some sections, concentrated focally at the periphery of the follicle (Fig. 6d).
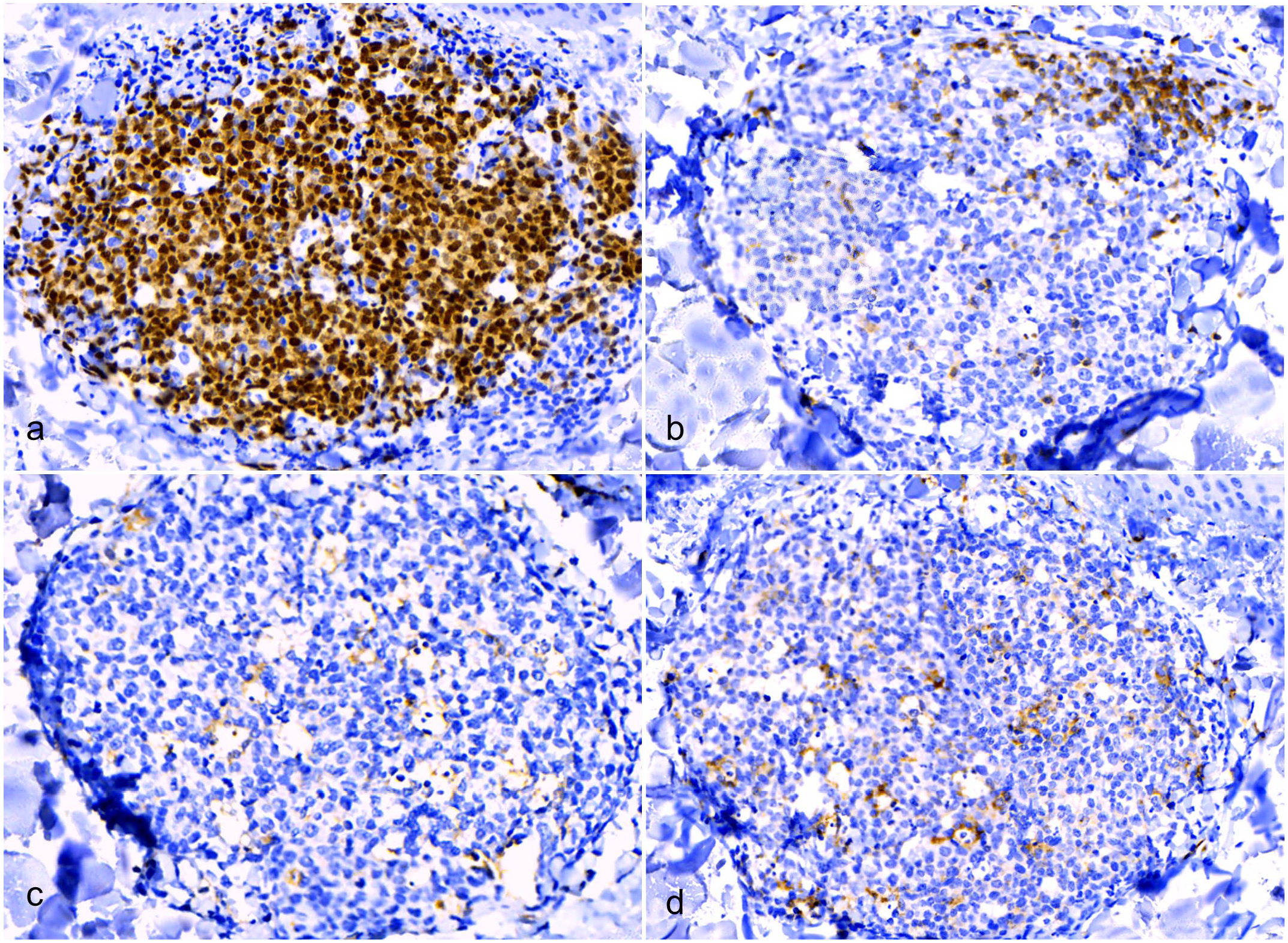
Immune cell organization in lymphohistiocytic proliferative cutaneous lesions (LPCLs), American alligator, skin. Immunohistochemistry (IHC). (a) LPCL is mainly composed of densely packed PAX5+ B lymphocytes. IHC for PAX5. (b) CD3+ T-cells are segregated at the periphery and are often associated with the dermal vasculature. IHC for CD3. (c) IBA1+ histiocytes are scattered throughout the lymphoid population, forming a delicate web. Tingible body macrophages are IBA1+ IHC for IBA1. (d) MHC-II+ antigen-presenting cells form a network within the lymphoid population and are also present in the perifollicular areas (upper right), together with CD3+ T-cells. IHC for MHC-II.
