Abstract
Insulinoma-associated protein 1 (INSM1), a recently identified neuroendocrine marker, is a transcriptional regulator with highly conserved INSM1 homologues in various species. This study investigated the immunohistochemical reactivity of the INSM1 antibody in 20 normal canine neuroendocrine tissues from various anatomical locations, 87 hyperplastic or neoplastic tissues of neuroendocrine origin, and 62 non-neuroendocrine neoplasms and compared the results with those of chromogranin A and synaptophysin in neuroendocrine neoplasms. Western blot was performed on fresh canine pituitary glands and canine parathyroid glands to confirm the specificity of the anti-INSM1 antibody. The results showed that the anti-INSM1 antibody could detect nuclear expression in normal canine neuroendocrine tissues, except for the parathyroid glands. INSM1 was detectable in 79/87 (91%) of the hyperplastic and neoplastic neuroendocrine lesions, but all parathyroid carcinomas and parathyroid adenomas (three samples each) were negative for INSM1. In contrast, INSM1 was detected in only one of 62 (2%) non-neuroendocrine neoplasms. The overall percentage of neuroendocrine neoplasms that immunolabeled positively for all three markers was 89%. In addition, the nuclear expression of INSM1 was easier to interpret than that of chromogranin A or synaptophysin. These findings confirm that INSM1 is a useful immunohistochemical marker for diagnosing canine neuroendocrine neoplasms, except for parathyroid neoplasms, and should be considered as part of immunohistochemistry panels to improve diagnostic capability.
Keywords
Neuroendocrine (NE) cells are a group of specialized cells that can synthesize, store, and release hormones in response to stimulation of the nervous system. NE cells are primarily localized in the endocrine system, while a small proportion is found in other body systems. NE neoplasms are derived from NE cells and can arise in almost any anatomical location. Histologically, well-differentiated NE neoplasms have NE packeting,19,20,24 but poorly differentiated NE neoplasms may be misdiagnosed as other neoplasms of epithelial origin or even round cell tumors.10,16,24
The diagnosis of NE neoplasms relies on confirmation of NE differentiation, which can be accomplished using several NE immunohistochemical (IHC) markers, including chromogranin A (CGA) and synaptophysin (SYP).9,20 Both CGA and SYP are cytoplasmic IHC markers and have different sensitivities and specificities for various NE neoplasms.20,24 Other IHC assays for detecting hormones produced by neoplastic cells are also valuable for diagnosing specific NE neoplasms, such as insulinomas and somatostatinomas. 24 In human medicine, second-generation NE markers have been proposed for expanding diagnostic capacity.11,12 The insulinoma-associated protein 1 (INSM1) antibody, a member of the second-generation NE markers, is thought to be highly sensitive and specific for NE neoplasms irrespective of their differentiation status or anatomical location. 20
The INSM1 antibody was first introduced as a diagnostic aid in 2015. 22 Since its first publication, the utility of the INSM1 antibody as a diagnostic marker for NE differentiation has been reported in a growing number of studies. The expression of INSM1 has been demonstrated in human pancreatic NE tumors,15,22,27 medullary thyroid carcinomas,8,22,26 pheochromocytomas, 22 and Merkel cell carcinomas.14,22 The nuclear expression of INSM1 provides an additional way to detect NE differentiation.
INSM1 is a transcription factor that plays a critical role in NE cell development6,7,13,32 and was originally isolated from human pancreatic insulinomas. 7 In humans and mice, INSM1 is mainly found in the NE tissues and nervous system of developing embryos, with only limited expression in healthy adult tissues.7,13,30 INSM1 is detected in several NE tumor cell lines, while its expression is not observed in non-NE epithelial or mesenchymal tumors in humans. 7
Highly conserved INSM1 homologues in various species have been confirmed previously. 13 At the protein level, the degree of relatedness of mouse INSM1 to human INSM1 is 86%. 30 The phylogenetic tree generated by Cai et al 3 reveals that, compared to the mouse Insm1, the canine INSM1 is more closely related to the human INSM1 gene. In the veterinary literature, the only published research mentioning INSM1 was published in 2020 by Capodanno et al. 4 They found an increased expression of INSM1 in canine insulinoma compared to normal pancreas using a quantitative real-time reverse transcription polymerase chain reaction assay. 4 Despite these prior studies, INSM1 has not been evaluated as an NE IHC marker for diagnostic use in veterinary pathology.
We hypothesized that the structure of the INSM1 protein in dogs is identical to that of human INSM1 and that the expression of INSM1 protein could be detected in canine NE neoplasms using IHC. The main objective of this study was to investigate the IHC reactivity of the INSM1 antibody in formalin-fixed, paraffin-embedded (FFPE) canine tissues and to introduce the INSM1 antibody as a nuclear NE marker for diagnosing canine NE neoplasms. We also compared the expression of INSM1 to that of common IHC markers of NE neoplasms in dogs.
Materials and Methods
Canine Case Selection
Only canine cases were included in this research. A retrospective screening was conducted on the electronic database of the Graduate Institute of Molecular and Comparative Pathobiology at National Taiwan University and the HOPE Veterinary Pathology Diagnostic Center, spanning from January 2014 to June 2023, utilizing electronic searches with specific keywords. The keywords used for NE proliferative lesions included aortic body, C-cell, carcinoid, carotid body, chemodectoma, insulinoma, islet, medullary carcinoma, Merkel, neuroendocrine, parafollicular, paraganglioma, parathyroid, pheochromocytoma, pituitary, and thyroid. To examine INSM1 IHC reactivity in non-NE neoplasms, cases were selected based on the following criteria: (1) cases previously suspected to be NE neoplasms based on histological features but were subsequently excluded using IHC; (2) neoplasms arising in endocrine organs that were not derived from NE cells, such as thyroid follicular neoplasms and adrenocortical neoplasms; and (3) tumors that have been suggested to be carefully differentiated from NE neoplasms in the literature, including anal sac adenocarcinomas, 17 malignant melanomas, 28 mammary gland carcinomas, 16 olfactory neuroblastomas, 25 round cell tumors, 10 and trichoblastomas. 29
Specimens with formalin fixation, available paraffin blocks, and adequate sample size were selected for inclusion in this study. The selected cases were grouped into 3 categories: histologically normal tissue, NE proliferation (hyperplasia, adenoma, or carcinoma), and non-NE neoplasm. Canine cases with NE neoplasms of the pituitary gland, thyroid gland, parathyroid gland, pancreas islets, adrenal medulla, and carotid and aortic bodies, or those diagnosed as Merkel cell carcinoma, neuroendocrine tumor, or neuroendocrine carcinoma were placed in the NE proliferation group. In all groups, the signalment and clinical and pathologic findings were recorded from pathology reports.
