Abstract
This report describes subacute and chronic toxic hepatopathy in cattle due to Crotalaria spectabilis poisoning. A total of 200 male Nellore cattle were introduced into a paddock contaminated with C. spectabilis. After spending 20 days grazing in this area, 6 cattle became ill and died. The remaining 194 cattle were moved to non-contaminated pasture in a nearby farm and, 45 days after arrival, 15 cattle became ill and died. Three affected cattle were necropsied. The main clinical changes consisted of anorexia, isolation from the herd, weight loss, jaundice, recumbency, and death. The primary lesions were observed in the liver. Subacutely poisoned cattle had slightly firm livers with an accentuated lobular pattern. Histologically, hepatocyte loss with dilated sinusoids, hepatomegalocytosis, and fibrosis was observed. Cattle with chronic disease had small, pale, firm livers with an irregular hepatic capsular surface. Microscopic changes included hepatocyte loss, hepatomegalocytosis, bile duct proliferation, and fibrosis.
The genus Crotalaria includes approximately 690 species, some of which are known to cause livestock poisonings due to pyrrolizidine alkaloids (PAs).2,7,15,19 The type of PAs present in the plant may vary with the species of Crotalaria.2,3,6,9,13 Consequently, the lesions caused by PAs’ toxic effects vary according to alkaloids in the plant and animal species poisoned.3,9,17 Crotalaria burkeana and Crotalaria barkae cause laminitis in cattle, whereas Crotalaria juncea has been associated with laminitis in cattle and a break in the wool in sheep. 7 Others, such as Crotalaria dura, Crotalaria globifera, Crotalaria juncea, and Crotalaria pallida, were described as causing respiratory lesions in animals,7,15,19 whereas Crotalaria spectabilis, Crotalaria retusa, Crotalaria incana, and Crotalaria sagittalis were associated with hepatic injury.1,7,10,12,15,16
Among the hepatotoxic Crotalaria species, C. spectabilis is considered one of the most toxic.2,5,6 The first outbreak of C. spectabilis poisoning was described in chickens in the United States in 1931, 18 and serious losses in livestock occurred subsequently.2,5,12,16 Because of subsequent attempts to eliminate its use, it has become much less of a threat, although the disease still occurs occasionally. 2
Studies of C. spectabilis poisoning in cattle are rare, and most were reported between 1930 and 1940,1,12,16,18 some of which had no reported histopathologic analysis.1,12,18 Therefore, this study aimed to describe an outbreak of C. spectabilis poisoning in cattle that occurred in the state of Mato Grosso, Brazil. This study followed the criteria established by the Ethics Committee for the Use of Animals (CEUA) of the Universidade Federal de Mato Grosso under protocol no. 23108.918801/2017-87.
Clinical and epidemiologic information was obtained directly from field veterinarians during the onsite visits. The outbreak began in a backgrounding farm from Sapezal County (farm A). This farm works in an integrated crop-livestock system, intercalating cotton, Crotalaria spp., and pasture cultures. After the cotton harvesting, C. spectabilis was planted on 309 acres as a soil builder and to combat root nematodes. After Crotalaria cultivation and incorporation into the soil, Urochloa brizantha was planted as a pasture. Furthermore, volunteer growth from Crotalaria “hard seed” was observed concomitantly with the pasture sprouting, resulting in intense pasture contamination. In early August 2017, Crotalaria plants were mature with flowers and pods (Fig. 1a), and 200 Nellore steers of approximately 18 months of age were introduced into this paddock. After spending 20 days grazing in this area, 6 cattle became ill and died. One of them was necropsied (case 1). As the 194 remaining Nellore cattle were ready for the finishing phase, in early September, they were moved to a semi-intensive feedlot farm located in São José do Rio Claro County (farm B), approximately 300 km away from farm A (Supplemental Figure S1). They were introduced in a 5-acre pasture composed of U. brizantha without contamination with Crotalaria species and received additional a total mixed ration diet in the feeder. Until mid-October (45 days after arrival on Farm B), 15 cattle became ill and died. Two were necropsied (cases 2 and 3). The rest of the cattle did not get sick. When the last bovine died, as the cattle had reached slaughter weight, they were sent to a slaughterhouse. However, it was not possible to monitor them during slaughter to check for possible hepatic injuries.