In Situ Hybridization
In the intestines, WNV genome was evident exclusively within the lymphoid follicles of the colonic GALT. The labeling was consistently identified in the cytoplasm of cells with morphology compatible with macrophages (Fig. 7a, b). Low amounts of WNV genome were also present in the macrophages within LPCLs (Fig. 7c–e) and splenic macrophages of the red pulp and PELS (Fig. 7f–h). No labeling was present in the intestines, spleens, or skin of control alligators (Supplemental Fig. S1). No labeling was evident in the skin without LPCLs of either WNV-positive alligators or controls. Positive controls showed diffuse intense hybridization for ubiquitin C within the GALT and enterocytes (Supplemental Fig. S2a, b). No labeling was detected in the dapB probe negative controls (Supplemental Fig. S2b, c).
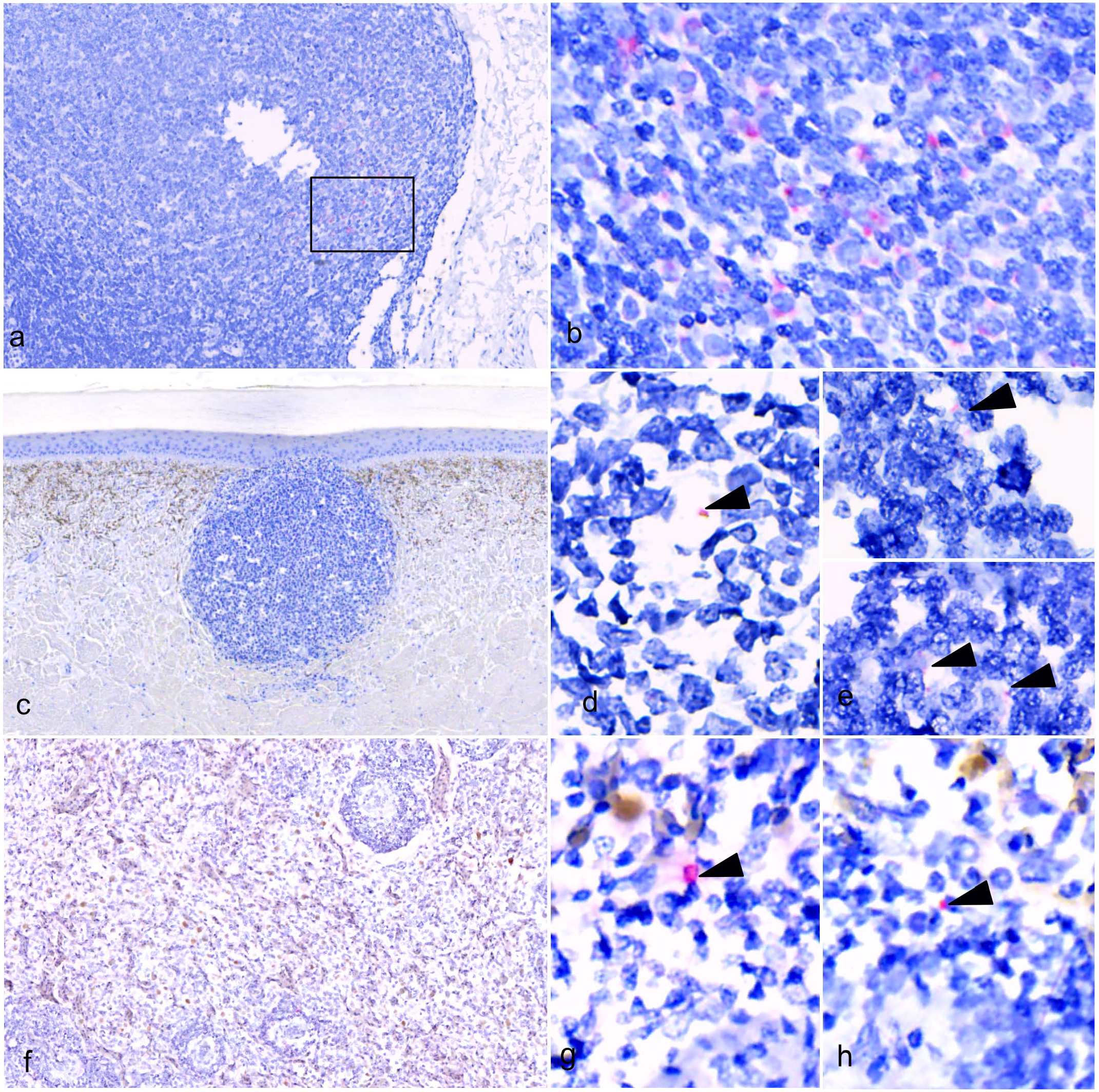
Distribution of West Nile virus (WNV) genomic RNA in American alligators with lymphohistiocytic proliferative cutaneous lesions (LPCLs). In situ hybridization (ISH) for WNV NY99P2. (a-b) Colon. (a) Low magnification of a lymphoid follicle of the colonic gut-associated lymphoid tissue (GALT). (b) A higher magnification of the section indicated in (a) by a black square. WNV RNA labeling is visible in the cytoplasm of the macrophages. (c-e) Skin from ventral abdomen. (c) Low magnification of LPCL. (d-e) WNV genome is present in low amounts within the macrophages of a dermal LPCL (arrowheads). (f-h) Spleen. (f) Low magnification of white and red pulp. (g-h) WNV genome was also within splenic macrophages of the red pulp and periellipsoid lymphoid sheath (arrowheads).
Discussion