Histologic Analysis
The FFPE specimens were routinely sectioned and stained with hematoxylin and eosin. The hematoxylin and eosin-stained slides of all selected cases were reviewed by 2 veterinary pathologists (W.-H. H. [board-certified by the Chinese Society of Veterinary Pathology] and F.-H. Y. [a third-year resident veterinary pathologist]) to confirm the diagnosis and achieve diagnostic consistency.
Immunohistochemistry
IHC labeling (see Supplemental Table S1 for antibodies and reagents used in this study) for INSM1, CGA, and SYP was performed on 4-μm-thick sections from all selected cases with the following primary antibodies: mouse monoclonal anti-INSM1 antibody (clone A-8, 1:1000 dilution; Santa Cruz, USA), rabbit anti-CGA antibody (1:100 dilution; Genemed, USA), and mouse monoclonal anti-SYP antibody (clone SY38; 1:1000 dilution; Dako, USA). Neuron-specific enolase (clone M0873; 1:4000 dilution; Dako, USA) was only used in cases where the tumor exclusively expressed either INSM1, CGA, or SYP to confirm or exclude an NE cell origin. For each tissue section, xylene and ethanol were used for deparaffinization, and heated Trilogy buffer (Cell Marque, USA) was used for antigen retrieval. Antigen retrieval was performed at 80°C in a digital pressure cooker for 30 minutes. The IHC protocol used the optimal dilution, retrieval method, and detection system with the best signal-to-noise ratio. The criteria for selecting positive control cases were: (1) cases with hormone-related clinical features and (2) expression of both CGA and SYP recorded in previous pathological reports. Additionally, normal tissues adjacent to the lesions served as internal negative controls. To evaluate the specificity of the INSM1 antibody for NE tissues and their derived neoplasms, the immunoassay included a broad range of canine non-NE normal tissues from various anatomic locations, encompassing all tissue sections of the selected cases, as well as non-NE neoplasms chosen according to the aforementioned criteria.
Interpretation of IHC was conducted by 2 veterinary pathologists (W.-H. H. and F.-H. Y.) to ensure interobserver consistency. The extent of immunolabeling, defined as the percentage of immunolabeled cells, for each tumor was assigned to 1 of 4 levels: negative (absence of positive labeling) or variable degrees of positivity (1%–19%, 20%–50%, or >50% immunopositive cells) (Supplemental Table S2). This semiquantitative immunolabeling scoring system was modified from the first report on the application of INSM1 IHC as a diagnostic aid. 20 The intensity of the antibody immunolabeling was subjectively scored according to nuclear/cytoplasmic reactivity as follows: 0 = absent labeling, 1 = weak, 2 = moderate, and 3 = strong positivity (Supplemental Figure S1).
Antigen Sequence Alignments and Western Blotting
Antigen sequence alignment and Western blotting were conducted to ascertain the cross-reactivity of the antibody employed in this study in dogs. The antigen sequence was identified according to the datasheet of the anti-INSM1 antibody used in this study and the total INSM1 sequence retrieved from the National Center for Biotechnology Information (NCBI) protein blast database. The canine and murine INSM1 protein sequences were also sourced using the same NCBI protein blast database. The amino acid sequence alignment of the INSM1 protein across these species was conducted using the Mega software.
Western blotting was performed to further validate the specificity and cross-reactivity of the monoclonal INSM1 antibody on canine specimens. Fresh tissues from the canine pituitary glands and parathyroid glands were collected from the necropsy cases submitted to our institute. Fresh mouse pituitary tissues were collected from euthanized healthy laboratory mice and served as positive control. Total protein was prepared by homogenized tissues in RIPA lysis buffer (VWR Chemicals, Sohn, Ohio, USA) on ice and mixed with cOmplete EDTA-free protease inhibitor cocktail (Roche Molecular Biochemicals, Laval, Quebec, Canada) and PhosSTOPTM phosphatase inhibitor (Roche Molecular Biochemicals). The solution was clarified by centrifugation at 14,000g for 15 minutes, and the supernatant was stored in aliquots at −80°C. For the Western blotting assay, protein lysate was size-fractionated by 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis gel, blotted onto a polyvinylidene difluoride membrane (Bio-Rad, Hercules, CA, USA) and blocked with BlockPROTM 1 Min Protein-Free Blocking Buffer (Energenesis Biomedical, Taipei City, Taiwan) for 3 minutes. The blots were subsequently probed at 4°C overnight with antibodies against INSM1 (1:1000 dilution in Tris-buffered saline with Tween 20; Santa Cruz, USA) or β-actin (1:2000 dilution in T Tris-buffered saline with Tween 20, GeneTex, USA). Following incubation with horseradish peroxidase-conjugated secondary anti-rabbit antibody (1:10,000 dilution in blocking buffer; Jackson ImmunoResearch Laboratories, Philadelphia, PA, USA), the protein bands were visualized with the ClarityTM Western ECL Blotting Substrates (Bio-Rad) and detected by ChemiDocTM Imaging Systems (Bio-Rad).