Crotalaria spectabilis poisoning in cattle. (a) Mature C. spectabilis plants with flowers and pods. Upper inset: detail of the flower. Lower inset: detail of the pods. (b-d) Liver, gross lesions. (b) Multifocal to coalescent red mottled areas on the liver cut surface (nutmeg liver). Case 1. (c-d) Liver from case 3. (c) Small liver with irregular capsular surface and multifocal to coalescent pale yellow areas (asterisks). (d) Cut section showing multifocal to coalescent, pale yellow areas (asterisks).
The affected cattle (cases 1, 2, and 3) were necropsied shortly after death. Samples of organs were collected and fixed in 10% buffered formalin. All tissues were prepared for histopathologic analysis and stained with hematoxylin and eosin. Additional representative sections of the livers were stained with Masson’s trichrome stain. 14 Dried samples of Crotalaria collected from farm A were sent to the Central Herbarium of the Universidade Federal de Mato Grosso and were identified as C. spectabilis.
The morbidity, mortality, and lethality rates were 10.5%, 10.5%, and 100%, respectively. Changes noted on external examination were similar in all affected cattle and consisted of rough hair coat, anorexia, isolation from the herd, weight loss, jaundice, recumbency, and death. In addition, cattle from farm B exhibited mild-to-moderate pear-shaped enlargement of the abdomen and sloshing of fluid on abdominal succussion. The clinical course lasted 1 week on farm A and 3 to 4 weeks on farm B.
At necropsy, significant lesions were identified in the livers of all cattle. In case 1, the liver was slightly firm with multifocal to coalescent red mottled areas on the hepatic capsular and cut surfaces with an accentuated lobular pattern (“nutmeg” appearance) (Fig. 1b). Cases 2 and 3 had small, firm livers with irregular capsular surfaces (Fig. 1c). On the capsular surface and cut section, the predominantly brown parenchyma was crisscrossed by a network of fine white strands (fibrosis) that sometimes converged to form pale yellow areas (Fig. 1d). In addition, cases 2 and 3 had mild-to-moderate edema of the mesentery and abomasal folds, mild-to-moderate ascites (causing enlargement of the abdomen), hydrothorax, and hydropericardium.