LPCLs, as implied by their name, are proliferations of lymphocytes and histiocytes displaying a distinctive arrangement. Instances of inflammatory structures formed by lymphocytes and histiocytes include granulomas and lymphoid follicles. Given the morphological similarities between LPCLs and GALT, we hypothesized that LPCLs might be akin to lymphoid follicles.
In contrast to diffuse lymphatic tissue, which is characterized by a moderate concentration of scattered lymphocytes, lymphoid follicles present a certain degree of organization. As they are plastic structures, their morphology and organization can change depending on their developmental stage. 15 On light microscopy, lymphoid follicles present as round, oval, or irregularly circumscribed aggregates of mostly small, densely packed lymphocytes. 15 From histological examination with routine stains, many features of LPCLs corresponded to the description of lymphoid follicles. In fact, they were discrete, dense aggregates of lymphocytes with scattered tingible body macrophages, responsible for the typical “starry night pattern” of proliferating lymphoid tissue. 38
A thorough histological analysis of alligators’ GALT also showed that lymphoid follicles in the intestinal lamina propria and submucosa have an overlapping morphology to LPCLs. In addition, GALT lymphoid follicles occasionally presented areas that somewhat resembled the light and dark zones of mammalian germinal centers. Mammalian lymphoid follicles, when activated, form germinal centers with a central pale area, featuring larger lymphocytes with more cytoplasm and lightly staining nuclei (centrocytes), in contrast to a dense marginal zone of small lymphocytes (centroblasts). 20 These distinct areas were not evident in the LPCLs, and their significance in alligators’ lymphoid tissue is currently unclear.