Results
Antigen Sequence Alignments and Western Blotting
According to the datasheet of anti-INSM1 antibody used in this study, the antigen was 81–125 amino acids near the N terminus of human INSM1; upon comparison, the 82–125 sequence completely matched the 104–147 amino acids near the N terminus of canine INSM1. The detailed sequence and its comparison with those of dogs and mice are presented in Table 1. Western blot analysis demonstrated that the INSM1 antibody identified two bands at about 65 and 50 kDa when tested against the entire canine pituitary protein extract. In the mouse pituitary, the INSM1 antibody identified 2 distinct bands at around 52 and 50 kDa. The Western blot did not detect the INSM1 protein in the canine parathyroid protein extract (Supplemental Figure S2).
Alignment of human insulinoma-associated protein 1 (INSM1) antigen amino acid sequences with canine and murine counterparts.

The sequence is the antigen source for the antibody utilized in this study.
Normal Canine Tissues
To confirm the utility of IHC for detecting INSM1 in canine FFPE specimens, we first demonstrated INSM1 expression in histologically normal NE tissues. Using an anti-INSM1 antibody, INSM1 was detected in canine pancreatic islets, anterior pituitary, adrenal medulla, thyroid C cells, enteroendocrine cells of the stomach and intestine, and Merkel cells of tactile hair (Fig. 1a–f). In contrast, IHC labeling for INSM1 was absent from the exocrine pancreas, posterior pituitary, adrenal cortex, thyroid follicular cells, parathyroid gland, non-NE elements of the digestive tracts and skin, and other non-NE tissues or cell types (Supplemental Table S4 and Supplemental Figure S3). The IHC labeling was limited to the nucleus, except for occasional intracytoplasmic labeling in cells in pancreatic islets, the anterior pituitary, and the adrenal medulla. Background labeling was minimal, except for nonspecific binding of the anti-INSM1 antibody in the colloid of thyroid follicles (Fig. 1d). Small cell-to-cell differences in labeling intensities were frequently observed between NE cells arranged in clusters, especially in the pancreatic islets (Fig. 1c).
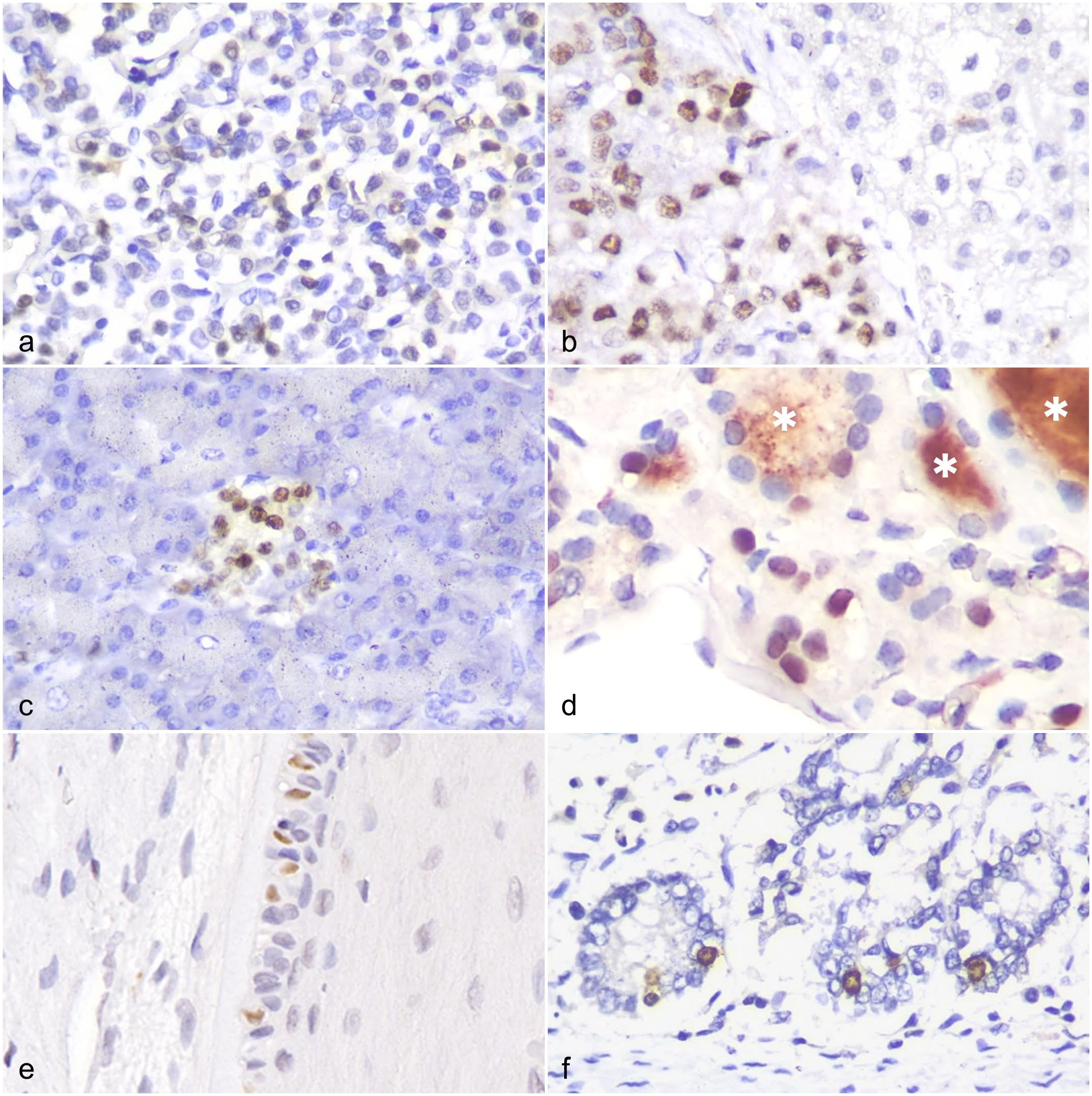
The anti-insulinoma-associated protein 1 (INSM1) antibody detects INSM1 expression in canine neuroendocrine tissues. INSM1 immunohistochemistry. (a) INSM1 is expressed in the anterior pituitary gland. (b) INSM1 is detected in the adrenal medulla (left side of the panel) but not in the adrenal cortex (right side of the panel). (c) INSM1 is strongly expressed in pancreatic islets but not in pancreatic acini. (d) Thyroid C cells express INSM1 in their nuclei; in contrast, thyroid follicular cells do not express INSM1. Nonspecific binding between anti-INSM1 antibody and the colloid of thyroid tissue is observed (asterisk). Scattered INSM1 labeling in (e) Merkel cells of tactile hairs and (f) enteroendocrine cells of intestine crypts.