Histologically, the hepatic lesions of case 1 consisted of centrilobular and bridging hepatocyte loss with dilated sinusoids and mild fibrosis (Fig. 2a). The adjacent areas were composed of hepatocytes with cytoplasmatic vacuolation, severe hepatomegalocytosis, and cholestasis (Fig. 2b-c). Microscopic liver changes in cases 2 and 3 consisted of hepatocyte loss with severe hepatomegalocytosis, bile duct proliferation, and fibrosis (dissecting type), causing disorganization of the typical hepatic lobe architecture (Fig. 2d-f). Furthermore, cases 2 and 3 had alterations in the brain (hepatic encephalopathy) characterized by mild-to-moderate vacuolization at the junction between the gray matter and the subcortical white matter of the frontal, parietal, and occipital telencephalon and the white matter of the mesencephalon and cerebellar peduncles.
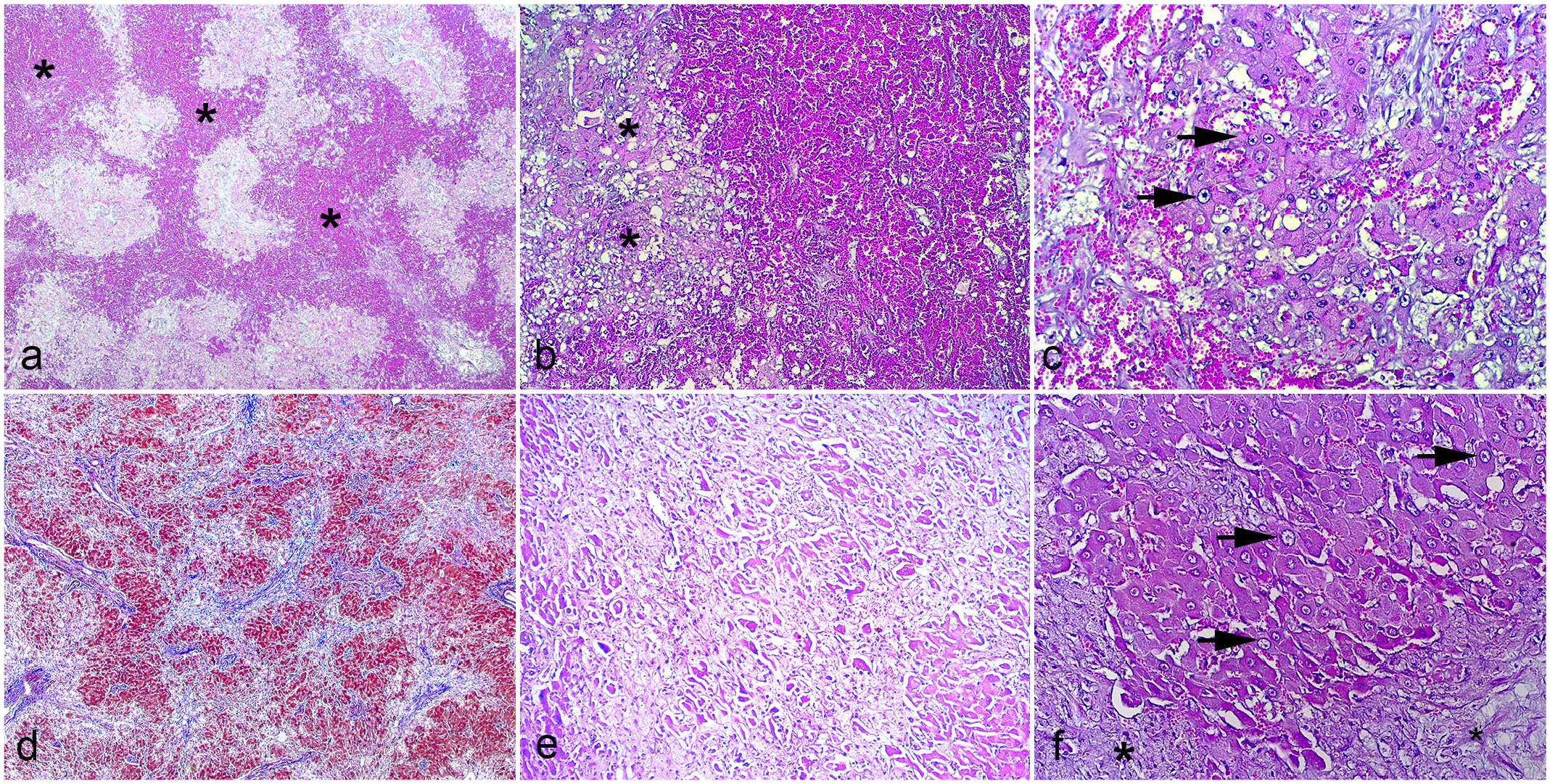
Histopathologic features of Crotalaria spectabilis poisoning in cattle. (a-c) Liver from case 1. Hematoxylin and eosin (HE). (a) Centrilobular and bridging hepatocyte loss with dilated sinusoids (asterisks). (b) The areas adjacent to necrosis are comprised of hepatocytes with moderate to severe cytoplasmatic vacuolation (asterisks) and (c) severe hepatomegalocytosis (arrows). (d-f) Liver from case 3. (d) Hepatocyte loss with multifocal to coalescent fibrosis (blue). Masson’s trichrome. (e) Hepatocyte loss and fibrosis. HE. (f) Fibrosis (asterisks) and severe hepatomegalocytosis (arrows). HE.
The diagnosis of C. spectabilis intoxication was based on epidemiological data, clinical signs, postmortem and histopathological findings, and the plant identification in the pasture.