The primary role of lymphoid follicles is to generate antibody responses with high affinity and class-switched immunoglobulins. This process necessitates well-defined B- and T-cell regions, specifically arranged in B-cell follicles and perifollicular T-cells. Within this organizational framework, CD4 T-cells play a crucial role in coordinating the proliferation, somatic mutation, and selection of B-cells with increased antibody affinity toward the antigen that triggered the response. 16
In addition, the organization of lymphoid tissue is essential for the formation of memory antibodies, as memory B- and T-cells locate each other in the outer T-zone of secondary lymphoid structures. 16
Results from immunolabeling showed that lymphoid follicles in alligator GALT maintain this organization, and so do LPCLs dermal nodules. In fact, both GALT lymphoid follicles and LPCLs were composed of a nodule of densely packed PAX5+ B lymphocytes and a peripheric area populated by CD3+ T lymphocytes. In addition, lower numbers of MHC-II+ APCs were also present through the follicle, in the form of a delicate network. In some sections, MHC-II+ cells also accumulated in the peripheral area of the dermal nodule together with T-cells. IBA+ macrophages were scattered in both B-cell and T-cell areas. This organization recapitulates the morphology of isolated lymphatic follicles of mammals and birds with a well-defined compartmentalization of B- and T-cell zones, associated with the presence of APCs (MHC-II+ cells). The APCs are specialized for activating T-cells by acquiring and presenting antigens. 31 Among these, dendritic cells, macrophages, and B-cells are recognized as “professional APCs,” with dendritic cells being the most specialized for antigen presentation. 31 The co-localization of APC and T lymphocytes in LPCLs supports the theory that they may be sites for the development of T-cell-dependent humoral immunity. Among APCs, follicular dendritic cells are considered the primary cellular players in the affinity maturation process in mammals and birds. 10 In contrast, amphibians lack follicular dendritic cells; in these species, conventional antigen-presenting cells stimulate both T- and B-cells, 10 and increases in immunoglobulin affinity are limited to 10 and 100-fold for a hapten-specific responses compared with an affinity increase of 1000 or more in mammals. 23 In reptiles, the presence of follicular dendritic cells remains uncertain, and there is no clear evidence of affinity maturation in reptile B-cells thus far.23,39 However, somatic modifications have been observed in turtle immunoglobulins.2,9 Reptiles have also demonstrated isotype switching capabilities 33 and the ability to generate immunological memory.3,6,18,28
Lymphoid follicles are the basic anatomic component of the adaptive immune system in both secondary and tertiary lymphoid organs. Secondary lymphoid organs develop in predetermined anatomic sites based on developmental signals, like the spleen, lymph nodes, Peyer’s patches, and GALT. 34 Instead, tertiary lymphoid organs form postnatally, through a process referred as lymphoid neogenesis, in response to chronic antigenic stimulation, inflammation, or persistent infection. 15 They consist uniquely of isolated lymphoid follicles tissue that can be induced in any tissue, including the brain and skin. 8 For these characteristics, tertiary lymphoid organs are also referred as ectopic or tertiary lymphoid follicles. Considering this distinction, we suggest LPCL lesions should be considered as tertiary lymphoid follicles forming de novo in the dermis.
If LPCLs are antibody-producing tertiary lymphoid follicles, they could be the result of localized or systemic antigenic stimulation. Tertiary lymphoid organs in mammals frequently develop locally in adults in response to non-resolving inflammation because of infection, autoimmunity, cancer, and allograft rejection.15,20 In birds, upon stimulation, the lymphoid system has an enhanced capacity to respond with the formation of new, widely distributed lymphoid foci, 1 especially in young animals. 22 We suggest that systemic infections, like those with WNV, may induce the crocodilian lymphoid system to respond with lymphoid neogenesis.
Lymphoid neogenesis resulting in LPCLs and lymphoid hyperplasia is supported by previous reports of lymphoid follicles formation in multiple organs and tissues in WNV-infected American alligators21,25,27 and saltwater crocodiles.11,13 In this study, a marked lymphofollicular proliferation was also evident in the intestines and spleens of alligators. In detail, GALT lymphoid follicles in WNV-positive alligators were 5.8 times more numerous than in LPCL-free animals. In the former, they were often clustered, forming structures of up to 2.5 mm at the largest diameter, whereas in the latter, they were smaller (up to 0.5 mm) and never clustered.