In addition, the normal canine NE tissue specimens were immunolabeled for CGA and SYP, 2 common NE IHC markers. There was an absence of variable expression of INSM1, CGA, or SYP in NE cells in several anatomical sites, including the parathyroid gland, small intestine, and cutaneous tactile hairs (Fig. 2a–i). INSM1 and SYP expression was not detected in any of the 7 canine parathyroid gland specimens. Scattered positive labeling for INSM1 and CGA was detected in the intestinal crypts of 5 canine small intestine specimens, but SYP expression was not detected. Merkel cells of 3 canine tactile hair specimens showed scattered positive labeling for INSM1 and SYP, but no expression of CGA.
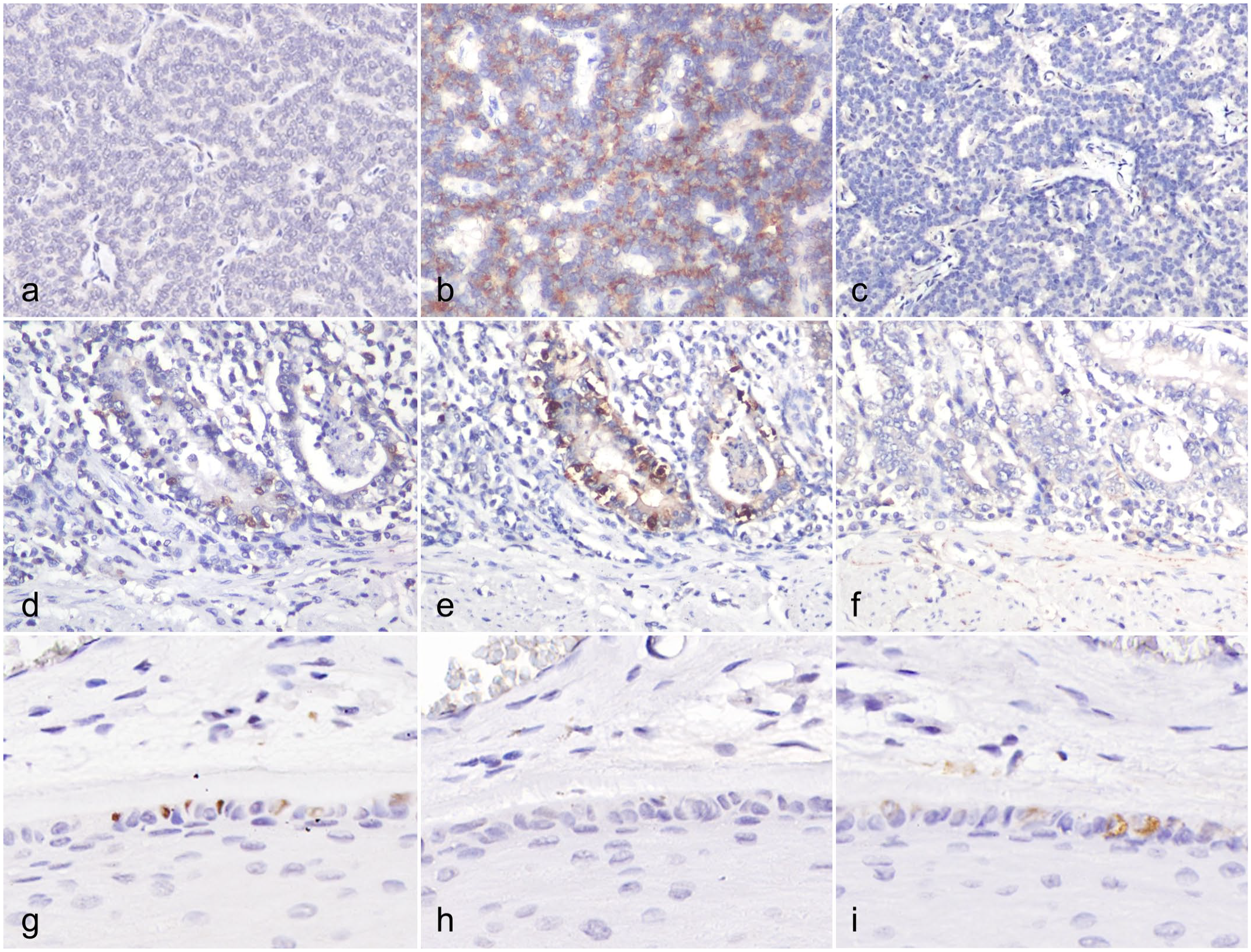
Variable immunohistochemical (IHC) labeling of (a, d, g) insulinoma-associated protein 1 (INSM1), (b, e, h) chromogranin A (CGA), and (c, f, i) synaptophysin (SYP) in the (a–c) parathyroid gland, (d–f) small intestine, and (g–i) cutaneous tactile hair, dog. (a–c) The parathyroid gland lacks expression of (a) INSM1 and (c) SYP but is labeled for (b) CGA. (d–f) Enteroendocrine cells of intestine crypts are labeled for (d) INSM1 and (e) CGA but lack labeling for (f) SYP. (g–i) Merkel cells of tactile hairs are labeled for (g) INSM1 and (i) SYP but are negative for (h) CGA.