C. spectabilis dissemination is associated with its ability as a soil builder and root nematodes control. 15 The main active principle is monocrotaline, a PA responsible for hepatic lesions.2,6 Among the Crotalaria species, C. spectabilis is considered one of the most toxic due to its high concentration of this substance.6,13 In this study, cattle fed on the plant in the flower and pod stages. This is the most toxic stage of Crotalaria species because the flowers and seeds (inside pods) have the highest concentrations of monocrotaline.6,13
Monocrotaline is the most common PA in the hepatotoxic Crotalaria species and primarily has hepatotoxic effects. As the hepatic cytochrome P450 system is responsible for metabolizing PAs into toxic pyrroles, the liver is the primary target organ. Pyrroles injure hepatocytes by irreversibly binding to DNA and inhibiting hepatocyte mitosis. The DNA continues to be synthesized in the nucleus, and the nuclear and cytoplasmic volumes of the non-dividing cell continue to increase. Despite their increased volume, hepatic megalocytes are not able to function as normal hepatocytes. Their metabolism is considerably diminished. Those attempting mitosis die and are replaced by fibrous connective tissue.2 –4,9,17 Liver failure occurs when the lesion extends beyond 70% to 80% of the parenchyma, resulting in the animal’s death. 4
Poisoned cattle mainly exhibit chronic toxic hepatopathy. Acute cases are uncommon and are restricted to experimental trials.5,10,12,15,16,19 The gross appearance (nutmeg liver) observed in the liver of case 1 is frequently reported in acute hepatic intoxication due to poisonous plant in cattle4,7,15,19 (including PA-containing plants,8,11 C. spectabilis5,18 and Crotalaria spp.15,19) and other animal species.4,19 Changes described in acute toxic liver injury include coagulation necrosis of centrilobular hepatocytes and hemorrhage. There are slightly shrunken hepatocytes that stain intensely with eosin and may have visible but distorted nuclei with pyknosis and karyorrhexis. 4 The histologic pattern in the case 1 liver consisted of hepatocyte loss with dilated sinusoids, mild fibrosis, and hepatomegalocytosis. We propose that a sublethal acute hepatic necrosis occurred in this case. The dead hepatocytes were removed by macrophages (Kupffer cells), and the absence of hepatocytes resulted in dilated sinusoids, leading to the nutmeg liver pattern. Later, fibrosis and megalocytosis developed. The gross and microscopic changes in cases 2 and 3 are consistent with those reported in chronic cases of hepatopathy in cattle poisoned by PA-containing plants, including C. spectabilis.5,7,8,11,12,16,18
During the hepatic metabolism of monocrotaline, a portion of the pyrroles may escape into the circulation, causing damage to other tissues, including the kidney and lungs.3,9,17 Although kidney and lung changes were described in monocrotaline poisoning in pigs and chickens, 9 these lesions were not observed in this study nor in other studies with cattle.8 –10,16 The toxic effect of PAs in livestock varies considerably with the animal species and the nature of the alkaloids involved.3,9,17
The clinical signs observed in this study were similar to those reported in the cattle hepatopathy due to consumption of Crotalaria spp. and others PA-containing plants.7,11,12,16,18 As described in this study, clinical signs or death of cattle grazing hepatotoxic PA-containing plants may occur after a latent period of months to years after the last intake of the green plant and seeds or after the plant has wilted and is no longer evident in the pasture.11,12,16 In most instances, poisoning develops after consumption of fresh plants, contaminated hay, or ration produced with seed-contaminated grains. Although it can cause disease in other species, 15 the consumption of seeds is a major problem among poultry and swine,2,15,18 whereas fresh plants and contaminated hay affect mainly cattle, horses, and small ruminants.2,15,16,18 C. spectabilis and other Crotalaria species are not usually palatable to grazing cattle1,7,12,16 and ingestion directly from the pasture is not common except where no other food is available or in highly contaminated pasture,12,16 as observed in this study.
Repeated ingestion of small doses of green plants or seeds over a prolonged period causes progressive liver injury over weeks or months until the lesion becomes sufficiently severe and signs of liver failure ensue. In such cases, cattle ingest sublethal doses insufficient to induce the acute form of the disease. However, small portions can produce negligible lesions that, over extended periods, can merge into significant chronic injuries that result in clinical signs of liver failure.11,12,16 Morbidity and mortality in the poisoning by C. spectabilis, 16 other Crotalaria species,7,8,10 and PAs-containing plants are variable, 8 but frequently the lethality is closer to 100%, which it was in this case.
In conclusion, C. spectabilis poisoning was only described in the United States and Brazil. Despite this intoxication occurring occasionally in the southern states of the United States, 2 C. spectabilis poisoning has increased in Brazil in the last few years. 15 Finally, along with Senecio spp. (in the south region) and C. retusa (in the northeast region), C. spectabilis (in the central west region) is an important PA-containing plant that can cause subacute and chronic cattle disease in Brazil.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858241281899 – Supplemental material for Subacute and chronic toxic hepatopathy in cattle grazing pasture with Crotalaria spectabilis
Supplemental material, sj-pdf-1-vet-10.1177_03009858241281899 for Subacute and chronic toxic hepatopathy in cattle grazing pasture with Crotalaria spectabilis by Marlon Ribeiro, Isadora N. Bianchi, Wuglenya D. M. Silva, João Paulo S. Cavasani, Ícaro G. Santos, Luana Dias, Edson M. Colodel and Fernando H. Furlan in Veterinary Pathology
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors are grateful to the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), and Fundação de Amparo à Pesquisa do Estado de Mato Grosso (FAPEMAT) and Pró Reitoria de Pesquisa da Universidade Federal de Mato Grosso (PROPeq).
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