Seventy percent of the immune system of reptilians resides in the gut in the form of isolated lymphoid follicles. 30 Gut-associated lymphatic follicles differ from GALT tissues such as Peyer Patches or mesenteric lymph nodes of mammals in the way that they are smaller and less obvious but are more numerous. 12 Also, they appear earlier in development and are primarily composed of IgA-producing B-cells. 12 In crocodiles, IgA is also expressed at high levels in the intestine, indicating an evolutionary conserved role in mucosal immunity.5,7
The spleen is the primordial secondary lymphoid organ that evolved concurrently with adaptive immunity. 23 Secondary lymphoid organs of warm-blooded species, such as mammalian and birds, display a highly organized cellular architecture, consisting of segregated T-cell zones and B-cell follicles comprising active germinal centers, follicular dendritic cell networks, and high endothelial venules (or fenestrated vessels in birds) 22 that allow naïve and central memory T- and B-cell homing and a stromal reticular network. 15 Very limited information is available concerning T- and B-like lymphocyte distribution in the reptile spleen. 23 Among crocodilians, the anatomy of the spleen has been briefly described for the American alligator. 32 As in chelonians, the white and red pulps are distinguishable, and the lymphoid tissue of the white pulp forms PALS and PELS. 32 So far, germinal centers have not been observed in the reptilian spleen. 17
In this study, some additional white pulp was observed between and adjacent to the PALS and PELS, the regions where avian germinal centers are located. These lymphoid structures were round, composed of large cells that were partly contained by a thin capsule, as seen in avian species, although less well defined. The frequent mitosis and apoptosis accompanied by the presence of scattered tingible body macrophages suggest these may be reactive lymphoid follicles, possibly akin to germinal centers. Supporting this hypothesis was the fact that these splenic germinal center-like formations were more frequent in WNV-positive than LPCL-free alligators (80% vs 16.6%, respectively).
Finally, to explore the hypothesis that LPCLs and GALT hyperplasia in alligators are induced by exposure to WNV antigens during infection, we looked for the viral genome within these tissues. Results from ISH highlight that WNV genomic RNA was present in GALT lymphoid tissue, as well as in the dermal LPCLs and in the spleen. In all samples, WNV labeling was present in the cytoplasm of cells with morphology consistent with macrophages. In LPCLs and splenic macrophages though, the signal was weaker and less frequent compared to macrophages in the GALT. The low level of virus in the skin was probably below the detection level of IHC, possibly explaining why previous studies detected WNV antigens in the gut 27 but not in the skin 26 using this technique. Another possibility is that while viral antigens were already destroyed or processed, the viral genome persists in LPCLs. The persistence of WNV material in the LPCLs and GALT suggests that this might be triggering the lymphoid follicle formation.
Conclusions
This study provides insights into the pathogenesis of WNV-associated LPCLs. Our findings suggest that longstanding WNV infection in alligators induces an immune response characterized by lymphoid follicular hyperplasia and neogenesis in several tissues, and LPCLs in the dermis is likely a part of this response. These findings align with the propositions of certain authors, suggesting that these structures likely manifest as a localized immune response to WNV infection. 13 In addition, our study presents morphological details concerning the organization of the GALT and spleen, serving as potential references for future studies and diagnostic applications related to the secondary lymphoid organs in crocodilians.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858251317481 – Supplemental material for The pathogenesis of West Nile virus-associated lymphohistiocytic proliferative cutaneous lesions of American alligators (Alligator mississippiensis)
Supplemental material, sj-pdf-1-vet-10.1177_03009858251317481 for The pathogenesis of West Nile virus-associated lymphohistiocytic proliferative cutaneous lesions of American alligators (Alligator mississippiensis) by Ilaria M. Piras, Javier G. Nevarez, Lynn Stevenson, Frazer Bell, Georgios Ilia, Susan Peters, Deirdre Slawski and Pamela A. Kelly in Veterinary Pathology
Footnotes
Acknowledgements
We thank Mr Brian Cloak, University College Dublin, School of Veterinary Medicine for his contributions on the improvement of image quality. We also wish to thank Professor Massimo Palmarini and Dr Vanessa Herder, Centre for Virus Research, University of Glasgow, for their support on the set up of in situ hybridization in alligator tissues.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research of this article was funded by the Overhead Investment Program, University College Dublin Veterinary Hospital.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