Canine NE Proliferation
To demonstrate the utility of IHC for detecting INSM1 in canine NE proliferation tissues, 87 FFPE specimens from 70 dogs were examined (Supplemental Table S3). Sixty-five specimens were primary malignant neoplasms, 14 were metastatic lesions, 6 were benign tumors, and 2 were hyperplastic lesions. The specimens originated from the following tissues: pancreas (20), thyroid glands (18), adrenal glands (11), lymph nodes (8), parathyroid glands (6), liver (5), spleen (3), skin (3), pituitary (2), lung (2), aortic body (2), gallbladder (2), carotid body (1), mediastinum (1), abdomen (1), duodenum (1), and maxilla (1). Two canine islet cell carcinomas associated with severe hypoglycemia and previously found to express CGA and SYP served as positive controls.
The immunolabeling pattern for INSM1 in canine NE hyperplastic and neoplastic lesions was distinct and exclusively nuclear (Fig. 3a–i). Nonspecific labeling was not observed in intratumoral necrotic regions, tumor stroma, or extra-neoplastic tissues. Across the 87 specimens tested, INSM1 showed positive expression in 79. The NE neoplasms that were negative for INSM1 included the 6 parathyroid neoplasms (Fig. 3i), 1 malignant pheochromocytoma, and 1 pituitary chromophobe adenoma. In 72% (63/87) of the cases, INSM1 immunolabeling was observed in more than 50% of the proliferative cells. A strong labeling intensity (score: 3) for INSM1 expression was observed in 63% (55/87) of the cases. Compared with cytoplasmic NE markers, the nuclear labeling of INSM1 was more easily distinguishable in neoplastic cells with intracytoplasmic pigments, vacuolated cytoplasm, or a high nuclear/cytoplasmic ratio.
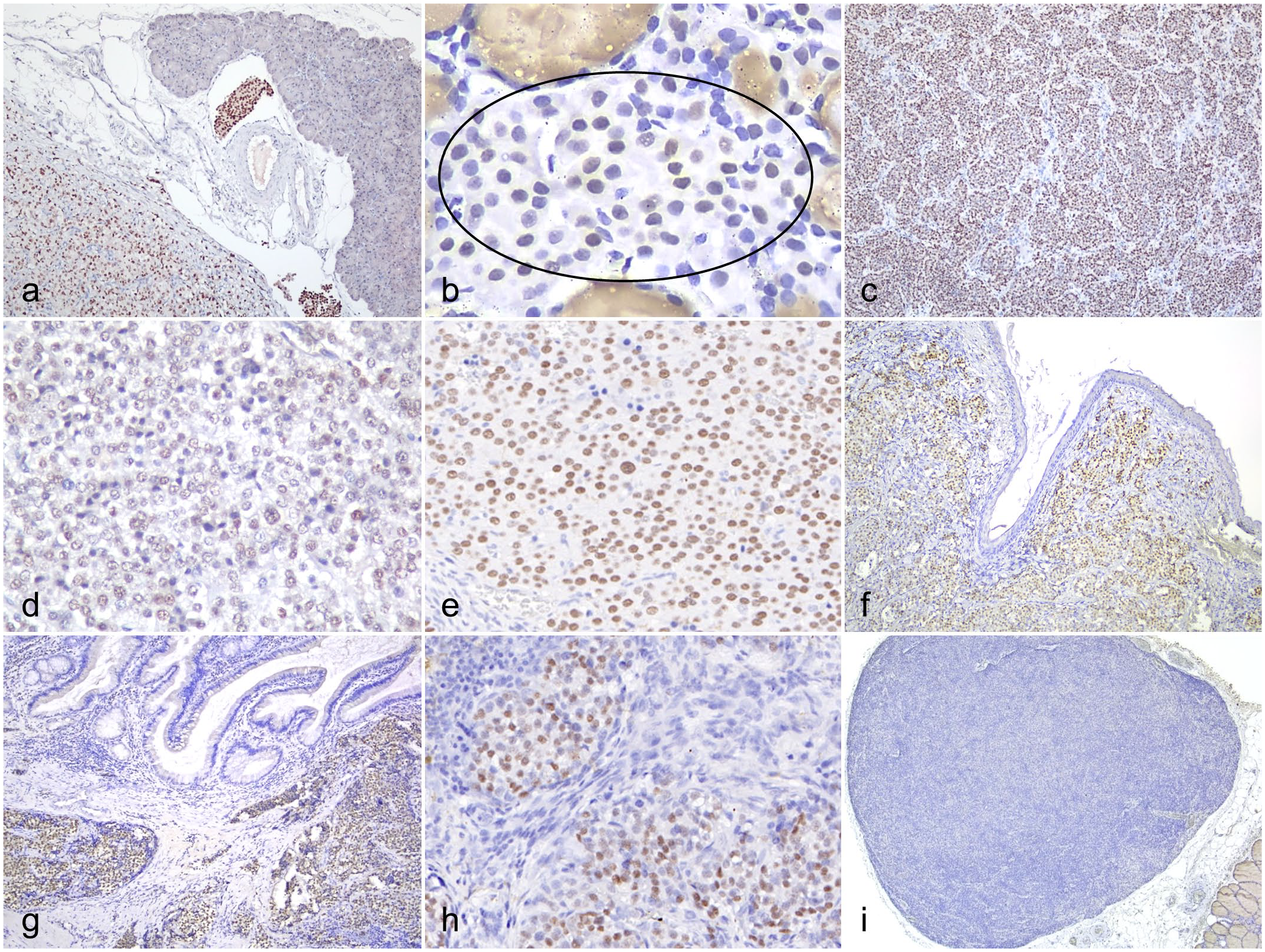
Insulinoma-associated protein 1 (INSM1) expression in hyperplastic and neoplastic neuroendocrine lesions in dogs. INSM1 immunohistochemistry. (a) Neoplastic cells and tumor emboli in an islet cell carcinoma are labeled for INSM1. (b) Hyperplastic thyroid C cells show variable positive labeling (oval). Nonspecific binding between the anti-INSM1 antibody and the colloid of thyroid tissue is observed. (c) C-cell carcinoma labeled for INSM1. (d) Pheochromocytoma labeled for INSM1. (e) Malignant chemodectoma labeled for INSM1. (f) Merkel cell carcinoma labeled for INSM1. (g) Neuroendocrine carcinoma arising from the gall bladder labeled for INSM1. (h) Metastatic neuroendocrine carcinoma (unknown primary origin) in a lymph node labeled for INSM1. (i) Parathyroid adenoma does not express INSM1.
The IHC results for INSM1 were compared to those for the traditional NE markers CGA and SYP (Table 2). INSM1 expression was detected in 91% (79/87) of the canine NE hyperplastic lesions and neoplasms examined. CGA was positive in 99% (86/87) of these cases and SYP in 90% (78/87). The percentage of NE neoplasms that were immunolabeled positively for all 3 markers was 89% (77/87), while all cases were immunolabeled for at least 1 of the 3 markers. Notably, INSM1 and SYP were not detected in any parathyroid neoplasms (3 parathyroid adenomas and 3 parathyroid carcinomas) or in 1 pituitary chromophobe adenoma. One of ten malignant pheochromocytomas did not label for INSM1. Of the malignant chemodectomas, one was negative for SYP and the other for both CGA and SYP (but positive for neuron-specific enolase).
Comparison of immunohistochemical expression of insulinoma-associated protein 1 (INSM1), chromogranin A (CGA), and synaptophysin (SYP) in canine neuroendocrine neoplasms.
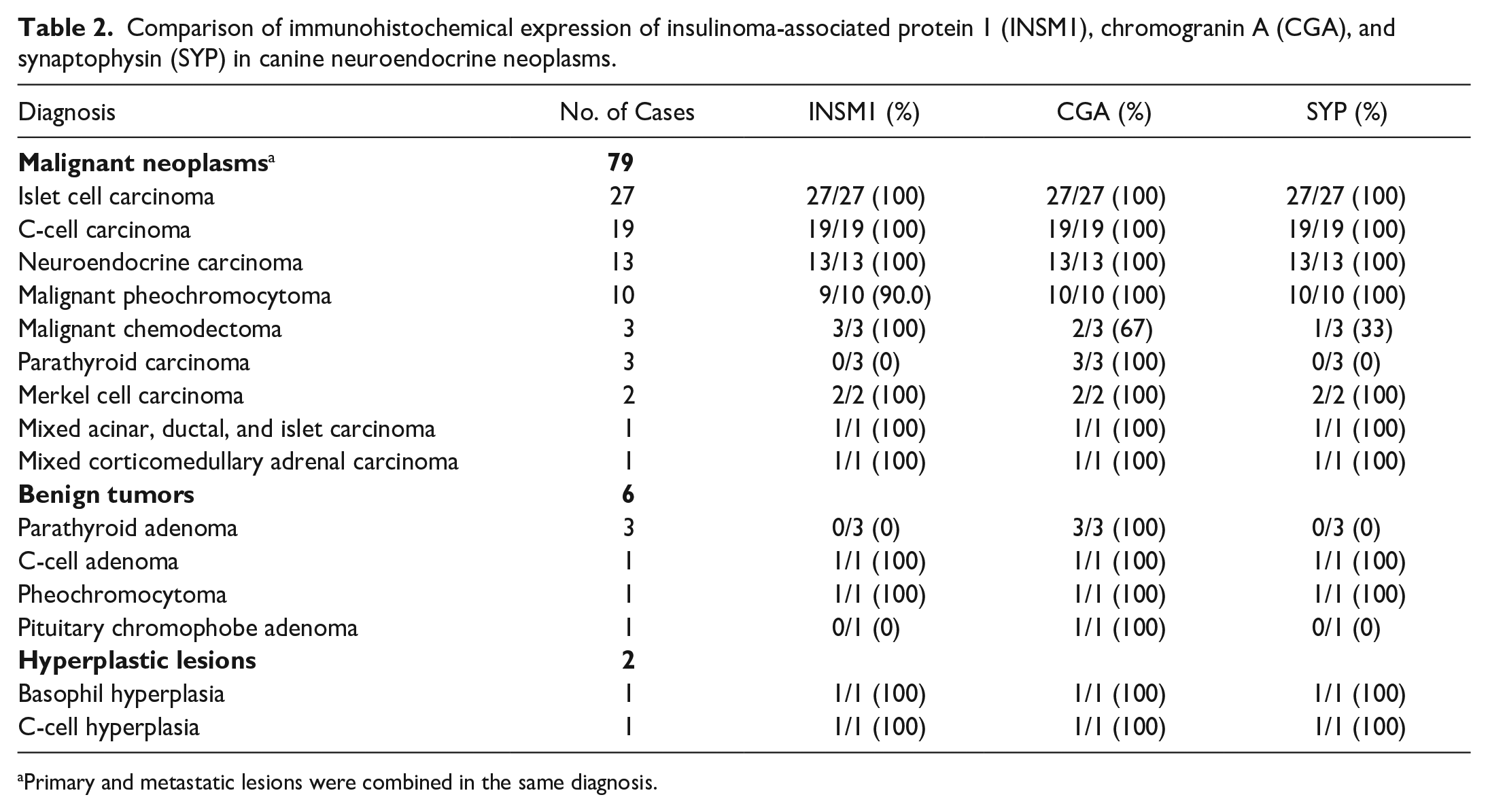
Primary and metastatic lesions were combined in the same diagnosis.
Canine Non-NE Neoplasms
In the canine non-NE neoplasm group, 98% (61/62) of cases were negative for INSM1 by IHC. The final diagnosis of these 62 cases included 19 carcinomas not otherwise specified, 10 thyroid follicular carcinomas, 6 sarcomas not otherwise specified, 3 adrenal cortical adenomas, 3 adrenocortical carcinomas, 3 amelanotic melanomas, 3 mammary gland carcinomas, 3 nasal transitional carcinomas, 2 anal sac adenocarcinomas, 2 lymphomas, 2 trichoblastomas, and 1 case each of hamartoma, histiocytoma, pigmented malignant melanoma, malignant seminoma, olfactory neuroblastoma, and meningioma. Only 1 amelanotic melanoma, which labeled positively for vimentin and Melan-A, showed weak positive labeling with the INSM1 antibody in less than 10% of the neoplastic cells (Supplemental Figure S4).
Discussion
In this study, the expression of INSM1 was detected in the nuclei of normal canine NE tissues, with the exception of the parathyroid gland. Furthermore, lack of or variable expression of 3 NE markers, INSM1, CGA, and SYP, was observed in the specimens from the parathyroid gland, small intestine, and tactile hairs. This finding indicates a potential tissue-specific pattern among different NE markers that could affect their diagnostic sensitivity and specificity.
The majority of the cases in the NE proliferation group were malignant tumors, of which islet cell carcinomas and medullary thyroid carcinomas accounted for more than half. The fact that INSM1 was consistently expressed in 100% of the 2 most common tumor types in our cases suggests that the INSM1 antibody could be a valuable diagnostic tool. For example, differentiating medullary thyroid carcinoma from thyroid follicular or parathyroid carcinoma may pose a diagnostic challenge.1,8 When faced with this situation, the combined usage of INSM1 and thyroid transcription factor-1 could be useful. The latter may be positive in both medullary thyroid and thyroid follicular carcinoma, 18 while INSM1 is only positive in medullary thyroid carcinoma.
INSM1 was not detectable in normal, hyperplastic, or neoplastic canine parathyroid glands, which is consistent with previous reports in human medicine.21,22,31 There is no conclusive explanation for why parathyroids and their derivative neoplasms do not express INSM1. One study stated that perhaps there was an alternative transcriptional mechanism in the parathyroid gland. 22 Another study suggested that parathyroid tumors should be classified as endocrine rather than NE because of their consistent hormone production and inconsistent expression of NE markers. 21 In our study, parathyroid glands and their derivative neoplasms were always INSM1 negative, making INSM1 a potentially useful marker for excluding possible parathyroid neoplasms.
With parathyroid neoplasms excluded, INSM1 was detected using IHC in 98% of the canine NE hyperplastic and neoplastic specimens. One malignant pheochromocytoma in our study was negative for INSM1 but positive for CGA and SYP. According to the human literature, most reported pheochromocytomas are positive for INSM1,12,22 but a minority of pheochromocytomas lack detectable INSM1 expression. 5 On the other hand, some pheochromocytomas in dogs have been reported that do not label for CGA or/and SYP. 2 Therefore, we recommend using at least 2 NE markers for supporting the diagnosis of canine malignant pheochromocytomas.
The other neoplasm that had a negative result for INSM1 was a pituitary chromophobe adenoma, which was also negative for SYP. One explanation for this result is that not all cells exhibit a positive labeling for INSM1 in normal pituitary tissue, as shown in Fig 1a. Another explanation is that the pituitary chromophobe adenoma included in our study was considered nonfunctional due to the lack of clinical findings compatible with specific hormone-related disorders. Since nonfunctional chromophobes appear to be derived from less differentiated pituitary cells, 23 we suggest that this may affect the expression of some immunohistochemical markers. However, owing to the extremely rare occurrence of pituitary gland tumors in dogs in our institution, the expression pattern of INSM1 in the pituitary gland could not be fully elucidated in this study.
The IHC positivity of INSM1 should be limited to the nucleus, but intracytoplasmic labeling in normal NE cells of pancreatic islets, anterior pituitary, and adrenal medulla was occasionally observed in our cases. Cytoplasmic INSM1 immunolabeling has been recorded in exocrine pancreatic tissue after 2 weeks of fixation. 12 Considering that INSM1 is a transcription factor with nuclear expression, the positivity of INSM1 outside of the nucleus should be regarded as nonspecific binding. Thus, the interpretation of the INSM1 immunolabeling is more straightforward than that of cytoplasmic NE markers. That is, when encountering small-sized specimens, intratumoral pigments (such as melanin or hemosiderin), neoplastic cells with a high nuclear/cytoplasmic ratio, and neoplastic cells with intracytoplasmic vacuoles, INSM1, with a clear nuclear pattern, offers advantages over common cytoplasmic NE markers.
One of the limitations of this study is the limited number of certain canine NE neoplasm subgroups in different anatomic locations, such as chemodectomas, Merkel cell carcinoma, and pituitary NE tumors. The INSM1 positivity rate for these neoplasms should be further investigated using a larger case series. Furthermore, additional studies on the potential diagnostic value of the anti-INSM1 antibody, such as INSM1 expression in NE neoplasms of other species, cytology specimens, and tissue microarrays from various non-NE neoplasms, are needed.
In summary, the anti-INSM1 antibody is a reliable diagnostic marker for NE neoplasms in various canine FFPE specimens with multiple advantages, such as its high sensitivity and the nature of nuclear immunolabeling. According to the results of this study, IHC for INSM1 could be used as a first-choice marker for suspected NE tumors from various anatomical sites (Fig. 4). IHC for INSM1 in combination with thyroid transcription factor-1 could help differentiate between thyroid follicular carcinoma, thyroid C-cell carcinoma, and parathyroid carcinoma. The combination of immunolabeling for INSM1, CGA, and SYP can enhance the diagnostic accuracy of identifying canine NE tumors.
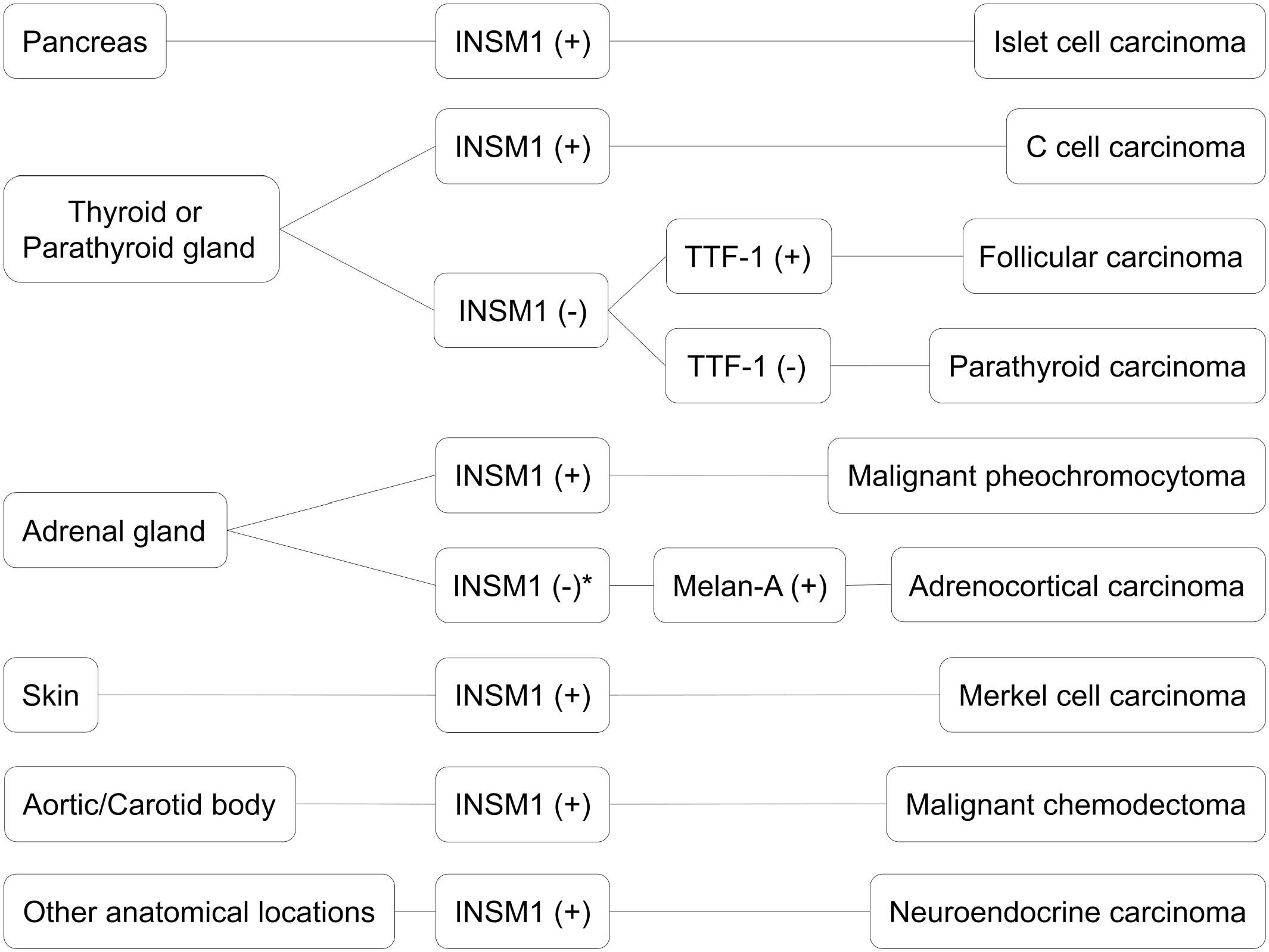
Diagnostic algorithm for insulinoma-associated protein 1 (INSM1) immunohistochemistry of canine neuroendocrine neoplasms with malignant biological behaviors. Thyroid transcription factor-1 (TTF-1) and Melan-A (not included in this study) are suggested for differentiating NE tumors from endocrine neoplasms. *Of note, an INSM1-negative malignant pheochromocytoma was recorded in this study. Therefore, examination of additional neuroendocrine markers is recommended for INSM1 and Melan-A double-negative adrenal neoplasms.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858241279127 – Supplemental material for Insulinoma-associated protein 1 (INSM1) immunohistochemical expression in normal, hyperplastic, and neoplastic canine neuroendocrine tissues
Supplemental material, sj-pdf-1-vet-10.1177_03009858241279127 for Insulinoma-associated protein 1 (INSM1) immunohistochemical expression in normal, hyperplastic, and neoplastic canine neuroendocrine tissues by Fu-Hua Yang, Ya-Pei Chang, Yen-Chen Chang, Hui-Wen Chang, Chian-Ren Jeng, Jia-Wei Jiang, Wen-Tien Tsao, I-Chi Luo and Wei-Hsiang Huang in Veterinary Pathology
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research was funded by the National Taiwan University under grant nos. 110L892903, 111L892303, and 112L891203. The personnel costs were partially supported by the Ministry of Agriculture, Executive Yuan, Taiwan under grant no. 110AS-2.7.1-AD-U1 and 111AS-2.7.1-AD-U1. The funding agency had no role in the study design, data acquisition, analysis, and interpretation of this work.
Supplemental Material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
